Kuhn H, Banthiya S, van Leyen K. Mammalian lipoxygenases and their biological relevance. Biochim Biophys Acta. 2015;1851:308–30. https://doi.org/10.1016/j.bbalip.2014.10.002.
Jisaka M, Kim RB, Boeglin WE, Nanney LB, Brash AR. Molecular cloning and functional expression of a phorbol ester-inducible 8S-lipoxygenase from mouse skin. J Biol Chem. 1997;272:24410–6. https://doi.org/10.1074/jbc.272.39.24410.
Palmer MA, Benatzy Y, Brüne B. Murine Alox8 versus the human ALOX15B ortholog: differences and similarities. Pflugers Arch. 2024;476:1817–32. https://doi.org/10.1007/s00424-024-02961-w.
Magnusson LU, Lundqvist A, Karlsson MN, Skålén K, Levin M, Wiklund O, et al. Arachidonate 15-lipoxygenase type B knockdown leads to reduced lipid accumulation and inflammation in atherosclerosis. PLoS ONE. 2012;7:e43142 https://doi.org/10.1371/journal.pone.0043142.
Benatzy Y, Palmer MA, Lütjohann D, Ohno R-I, Kampschulte N, Schebb NH, et al. ALOX15B controls macrophage cholesterol homeostasis via lipid peroxidation, ERK1/2 and SREBP2. Redox Biol. 2024;72:103149. https://doi.org/10.1016/j.redox.2024.103149.
Kim E, Rundhaug JE, Benavides F, Yang P, Newman RA, Fischer SM. An antitumorigenic role for murine 8S-lipoxygenase in skin carcinogenesis. Oncogene. 2005;24:1174–87. https://doi.org/10.1038/sj.onc.1208269.
Gras D, Bourdin A, Vachier I, Senneville L, de, Bonnans C, et al. An ex vivo model of severe asthma using reconstituted human bronchial epithelium. J Allergy Clin Immunol. 2012;129:1259–1266.e1. https://doi.org/10.1016/j.jaci.2012.01.073.
Alfardan R, Guo C, Toth LA, Nie D. Impaired Recovery from Influenza A/X-31(H3N2) Infection in Mice with 8-Lipoxygenase Deficiency. Med Sci. 2019. https://doi.org/10.3390/medsci7040060.
Suraneni MV, Schneider-Broussard R, Moore JR, Davis TC, Maldonado CJ, Li H, et al. Transgenic expression of 15-lipoxygenase 2 (15-LOX2) in mouse prostate leads to hyperplasia and cell senescence. Oncogene. 2010;29:4261–75. https://doi.org/10.1038/onc.2010.197.
Sieminska I, Pieniawska M, Grzywa TM. The immunology of psoriasis-current concepts in pathogenesis. Clin Rev Allergy Immunol. 2024;66:164–91. https://doi.org/10.1007/s12016-024-08991-7.
Sorokin AV, Domenichiello AF, Dey AK, Yuan Z-X, Goyal A, Rose SM, et al. Bioactive lipid mediator profiles in human psoriasis skin and blood. J Invest Dermatol. 2018;138:1518–28. https://doi.org/10.1016/j.jid.2018.02.003.
Nicolaou A. Eicosanoids in skin inflammation. Prostaglandins Leukot Essent Fatty Acids. 2013;88:131–8. https://doi.org/10.1016/j.plefa.2012.03.009.
Kragballe K, Pinnamaneni G, Desjarlais L, Duell EA, Voorhees JJ. Dermis-derived 15-hydroxy-eicosatetraenoic acid inhibits epidermal 12-lipoxygenase activity. J Investig Dermatol. 1986;87:494–8. https://doi.org/10.1111/1523-1747.ep12455564.
Singh S, Ahmed F, Fatima N, Cholleti Sai N, Bhuktar H, Khan RA, et al. ALOX12B overexpression in the skin drives inflammasome/Th17 signaling axis to promote inflammation in the mouse model and human patients. 2025. Preprint available at https://doi.org/10.1101/2025.06.09.658245.
Schebb NH, Kühn H, Kahnt AS, Rund KM, O’Donnell VB, Flamand N, et al. Formation, signaling and occurrence of specialized pro-resolving lipid mediators-what is the evidence so far? Front Pharmacol. 2022;13:838782. https://doi.org/10.3389/fphar.2022.838782.
Schebb NH, Kampschulte N, Hagn G, Plitzko K, Meckelmann SW, Ghosh S, et al. Technical recommendations for analyzing oxylipins by liquid chromatography-mass spectrometry. Sci Signal. 2025;18:eadw1245. https://doi.org/10.1126/scisignal.adw1245.
Saito P, Melo CPB, Martinez RM, Fattori V, Cezar TLC, Pinto IC, et al. The lipid mediator resolvin D1 reduces the skin inflammation and oxidative stress induced by UV irradiation in hairless mice. Front Pharmacol. 2018;9:1242. https://doi.org/10.3389/fphar.2018.01242.
Hellmann J, Sansbury BE, Wong B, Li X, Singh M, Nuutila K, et al. Biosynthesis of D-series resolvins in skin provides insights into their role in tissue repair. J Investig Dermatol. 2018;138:2051–60. https://doi.org/10.1016/j.jid.2018.03.1498.
Park K-D, Kim N, Kang J, Dhakal H, Kim JY, Jang YH, et al. Protectin D1 reduces imiquimod-induced psoriasiform skin inflammation. Int Immunopharmacol. 2021;98:107883. https://doi.org/10.1016/j.intimp.2021.107883.
Hu F, Qu Z, Chen K, Zhang P, Wang B, Jiang R, et al. Lipoxin A4 ameliorates imiquimod-induced psoriasis-like dermatitis via promoting the regression of inflammation. Clin Cosmet Investig Dermatol. 2023;16:2103–11. https://doi.org/10.2147/CCID.S418467.
Palmer MA, Kirchhoff R, Buerger C, Benatzy Y, Schebb NH, Brüne B. RNAi-based ALOX15B silencing augments keratinocyte inflammation in vitro via EGFR/STAT1/JAK1 signalling. Cell Death Dis. 2025;16:39. https://doi.org/10.1038/s41419-025-07357-x.
Seiringer P, Hillig C, Schäbitz A, Jargosch M, Pilz AC, Eyerich S, et al. Spatial transcriptomics reveals altered lipid metabolism and inflammation-related gene expression of sebaceous glands in psoriasis and atopic dermatitis. Front Immunol. 2024;15:1334844. https://doi.org/10.3389/fimmu.2024.1334844.
Wang X, Hao Y, Wang X, Wang L, Chen Y, Sun J, et al. A PPARδ-selective antagonist ameliorates IMQ-induced psoriasis-like inflammation in mice. Int Immunopharmacol. 2016;40:73–8. https://doi.org/10.1016/j.intimp.2016.08.027.
Kakularam KR, Canyelles-Niño M, Chen X, Lluch JM, González-Lafont À, Kuhn H. Functional characterization of mouse and human arachidonic acid lipoxygenase 15B (ALOX15B) orthologs and of their mutants exhibiting humanized and murinized reaction specificities. Int J Mol Sci 2023. https://doi.org/10.3390/ijms241210046.
Vietri Rudan M, Watt FM. Mammalian epidermis: a compendium of lipid functionality. Front Physiol. 2021;12:804824. https://doi.org/10.3389/fphys.2021.804824.
Lee JL, Mukhtar H, Bickers DR, Kopelovich L, Athar M. Cyclooxygenases in the skin: pharmacological and toxicological implications. Toxicol Appl Pharmacol. 2003;192:294–306. https://doi.org/10.1016/s0041-008x(03)00301-6.
Tripp CS, Blomme EAG, Chinn KS, Hardy MM, LaCelle P, Pentland AP. Epidermal COX-2 induction following ultraviolet irradiation: suggested mechanism for the role of COX-2 inhibition in photoprotection. J Investig Dermatol. 2003;121:853–61. https://doi.org/10.1046/j.1523-1747.2003.12495.x.
Gschwendt M, Fürstenberger G, Kittstein W, Besemfelder E, Hull WE, Hagedorn H, et al. Generation of the arachidonic acid metabolite 8-HETE by extracts of mouse skin treated with phorbol ester in vivo; identification by 1H-n.m.r. and GC-MS spectroscopy. Carcinogenesis. 1986;7:449–55. https://doi.org/10.1093/carcin/7.3.449.
Schneider C, Strayhorn WD, Brantley DM, Nanney LB, Yull FE, Brash AR. Upregulation of 8-lipoxygenase in the dermatitis of IkappaB-alpha-deficient mice. J Investig Dermatol. 2004;122:691–8. https://doi.org/10.1111/j.0022-202X.2004.22329.x.
Bender G, Schexnaydre EE, Murphy RC, Uhlson C, Newcomer ME. Membrane-dependent activities of human 15-LOX-2 and its murine counterpart: IMPLICATIONS FOR MURINE MODELS OF ATHEROSCLEROSIS. J Biol Chem. 2016;291:19413–24. https://doi.org/10.1074/jbc.M116.741454.
Jarocka-Karpowicz I, Biernacki M, Wroński A, Gęgotek A, Skrzydlewska E. Cannabidiol effects on phospholipid metabolism in keratinocytes from patients with psoriasis vulgaris. Biomolecules 2020. https://doi.org/10.3390/biom10030367.
Shou Y, Yang L, Yang Y, Xu J. Inhibition of keratinocyte ferroptosis suppresses psoriatic inflammation. Cell Death Dis. 2021;12:1009. https://doi.org/10.1038/s41419-021-04284-5.
Awasthi YC, Sharma R, Cheng JZ, Yang Y, Sharma A, Singhal SS, et al. Role of 4-hydroxynonenal in stress-mediated apoptosis signaling. Mol Asp Med. 2003;24:219–30. https://doi.org/10.1016/S0098-2997(03)00017-7.
Ji Y, Dai Z, Wu G, Wu Z. 4-Hydroxy-2-nonenal induces apoptosis by activating ERK1/2 signaling and depleting intracellular glutathione in intestinal epithelial cells. Sci Rep. 2016;6:32929. https://doi.org/10.1038/srep32929.
Swiader A, Camaré C, Guerby P, Salvayre R, Negre-Salvayre A. 4-hydroxynonenal contributes to fibroblast senescence in skin photoaging evoked by UV-A radiation. Antioxidants 2021. https://doi.org/10.3390/antiox10030365.
Shearn CT, Reigan P, Petersen DR. Inhibition of hydrogen peroxide signaling by 4-hydroxynonenal due to differential regulation of Akt1 and Akt2 contributes to decreases in cell survival and proliferation in hepatocellular carcinoma cells. Free Radic Biol Med. 2012;53:1–11. https://doi.org/10.1016/j.freeradbiomed.2012.04.021.
Polese B, Thurairajah B, Zhang H, Soo CL, McMahon CA, Fontes G, et al. Prostaglandin E2 amplifies IL-17 production by γδ T cells during barrier inflammation. Cell Rep. 2021;36:109456. https://doi.org/10.1016/j.celrep.2021.109456.
Lee J, Aoki T, Thumkeo D, Siriwach R, Yao C, Narumiya S. T cell-intrinsic prostaglandin E2-EP2/EP4 signaling is critical in pathogenic TH17 cell-driven inflammation. J Allergy Clin Immunol. 2019;143:631–43. https://doi.org/10.1016/j.jaci.2018.05.036.
Voglis S, Moos S, Kloos L, Wanke F, Zayoud M, Pelczar P, et al. Regulation of IL-22BP in psoriasis. Sci Rep. 2018;8:5085. https://doi.org/10.1038/s41598-018-23510-3.
Qi L, Pan X, Chen X, Liu P, Chen M, Zhang Q, et al. COX-2/PGE2 upregulation contributes to the chromosome 17p-deleted lymphoma. Oncogenesis. 2023;12:5. https://doi.org/10.1038/s41389-023-00451-9.
Knox S, O’Boyle NM. Skin lipids in health and disease: a review. Chem Phys Lipids. 2021;236:105055. https://doi.org/10.1016/j.chemphyslip.2021.105055.
Lew B-L, Cho Y, Kim J, Sim W-Y, Kim N-I. Ceramides and cell signaling molecules in psoriatic epidermis: reduced levels of ceramides, PKC-alpha, and JNK. J Korean Med Sci. 2006;21:95–9. https://doi.org/10.3346/jkms.2006.21.1.95.
Farwanah H, Raith K, Neubert RHH, Wohlrab J. Ceramide profiles of the uninvolved skin in atopic dermatitis and psoriasis are comparable to those of healthy skin. Arch Dermatol Res. 2005;296:514–21. https://doi.org/10.1007/s00403-005-0551-2.
Cho Y, Lew B-L, Seong K, Kim N-I. An inverse relationship between ceramide synthesis and clinical severity in patients with psoriasis. J Korean Med Sci. 2004;19:859–63. https://doi.org/10.3346/jkms.2004.19.6.859.
Uchida Y, Murata S, Schmuth M, Behne MJ, Lee JD, Ichikawa S, et al. Glucosylceramide synthesis and synthase expression protect against ceramide-induced stress. J Lipid Res. 2002;43:1293–302.
Wakita H, Tokura Y, Yagi H, Nishimura K, Furukawa F, Takigawa M. Keratinocyte differentiation is induced by cell-permeant ceramides and its proliferation is promoted by sphingosine. Arch Dermatol Res. 1994;286:350–4. https://doi.org/10.1007/BF00402228.
Jiang YJ, Kim P, Uchida Y, Elias PM, Bikle DD, Grunfeld C, et al. Ceramides stimulate caspase-14 expression in human keratinocytes. Exp Dermatol. 2013;22:113–8. https://doi.org/10.1111/exd.12079.
Sayama K, Hanakawa Y, Shirakata Y, Yamasaki K, Sawada Y, Sun L, et al. Apoptosis signal-regulating kinase 1 (ASK1) is an intracellular inducer of keratinocyte differentiation. J Biol Chem. 2001;276:999–1004. https://doi.org/10.1074/jbc.M003425200.
Picardo M, Ottaviani M, Camera E, Mastrofrancesco A. Sebaceous gland lipids. Dermatoendocrinol. 2009;1:68–71. https://doi.org/10.4161/derm.1.2.8472.
Jisaka M, Iwanaga C, Takahashi N, Goto T, Kawada T, Yamamoto T, et al. Double dioxygenation by mouse 8S-lipoxygenase: specific formation of a potent peroxisome proliferator-activated receptor alpha agonist. Biochem Biophys Res Commun. 2005;338:136–43. https://doi.org/10.1016/j.bbrc.2005.08.029.
Yamada H, Oshiro E, Kikuchi S, Hakozaki M, Takahashi H, Kimura K-I. Hydroxyeicosapentaenoic acids from the Pacific krill show high ligand activities for PPARs. J Lipid Res. 2014;55:895–904. https://doi.org/10.1194/jlr.M047514.
Hanley K, Kömüves LG, Ng DC, Schoonjans K, He SS, Lau P, et al. Farnesol stimulates differentiation in epidermal keratinocytes via PPARalpha. J Biol Chem. 2000;275:11484–91. https://doi.org/10.1074/jbc.275.15.11484.
Kömüves LG, Hanley K, Lefebvre AM, Man MQ, Ng DC, Bikle DD, et al. Stimulation of PPARalpha promotes epidermal keratinocyte differentiation in vivo. J Investig Dermatol. 2000;115:353–60. https://doi.org/10.1046/j.1523-1747.2000.00073.x.
Muga SJ, Thuillier P, Pavone A, Rundhaug JE, Boeglin WE, Jisaka M, et al. 8S-lipoxygenase products activate peroxisome proliferator-activated receptor alpha and induce differentiation in murine keratinocytes. Cell Growth Differ. 2000;11:447–54.
Mao-Qiang M, Fowler AJ, Schmuth M, Lau P, Chang S, Brown BE, et al. Peroxisome-proliferator-activated receptor (PPAR)-gamma activation stimulates keratinocyte differentiation. J Investig Dermatol. 2004;123:305–12. https://doi.org/10.1111/j.0022-202X.2004.23235.x.
Kim DJ, Bility MT, Billin AN, Willson TM, Gonzalez FJ, Peters JM. PPARbeta/delta selectively induces differentiation and inhibits cell proliferation. Cell Death Differ. 2006;13:53–60. https://doi.org/10.1038/sj.cdd.4401713.
Michalik L, Desvergne B, Tan NS, Basu-Modak S, Escher P, Rieusset J, et al. Impaired skin wound healing in peroxisome proliferator-activated receptor (PPAR)alpha and PPARbeta mutant mice. J Cell Biol. 2001;154:799–814. https://doi.org/10.1083/jcb.200011148.
Schweiger D, Fürstenberger G, Krieg P. Inducible expression of 15-lipoxygenase-2 and 8-lipoxygenase inhibits cell growth via common signaling pathways. J Lipid Res. 2007;48:553–64. https://doi.org/10.1194/jlr.M600311-JLR200.
van der Fits L, Mourits S, Voerman JSA, Kant M, Boon L, Laman JD, et al. Imiquimod-induced psoriasis-like skin inflammation in mice is mediated via the IL-23/IL-17 axis. J Immunol. 2009;182:5836–45. https://doi.org/10.4049/jimmunol.0802999.
Amberg N, Holcmann M, Stulnig G, Sibilia M. Effects of imiquimod on hair follicle stem cells and hair cycle progression. J Investig Dermatol. 2016;136:2140–9. https://doi.org/10.1016/j.jid.2016.06.613.
Koch E, Mainka M, Dalle C, Ostermann AI, Rund KM, Kutzner L, et al. Stability of oxylipins during plasma generation and long-term storage. Talanta. 2020;217:121074. https://doi.org/10.1016/j.talanta.2020.121074.
Hartung NM, Mainka M, Pfaff R, Kuhn M, Biernacki S, Zinnert L, et al. Development of a quantitative proteomics approach for cyclooxygenases and lipoxygenases in parallel to quantitative oxylipin analysis allowing the comprehensive investigation of the arachidonic acid cascade. Anal Bioanal Chem. 2023;415:913–33. https://doi.org/10.1007/s00216-022-04489-3.
Ostermann AI, Koch E, Rund KM, Kutzner L, Mainka M, Schebb NH. Targeting esterified oxylipins by LC–MS – effect of sample preparation on oxylipin pattern. Prostaglandins Other Lipid Mediat. 2020;146:106384. https://doi.org/10.1016/j.prostaglandins.2019.106384.
Kutzner L, Rund KM, Ostermann AI, Hartung NM, Galano J-M, Balas L, et al. Development of an optimized LC-MS method for the detection of specialized pro-resolving mediators in biological samples. Front Pharmacol. 2019;10:169. https://doi.org/10.3389/fphar.2019.00169.
Rund KM, Ostermann AI, Kutzner L, Galano J-M, Oger C, Vigor C, et al. Development of an LC-ESI(-)-MS/MS method for the simultaneous quantification of 35 isoprostanes and isofurans derived from the major n3- and n6-PUFAs. Anal Chim Acta. 2018;1037:63–74. https://doi.org/10.1016/j.aca.2017.11.002.
Sens A, Rischke S, Hahnefeld L, Dorochow E, Schäfer SMG, Thomas D, et al. Pre-analytical sample handling standardization for reliable measurement of metabolites and lipids in LC-MS-based clinical research. J Mass Spectrom Adv Clin Lab. 2023;28:35–46. https://doi.org/10.1016/j.jmsacl.2023.02.002.
Dorochow E, Gurke R, Rischke S, Geisslinger G, Hahnefeld L. Effects of different storage conditions on lipid stability in mice tissue homogenates. Metabolites 2023. https://doi.org/10.3390/metabo13040504.
Pang Z, Lu Y, Zhou G, Hui F, Xu L, Viau C, et al. MetaboAnalyst 6.0: towards a unified platform for metabolomics data processing, analysis and interpretation. Nucleic Acids Res. 2024;52:W398–W406. https://doi.org/10.1093/nar/gkae253.
Bankhead P, Loughrey MB, Fernández JA, Dombrowski Y, McArt DG, Dunne PD, et al. QuPath: open source software for digital pathology image analysis. Sci Rep. 2017;7:16878. https://doi.org/10.1038/s41598-017-17204-5.
Schmidt U, Weigert M, Broaddus C, Myers G. Cell detection with star-convex polygons. In: Frangi A, Schnabel J, Davatzikos C, Alberola-López C, Fichtinger G, editors. Medical image computing and computer assisted intervention – MICCAI 2018. MICCAI 2018. Lecture notes in computer science. Springer; Cham: 2018. p 265–273.





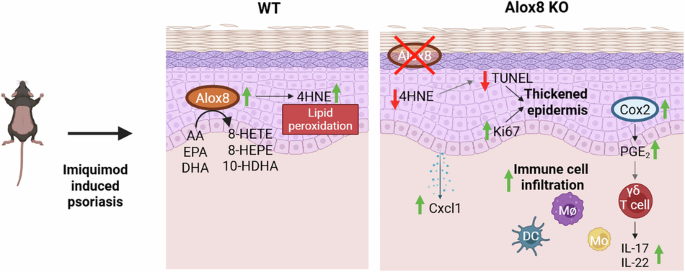







Leave a Reply