Elrobaa IH, New KJ. COVID-19: pulmonary and extra pulmonary manifestations. Front Public Health. 2021;9:711616.
Yeo S, Kim H, Lee J, Yi J, Chung YR. Retinal vascular occlusions in COVID-19 infection and vaccination: a literature review. Graefes Arch Clin Exp Ophthalmol. 2023;261:1793–808.
Goyal M, Murthy SI, Annum S. Retinal manifestations in patients following COVID-19 infection: a consecutive case series. Indian J Ophthalmol. 2021;69:1275–82.
Farshbafnadi M, Kamali Zonouzi S, Sabahi M, Dolatshahi M, Aarabi MH. Aging & COVID-19 susceptibility, disease severity, and clinical outcomes: the role of entangled risk factors. Exp Gerontol. 2021;154:111507.
Wilson B, Geetha KM. Lipid nanoparticles in the development of mRNA vaccines for COVID-19. J Drug Deliv Sci Technol. 2022;74:103553.
Smith E, Tran T, Gillies A, Yeung S, Ma PE. Multiple evanescent white dot syndrome following COVID-19 mRNA vaccination in two patients. Ocul Immunol Inflamm. 2022;30:1240–3.
Kaneda Y, Kawasaki C, Namba M, Kaneda U, Ozaki A, Tanimoto T. Optic neuritis after mRNA vaccination against COVID-19: a secondary analysis of post-approval national safety data by the Japanese government. Ann Eye Sci. 2023;8:1–4.
Lee DS, Kim JW, Lee KL, Jung YJ, Kang HW. Adverse events following COVID-19 vaccination in South Korea between February 28 and August 21, 2021: a nationwide observational study. J Infect Dis. 2022;118:173.
Odani S, Honda H, Tabuchi T. Association of COVID-19 vaccine intake with diagnosis, hospitalization, and oxygenation/ventilation: a longitudinal analysis, 2021-2022. Japan Vaccine. 2024;12:1264.
Hashimoto Y, Yamana H, Iwagami M, Ono S, Takeuchi Y, Michihata N, et al. Ocular adverse events after coronavirus disease 2019 mRNA vaccination: matched cohort and self-controlled case series studies using a large database. Ophthalmol. 2023;130:256–64.
Ng XL, Betzler BK, Testi I, Ho SL, Tien M, Ngo WK, et al. Ocular adverse events after COVID-19 vaccination. Ocul Immunol Inflamm. 2021;29:1.
Hurissi EA, et al. Ocular complications after COVID-19 vaccination: a systematic review. Medicina. 2024;60:249.
Zhang Y, Zhang H, Lv K, Lin X, Chen F, Cao H, et al. Retinal vascular occlusion following COVID-19 vaccination: a comprehensive review of observational study and pathophysiological mechanisms. Vaccines. 2025;13:733.
Leung HM, Au SCL. Retinal vein occlusion after COVID-19 vaccination—a review. Vaccines. 2023;11:1281.
World Health Organization. Causality Assessment of an Adverse Event Following Immunization (AEFI) : User Manual for the Revised WHO Classification, 2nd ed., 2019 Update. Geneva, Switzerland: World Health Organization; 2021.
Montano D. Frequency and associations of adverse reactions of COVID-19 vaccines reported to pharmacovigilance systems in the European Union and the United States. Front Public Health. 2022;9:756633.
Raethke M, van Hunsel F, Luxi N, Lieber T, Bellitto C, Mulder E, et al. Frequency and timing of adverse reactions to COVID-19 vaccines; A multi-country cohort event monitoring study. Vaccine. 2024;42:2357–69.
Guyatt G, Oxyman AD, Akl EA, Kunz R, Vist G, Brozek J, et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J Clin Epidemiol. 2011;4:383–94.
DerSimonian R, Laird N. Meta-analysis in clinical trials revisited. Contemp Clin Trials. 2015;45:139.
Ouranos K, Avila DV, Mylona EK, Vassilopoulos A, Vassilopoulos S, Shehadeh F, et al. Cumulative incidence and risk of infection in patients with rheumatoid arthritis treated with janus kinase inhibitors: a systematic review and meta-analysis. PLoS One. 2024;19: e0306548 .
Greenland S, Longnecker MP. Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am J Epidemiol. 1992;135:1301–9.
Dorney I, Shaia J, Kaelber DC, Talcott KE, Singh RP. Risk of new retinal vascular occlusion after mRNA COVID-19 vaccination within aggregated electronic health record data. JAMA Ophthalmol. 2023;141:441EP–447.
Coronavirus (COVID-19) vaccinations – our world in data. 2025. https://ourworldindata.org/covid-vaccinations.
Mohammadi D, Ghasemi M, Manouchehrian N, Zafarmand M, Akbari M, Boroumand AB. COVID-19 vaccines: current and future challenges. Front Pharmacol. 2024;15:1434181.
Mendonça SA, Lorincz R, Boucher P, Curiel DT. Adenoviral vector vaccine platforms in the SARS-CoV-2 pandemic. Vaccine. 2021;6:1–14.
Krellenstein J, Wilkinson G, Osmundson J, Keshanjee S, Rasmussen AL, El-Sadr W. 22 Billion in the Hole: Omicron’s Implications for Global mRNA Vaccine Needs in 2022. Brooklyn, NY: Prep4All; 2022.
Li Y, Tenchov R, Smoot J, Liu C, Watkins S, Zhou Q. A comprehensive review of the global efforts on COVID-19 vaccine development. ACS Cent Sci. 2021;7:512.
Sandoval C, Guerrero D, Munoz J, Godoy K, Souza-Mello V, Farias J. Effectiveness of mRNA, protein subunit vaccine and viral vectors vaccines against SARS-CoV-2 in people over 18 years old: a systematic review. Expert Rev Vaccines. 2023;22:35–53.
King ML. How manufacturing won or lost the COVID-19 vaccine race. Vaccine. 2024;42:1004–12.
Pelle MC, Provenzano M, Zaffina I, Pujia R, Giofre F & Luca S, et al. Endothelial dysfunction in COVID-19: potential mechanisms and possible therapeutic options. Life. 2022;12:1605.
Takacs A, Ecsedy M, Nagy ZZ. Possible COVID-19 MRNA vaccine-induced case of unilateral central retinal vein occlusion. Ocul Immunol Inflamm. 2023;31:1145–50.
Panigada M, Bottino N, Tagliabue P, Grasselli G, Novembrino C, Chantarangkul V, et al. Hypercoagulability of COVID-19 patients in intensive care unit: a report of thromboelastography findings and other parameters of hemostasis. J Thromb Haemost. 2020;18:1738–42.
Mani A, Ojha V. Thromboembolism after COVID-19 vaccination: a systematic review of such events in 286 patients. Ann Vasc Surg. 2022;84:12–20.e1.
Abrignani MG, Murrone A, De Luca L, Roncon L, Di Lenarda A, Valente A, et al. COVID-19, vaccines, and thrombotic events: a narrative review. J Clin Med. 2022;11:948.
Li JX, Wang YH, Bair H, Hsu SB, Chen C, Wei JCC, et al. Risk assessment of retinal vascular occlusion after COVID-19 vaccination. Vaccine. 2023;8:1–10.
Faksova K, Walsh D, Jiang Y, Griffin J, Philips A, Gentile A, et al. COVID-19 vaccines and adverse events of special interest: a multinational Global Vaccine Data Network (GVDN) cohort study of 99 million vaccinated individuals. Vaccine. 2024;42:2200–11.
Francis AI, Ghany S, Gilkes T, Umakanthan S. Review of COVID-19 vaccine subtypes, efficacy and geographical distributions. Postgrad Med J. 2022;98:389–94.
Trougakos IP, Terpos E, Alexopopulos H, Politou M, Paraskevis D, Scorilas A, et al. Adverse effects of COVID-19 mRNA vaccines: the spike hypothesis. Trends Mol Med. 2022;28:542.
Kim S, Jeon JH, Kim M, Lee Y, Hwang YH, Park M, et al. Innate immune responses against mRNA vaccine promote cellular immunity through IFN-β at the injection site. Nat Commun. 2024;15:1–15.
Canale MP, Menghini R, Martelli E, Federici M. COVID-19–associated endothelial dysfunction and microvascular injury: From pathophysiology to clinical manifestations. Card Electrophysiol Clin. 2021;14:21.
Fodor A, Tiperciuc B, Login C, Orasan OH, Lazar AL, Buchman C, et al. Endothelial dysfunction, inflammation, and oxidative stress in COVID-19—mechanisms and therapeutic targets. Oxid Med Cell Longev. 2021;2021:8671713.
Sekulovski M, Mileva N, Vasilev GV, Miteva D, Gulinac M, Preshevska-Sekulovska M, et al. Blood coagulation and thrombotic disorders following SARS-CoV-2 infection and COVID-19 vaccination. Biomedicines. 2023;11:2813.
Haseeb AA, Solyman O, Abushanab MM, Obaia ASA, Elhusseiny AM. Ocular complications following vaccination for COVID-19: a one-year retrospective. Vaccine. 2022;10:342.
Kyriakidis NC, López-Cortés A, González EV, Grimaldos AB, Prado EO. SARS-CoV-2 vaccines strategies: a comprehensive review of phase 3 candidates. Vaccine. 2021;6:28.
Zou Y, Kamoi K, Zong Y, Zhang J, Yang M, Ohno-Matsui K. Ocular inflammation post-vaccination. Vaccine. 2023;11:1626.
Sejournet L, Kodjikian L, Rezkallah A, Denis P, Mathis T, Loria O. Multiple evanescent white dot syndrome relapse following BNT162b2 mRNA COVID-19 vaccination. Int J Ophthalmol. 2023;16:1724.
Caldarelli M, Rio P, Giambra V, Gasbarrini A, Gambassi G, Cianci R. ASIA syndrome: State-of-the-art and future perspectives. Vaccine. 2024;12:1183.
Zhou L, Xu Z, Guerra J, Rosenberg AZ, Fenaroli P, Eberhart CG, et al. Expression of the SARS-CoV-2 receptor ACE2 in human retina and diabetes—implications for retinopathy. Invest Ophthalmol Vis Sci. 2021;62:6.
Karagöz IK, Tokuc EO, Karabas L, Ruckert R, Kaya Mucahit, Munk MR. Expression of key SARS-CoV-2 entry molecules in surgically obtained human retinal biopsies. Int Ophthalmol. 2023;43:5055–62.
Menuchin-Lasowski Y, Schreiber A, Lecanda A, Mecate-Zambrano A, Brunotte L, Psathaki OE, et al. SARS-CoV-2 infects and replicates in photoreceptor and retinal ganglion cells of human retinal organoids. Stem Cell Rep. 2022;17:789–803.
Gong J, Ge L, Zeng Y, Yang C, Luo Y, Kang J, et al. The influence of SARS-CoV-2 spike protein exposure on retinal development in the human retinal organoids. Cell Biosci. 2025;15:1–19.
World Health Organization. COVID-19 Epidemiological Update. Geneva, Switzerland: World Health Organization; 2025.





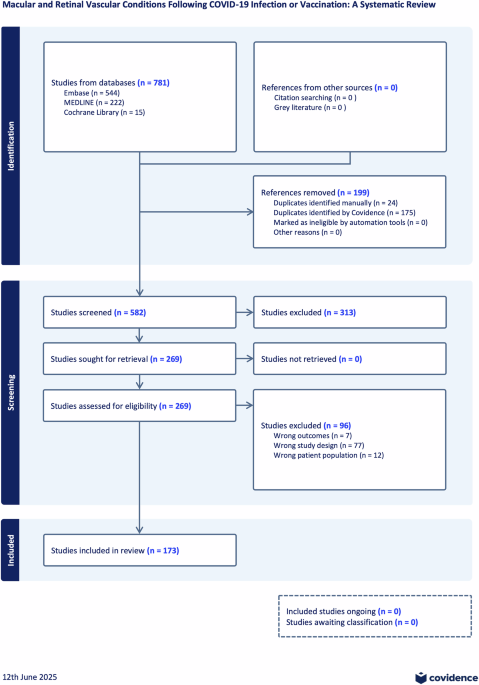










Leave a Reply