This study is a comprehensive narrative review of contemporary drug causes of inflammatory colitis. A literature search was conducted using Ovid (Medline). 758 studies were screened by a single author and 26 studies were included in the results of this review. Search terms included “colitis,” “colitides,” “colonic inflammation,” “non-infectious colitis,” “macroscopic colitis,” “drug induced,” “medication induced” and “adverse event.”
Original studies, meta-analyses, systematic reviews, review articles and case series n ≥ 2 were included in this review. Single case reports were included where case series n ≥ 2 and/or other larger studies existed for colitis secondary to the same drug class. Studies published from 2020 onwards were included given the focus of this review on emerging drug classes.
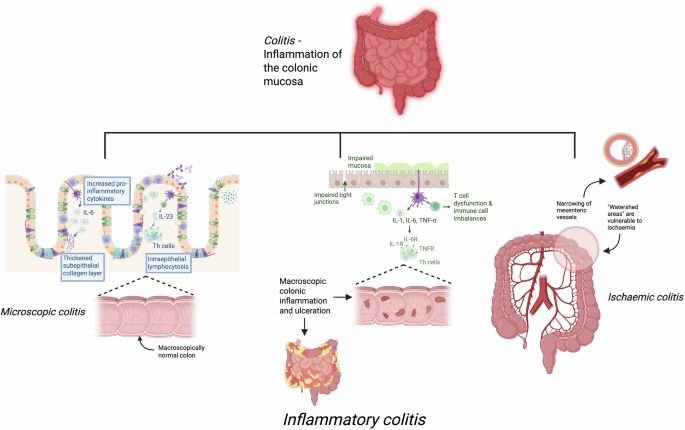
Studies on microscopic colitis, ischaemic colitis, infectious colitis and ICI-induced colitis were excluded. Exclusion search terms including “microscopic colitis”, “ischaemic colitis” and “immune checkpoint inhibitor” were incorporated into the search strategy. Editorials, book chapters and comments were excluded. Studies published in a language other than English were excluded. All case reports were screened and where there were no larger studies for the same drug class, single case reports was excluded. Studies published prior to 2020 were excluded.
Tyrosine kinase inhibitors
Drug indication
Tyrosine kinase inhibitors (TKI) inhibit the phosphorylation of tyrosine kinase enzymes, disrupting cancer cell growth, differentiation and apoptosis4,17. This drug class continues to expand rapidly and has revolutionised cancer treatment over the past two decades. TKIs are approved primarily for the treatment of haematological malignancies, including chronic myeloid leukaemia (CML) and solid tumours, such as lung, breast, gastrointestinal and genitourinary malignancies4,17,18.
Mechanism of colitis
The mechanisms of TKI-induced colitis are not well characterised. An increase in gastrointestinal inflammation, direct mucosal toxicity and secretory diarrhoea secondary to the inhibition of luminal epidermal growth factor receptors (EGFR) are possible contributors17. Reactivation of cytomegalovirus (CMV) has been implicated in the pathogenesis of dasatinib-induced colitis specifically18. It is likely that CMV acts as an inflammatory promotor in the colonic mucosa, although the exact mechanism is unclear18.
Prevalence
TKI-induced diarrhoea occurs secondary to nearly every TKI class with varying incidences4. In a large single centre study of patients on over 12 different TKIs, 10.5% developed gastrointestinal adverse effects17. Of these patients, the majority (91.7%) presented with Common Terminology Criteria for Adverse Events (CTCAE) grade 1–2 diarrhoea17. Up to 90% of patients treated with afatinib have also been reported to develop diarrhoea4. Furthermore, a randomised trial of tucatinib observed diarrhoea in 80.9% of patients, with CTCAE grade 3-4 diarrhoea being reported in 12.9% of patients19. In another study of patients receiving dasatinib, 17.6% were diagnosed with dasatinib-induced colitis18.
Clinical presentation
The clinical characteristics and severity of TKI-induced colitis are similar across different TKI classes17. Mild diarrhoea (CTCAE grade 1-2) is the most common symptom reported, as well as nausea, vomiting, abdominal pain and haematochezia4,17,18,19. Median time to onset of gastrointestinal adverse events, including colitis, was 68 days in one single centre study17. Patients with dasatinib-induced haemorrhagic colitis specifically often present with severe haematochezia18. A case of severe colitis with grade 3 CTCAE diarrhoea and intestinal haemorrhage secondary to osimertinib has also been described20.
Endoscopy/histology
Endoscopic findings are variable. Liu et al. described left-sided non-ulcerative inflammation (33.3%), mucosal ulcers (13.3%), and normal colonoscopy findings in the remaining study population17. Dasatinib-induced colitis presented with loss of vascular pattern (100%) and multiple erosions (83.3%) in another recent analysis18. Dasatinib-induced haemorrhagic colitis may present as oedematous, erythematous and friable mucosa with areas of erosion and spontaneous bleeding18. Histologically, findings of acute colitis, chronic inflammatory cell infiltrate, ischaemic change in the lamina propria, nuclear inclusion bodies and positive CMV immunostaining have been described17,18,20. Infiltrates of CMV-positive cells, natural killer (NK) cells and cytotoxic T cells in the colonic mucosa of patients with dasatinib-induced colitis have been observed, suggestive of an active immune response to CMV18. A recent case series also highlights a novel Crohn’s-like histopathological pattern in 4 patients on dasatinib13. In this study, discontinuous inflammation was reported in all cases and clear microgranulomas in 2 patients13. Ileal, right colon and rectal tissues showed either no or mild, patchy inflammation13.
Phosphatidylinositol 3-kinase inhibitors
Drug indication
Hyperactive and dysregulated phosphatidylinositol 3-kinase (Pi3K) signaling is critical in the development of many cancers21. Pi3K-inhibitors target Pi3K signaling pathways and are widely approved for the treatment of advanced metastatic breast cancers and indolent B-cell malignancies22.
Mechanism of colitis
Inhibition of the Pi3K pathway alters the downstream activation and differentiation of B cells and T cells and is thought to contribute to immune mediated toxicities, including colitis, interstitial pneumonitis and hepatitis22. Gastrointestinal inflammation and damage may be a result of decreased regulatory T cell activity and enhanced cytotoxic T cell activity specifically22.
Prevalence
In a review of the FDA Adverse Event Reporting System (FAERS) database, 20 cases of alpelisib-induced colitis were identified3. Causality assessment revealed only 4 cases with a probable causal link, while the remaining 16 cases were reported as having a possible causal link3. Idelalisib-induced colitis occurred in 6 of 15 patients (40%) treated with idelalisib in a recent single centre cohort study22. This is comparatively higher than a prior, larger study which observed CT CAE grade 3 and 4/5 diarrhoea at frequencies of 10.9% and 8.2% respectively23. CT CAE grade 3-4 diarrhoea and/or colitis was identified in 15% of patients on idelalisib in another retrospective analysis of nine clinical trials4,16,24.
Clinical presentation
Pi3K-induced colitis is characterised by severe diarrhoea (CT CAE grade 3-5) and delayed symptom onset of median 4 months22. Alpelisib-induced colitis specifically is reported to present with CT CAE grade 2-5 diarrhoea within 6-months of drug initiation3. In this study, 95% of patients required hospitalisation and 20% of patients presented with intestinal perforation3. Another single center study reported that up to half of patients with Pi3K-induced colitis required intensive care admission22.
Endoscopy/histology
Specific endoscopic findings have not been widely described. Erythema, absence of vascular pattern and superficial ulceration were seen in some cases of idelalisib-induced colitis22. Four histological patterns of idelalisib-induced colitis have been identified, including an apoptotic pattern, inflammatory pattern, mixed inflammatory and apoptotic pattern and normal mucosal pattern16. In the same study, infectious pathogens, including CMV, adenovirus, and herpesvirus 6, were detected in 31% of samples; suggesting an infectious overlay16.
IL-6 receptor antagonists – Tocilizumab
Drug indication
Tocilizumab (TCZ) is a monoclonal antibody targeting the interleukin-6 (IL-6) receptor25. By blocking the pro-inflammatory effects of IL-6, TCZ is thought to induce remission and reduce steroid exposure in patients with treatment refractory rheumatological conditions including giant cell arteritis, rheumatoid arthritis and juvenile idiopathic arthritis25,26. Off-label use as a steroid-sparing agent in Takayasu arteritis has also been reported25.
Mechanism of colitis
IL-6 exhibits pleiotropic effects by activating the inflammatory response, maintaining crypt homeostasis and promoting intestinal epithelial cell proliferation, angiogenesis and tissue repair25,27,28. IL-6 deficient mice appear to have impaired intestinal epithelial repair processes and higher risk of epithelial erosion and mortality25. Thus, the inhibition of IL-6 by TCZ likely blunts its protective effects on the colon and may subsequently lead to inflammatory colitis25.
Prevalence
There is no wide scale prevalence data for TCZ-induced colitis. A single centre study of 34 patients treated with TCZ reported colitis requiring hospitalisation in 8.8% of patients5.
Clinical presentation
In a study of three patients with TCZ-induced colitis, abdominal pain, watery diarrhoea, fever and nausea were the main presenting symptoms; associated with non-elevated CRP levels in all cases5. Time to onset of colitis varied widely from 24 days to 18 months and one case was complicated by colonic perforation5. Borghini et al. reported a single case of a first presentation of UC following TCZ therapy28. In this case, the patient presented 3 months following TCZ initiation with bloody diarrhoea and raised CRP and transaminase levels28. Hanioka et al. described a case of new onset severe UC complicated by sigmoid perforation and proctocolectomy following 4 weeks of treatment with TCZ25. The same study described a patient with pre-existing UC who experienced a worsening of intestinal symptoms following TCZ commencement and steroid weaning25.
Endoscopy/histology
A recent case series described three cases of ascending colitis, two of which also had ileocaecal ulceration5. Rather, Borghini et al. present a case of Mayo score 1-2 descending colitis, with repeat endoscopic evaluation showing total mucosal healing after discontinuation of TCZ28. A case of pan-colonic mucosal oedema and erosions leading to a new diagnosis of UC in a single patient has also been reported25. Histological findings consistent with UC have been described in two cases, including inflammatory cell infiltrates, mucosal oedema, crypt abscesses and crypt atrophy25.
IL-17 inhibitors
Drug Indication
IL-17 inhibitors, including Secukinumab (SEC) and Ixekizumab (IXE), are primarily used to treat immune-mediated inflammatory diseases including psoriasis, psoriatic arthritis, and ankylosing spondylitis29. IL-17 is produced by Th17 cells and plays a central role in disease pathogenesis by driving neutrophil-mediated inflammation and keratinocyte activation30,31.
Mechanism of colitis
IL-17 maintains intestinal epithelial barrier integrity and permeability through modulation of tight junction proteins32,33. Blockade of IL-17A or its receptor has been shown to impair the functioning of tight junctions and subsequently exacerbate colitis in a murine model32. Thus, it is proposed that the inhibition of IL-17 precipitates colitis by compromising mucosal immunity, increasing intestinal barrier permeability, and promoting inflammation32,33.
Prevalence
A review of the FAERS database identified a total of 388 cases of IL-17 inhibitor-induced IBD and colitis15. Among these, SEC accounted for 348 cases (90%) and IXE accounted for 36 cases (9%)15. The same authors conducted a descriptive analysis of the published literature, whereby 34 cases exhibited evidence of IBD during treatment with SEC (79.4%) and IXE (20.6%)15. Of these, 31 cases represented new-onset IBD15. A recent systematic review also identified 27 published cases of new onset or exacerbation of pre-existing IBD associated with IL-17 inhibitor therapy34.
Clinical presentation
Among the 34 cases identified by Deng et al., the majority presented with diarrhoea (90.9%), abdominal pain (57.6%) and bloody diarrhoea (51.5%), consistent with other recent studies15,34,35,36. Fever, weight loss, and fatigue are described less commonly15,34,35,36. Time to symptom onset is variable, ranging from a several weeks to 24 months after initiation of IL-17 inhibitor therapy15,34,35,36.
Endoscopy/histology
Endoscopically, Deng et al. reported deep ulceration (46.4%), superficial ulceration (21.4%), active inflammation (28.5%), friable mucosa (14.3%), and loss of vascular pattern (14.3%)15. These findings are consistent with smaller case series where subsequent diagnoses of pancolitis or left-sided colitis were made35,36. Histopathological evaluation of 25 patients revealed chronic inflammation (39.3%), cryptitis (32%), crypt abscesses (32%), and granulomatous lesions (28%)15. This is largely consistent with several smaller case series; however, histological features of both acute and chronic inflammation were also noted35,36.
Anti-CD 20 monoclonal antibodies
Drug indication
The anti-CD20 monoclonal antibodies are successfully used in the clinical setting for the depletion of B cells to treat various types of haematological malignancies and autoimmune conditions14. As a class, they induce tumour killing via various mechanisms such as direct induction of apoptosis, antibody-dependent cell-mediated cytotoxicity or complement-dependent lysis37. Rituximab, a chimeric mouse-human monoclonal antibody was one of the first monoclonal antibodies ever developed for clinical use and its clinical indications have extended from B cell lymphoma and rheumatoid arthritis to other off-label indications including anti-neutrophil cytoplasmic antibody (ANCA) vasculitis, myasthenia gravis and IgG4 disease38. Ocrelizumab is a humanised chimeric monoclonal antibody with less immunogenicity risk to rituximab, approved for the treatment for the relapsing and primary progressive forms of multiple sclerosis (MS)39,40.
Mechanism of colitis
Anti-CD20-induced colitis is an adverse effect associated with B-cell depletion therapies like rituximab and ocrelizumab. The precise mechanism is not fully understood, but current evidence suggests a combination of immune dysregulation and mucosal immune imbalance14.
The proposed mechanism of drug-induced colitis observed with rituximab is through depletion of CD20 + B-cells, which have been shown to play a protective role against colitis14. B cells help maintain the gut mucosal barrier by producing secretory immunoglobulins like IgA and IgM antibodies which regulate the gut microbiota14. Loss of B-cell support can therefore lead to dysbiosis, promoting intestinal inflammation14. B-cells also produce IL-10, an anti-inflammatory cytokine which helps maintain gut homeostasis14. A significant reduction in B cells and IL-10 mediated by anti CD20 therapies can lead to increases in production of pro-inflammatory cytokines (Th1/Th17), that disrupt gut homeostasis and cause colitis14.
Prevalence
Colitis induced by ocrelizumab is a rare but potentially serious adverse effect. According to Kim et al., 39 cases have been reported in the literature and in the FAERS database. Of these, only three were considered to have a probable causal link to the monoclonal antibody therapy, while the remaining 35 were classified as having a possible association12.
The incidence of rituximab-induced colitis is estimated at approximately 4%, with a median onset of 20 months after the first dose41. Most cases of rituximab-induced colitis are mild and respond to supportive care41.
Clinical presentation
The clinical characteristics of drug-induced colitis observed with anti-CD20 therapies can mimic that of UC and CD, including symptoms of diarrhoea, abdominal pain and bloody stool14,40. In the context of ocrelizumab, patients with MS are at an increased risk of IBD, and vice versa. This is likely due to systemic inflammation, genetic predisposition, environmental influences and contributions from the gastrointestinal-brain axis14,40. The differentiating factor is that a life-long relapsing remitting course is not observed upon cessation of the culprit agent14,40. It is important to consider that B-cell recovery from rituximab generally ranges from 4 days to 4 weeks however ocrelizumab causes complete B-cell depletion by 6 months and complete repletion of B cells may take 1-3 years which may be a cause for a longer symptomatic trajectory14,40.
Endoscopy/histology
The endoscopic appearance may often resemble UC or CD. In a case series and review correlating anti-CD monoclonal antibody therapy with colitis, according to the colonoscopy and histological findings 27% of patients were confirmed to have CD and 18% to have UC14. Histology often resembles features seen in classic IBD with some distinguishing features. Colonic biopsy histology in a patient with ocrelizumab-induced colitis showed chronic active colitis with cryptitis and lymphocytic infiltrates without granulomas which are generally characteristic of de novo IBD3.
Management
Determining the diagnosis of drug-induced colitis is challenging. Lack of clinician awareness may result in delayed diagnosis or misdiagnosis, subsequently leading to non-essential diagnostic procedures and prolonged exposure to the offending agent. The challenge of diagnosis is also in part, due to the difficulty in differentiating the clinical, endoscopic and histologic features of drug-induced colitis from a first presentation of IBD. Therefore, the disease course may be a useful differentiating factor. Rapid clinical and biochemical improvement following cessation of the culprit agent, as well as repeat endoscopic and histopathological evaluation showing complete mucosal healing, is suggestive of a drug-induced pathology. Conversely, underlying IBD would be highly suspected in individuals with a relapsing-remitting course after drug discontinuation, highlighting the importance of longitudinal follow-up.
Novel offending agents are also commonly utilised in the treatment of chronic immune-mediated conditions and it is known that patients with pre-existing immune-mediated diseases are predisposed to developing other similar conditions, including IBD42,43. Kosmidou et al. demonstrated a 50% increased risk of comorbidity between MS and IBD, likely reflecting shared immunopathogenic mechanisms, genetic susceptibility, and chronic systemic inflammation43. Consequently, distinguishing drug-induced inflammatory colitis from the natural evolution of underlying disease may be particularly complex in this cohort.
A pragmatic diagnostic approach should commence with a detailed medication history, including drug timelines, factoring in recent commencement of biologics or immunotherapies known to be implicated in drug-induced colitis. Following initial investigations to exclude infectious causes, including stool tests to exclude Clostridioides difficle and cytomegalovirus, patients with moderate to severe symptoms or those with persistent diarrhoea despite cessation of the suspected culprit agent should be considered for endoscopic assessment. Atypical distribution patterns, marked epithelial apoptosis and lack of chronic histological features argue against the classic IBD presentation and may support a drug-induced cause where infection has been excluded2,16,44,45.
Currently, no standardized treatment guidelines exist for drug-induced inflammatory colitis. The cornerstone of management is discontinuing the offending medication, which often results in significant clinical improvement, although it’s important to consider the pharmacokinetic properties of some of the culprits discussed in this paper. The B cell depletion after administration of ocrelizumab for example, may take a median of 72 weeks following the last infusion to return to baseline and subsequently, symptoms may persist despite discontinuation14,40. Various forms of systemic corticosteroids such as intravenous hydrocortisone and prednisolone are frequently initiated in the management pathway, particularly in moderate to severe cases, often employing the treatment algorithm of IBD management12,15,22.
Infliximab, a TNF-α inhibitor holds the strongest evidence base for successful outcomes in steroid-refractory severe cases of drug-induced colitis14,15. It’s rapid onset of action has seen positive outcomes in clinical, biochemical and endoscopic remission in multiple case series and cohort studies12,14,15,25.
Vedolizumab has emerged as an alternative biologic, particularly in patients with malignancy or significant baseline immunosuppression, owing to its gut-selective mechanism and favourable safety profile. Despite its slower onset of action, successful use has been reported in ocrelizumab-induced colitis following steroid failure in patients with multiple sclerosis, whereby TNF-α inhibitors are generally avoided due to concerns regarding demyelination and relapse risk2,12,14.
Strengths and limitations
This review explores emerging drug causes of inflammatory colitis that, to our knowledge, have not been reviewed and collated in detail elsewhere. This content is highly applicable in clinical practice across multiple medical specialisations. Further, we highlight current knowledge gaps, including the lack of clear diagnostic frameworks and standardised management guidelines in steroid-refractory cases, and emphasise the need for further research in this space.
However, there are notable limitations of this review and of the studies included. The literature search was limited to a single database. This increases the likelihood of relevant studies being missed in the screening process and may reduce the comprehensiveness of this narrative review. Despite increasing recognition, the current evidence base on drug-induced inflammatory colitis remains limited with much of the literature stemming from individual case reports and small cohort studies with a paucity of large prospective studies evaluating incidence, risk factors or optimal management strategies across different drug classes. This may limit the generalisability of the findings of this review to broader clinical settings. Further, the strength of the evidence remains unclear without a formal risk of bias evaluation.














Leave a Reply