Li WH, Gu Z, Wang H, Nekrutenko A. Evolutionary analyses of the human genome. Nature. 2001;409:847–9.
Pavlícek A, Paces J, Elleder D, Hejnar J. Processed pseudogenes of human endogenous retroviruses generated by LINEs: their integration, stability, and distribution. Genome Res. 2002;12:391–9.
Blond JL, Lavillette D, Cheynet V, Bouton O, Oriol G, Chapel-Fernandes S, et al. An envelope glycoprotein of the human endogenous retrovirus HERV-W is expressed in the human placenta and fuses cells expressing the type D mammalian retrovirus receptor. J Virol. 2000;74:3321–9.
Lee X, Keith JC, Stumm N, Moutsatsos I, McCoy JM, Crum CP, et al. Downregulation of placental syncytin expression and abnormal protein localization in pre-eclampsia. Placenta. 2001;22:808–12.
Frendo J-L, Olivier D, Cheynet V, Blond J-L, Bouton O, Vidaud M, et al. Direct involvement of HERV-W Env glycoprotein in human trophoblast cell fusion and differentiation. Mol. Cell Biol. 2003;23:3566–74.
Mi S, Lee X, Li X, Veldman GM, Finnerty H, Racie L, et al. Syncytin is a captive retroviral envelope protein involved in human placental morphogenesis. Nature. 2000;403:785–9.
Gholami Barzoki M, Shatizadeh Malekshahi S, Heydarifard Z, Mahmodi MJ, Soltanghoraee H. The important biological roles of Syncytin-1 of human endogenous retrovirus W (HERV-W) and Syncytin-2 of HERV-FRD in the human placenta development. Mol Biol Rep. 2023;50:7901–7.
Rasko JE, Battini JL, Gottschalk RJ, Mazo I, Miller AD. The RD114/simian type D retrovirus receptor is a neutral amino acid transporter. Proc Natl Acad Sci USA. 1999;96:2129–34.
Tailor CS, Nouri A, Zhao Y, Takeuchi Y, Kabat D. A sodium-dependent neutral-amino-acid transporter mediates infections of feline and baboon endogenous retroviruses and simian type D retroviruses. J Virol. 1999;73:4470–4.
Marin M, Lavillette D, Kelly SM, Kabat D. N-linked glycosylation and sequence changes in a critical negative control region of the ASCT1 and ASCT2 neutral amino acid transporters determine their retroviral receptor functions. J Virol. 2003;77:2936–45.
Lavillette D, Marin M, Ruggieri A, Mallet F, Cosset F-L, Kabat D. The envelope glycoprotein of human endogenous retrovirus type W uses a divergent family of amino acid transporters/cell surface receptors. J Virol. 2002;76:6442–52.
Cheynet V, Ruggieri A, Oriol G, Blond J-L, Boson B, Vachot L, et al. Synthesis, assembly, and processing of the Env ERVWE1/syncytin human endogenous retroviral envelope. J Virol. 2005;79:5585–93.
Kanai Y, Clémençon B, Simonin A, Leuenberger M, Lochner M, Weisstanner M, et al. The SLC1 high-affinity glutamate and neutral amino acid transporter family. Mol Asp Med. 2013;34:108–20.
Freidman N, Chen I, Wu Q, Briot C, Holst J, Font J, et al. Amino acid transporters and exchangers from the SLC1A family: structure, mechanism and roles in physiology and cancer. Neurochem Res. 2020;45:1268–86.
Finkelshtein D, Werman A, Novick D, Barak S, Rubinstein M. LDL receptor and its family members serve as the cellular receptors for vesicular stomatitis virus. Proc Natl Acad Sci. 2013;110:7306–11.
Nikolic J, Belot L, Raux H, Legrand P, Gaudin Y, Albertini A. Structural basis for the recognition of LDL-receptor family members by VSV glycoprotein. Nat. Commun. 2018;9:1029.
Amirache F, Lévy C, Costa C, Mangeot P-E, Torbett BE, Wang CX, et al. Mystery solved: VSV-G-LVs do not allow efficient gene transfer into unstimulated T cells, B cells, and HSCs because they lack the LDL receptor. Blood. 2014;123:1422–4.
Bernadin O, Amirache F, Girard-Gagnepain A, Moirangthem RD, Lévy C, Ma K, et al. Baboon envelope LVs efficiently transduced human adult, fetal, and progenitor T cells and corrected SCID-X1 T-cell deficiency. Blood Adv. 2019;3:461–75.
Maurice M, Verhoeyen E, Salmon P, Trono D, Russell SJ, Cosset F-L. Efficient gene transfer into human primary blood lymphocytes by surface-engineered lentiviral vectors that display a T cell–activating polypeptide. Blood. 2002;99:2342–50.
Colamartino ABL, Lemieux W, Bifsha P, Nicoletti S, Chakravarti N, Sanz J, et al. Efficient and robust NK-cell transduction with baboon envelope pseudotyped lentivector. Front Immunol. 2019;10:2873.
Levy C, Fusil F, Amirache F, Costa C, Girard-Gagnepain A, Negre D, et al. Baboon envelope pseudotyped lentiviral vectors efficiently transduce human B cells and allow active factor IX B cell secretion in vivo in NOD/SCIDγc-/- mice. J Thromb Haemost. 2016;14:2478–92.
Fusil F, Calattini S, Amirache F, Mancip J, Costa C, Robbins JB, et al. A lentiviral vector allowing physiologically regulated membrane-anchored and secreted antibody expression depending on B-cell maturation status. Mol Ther. 2015;23:1734–47.
Sandrin V, Boson B, Salmon P, Gay W, Nègre D, Le Grand R, et al. Lentiviral vectors pseudotyped with a modified RD114 envelope glycoprotein show increased stability in sera and augmented transduction of primary lymphocytes and CD34+ cells derived from human and nonhuman primates. Blood. 2002;100:823–32.
Girard-Gagnepain A, Amirache F, Costa C, Lévy C, Frecha C, Fusil F, et al. Baboon envelope pseudotyped LVs outperform VSV-G-LVs for gene transfer into early-cytokine-stimulated and resting HSCs. Blood. 2014;124:1221–31.
Piovan C, Marin V, Scavullo C, Corna S, Giuliani E, Bossi S, et al. Vectofusin-1 promotes RD114-TR-pseudotyped lentiviral vector transduction of human HSPCs and T lymphocytes. Mol Ther Methods Clin Dev. 2017;5:22–30.
Ward M, Sattler R, Grossman IR, Bell AJ, Skerrett D, Baxi L, et al. A stable murine-based RD114 retroviral packaging line efficiently transduces human hematopoietic cells. Mol Ther. 2003;8:804–12.
Radek C, Bernadin O, Drechsel K, Cordes N, Pfeifer R, Sträßer P, et al. Vectofusin-1 improves transduction of primary human cells with diverse retroviral and lentiviral pseudotypes, enabling robust, automated closed-system manufacturing. Hum Gene Ther. 2019;30:1477–93.
Labenski V, Suerth JD, Barczak E, Heckl D, Levy C, Bernadin O, et al. Alpharetroviral self-inactivating vectors produced by a superinfection-resistant stable packaging cell line allow genetic modification of primary human T lymphocytes. Biomaterials. 2016;97:97–109.
Ren Q, Zu Y, Su H, Lu Q, Xiang B, Luo Y, et al. Single VHH-directed BCMA CAR-NK cells for multiple myeloma. Exp Hematol Oncol. 2023;12:98.
Ciulean IS, Fischer J, Quaiser A, Bach C, Abken H, Tretbar US, et al. CD44v6 specific CAR-NK cells for targeted immunotherapy of head and neck squamous cell carcinoma. Front Immunol. 2023;14:1290488.
Weidner T, Agarwal S, Perian S, Fusil F, Braun G, Hartmann J, et al. Genetic in vivo engineering of human T lymphocytes in mouse models. Nat Protoc. 2021;16:3210–40.
Bari R, Granzin M, Tsang KS, Roy A, Krueger W, Orentas R, et al. A distinct subset of highly proliferative and Lentiviral Vector (LV)-transducible NK cells define a readily engineered subset for adoptive cellular therapy. Front Immunol. 2019;10:2001.
Ito M, Hiramatsu H, Kobayashi K, Suzue K, Kawahata M, Hioki K, et al. NOD/SCID/gamma(c)(null) mouse: an excellent recipient mouse model for engraftment of human cells. Blood. 2002;100:3175–82.
McIntosh BE, Brown ME, Duffin BM, Maufort JP, Vereide DT, Slukvin II, et al. Nonirradiated NOD,B6.SCID Il2rγ-/- Kit(W41/W41) (NBSGW) mice support multilineage engraftment of human hematopoietic cells. Stem Cell Rep. 2015;4:171–80.
Miller PH, Rabu G, MacAldaz M, Knapp DJHF, Cheung AMS, Dhillon K, et al. Analysis of parameters that affect human hematopoietic cell outputs in mutant c-kit-immunodeficient mice. Exp Hematol. 2017;48:41–49.
Brendel C, Rio P, Verhoeyen E. Humanized mice are precious tools for evaluation of hematopoietic gene therapies and preclinical modeling to move towards a clinical trial. Biochem Pharmacol. 2020;174:113711.
Rogers GL, Huang C, Mathur A, Huang X, Chen H-Y, Stanten K, et al. Reprogramming human B cells with custom heavy-chain antibodies. Nat Biomed Eng. 2024;8:1700–14.
Luo B, Zhan Y, Luo M, Dong H, Liu J, Lin Y, et al. Engineering of α-PD-1 antibody-expressing long-lived plasma cells by CRISPR/Cas9-mediated targeted gene integration. Cell Death Dis. 2020;11:973.
Nahmad AD, Raviv Y, Horovitz-Fried M, Sofer I, Akriv T, Nataf D, et al. Engineered B cells expressing an anti-HIV antibody enable memory retention, isotype switching and clonal expansion. Nat Commun. 2020;11:5851.
Jamil A, Qureshi Z, Siddique R, Altaf F, Jamil R, Wali N. A meta-analysis on effects of chimeric antigen receptor T-cell therapy in relapsed or refractory B-cell acute lymphoblastic leukemia. Am. J. Clin. Oncol. https://doi.org/10.1097/COC.0000000000001176 2025.
Ibrahiam AT, Geddada S, Ullah N, Al-Qassab ZM, Ahmed O, Khan S. Chimeric Antigen Receptor (CAR) T-cell therapy in the treatment of diffuse large B-cell Lymphoma (DLBCL): a systematic review. Cureus. 2024;16:e75854.
Canichella M, de Fabritiis P. CAR-T therapy beyond B-cell hematological malignancies. Cells. 2025;14:41.
Robinson WH, Fiorentino D, Chung L, Moreland LW, Deodhar M, Harler MB, et al. Cutting-edge approaches to B-cell depletion in autoimmune diseases. Front Immunol. 2024;15:1454747.
Testa U, Chiusolo P, Pelosi E, Castelli G, Leone G. CAR-T cell therapy for T-Cell malignancies. Mediterr J Hematol Infect Dis. 2024;16:e2024031.
Sanber K, Savani B, Jain T. Graft-versus-host disease risk after chimeric antigen receptor T-cell therapy: the diametric opposition of T cells. Br J Haematol. 2021;195:660–8.
Mohammad A, Yurina A, Simonyan T, Chistyakov D, Salman R, Zornikova K, et al. Modular (universal) CAR-T platforms in vivo: a comprehensive systematic review. Front Immunol. 2024;15:1409665.
Carlson S, Lin TL, Larson SM. Allogeneic chimeric antigen receptors (CARs) as an ‘off-the-shelf’ therapy in multiple myeloma. Immunotherapy 17, 211–222 (2025).
Krug A, Martinez-Turtos A, Verhoeyen E. Importance of T, NK, CAR T and CAR NK cell metabolic fitness for effective anti-cancer therapy: a continuous learning process allowing the optimization of T, NK and CAR-based anti-cancer therapies. Cancers. 2021;14:183.
Tucci F, Galimberti S, Naldini L, Valsecchi MG, Aiuti A. A systematic review and meta-analysis of gene therapy with hematopoietic stem and progenitor cells for monogenic disorders. Nat Commun. 2022;13:1315.
Ghani K, Wang X, de Campos-Lima PO, Olszewska M, Kamen A, Rivière I, et al. Efficient human hematopoietic cell transduction using RD114- and GALV-pseudotyped retroviral vectors produced in suspension and serum-free media. Hum Gene Ther. 2009;20:966–74.
Mock U, Thiele R, Uhde A, Fehse B, Horn S. Efficient lentiviral transduction and transgene expression in primary human B cells. Hum Gene Ther Methods. 2012;23:408–15.
Bovia F, Salmon P, Matthes T, Kvell K, Nguyen TH, Werner-Favre C, et al. Efficient transduction of primary human B lymphocytes and nondividing myeloma B cells with HIV-1-derived lentiviral vectors. Blood. 2003;101:1727–33.
Serafini M, Naldini L, Introna M. Molecular evidence of inefficient transduction of proliferating human B lymphocytes by VSV-pseudotyped HIV-1-derived lentivectors. Virology. 2004;325:413–24.
Funke S, Schneider IC, Glaser S, Mühlebach MD, Moritz T, Cattaneo R, et al. Pseudotyping lentiviral vectors with the wild-type measles virus glycoproteins improves titer and selectivity. Gene Ther. 2009;16:700–5.
Frecha C, Costa C, Lévy C, Nègre D, Russell SJ, Maisner A, et al. Efficient and stable transduction of resting B lymphocytes and primary chronic lymphocyte leukemia cells using measles virus gp displaying lentiviral vectors. Blood. 2009;114:3173–80.
Marofi F, Saleh MM, Rahman HS, Suksatan W, Al-Gazally ME, Abdelbasset WK, et al. CAR-engineered NK cells; a promising therapeutic option for treatment of hematological malignancies. Stem Cell Res Ther. 2021;12:374.
Pfefferle A, Contet J, Wong K, Chen C, Verhoeyen E, Slichter CK, et al. Optimisation of a primary human CAR-NK cell manufacturing pipeline. Clin Transl Immunol. 2024;13:e1507.
Bernard P-L, Delconte R, Pastor S, Laletin V, Costa Da Silva C, Goubard A, et al. Targeting CISH enhances natural cytotoxicity receptor signaling and reduces NK cell exhaustion to improve solid tumor immunity. J Immunother Cancer. 2022;10:e004244.
Müller S, Bexte T, Gebel V, Kalensee F, Stolzenberg E, Hartmann J, et al. High cytotoxic efficiency of lentivirally and alpharetrovirally engineered cd19-specific chimeric antigen receptor natural killer cells against acute lymphoblastic leukemia. Front Immunol. 2019;10:3123.
Costa C, Hypolite G, Bernadin O, Lévy C, Cosset F-L, Asnafi V, et al. Baboon envelope pseudotyped lentiviral vectors: a highly efficient new tool to genetically manipulate T-cell acute lymphoblastic leukaemia-initiating cells. Leukemia. 2017;31:977–80.
Štafl K, Trávníček M, Janovská A, Kučerová D, Pecnová Ľ, Yang Z, et al. Receptor usage of Syncytin-1: ASCT2, but not ASCT1, is a functional receptor and effector of cell fusion in the human placenta. Proc Natl Acad Sci USA. 2024;121:e2407519121.





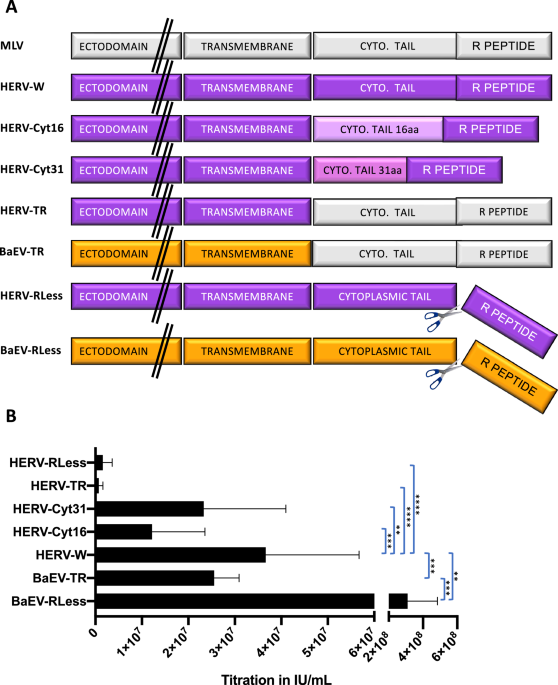









Leave a Reply