Jang, C., Chen, L. & Rabinowitz, J. D. Metabolomics and Isotope Tracing. Cell 173, 822–837 (2018).
Fischbach, M. A. Microbiome: focus on causation and mechanism. Cell 174, 785–790 (2018).
Wells, J. M., Rossi, O., Meijerink, M. & van Baarlen, P. Epithelial crosstalk at the microbiota-mucosal interface. Proc. Natl Acad. Sci. USA 108, 4607–4614 (2011).
Mongia, M. et al. Fast mass spectrometry search and clustering of untargeted metabolomics data. Nat. Biotechnol. https://doi.org/10.1038/s41587-023-01985-4 (2024).
Mohanty, I. et al. The underappreciated diversity of bile acid modifications. Cell https://doi.org/10.1016/j.cell.2024.02.019 (2024).
Meijnikman, A. S., Nieuwdorp, M. & Schnabl, B. Endogenous ethanol production in health and disease. Nat. Rev. Gastroenterol. Hepatol. https://doi.org/10.1038/s41575-024-00937-w (2024).
Mann, E. R., Lam, Y. K. & Uhlig, H. H. Short-chain fatty acids: linking diet, the microbiome and immunity. Nat. Rev. Immunol. https://doi.org/10.1038/s41577-024-01014-8 (2024).
Arifuzzaman, M., Collins, N., Guo, C. J. & Artis, D. Nutritional regulation of microbiota-derived metabolites: implications for immunity and inflammation. Immunity 57, 14–27 (2024).
Levy, M., Thaiss, C. A. & Elinav, E. Metabolites: messengers between the microbiota and the immune system. Genes Dev. 30, 1589–1597 (2016).
Aguilar-Toala, J. E. et al. Postbiotics — when simplification fails to clarify. Nat. Rev. Gastroenterol. Hepatol. 18, 825–826 (2021).
Levy, M. et al. Microbiota-modulated metabolites shape the intestinal microenvironment by regulating NLRP6 inflammasome signaling. Cell 163, 1428–1443 (2015).
Levy, H. L. et al. Efficacy of sapropterin dihydrochloride (tetrahydrobiopterin, 6R-BH4) for reduction of phenylalanine concentration in patients with phenylketonuria: a phase III randomised placebo-controlled study. Lancet 370, 504–510 (2007).
Cushing, K. & Higgins, P. D. R. Management of crohn disease-reply. JAMA 325, 1794–1795 (2021).
Gros, B. & Kaplan, G. G. Ulcerative colitis in adults: a review. JAMA 330, 951–965 (2023).
Bogale, K. et al. Symptoms associated with healthcare resource utilization in the setting of inflammatory bowel disease. Sci. Rep. 12, 10577 (2022).
Johansen, I. et al. Symptoms and symptom clusters in patients newly diagnosed with inflammatory bowel disease: results from the IBSEN III Study. BMC Gastroenterol. 23, 255 (2023).
Franzosa, E. A. et al. Gut microbiome structure and metabolic activity in inflammatory bowel disease. Nat. Microbiol. 4, 293–305 (2019).
Lloyd-Price, J. et al. Multi-omics of the gut microbial ecosystem in inflammatory bowel diseases. Nature 569, 655–662 (2019).
Michaudel, C. & Sokol, H. The gut microbiota at the service of immunometabolism. Cell Metab. 32, 514–523 (2020).
Pereira, G. V. et al. Opposing diet, microbiome, and metabolite mechanisms regulate inflammatory bowel disease in a genetically susceptible host. Cell Host Microbe 32, 527–542.e9 (2024).
Koh, A., De Vadder, F., Kovatcheva-Datchary, P. & Backhed, F. From dietary fiber to host physiology: short-chain fatty acids as key bacterial metabolites. Cell 165, 1332–1345 (2016).
Chang, P. V., Hao, L., Offermanns, S. & Medzhitov, R. The microbial metabolite butyrate regulates intestinal macrophage function via histone deacetylase inhibition. Proc. Natl Acad. Sci. USA 111, 2247–2252 (2014).
Ji, J. et al. Microbial metabolite butyrate facilitates M2 macrophage polarization and function. Sci. Rep. 6, 24838 (2016).
Byndloss, M. X. et al. Microbiota-activated PPAR-γ signaling inhibits dysbiotic Enterobacteriaceae expansion. Science 357, 570–575 (2017).
Singh, N. et al. Activation of Gpr109a, receptor for niacin and the commensal metabolite butyrate, suppresses colonic inflammation and carcinogenesis. Immunity 40, 128–139 (2014).
Cohen, L. J. et al. Commensal bacteria make GPCR ligands that mimic human signalling molecules. Nature 549, 48–53 (2017).
Dodd, D. et al. A gut bacterial pathway metabolizes aromatic amino acids into nine circulating metabolites. Nature 551, 648–652 (2017).
Lamas, B. et al. CARD9 impacts colitis by altering gut microbiota metabolism of tryptophan into aryl hydrocarbon receptor ligands. Nat. Med. 22, 598–605 (2016).
Islam, J. et al. Dietary tryptophan alleviates dextran sodium sulfate-induced colitis through aryl hydrocarbon receptor in mice. J. Nutr. Biochem. 42, 43–50 (2017).
Nikolaus, S. et al. Increased tryptophan metabolism is associated with activity of inflammatory bowel diseases. Gastroenterology 153, 1504–1516.e2 (2017).
Nieves, K. M. et al. Indole-3-propionic acid protects medium-diversity colitic mice via barrier enhancement preferentially over anti-inflammatory effects. Am. J. Physiol. Gastrointest. Liver Physiol. 328, G696–G715 (2025).
Alexeev, E. E. et al. Microbiota-derived indole metabolites promote human and murine intestinal homeostasis through regulation of interleukin-10 receptor. Am. J. Pathol. 188, 1183–1194 (2018).
Li, M. et al. Indole-3-acetic acid alleviates DSS-induced colitis by promoting the production of R-equol from Bifidobacterium pseudolongum. Gut Microbes 16, 2329147 (2024).
Iyer, S. S. et al. Dietary and microbial oxazoles induce intestinal inflammation by modulating aryl hydrocarbon receptor responses. Cell 173, 1123–1134.e11 (2018).
Michaudel, C. et al. Rewiring the altered tryptophan metabolism as a novel therapeutic strategy in inflammatory bowel diseases. Gut 72, 1296–1307 (2023).
Song, X. et al. Microbial bile acid metabolites modulate gut RORγ+ regulatory T cell homeostasis. Nature 577, 410–415 (2020).
Arifuzzaman, M. et al. Inulin fibre promotes microbiota-derived bile acids and type 2 inflammation. Nature 611, 578–584 (2022).
Kushkevych, I., Dordevic, D. & Vitezova, M. Possible synergy effect of hydrogen sulfide and acetate produced by sulfate-reducing bacteria on inflammatory bowel disease development. J. Adv. Res. 27, 71–78 (2021).
Flannigan, K. L. et al. Impaired hydrogen sulfide synthesis and IL-10 signaling underlie hyperhomocysteinemia-associated exacerbation of colitis. Proc. Natl Acad. Sci. USA 111, 13559–13564 (2014).
Mielke, L. A. et al. Retinoic acid expression associates with enhanced IL-22 production by γδ T cells and innate lymphoid cells and attenuation of intestinal inflammation. J. Exp. Med. 210, 1117–1124 (2013).
Huang, L. J. et al. Multiomics analyses reveal a critical role of selenium in controlling T cell differentiation in Crohn’s disease. Immunity 54, 1728–1744.e7 (2021).
Dong, L. et al. Mannose ameliorates experimental colitis by protecting intestinal barrier integrity. Nat. Commun. 13, 4804 (2022).
Schwarzler, J. et al. PUFA-induced metabolic enteritis as a fuel for Crohn’s disease. Gastroenterology 162, 1690–1704 (2022).
Wang, Z. et al. Synergistic role of gut-microbial L-ornithine in enhancing ustekinumab efficacy for Crohn’s disease. Cell Metab. https://doi.org/10.1016/j.cmet.2025.01.007 (2025).
Canavan, C., West, J. & Card, T. The epidemiology of irritable bowel syndrome. Clin. Epidemiol. 6, 71–80 (2014).
Enck, P. et al. Irritable bowel syndrome. Nat. Rev. Dis. Primers 2, 16014 (2016).
Lovell, R. M. & Ford, A. C. Global prevalence of and risk factors for irritable bowel syndrome: a meta-analysis. Clin. Gastroenterol. Hepatol. 10, 712–721.e4 (2012).
Oka, P. et al. Global prevalence of irritable bowel syndrome according to Rome III or IV criteria: a systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 5, 908–917 (2020).
Zhang, T. et al. Global research trends in irritable bowel syndrome: a bibliometric and visualized study. Front. Med. 9, 922063 (2022).
Staudacher, H. M., Black, C. J., Teasdale, S. B., Mikocka-Walus, A. & Keefer, L. Irritable bowel syndrome and mental health comorbidity — approach to multidisciplinary management. Nat. Rev. Gastroenterol. Hepatol. 20, 582–596 (2023).
Mars, R. A. T. et al. Longitudinal multi-omics reveals subset-specific mechanisms underlying irritable bowel syndrome. Cell 182, 1460–1473.e17 (2020).
Crowell, M. D. Role of serotonin in the pathophysiology of the irritable bowel syndrome. Br. J. Pharmacol. 141, 1285–1293 (2004).
Gros, M., Gros, B., Mesonero, J. E. & Latorre, E. Neurotransmitter dysfunction in irritable bowel syndrome: emerging approaches for management. J. Clin. Med. 10, 3429 (2021).
Agus, A., Planchais, J. & Sokol, H. Gut microbiota regulation of tryptophan metabolism in health and disease. Cell Host Microbe 23, 716–724 (2018).
Cremon, C. et al. Intestinal serotonin release, sensory neuron activation, and abdominal pain in irritable bowel syndrome. Am. J. Gastroenterol. 106, 1290–1298 (2011).
Gershon, M. D. Serotonin is a sword and a shield of the bowel: serotonin plays offense and defense. Trans. Am. Clin. Climatol. Assoc. 123, 268–280 (2012).
Keszthelyi, D., Troost, F. J. & Masclee, A. A. Understanding the role of tryptophan and serotonin metabolism in gastrointestinal function. Neurogastroenterol. Motil. 21, 1239–1249 (2009).
Spiller, R. Serotonin and GI clinical disorders. Neuropharmacology 55, 1072–1080 (2008).
Zhai, L. et al. Ruminococcus gnavus plays a pathogenic role in diarrhea-predominant irritable bowel syndrome by increasing serotonin biosynthesis. Cell Host Microbe 31, 33–44.e5 (2023).
Zhai, L. et al. Gut microbiota-derived tryptamine and phenethylamine impair insulin sensitivity in metabolic syndrome and irritable bowel syndrome. Nat. Commun. 14, 4986 (2023).
Bhattarai, Y. et al. Gut microbiota-produced tryptamine activates an epithelial g-protein-coupled receptor to increase colonic secretion. Cell Host Microbe 23, 775–785.e5 (2018).
Yano, J. M. et al. Indigenous bacteria from the gut microbiota regulate host serotonin biosynthesis. Cell 161, 264–276 (2015).
Shufflebotham, J. et al. Acute tryptophan depletion alters gastrointestinal and anxiety symptoms in irritable bowel syndrome. Am. J. Gastroenterol. 101, 2582–2587 (2006).
Wong, A. C. et al. Serotonin reduction in post-acute sequelae of viral infection. Cell 186, 4851–4867.e20 (2023).
Fritsch, P., Kolber, M. R. & Korownyk, C. Antidepressants for irritable bowel syndrome. Can. Fam. Physician 66, 265 (2020).
Bautzova, T. et al. 5-OxoETE triggers nociception in constipation-predominant irritable bowel syndrome through MAS-related G protein-coupled receptor D. Sci. Signal. 11, eaal2171 (2018).
De Palma, G. et al. Histamine production by the gut microbiota induces visceral hyperalgesia through histamine 4 receptor signaling in mice. Sci. Transl. Med. 14, eabj1895 (2022).
Macia, L. et al. Metabolite-sensing receptors GPR43 and GPR109A facilitate dietary fibre-induced gut homeostasis through regulation of the inflammasome. Nat. Commun. 6, 6734 (2015).
Shaidullov, I. F. et al. Short chain fatty acids and colon motility in a mouse model of irritable bowel syndrome. BMC Gastroenterol. 21, 37 (2021).
Eng, C. et al. Colorectal cancer. Lancet 404, 294–310 (2024).
Ramaboli, M. C. et al. Diet changes due to urbanization in South Africa are linked to microbiome and metabolome signatures of Westernization and colorectal cancer. Nat. Commun. 15, 3379 (2024).
Cong, J. et al. Bile acids modified by the intestinal microbiota promote colorectal cancer growth by suppressing CD8+ T cell effector functions. Immunity https://doi.org/10.1016/j.immuni.2024.02.014 (2024).
Li, D. et al. Gut microbiota-derived metabolite trimethylamine-N-oxide and multiple health outcomes: an umbrella review and updated meta-analysis. Am. J. Clin. Nutr. 116, 230–243 (2022).
Luu, M. et al. Microbial short-chain fatty acids modulate CD8+ T cell responses and improve adoptive immunotherapy for cancer. Nat. Commun. 12, 4077 (2021).
Dmitrieva-Posocco, O. et al. β-Hydroxybutyrate suppresses colorectal cancer. Nature 605, 160–165 (2022).
Sugimura, N. et al. Lactobacillus gallinarum modulates the gut microbiota and produces anti-cancer metabolites to protect against colorectal tumourigenesis. Gut 71, 2011–2021 (2022).
Cuevas-Ramos, G. et al. Escherichia coli induces DNA damage in vivo and triggers genomic instability in mammalian cells. Proc. Natl Acad. Sci. USA 107, 11537–11542 (2010).
Wilson, M. R. et al. The human gut bacterial genotoxin colibactin alkylates DNA. Science 363, eaar7785 (2019).
Dziubańska-Kusibab, P. J. et al. Colibactin DNA-damage signature indicates mutational impact in colorectal cancer. Nat. Med. 26, 1063–1069 (2020).
Nougayrède, J.-P. et al. Escherichia coli induces DNA double-strand breaks in eukaryotic cells. Science 313, 848–851 (2006).
Lara-Tejero, M. & Galán, J. E. A bacterial toxin that controls cell cycle progression as a deoxyribonuclease I-like protein. Science 290, 354–357 (2000).
Pöltl, L. et al. Microbiota-derived genotoxin tilimycin generates colonic stem cell mutations. Cell Rep. 42, 112199 (2023).
Cao, Y. et al. Commensal microbiota from patients with inflammatory bowel disease produce genotoxic metabolites. Science 378, eabm3233 (2022).
Boleij, A. et al. The Bacteroides fragilis toxin gene is prevalent in the colon mucosa of colorectal cancer patients. Clin. Infect. Dis. 60, 208–215 (2015).
Cheng, W. T., Kantilal, H. K. & Davamani, F. The mechanism of Bacteroides fragilis toxin contributes to colon cancer formation. Malays. J. Med. Sci. 27, 9–21 (2020).
Arner, E. N. & Rathmell, J. C. Metabolic programming and immune suppression in the tumor microenvironment. Cancer Cell 41, 421–433 (2023).
Mowat, C., Dhatt, J., Bhatti, I., Hamie, A. & Baker, K. Short chain fatty acids prime colorectal cancer cells to activate antitumor immunity. Front. Immunol. 14, 1190810 (2023).
He, Y. et al. Gut microbial metabolites facilitate anticancer therapy efficacy by modulating cytotoxic CD8+ T cell immunity. Cell Metab. 33, 988–1000.e7 (2021).
Katkeviciute, E. et al. Bacteria-derived 3-hydroxydodecanoic acid induces a potent anti-tumor immune response via the GPR84 receptor. Cell Rep. 44, 115357 (2025).
Zhang, Q. et al. Lactobacillus plantarum-derived indole-3-lactic acid ameliorates colorectal tumorigenesis via epigenetic regulation of CD8+ T cell immunity. Cell Metab. 35, 943–960.e9 (2023).
Fong, W. et al. Lactobacillus gallinarum-derived metabolites boost anti-PD1 efficacy in colorectal cancer by inhibiting regulatory T cells through modulating IDO1/Kyn/AHR axis. Gut 72, 2272–2285 (2023).
Jiang, S. S. et al. Fusobacterium nucleatum-derived succinic acid induces tumor resistance to immunotherapy in colorectal cancer. Cell Host Microbe 31, 781–797.e789 (2023).
Bell, H. N. et al. Microenvironmental ammonia enhances T cell exhaustion in colorectal cancer. Cell Metab. 35, 134–149.e6 (2023).
Imray, C. H. et al. Faecal unconjugated bile acids in patients with colorectal cancer or polyps. Gut 33, 1239–1245 (1992).
Ou, J. et al. Diet, microbiota, and microbial metabolites in colon cancer risk in rural Africans and African Americans. Am. J. Clin. Nutr. 98, 111–120 (2013).
Reddy, B. S. Role of bile metabolites in colon carcinogenesis. Animal models. Cancer 36, 2401–2406 (1975).
Zeng, H., Hamlin, S. K., Safratowich, B. D., Cheng, W. H. & Johnson, L. K. Superior inhibitory efficacy of butyrate over propionate and acetate against human colon cancer cell proliferation via cell cycle arrest and apoptosis: linking dietary fiber to cancer prevention. Nutr. Res. 83, 63–72 (2020).
Li, Q. et al. Carnobacterium maltaromaticum boosts intestinal vitamin D production to suppress colorectal cancer in female mice. Cancer Cell 41, 1450–1465.e8 (2023).
Jin, D. et al. Deoxycholic acid induces gastric intestinal metaplasia by activating STAT3 signaling and disturbing gastric bile acids metabolism and microbiota. Gut Microbes 14, 2120744 (2022).
Zhang, W. et al. Microbial metabolite trimethylamine-N-oxide induces intestinal carcinogenesis through inhibiting farnesoid X receptor signaling. Cell. Oncol. https://doi.org/10.1007/s13402-024-00920-2 (2024).
Liu, Y. et al. Secondary bile acids and tumorigenesis in colorectal cancer. Front. Oncol. 12, 813745 (2022).
Modica, S., Murzilli, S., Salvatore, L., Schmidt, D. R. & Moschetta, A. Nuclear bile acid receptor FXR protects against intestinal tumorigenesis. Cancer Res. 68, 9589–9594 (2008).
Satoh, K. et al. Global metabolic reprogramming of colorectal cancer occurs at adenoma stage and is induced by MYC. Proc. Natl Acad. Sci. USA 114, E7697–E7706 (2017).
Brown, R. E., Short, S. P. & Williams, C. S. Colorectal cancer and metabolism. Curr. Colorectal Cancer Rep. 14, 226–241 (2018).
Bell, H. N. et al. Reuterin in the healthy gut microbiome suppresses colorectal cancer growth through altering redox balance. Cancer Cell 40, 185–200.e6 (2022).
Vellinga, T. T. et al. SIRT1/PGC1α-dependent increase in oxidative phosphorylation supports chemotherapy resistance of colon cancer. Clin. Cancer Res. 21, 2870–2879 (2015).
Denise, C. et al. 5-Fluorouracil resistant colon cancer cells are addicted to OXPHOS to survive and enhance stem-like traits. Oncotarget 6, 41706–41721 (2015).
Liang, Y. et al. Dichloroacetate restores colorectal cancer chemosensitivity through the p53/miR-149-3p/PDK2-mediated glucose metabolic pathway. Oncogene 39, 469–485 (2020).
Ghosh, S. et al. Microbial metabolite restricts 5-fluorouracil-resistant colonic tumor progression by sensitizing drug transporters via regulation of FOXO3-FOXM1 axis. Theranostics 12, 5574–5595 (2022).
Petri, W. A. Jr. et al. Enteric infections, diarrhea, and their impact on function and development. J. Clin. Invest. 118, 1277–1290 (2008).
Sassone-Corsi, M. & Raffatellu, M. No vacancy: how beneficial microbes cooperate with immunity to provide colonization resistance to pathogens. J. Immunol. 194, 4081–4087 (2015).
Bosnjak, M. et al. Multi-omics analysis of hospital-acquired diarrhoeal patients reveals biomarkers of enterococcal proliferation and Clostridioides difficile infection. Nat. Commun. 14, 7737 (2023).
Rogers, A. W. L. et al. Salmonella re-engineers the intestinal environment to break colonization resistance in the presence of a compositionally intact microbiota. Cell Host Microbe 32, 1774–1786.e9 (2024).
Tovaglieri, A. et al. Species-specific enhancement of enterohemorrhagic E. coli pathogenesis mediated by microbiome metabolites. Microbiome 7, 43 (2019).
Hansen, Z. A. et al. Shifts in the functional capacity and metabolite composition of the gut microbiome during recovery from enteric infection. Front. Cell Infect. Microbiol. 14, 1359576 (2024).
Schulthess, J. et al. The short chain fatty acid butyrate imprints an antimicrobial program in macrophages. Immunity 50, 432–445.e7 (2019).
Stacy, A. et al. Infection trains the host for microbiota-enhanced resistance to pathogens. Cell 184, 615–627.e7 (2021).
Buffie, C. G. et al. Precision microbiome reconstitution restores bile acid mediated resistance to Clostridium difficile. Nature 517, 205–208 (2015).
Fachi, J. L. et al. Butyrate protects mice from Clostridium difficile-induced colitis through an HIF-1-dependent mechanism. Cell Rep. 27, 750–761.e7 (2019).
Jacobson, A. et al. A gut commensal-produced metabolite mediates colonization resistance to Salmonella infection. Cell Host Microbe 24, 296–307.e7 (2018).
Scott, S. A., Fu, J. & Chang, P. V. Dopamine receptor D2 confers colonization resistance via microbial metabolites. Nature 628, 180–185 (2024).
Zelante, T. et al. Tryptophan catabolites from microbiota engage aryl hydrocarbon receptor and balance mucosal reactivity via interleukin-22. Immunity 39, 372–385 (2013).
Goldberg, M. R. et al. Microbial signature in IgE-mediated food allergies. Genome Med. 12, 92 (2020).
Leonard, M. et al. Microbiome signatures of progression toward celiac disease onset in at-risk children in a longitudinal prospective cohort study. Proc. Natl Acad. Sci. USA 118, e2020322118 (2021).
Bao, R. et al. Fecal microbiome and metabolome differ in healthy and food-allergic twins. J. Clin. Invest. 131, e141935 (2021).
Tan, J. et al. Dietary fiber and bacterial scfa enhance oral tolerance and protect against food allergy through diverse cellular pathways. Cell Rep. 15, 2809–2824 (2016).
Wang, R. et al. Treatment of peanut allergy and colitis in mice via the intestinal release of butyrate from polymeric micelles. Nat. Biomed. Eng. 7, 38–55 (2023).
Paparo, L. et al. Butyrate as a bioactive human milk protective component against food allergy. Allergy 76, 1398–1415 (2021).
Lamas, B. et al. Aryl hydrocarbon receptor ligand production by the gut microbiota is decreased in celiac disease leading to intestinal inflammation. Sci. Transl. Med. 12, eaba0624 (2020).
Stephen-Victor, E. et al. RELMβ sets the threshold for microbiome-dependent oral tolerance. Nature 638, 760–768 (2025).
Hufnagl, K. & Jensen-Jarolim, E. Does a carrot a day keep the allergy away? Immunol. Lett. 206, 54–58 (2019).
DePaolo, R. W. et al. Co-adjuvant effects of retinoic acid and IL-15 induce inflammatory immunity to dietary antigens. Nature 471, 220–224 (2011).
Chew, N. W. S. et al. The global burden of metabolic disease: data from 2000 to 2019. Cell Metab. 35, 414–428.e3 (2023).
Peng, J., Lu, M., Wang, P., Peng, Y. & Tang, X. The global burden of metabolic disease in children and adolescents: data from the Global Burden of Disease 2000-2019. Metabolism 148, 155691 (2023).
Cirulli, E. T. et al. Profound perturbation of the metabolome in obesity is associated with health risk. Cell Metab. 29, 488–500.e2 (2019).
Asnicar, F. et al. Microbiome connections with host metabolism and habitual diet from 1,098 deeply phenotyped individuals. Nat. Med. 27, 321–332 (2021).
Chanda, D. & De, D. Meta-analysis reveals obesity associated gut microbial alteration patterns and reproducible contributors of functional shift. Gut Microbes 16, 2304900 (2024).
Thingholm, L. B. et al. Obese individuals with and without type 2 diabetes show different gut microbial functional capacity and composition. Cell Host Microbe 26, 252–264.e10 (2019).
Canfora, E. E., Meex, R. C. R., Venema, K. & Blaak, E. E. Gut microbial metabolites in obesity, NAFLD and T2DM. Nat. Rev. Endocrinol. 15, 261–273 (2019).
Miyamoto, J. et al. Gut microbiota confers host resistance to obesity by metabolizing dietary polyunsaturated fatty acids. Nat. Commun. 10, 4007 (2019).
Li, V. L. et al. An exercise-inducible metabolite that suppresses feeding and obesity. Nature 606, 785–790 (2022).
Moya-Garzon, M. D. et al. A β-hydroxybutyrate shunt pathway generates anti-obesity ketone metabolites. Cell 188, 175–186.e20 (2025).
Xiao, S. et al. Lac-Phe mediates the effects of metformin on food intake and body weight. Nat. Metab. 6, 659–669 (2024).
De Vadder, F. et al. Microbiota-produced succinate improves glucose homeostasis via intestinal gluconeogenesis. Cell Metab. 24, 151–157 (2016).
Shelton, C. D. et al. An early-life microbiota metabolite protects against obesity by regulating intestinal lipid metabolism. Cell Host Microbe 31, 1604–1619.e10 (2023).
Virtue, A. T. et al. The gut microbiota regulates white adipose tissue inflammation and obesity via a family of microRNAs. Sci. Transl. Med. 11, eaav1892 (2019).
Du, W. et al. The microbiota-dependent tryptophan metabolite alleviates high-fat diet-induced insulin resistance through the hepatic AhR/TSC2/mTORC1 axis. Proc. Natl Acad. Sci. USA 121, e2400385121 (2024).
Li, X. et al. Ketogenic diet-induced bile acids protect against obesity through reduced calorie absorption. Nat. Metab. 6, 1397–1414 (2024).
Chen, Y. et al. A small intestinal bile acid modulates the gut microbiome to improve host metabolic phenotypes following bariatric surgery. Cell Host Microbe 32, 1315–1330.e5 (2024).
Jahansouz, C. et al. Bile acids increase independently from hypocaloric restriction after bariatric surgery. Ann. Surg. 264, 1022–1028 (2016).
Patti, M. E. et al. Serum bile acids are higher in humans with prior gastric bypass: potential contribution to improved glucose and lipid metabolism. Obesity 17, 1671–1677 (2009).
Sun, L. et al. Gut microbiota and intestinal FXR mediate the clinical benefits of metformin. Nat. Med. 24, 1919–1929 (2018).
Takeuchi, T. et al. Fatty acid overproduction by gut commensal microbiota exacerbates obesity. Cell Metab. 35, 361–375.e9 (2023).
Kong, L. et al. Trimethylamine N-oxide impairs β-cell function and glucose tolerance. Nat. Commun. 15, 2526 (2024).
Schugar, R. C. et al. The TMAO-producing enzyme flavin-containing monooxygenase 3 regulates obesity and the beiging of white adipose tissue. Cell Rep. 19, 2451–2461 (2017).
Tang, W. H. et al. Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. N. Engl. J. Med. 368, 1575–1584 (2013).
Chen, S. et al. Trimethylamine N-oxide binds and activates PERK to promote metabolic dysfunction. Cell Metab. 30, 1141–1151.e5 (2019).
Liu, G. W., Su, R., Osterling, B. L. G., Carrazco, R. & Feig, V. R. Toward next-generation ingestible hydrogels. Biomacromolecules https://doi.org/10.1021/acs.biomac.4c00902 (2025).
Wang, Z. et al. Non-lethal inhibition of gut microbial trimethylamine production for the treatment of atherosclerosis. Cell 163, 1585–1595 (2015).
Monteleone, I. et al. Aryl hydrocarbon receptor-induced signals up-regulate IL-22 production and inhibit inflammation in the gastrointestinal tract. Gastroenterology 141, 237–248 (2011).
Litvak, Y., Byndloss, M. X. & Baumler, A. J. Colonocyte metabolism shapes the gut microbiota. Science 362, eaat9076 (2018).
Etchegaray, J. P. & Mostoslavsky, R. Interplay between metabolism and epigenetics: a nuclear adaptation to environmental changes. Mol. Cell 62, 695–711 (2016).
Berger, S. L. & Sassone-Corsi, P. Metabolic signaling to chromatin. Cold Spring Harb. Perspect. Biol. 8, a019463 (2016).
Dai, Z., Ramesh, V. & Locasale, J. W. The evolving metabolic landscape of chromatin biology and epigenetics. Nat. Rev. Genet. 21, 737–753 (2020).
Shalon, D. et al. Profiling the human intestinal environment under physiological conditions. Nature 617, 581–591 (2023).





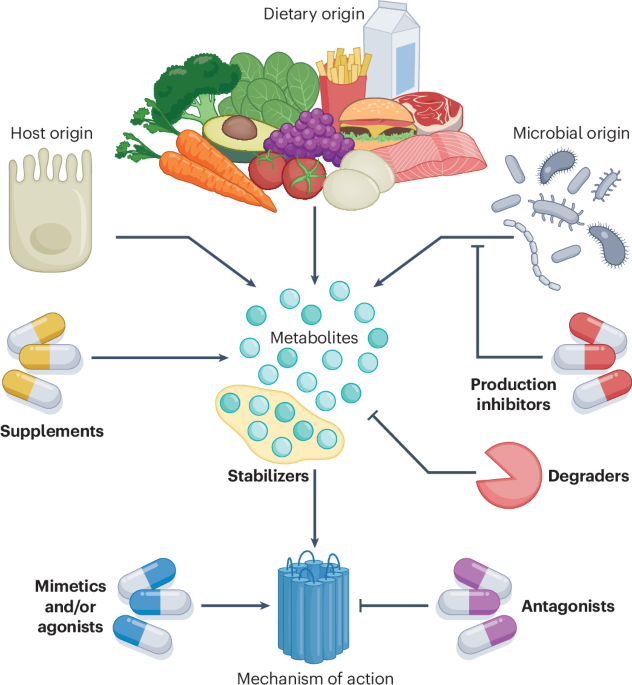












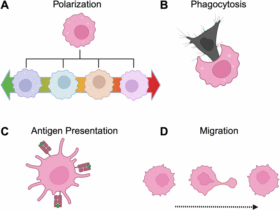
Leave a Reply