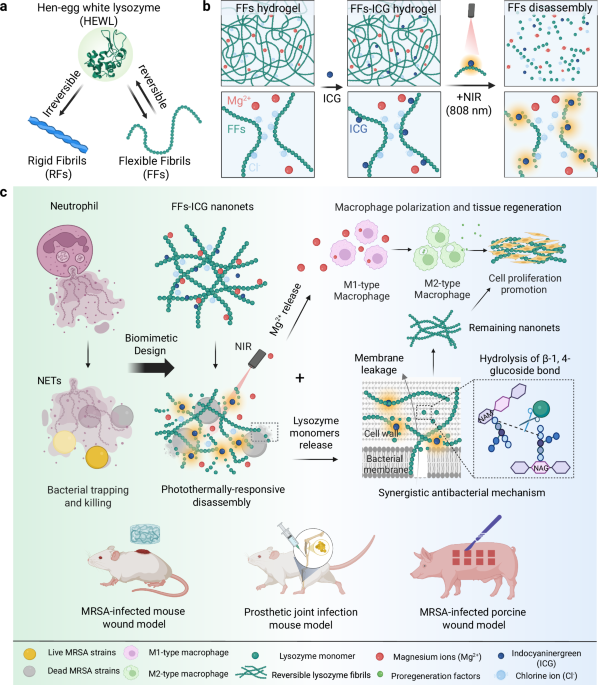
Photothermal reversibility characterization of lysozyme flexible fibrils (FFs) nanoNETs
Atomic force microscopy (AFM) images (Fig. 2a and Supplementary Fig. 1) show the morphologies of lysozyme FFs and RFs, in the absence and the presence of ICG. FFs exhibit a short and curly shape whereas RFs feature a long and rigid morphology, with diameters of 2.5 and 7.8 nm, respectively. Notably, ICG modification induced no significant morphological differences (Fig. 2b). However, the persistence length (Fig. 2c), representing fibril rigidity, showed a notable increase in both fibrils upon ICG incorporation: FFs double from 60 to 120 nm while RFs increase from 6 to 10 µm. These findings indicate the successful incorporation of ICG molecules into both FFs and RFs33, enhancing fibril stiffness with minimal changes in fibril diameter. Secondary structural investigation via circular dichroism (CD) and Thioflavin T (ThT) fluorescence assay (Supplementary Fig. 2) showed that FFs are rich in both α-helical and β-sheet contents, whereas RFs were predominantly composed of β-sheet structures. These results confirm that FFs are composed of partially unfolded lysozyme with preserved functional domains necessary to enzymatic activity, whereas RFs contain a robust β-sheet-rich amyloid core31.

a AFM images of lysozyme FFs and RFs in the absence and the presence of ICG (scale bar: 500 nm). Statistical analysis on the height (b) of FFs and RFs based on AFM investigation. A large number of fibrils (n = 449) were included in the height analysis. The data were expressed as mean ± standard deviation (S.D). Statistical significance was analyzed by one-way ANOVA using GraphPad Prism 8, followed by Tukey’s post-hoc test for pairwise comparisons. Statistical significance was defined as *p < 0.05, **p < 0.01, and ***p < 0.001. FFs vs. FFs-ICG (p < 0.0001), RFs vs. RFs-ICG (p < 0.0001). Fibril rigidity was studied by measuring the mean square end-to-end distance (MSE2ED) of their polymeric chain47. The morphological data of these fibrils were collectively fitted to obtain MSE2ED value (c) of FFs/FFs-ICG and RFs/RFs-ICG, with the error bar representing fitting error using the 2D worm-like chain model48. FFs vs. FFs-ICG (p < 0.0001), RFs vs. RFs-ICG (p < 0.0001). d Photo of FFs hydrogel and RFs hydrogel. Representative photothermal imaging (e) and the corresponding photothermal curve (f) of FFs, ICG, and FFs-ICG after 808 nm NIR irradiation. g, Comparison of ICG and FFs-ICG in the photothermal conversion stability. h AFM images of FFs and RFs (scale bar: 800 nm) under the heating and cooling treatments. i CD analysis of the secondary structural transition of FFs and RFs before and after heating at 60 oC. j Temperature-dependent rheological changes of FFs and RFs hydrogel (1.0 wt.% concentration) ranging from 20 to 60 oC. Source data are provided as a Source Data file.
Subsequently, hydrogels composed of either FFs or RFs were fabricated by introducing magnesium chloride (Fig. 2d), which screen electrostatic charges by reducing the Debye length34. These hydrogels show tunable mechanical strength, proportional to lysozyme and MgCl2 concentrations (Supplementary Figs. 3-4). Both ICG-incorporated RFs (RFs-ICG) and FFs (FFs-ICG) exhibited strong absorbance in the NIR region (700–850 nm), distinct from free ICG (Supplementary Fig. 5), possibly due to ICG binding to the amyloid β-sheets. Consequently, FFs-ICG hydrogels demonstrated excellent photothermal conversion efficiency, dependent on both NIR intensity and ICG concentration (Fig. 2e-f and Supplementary Fig. 6). Additionally, FFs-ICG hydrogels demonstrated superior stability and cycling performance compared to pristine ICG molecule (Fig. 2g and Supplementary Fig. 7), which is attributed to the homogeneous dispersion of ICG within FF-hydrogel, preventing ICG aggregation and subsequent photobleaching upon NIR irradiation35.
We further explored the morphological and structural transformations of FFs-ICG hydrogels induced by photothermal treatments. After 10 min of NIR irradiation, FFs-ICG hydrogels exhibit a distinct gel-solution transition (Supplementary Fig. 8) and FFs disassemble into smaller aggregates or monomers (Fig. 2h). Notably, these disassembled building blocks reassemble into FFs after 2 h, resulting in a solution-to-gel reverse transition. In contrast, both RFs and its hydrogels showed no significant changes during the heating-cooling process. These results highlight the thermal- and photothermal-responsive reversible behavior of FFs-based hydrogels. Moreover, CD spectroscopy (Fig. 2i) revealed a decrease in the negative peak at 218 nm and an increase at 198 nm of FFs upon heating, indicating a structural transition from β-sheet to random coil31, whereas RFs exhibited no significant changes. Rheological study confirmed that RFs exhibited temperature-dependent decrease of mechanical strength, evidenced by the gradual decrease in both storage modulus (G’) and loss modulus (G”) with cyclic heating (Fig. 2j and Supplementary Fig. 9), due to gel melting36. On the other hand, FFs hydrogel showed a significantly more pronounced reduction of mechanical strength upon heating (Fig. 2j and Supplementary Fig. 10), suggesting a combined effect of gel softening and FF disassembly. This scenario is supported by the ThT-stained confocal imaging, as shown in Supplementary Fig. 11, showing a significant decrease in fluorescence intensity of FFs after photothermal treatments, whereas RFs remained stable. Notably, both RFs and FFs hydrogel retain the gel state, maintaining G’ > G” even at 60 oC, suggesting partial preservation of the architecture in their hydrogel networks.
In vitro characterization for the bioactivity of flexible fibrils (FFs) nanoNETs
Lysozyme possesses intrinsic antibacterial activity. However, its applications are limited due to the tendency to aggregation and potential cytotoxicity at high concentrations29,37. We hypothesized that the formation of FFs could mitigate cytotoxicity, while preserving intrinsic antibacterial activity. To confirm this, we first applied L-929 fibroblast in the assessment of FFs cytocompatibility (Fig. 3a). Results indicate that lysozyme monomers (1 wt.%) reduced cell viability to 50%, whereas both RFs and FFs maintain cell survival to 90%. This suggests that the linear alignment of lysozyme significantly mitigates the cytotoxic effect of native lysozyme. Live/dead-staining confocal imaging results verified the cytocompatibility of the FFs hydrogel (Supplementary Fig. 12). The concentration-dependent cytotoxicity in L-929 cells was also evaluated (Supplementary Fig. 13), demonstrating an excellent biocompatibility of the FFs hydrogel, even at high-concentration exceeding 1 wt.%. Similar results were observed in the cytotoxicity assays using human umbilical vein endothelial cells (HUVECs) (Supplementary Fig. 14). In addition, both FFs and RFs displayed a high level of hemocompatibility, maintaining the hemolysis ratio ranging from 0.1 to 1 wt.% concentration (Supplementary Fig. 15).

a Cytocompatibility assays (n = 6) of lysozyme monomers, FFs, and RFs using methyl thiazolyl tetrazolium (MTT) assay. b Quantification (n = 3) of released monomers from lysozyme FFs and RFs at 20 oC and 60 oC. RFs 20 oC vs. RFs 60oC (p = 0.3398), FFs 20 oC vs. FFs 60oC (p = 0.0196). Lysozyme activity assays (n = 3) of FFs-ICG at different concentrations after NIR irradiation (c) and RFs/FFs before and after NIR irradiation (d). RFs vs. FFs (p = 0.0583), RFs-ICG vs. FFs-ICG (p = 0.0031). e Schematic illustration for RFs/FFs bioactivity and thermally-induced disassembly. f Antibacterial efficiency assays (n = 3) of Lys, RFs, FFs, RFs-ICG and FFs-ICG against MRSA using CFU counting methods. Lys (p = 0.0001), RFs (p = 0.0013), FFs (p < 0.0001), ICG (p < 0.0001), RFs-ICG (p = 0.0004), FFs-ICG (p < 0.0001) vs. PBS control. SEM images (scale bar: 1 μm) (g) and live/dead bacteria staining confocal images (scale bar: 100 μm) (h) of MRSA strains after different treatments. i Inductively-coupled plasma mass spectrometry (ICP-MS) assays (n = 3) of released Mg2+ concentration before and after heating treatments. RFs 20 oC vs. FFs 20 oC (p = 0.0056), RFs 60 oC vs. FFs 60 oC (p = 0.0017). Flow cytometry analysis (n = 3) (j, k) and confocal microscopic images (scale bar: 50 μm) (l) of Raw 264.7 cells cultured with RFs, FFs, RFs-ICG, and FFs-ICG, with PBS and IL-4 as the negative and positive control, respectively. IL-4 (p = 0.0149), RFs (p = 0.0475), FFs (p = 0.1702), RFs-ICG (p = 0.0036), FFs-ICG (p = 0.0063) vs. PBS control. The data were expressed as mean ± standard deviation (S.D). Statistical significance was analyzed by one-way ANOVA using GraphPad Prism 8, followed by Tukey’s post hoc test for pairwise comparisons. Statistical significance was defined as *p < 0.05, **p < 0.01, and ***p < 0.001. Source data are provided as a Source Data file.
Unlike traditional protein nanofibrils that decrease their cytotoxicity by compromising biological activity38, the FFs effectively align partially-unfolded active lysozyme, avoiding high cytotoxicity while retaining biological activity. Notably, this enables the controllable release of active lysozyme monomers upon photothermal-stimulation. This release was quantified by measuring lysozyme monomer concentration in the RFs and FFs solution before and after photothermal treatments (Supplementary Fig. 16). Consistent with the observation above (Fig. 2h), RFs released negligible amounts of monomers after 10 min at 60oC, whereas FFs exhibited a fivefold increase in lysozyme monomer concentration, compared to their release at 20 oC (Fig. 3b). The released monomers retained enzymatic bioactivity, linearly correlated with the initial FFs concentrations (Fig. 3c). Upon NIR irradiation, FFs-ICG exhibited a significant increase in enzymatic activity, whereas RFs, FFs, and RFs-ICG remained with low enzymatic functionalities (Fig. 3d). These results confirm that the nanoconfined bioactive lysozyme in the FFs can be released upon photothermal treatment (Fig. 3e), while irreversible RFs remain intact upon heat treatment due to their stable misfolded β-sheet-rich structure derived from the fully-unfolding state.
To demonstrate the pathogen-trapping and killing capacity of our nanoNETs, we utilized MRSA and Pseudomonas aeruginosa (P. aeruginosa, a highly motile Gram-negative bacteria known for its swimming and swarming motility) as model strains. As shown in Supplementary Fig. 17, our nanoNETs can achieve the effective physical entrapment of both MRSA and P. aeruginosa within the fibrous network, visually confirmed by scanning electron microscope (SEM) images, which offer direct morphological evidence of bacterial capture, regardless of bacterial motility. We have also experimentally confirmed that both strains (MRSA and P. aeruginosa) exhibit negative surface charges, as measured by zeta potential in Supplementary Fig. 18. This characteristic enables them to be effectively captured through electrostatic adsorption by the positively charged nanoNETs29,38. We found that the lysozyme monomers exhibited a high antimicrobial efficiency (89.2 ± 2.5%), outperforming RFs (62.1 ± 5.5%) and FFs (71.9 ± 0.7%) (Fig. 3f and Supplementary Fig. 19). RFs-ICG achieved a higher antibacterial rate (81.5% ± 3.8%) partially increased by ICG, whereas FFs-ICG demonstrated the highest antibacterial performance (95.4% ± 0.6%) within low-power NIR irradiation (0.3 W/cm2). Notably, this low photothermal power allows to accurately assess the differences in antibacterial activity between RFs and FFs. These results are supported by the live/dead bacterial staining images (Fig. 3g). Further, SEM imaging (Fig. 3h) shows the MRSA membrane disruption, and the leakage of N-acetyl-beta-D-glucosidase (NAG) and K+ after treatments (Supplementary Fig. 20-21), indicating the increased bacterial membrane permeability as the antibacterial activity. Additionally, FFs-ICG nanoNETs exhibited an antimicrobial efficacy of 86.2 ± 4.3% for P. aeruginosa, and 89.8 ± 4.7% for Candida albicans (a typical fungal strain) (Supplementary Fig. 22), demonstrating the broad-spectrum antibacterial efficacy of nanoNETs beyond Gram-positive bacteria. These results indicate our FFs possess a synergistic effect against bacteria: NIR-induced FFs partially disassemble and release active lysozyme for bacteria degradation, while the residual FFs-ICG nanoNETs maintain the function of pathogen entrapment through the absorption-contact mechanism as conventional RFs39,40, collectively providing direct evidence to support the “trap-and-kill” mechanism of the nanoNETs.
In addition to lysozyme release, we also quantified the Mg2+ ion release from the hydrogel. Following NIR irradiation, Mg2+ concentrations in RFs and FFs solutions increased approximately six- and eight-fold, respectively, compared to pre-irradiation levels (Fig. 3i). The release of magnesium chloride indicates the partial dissociation of hydrogel network and the reduction of mechanical strength. On the other hand, magnesium ions are known to promote macrophage polarization from M1 to anti-inflammatory M2 phenotype and facilitate tissue regeneration41. To confirm this, we used Raw 264.7 cells as the macrophage model to assess the immunomodulatory capacity of our nanoNETs, with IL-4 as the positive control for M2 polarization. We observe a clear correlation between Mg2+ release and M2-type polarization (Fig. 3j–l), with the highest M2 polarization ratio (36.7 ± 1.3%) in FFs-ICG group due to the photothermal-responsive characteristics. Dysregulated or excessive native NETs release can lead to tissue damage and chronic inflammation, whereas our nanoNETs can avoid these detrimental effects and harnessing the beneficial antimicrobial mechanism. The data above provide definitive evidence for the role of released Mg2+ from nanoNETs in macrophage polarization to prevent the occurrence of these adverse outcomes. Additionally, we collected supernatant from macrophages treated with FFs-ICG, which was then added into the medium and co-incubated with L-929 cells. The result revealed that this supernatant significantly enhanced L-929 cells proliferation by 39.91% compared to the control group (Supplementary Fig. 23), suggesting the presence of reparative cytokines in the supernatant and thus highlighting the functional role of M2-polarized macrophages in promoting cellular growth and tissue regeneration. Collectively, FFs-ICG nanoNETs provides a multifunctional platform for anti-infection therapy: acting as the “host defense network” to trap bacteria, releasing bioactive lysozyme upon irradiation for antimicrobial effects, and enabling Mg2+ release for promoting tissue repair via macrophage polarization regulation.
In vivo evaluation of FFs-ICG nanoNETs in an MRSA-infected murine wound model
To evaluate the in vivo therapeutic efficiency of FFs-ICG hydrogel, we established a murine model of MRSA-infected wound healing (Fig. 4a). MRSA-infected wounds were treated with PBS, lysozyme, RFs, FFs, ICG, RFs-ICG, and FFs-ICG. Quantitative analysis revealed that NIR-irradiated FFs-ICG treatment achieved a nearly complete wound closure (approximately 100%) by day 14, significantly outperforming the PBS group (71.5% ± 8.2%) (Fig. 4b, c and Supplementary Fig. 24a). Correspondingly, bacterial enumeration demonstrated a > 95% reduction in MRSA viability in the FFs-ICG group (Fig. 4d). By contrast, RFs-ICG exhibited both relatively lower healing and antibacterial efficacy than the FFs-ICG, while lysozyme monomers demonstrated strong antibacterial activity (Fig. 4d) but impaired wound healing (Fig. 4b and Supplementary Fig. 24b), likely due to their cytotoxic effects.

a Timeline of the treatment and evaluation process of FFs-ICG hydrogel in an MRSA-infected wound healing mice model. Created in BioRender. Xuan, Q. (2025) https://BioRender.com/e9mjxve. Wound area quantification (n = 5) (b) and the corresponding representative macroscopic wound images (Scale bar: 1 cm) (c) of MRSA-infected mice after various treatments. d Quantification of MRSA survival rate (n = 3; data are presented as individual points) in the infected skin tissues after distinct treatments using CFU counting method. Lys (p = 0.0001), RFs (p = 0.0331), FFs (p = 0.0025), ICG (p < 0.0001), RFs-ICG (p < 0.0001), FFs-ICG (p < 0.0001) vs. PBS control. Representative H&E staining (e), Ki-67-staining (f), and Giemsa-staining (g), iNOS and CD206-staining (h) images of infected tissues from various treatment groups on day 14 for the analysis of dermis and epidermis regeneration, cell proliferation, survival bacteria, and macrophage polarization, respectively. In Fig. 4e, g represents granulation tissue, and NE represents newly formed epidermis, and ND represents newly formed dermis. In Fig. 4g, the red arrows represent the residual bacteria. the fluorescence intensity quantification (i) (n = 3; data are presented as individual points) and the corresponding representative ThT-staining images (j) of wounds for the analysis of in vivo thermal-induced disassembling behaviors of FFs-ICG hydrogel. Scale bars: (e) 2 mm (left), 500 μm (right); (f–h) 500 μm; (h) 100 μm; (j) 200 μm. The data were expressed as mean ± standard deviation (S.D). Statistical significance was analyzed by one-way ANOVA using GraphPad Prism 8, followed by Tukey’s post-hoc test for pairwise comparisons. Statistical significance was defined as *p < 0.05, **p < 0.01, and ***p < 0.001. Source data are provided as a Source Data file.
Histological analysis of Hematoxylin & eosin (H&E) stained sections (Fig. 4e and Supplementary Fig. 25a) further demonstrated accelerated regeneration in the FFs-ICG group compared to other groups, characterized by complete dermis and epidermis reconstruction and nascent hair follicle formation. Immunohistochemical detection of Ki-67 and Giemsa staining confirmed enhanced cellular proliferation and antimicrobial effect in the FFs-ICG treated tissues (Fig. 4f–g and Supplementary Fig. 25b–d). Notably, the substantial Mg2+ release induced by FFs-ICG under NIR exposure significantly promoted the macrophage polarization to M2 phenotype, particularly in the dermal layer, which contributed to sustained tissue regeneration (Fig. 4h). In contrast, PBS-treated wounds exhibited only granulation tissues formation, while FFs and RFs groups showed partial dermal regeneration (Fig. 4e and Supplementary Fig. 25). These groups also demonstrated inferior performances in cell proliferation, antibacterial efficacy, and immunomodulation (Supplementary Fig. 25), consistent with in-vitro results. We further performed quantitative PCR validation on wound tissues and observed significant upregulation in the mRNA expression of the genes related to key reparative cytokines and growth factors in the FFs-ICG group, including Vegfa, Hif1α, Il10, and Pdgfb, compared to the PBS control (Supplementary Fig. 26). This provides direct molecular evidence that FFs-ICG actively promotes a pro-regenerative transcriptional program within the wound microenvironment, perfectly aligning with the observed functional outcomes of accelerated healing. Furthermore, the therapeutic benefit of our FFs-ICG nanoNETs extends to the established infections (3 days) with mature biofilms (Supplementary Fig. 27). This significantly enhances the clinical relevance of our platform, as it demonstrates efficacy not just as a prophylactic or early intervention, but as a potential treatment for ongoing, resistant infections. Collectively, these results establish that FFs-ICG effectively eradicates MRSA infections while simultaneously promoting cell proliferative and tissue regenerative processes.
A comprehensive biosafety evaluation, including histological assessment of major organs and blood biochemical analysis, confirmed the excellent in vivo biocompatibility of all investigated biomaterials (Supplementary Fig. 28). Also, the hematological analysis demonstrated that the treatments of these prepared biomaterials, including our FFs-ICG nanoNETs, did not elicit significant systemic inflammation or disrupt leukocyte homeostasis, supporting their favorable safety profiles (Supplementary Fig. 29). More importantly, our FFs-ICG nanoNETs also exhibited a low risk of inducing thrombosis (including platelet activation and thrombus formation), supporting their safety for proposed topical applications (Supplementary Fig. 30). The lack of thrombogenicity is likely due to the biocompatible nature of lysozyme and the flexible, non-rigid structure of the FFs, which is less prone to mechanically triggering coagulation cascades compared to stiff fibrous materials. To further verify photothermal-triggered FFs disassembly in vivo, we performed the analysis of ThT-stained wound tissue sections from FFs-ICG and RFs-ICG groups on day 1 (Fig. 4i–j). Both groups exhibited high fluorescence signal without NIR irradiation. However, following irradiation, FFs-ICG-treated tissues showed significantly reduced ThT fluorescence, suggesting in vivo FFs disassembly. Partial post-irradiation reassembly was evidenced from the slight fluorescence recovery. In contrast, RFs-ICG maintained constant strong fluorescence, consistent with in vitro observations. These observations underline the dual functional mechanism of FFs-ICG hydrogel: (i) Positively charged FFs initially mimic NETs to entrap MRSA while NIR-triggered monomer release enables bacterial eradication; and (ii) Subsequent FFs reassembly templates an ordered scaffold that promotes cell adhesion and proliferation37. Additionally, our results facilitate to distinguish the contribution of photothermal effects and the release of bioactive lysozyme/Mg2+. The photothermal effect (from ICG) provides a strong, non-specific antibacterial action, while the FFs provides a passive “trap” function and a biocompatible scaffold. The synergistic effect was achieved in FFs-ICG + NIR, and the photothermal effect triggers the on-demand release of the specific biological “kill” agent (lysozyme) and the pro-healing agent (Mg2+), while the fibrous network provides a protective scaffold that mitigates the potential collateral damage of pure PTT and supports cell growth. It is the photothermal (ICG)-controlled dynamic functionality of the nanoNETs, the ability to convert from a passive trap to an active antimicrobial and regenerative factory, that delivers the optimal therapeutic outcome. This hypothesis was further verified by the results from a sterile (non-infected) murine full-thickness wound healing model (Supplementary Fig. 31), which confirmed that FFs-ICG nanoNETs platform possessed intrinsic pro-regenerative properties that operate independently of its antimicrobial function.
In vivo evaluation of FFs-ICG nanoNETs in an MRSA-infected periprosthetic joint infection murine model
Based on the potent antibacterial and tissue-regenerative capabilities of the FFs-ICG hydrogel, we further investigated its therapeutic potential against endogenous infections using a periprosthetic joint infection model in mice (Fig. 5a). Mice were administered a total dose of 500 mg/kg nanoNETs formulations on day 1. As a distinct feature of the infected wound model, the formation of a biofilm emerges as the predominant survival strategy for MRSA in periprosthetic joint infection (PJI) mice model, rather than the existence of free-floating bacterial cells within the bone tissues42. Therefore, the implants with biofilm were extracted and analyzed using SEM, and bacterial burden on the implant and the surrounding bone tissues was quantified by CFU counting on day 7 to evaluate antibacterial efficacy. SEM images (Fig. 5b and Supplementary Fig. 32a) revealed near-complete biofilm disruption on the implants in the FFs-ICG group, with only deformed bacteria remnants visible. Correspondingly, CFU analysis confirmed that FFs-ICG achieved a significant bacterial clearance efficacy of 99.7 ± 0.2% for peri-implant bacteria and 94.6 ± 1.1% antimicrobial efficiency for MRSA-infected tibias (Fig. 5c, d). In addition, the diameter of the infected knee greatly decreased in PJI mice after FFs-ICG treatment (Fig. 5e), indicating reduced knee swelling and alleviation of infection-induced inflammation. These findings demonstrate the effective and broad-spectrum antibacterial activity of our FFs-ICG nanoNETs, independent of the survival mode (free-floating or biofilm) and the pathogen source (endogenous or exogenous).

a Schematic timeline to assess the therapeutic efficiency of FFs-ICG hydrogel in the MRSA-infected PJI model. Created in BioRender. Xuan, Q. (2025) https://BioRender.com/e9mjxve. SEM images of MRSA biofilm (b) on the surface of knee prosthesis at day 14. (Scale bars: 1 μm) CFU quantification (n = 3) of the joint peri-implant tissues (c) and bone (d) at day 14 in the PJI model. Lys (p = 0.2910), RFs (p = 0.0102), FFs (p = 0.0022), ICG (p = 0.0057), RFs-ICG (p = 0.0047), FFs-ICG (p = 0.0007) vs. PBS control in (c). Lys (p = 0.0109), RFs (p = 0.0055), FFs (p = 0.0024), ICG (p = 0.0028), RFs-ICG (p = 0.0015), FFs-ICG (p = 0.0004) vs. PBS control in (d). e Diameter of infected knee joints in PJI mice at days 0, 3, 5, 7, and 14 (n = 3 for each time point). f Axial, coronal, sagittal, and 3D reconstruction micro-CT images 4 weeks after MRSA infection. The mouse tibias are shown in dark yellow, and the implant is shown in light yellow. Quantitative micro-CT analysis including the bone volume/total volume (BV/TV) (g), and trabecular thickness (TB.TH) (h) (n = 3; data are presented as individual points). Non-infected (p = 0.0017), Lys (p = 0.4128), RFs (p = 0.2015), FFs (p = 0.1380), ICG (p = 0.0546), RFs-ICG (p = 0.0483), FFs-ICG (p = 0.0210) vs. PBS control in (g). Non-infected (p = 0.0047), Lys (p = 0.3736), RFs (p = 0.1981), FFs (p = 0.3106), ICG (p = 0.0090), RFs-ICG (p = 0.0062), FFs-ICG (p < 0.0001) vs. PBS control in (h). i Quantitative analysis of mouse knee-bending angle 4 weeks after infection (n = 4; data are presented as individual points). Non-infected (p = 0.0049), Lys (p = 0.4772), RFs (p = 0.1855), FFs (p = 0.2831), ICG (p = 0.0083), RFs-ICG (p = 0.1757), FFs-ICG (p = 0.0207) vs. PBS control. j–k Gait analysis showing actual mice walking trajectory and footprint imaging. Green for left hind footprints; pink for right hind footprints (j). 3D reconstruction of left hind paw of mice in each group, with color-coded pressure intensity of the mouse’s footprints (k). Quantitative gait analysis and the parameters: support time (l), stride length (m), average intensity (n) (average pressure on the runway), and average speed (o) (n = 4, data are presented as individual points). Non-infected (p = 0.0081), Lys (p = 0.00394), RFs (p = 0.0026), FFs (p = 0.0016), ICG (p = 0.0039), RFs-ICG (p = 0.0068), FFs-ICG (p = 0.0008) vs. PBS control in (l). Non-infected (p = 0.0009), Lys (p = 0.0125), RFs (p = 0.0165), FFs (p = 0.0086), ICG (p = 0.0017), RFs-ICG (p = 0.0029), FFs-ICG (p = 0.0014) vs. PBS control in (m). Non-infected (p = 0.0005), Lys (p = 0.0183), RFs (p = 0.3766), FFs (p = 0.0069), ICG (p = 0.0010), RFs-ICG (p = 0.0018), FFs-ICG (p = 0.0033) vs. PBS control in n. Non-infected (p = 0.0104), Lys (p = 0.0329), RFs (p = 0.1638), FFs (p = 0.0182), ICG (p = 0.0368), RFs-ICG (p = 0.0319), FFs-ICG (p = 0.0323) vs. PBS control in (o). The data were expressed as mean ± standard deviation (S.D). Statistical significance was analyzed by one-way ANOVA using GraphPad Prism 8, followed by Tukey’s post hoc test for pairwise comparisons. Statistical significance was defined as *p < 0.05, **p < 0.01, and ***p < 0.001. Source data are provided as a Source Data file.
Orthopedic implant infections often lead to bone resorption and periprosthetic osteolysis43,44. To investigate whether FFs-ICG nanoNETs could prevent infection-induced peri-implant osteolysis, we performed micro-CT analysis of tibias osteolysis (Fig. 5f and Supplementary Fig. 32b). Compared to the non-infected group, a severe osteolysis was observed around the implant with increased intertrabecular spacing and reduced bone mass in the PBS group, whereas FFs-ICG group exhibited preserved bone mass and maintained normal bone architecture. Quantitative analysis further (Fig. 5g, h) revealed significantly higher trabecular thickness (TB.TH) and peri-implant bone volume fraction (bone volume/total volume, BV/TV) in the ICG, RFs-ICG, and FFs-ICG groups, compared to PBS group. The highest efficacy was observed in FFs-ICG treated group, with values approaching those in non-infected mice. Interestingly, MRSA-infection also reduced joint bending angle (Fig. 5i), indicating impaired mobility, whereas FFs-ICG treatment effectively restored the joint flexibility to the level of non-infected mice, outperforming other treatment groups. Taken together, these results indicate that the FFs-ICG nanoNET not only is effective in implant-associated infections but it also prevents purulent arthritis and protects bone integrity.
Orthopedic implant-associated infections often require a treatment approach that extends beyond bacterial eradication to alleviate pain and preserve joint function. To assess their impact on mobility44, we investigated the effects of FFs-ICG on the walking ability of mice with implant-associated infections through gait analysis (Fig. 5a). Mice in the PBS and RFs-ICG groups displayed irregular and scattered footprints, indicating their impaired mobility (Fig. 5j and Supplementary Fig. 32c). In contrast, mice in the FFs-ICG group demonstrated well-defined footprints similar to those in the non-infected group, indicating their restored walking ability. Three-dimensional footprint reconstruction results (Fig. 5k and Supplementary Fig. 32d) further confirmed a normalized force distribution between soles and toes in the FFs-ICG and non-infected group, while other treated groups (Lys, RFs, FFs, ICG, and RFs-ICG) showed incomplete foot engagement. Furthermore, significant improvements in support time, average intensity, and average speed (Fig. 5i–o) were observed in the FFs-ICG group, compared to the PBS group. These results highlight the efficacy of FFs-ICG in mitigating periprosthetic osteolysis and restoring joint functional mobility.
In vivo evaluation of FFs-ICG nanoNETs in an MRSA-infected porcine wound healing model
To further examine the translational potential of FFs-ICG nanoNETs, we employed a porcine wound healing model, which closely recapitulates human skin architecture and re-epithelialization dynamics during tissue repair. Porcine skin shares key structural and functional similarities with human skin, including analogous dermal and epidermis layers, subcutaneous adipose tissue, sparse hair follicles distribution, and the presence of rete ridges45,46. As shown in Fig. 6a, eighteen square dermal wounds (1.5 cm × 1.5 cm × 0.6 cm) were bilaterally created on the porcine dorsum. Following a 4 h MRSA inoculation period (except for the non-infected group), wounds were randomly assigned to various treatment groups. To minimize locational healing bias, the assignment was arranged in a randomized and crosswise manner. After treatments, the wounds were protected with sterile cotton gauze and a custom-made fenestrated jacket to prevent exogenous bacterial contamination. By day 15, the FFs-ICG-treated group demonstrated significantly enhanced wound closure efficacy compared to other treated groups (Fig. 6b, c and Supplementary Fig. 33a), and its residual wound areas were even smaller than those in non-infected controls. This superior regenerative and healing performance is understood to result from the cell proliferation-promoting properties of our FFs-ICG nanoNETs. Furthermore, the quantitative analysis revealed the lowest bacterial survival rate in the FFs-ICG group (Fig. 6d, e), confirming their robust in vivo antibacterial activity.

a Schematic timeline outlining the experimental design for assessing the therapeutic efficiency of FFs-ICG hydrogel in an MRSA-infected porcine wound model. Created in BioRender. Xuan, Q. (2025) https://BioRender.com/e9mjxve. Wound area quantification (n = 3) (b) and the corresponding representative wound images (Scale bar: 1.5 cm) (c) of MRSA-infected Bama pig after various treatments. The CFU plate images (d) and statistical analysis of MRSA survival rates (e) (n = 3; data are presented as individual points) in the infected skin tissues after different treatments. Non-infected (p = 0.0018), RFs (p = 0.0270), FFs (p = 0.0054), RFs-ICG (p = 0.0070), FFs-ICG (p = 0.0023) vs. PBS control. Representative H&E staining (scale bar: 1 mm) (f), Masson-staining (scale bar: 1 mm) (g), Giemsa-staining (Scale bar: 1 mm, top; 200 μm, down) (h), and Ki-67-staining (scale bar: 500 μm) (i) images of porcine wounds in different treatment groups on day 15 for the analysis of dermis and epidermis regeneration, collagen deposition, survival bacteria, and cell proliferation, respectively. In Fig. 6h, the red arrows represent the residual bacteria. Representative CD206-staining images (scale bar: 500 μm) (j) and the corresponding quantification (k) (n = 3; data are presented as individual points) of porcine wounds for the analysis of macrophage polarization in different treatment groups on day 15. Non-infected (p = 0.0084), RFs (p = 0.0225), FFs (p = 0.0472), RFs-ICG (p = 0.0010), FFs-ICG (p = 0.0022) vs. PBS control. The data were expressed as mean ± standard deviation (S.D). Statistical significance was analyzed by one-way ANOVA using GraphPad Prism 8, followed by Tukey’s post hoc test for pairwise comparisons. Statistical significance was defined as *p < 0.05, **p < 0.01, and ***p < 0.001. Source data are provided as a Source Data file.
Histological evaluation using H&E and Masson’s staining demonstrated enhanced tissue regeneration in the FFs-ICG group, showing complete restoration of dermis and epidermis architectures, and well-organized collagen deposition, in addition to the smallest wound area (Fig. 6f, g and Supplementary Fig. 33b, c). Giemsa and Ki-67 immunohistochemistry analysis further validated the dual functionality of FFs-ICG nanoNETs, highlighting both robust antibacterial action and enhanced cell proliferative activity (Fig. 6h–i and Supplementary Fig. 33d–f). Immunofluorescence analysis of CD206 (Fig. 6j–k and Supplementary Fig. 33g) indicated a significantly elevated ratio of M2-phenotype macrophage in the FFs-ICG group compared to controls, indicating its potent immunomodulatory and anti-inflammatory effects. Additionally, comprehensive biosafety assessment, including the histological analysis of major organs and the blood biochemistry analysis, confirmed the excellent in vivo biocompatibility of these lysozyme-based nanofibrils (Supplementary Fig. 33h, i). Together, these results demonstrate that FFs-ICG nanoNETs promote wound healing through a multifunctional mechanism combining: (i) NETs-like antibacterial mechanism, (ii) anti-inflammatory modulation via macrophage polarization, and (iii) stimulation of cellular proliferation.
This work introduces lysozyme reversible amyloid fibrils as an original and powerful class of stimuli-responsible scaffolds for therapeutic tissue regeneration in wounds affected by bacterial infections. The escalating prevalence of antibiotic resistance demands innovative antimicrobial strategies that surpass conventional therapeutic paradigms. The developed photothermally switchable lysozyme-based nanoNETs system recapitulates natural NETs immune mechanisms enabling effective infection control and tissue regeneration upon non-invasive triggers, such as NIR. The resulting outstanding performance in tissue regeneration of these nanoNETs occurs via the dual release of bioactive lysozyme monomers, which facilitate site-specific bactericidal activity, and Mg²⁺ ions, which modulate macrophage polarization, thus mitigating immune dysregulation. The potential of this approach was validated on three preclinical animal models, considering both soft tissue and orthopedic implant infections, demonstrating a significant reduction in bacterial infection, accelerated epithelialization, biofilm prevention, and joint function restoration. By leveraging natural immune strategies combined with photothermal activation, the biomimetic approach proposed here bridges host defense mechanisms with adaptable material design, a concept which may be further extended to other bioactive proteinaceous compounds -such as defensins and myeloperoxidase- greatly expanding the scope of next-generation antimicrobial therapies and regenerative medicine.

















Leave a Reply