Macrophages, first described over a century ago for their ability to engulf debris and pathogens, display remarkable plasticity and heterogeneity1,2,3. Present in nearly all tissues from early fetal development through adult life, most tissue-resident macrophages originate from yolk-sac-derived erythroid–myeloid progenitors (EMPs), with partial replacement by macrophages originating from bone marrow hematopoietic stem cell-derived monocytes4,5,6,7. Given the scale of the vertebrate vascular network and the need to maintain blood quality and endothelial integrity, it remains unclear whether a dedicated macrophage population resides inside blood vessels. Kupffer cells in hepatic sinusoids filter portal venous blood to remove gut-derived microbes and debris8, and red pulp macrophages phagocytose senescent erythrocytes and pathogens within the unique open circulatory system of the spleen9,10. However, these actions are restricted to these tissues’ specific vascular niches, leaving most blood vessels unmonitored. Patrolling monocytes can initiate early immune responses11, yet rapidly extravasate into the infected tissue and differentiate into macrophages11, emphasizing their transient nature. Here, we identify a bona fide population of intravascular macrophages that continuously safeguards blood and vessel integrity, which we term blood vessel-resident macrophages (bMΦs).
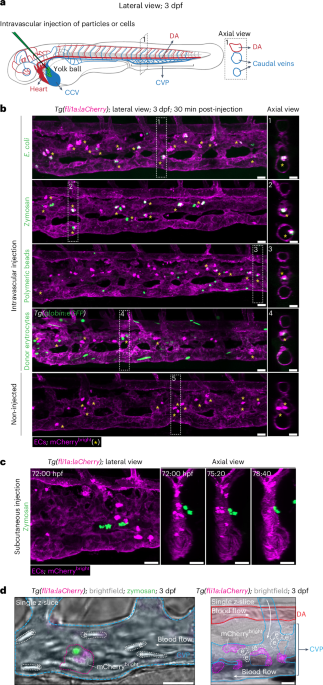
To determine how threats are cleared from the bloodstream, fluorescently labeled foreign particles and cells were injected into the circulation of zebrafish embryos 3 days post fertilization (dpf), in which the fli1a promoter drives mCherry expression and labels all vascular endothelial cells (ECs)12 (Fig. 1a). Within 30 min, particles and cells were exclusively phagocytosed by cells with an unusually bright mCherry signal (mCherrybright) (Fig. 1b, Extended Data Fig. 1a and Supplementary Video 1). These phagocytic mCherrybright cells were located inside venous blood vessels of the caudal part of the trunk, also known as the caudal vein plexus (CVP). Under homeostatic conditions, mCherrybright cells were already present in the CVP and their numbers did not change after foreign particle exposure (Fig. 1b, bottom, and Extended Data Fig. 1b). Zymosan injected into the tissue adjacent to the CVP did not result in extravasation of intravascular mCherrybright cells (Fig. 1c and Supplementary Video 2), indicating that they function within the vasculature. High-spatiotemporal-resolution imaging showed that mCherrybright cells were not embedded in vessel walls but crawled with an ameboid-like movement along the luminal surface, independently of blood flow direction (Fig. 1d and Supplementary Videos 3 and 4), often transiently obstructing flow, indicative of strong endothelial adhesion (Supplementary Video 4).
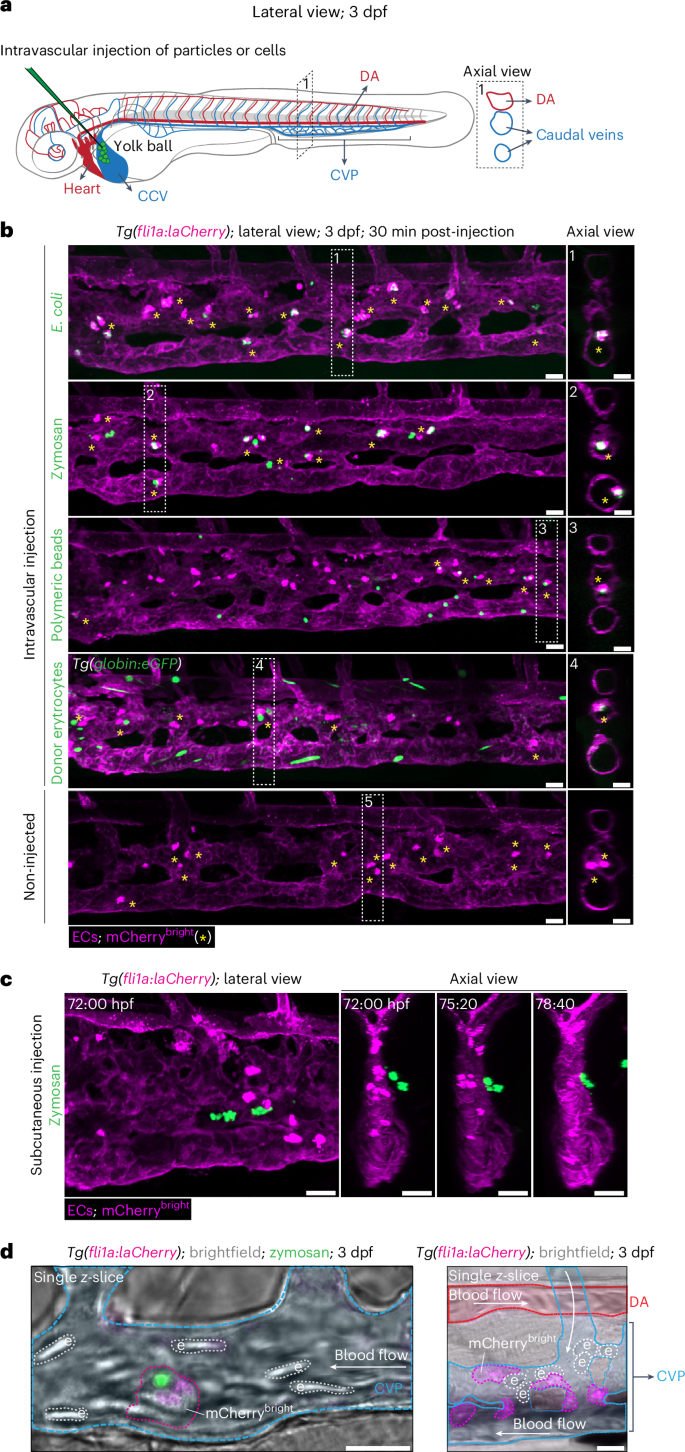
a, Schematic representation of a 3-dpf zebrafish embryo. The axial view (dashed box) is generated postprocessing to visualize the intraluminal space of the blood vessels. Anterior is oriented to the left for all imaging data in this study. b, Images of the CVP of 3-dpf Tg(fli1a:laCherry) embryos after intravascular injection of fluorescently labeled particles (E. coli, zymosan (S. cerevisiae bioparticles), polymeric beads) or cells (donor erythrocytes from adult Tg(globin:eGFP)) (green). Injection in the CCV as indicated in the scheme in a. All ECs (fli1a) are labeled with laCherry (magenta) that is fused N-terminally to lifeact (la), a small actin-binding peptide that is incorporated into the actin cytoskeleton. GFP+ particles and cells (green) were exclusively phagocytosed by mCherrybright cells. The bottom panel shows the non-injected control. mCherrybright cells are indicated by asterisks. c, Still images of Supplementary Video 2 in which zymosan particles (green) were injected into the tissue adjacent to the CVP of a Tg(fli1a:laCherry) embryo. d, Still images of Supplementary Videos 3 (left) and 4 (right), showing 3-dpf Tg(fli1a:laCherry) embryos injected with zymosan (left) or not injected (right). DA, CVP, erythrocytes and mCherrybright cells are outlined and colored on brightfield single z-slide images. Images are representative of n = 3 independent experiments (30 embryos analyzed in total) (b), n = 2 (10 embryos analyzed in total) (c), and 2 and 5 independent time-lapse experiments, respectively (d). Scale bars, 20 µm. e, erythrocyte.
To identify the nature of mCherrybright cells, we crossed our fli1a reporter line with the reporter transgenic lines Tg(mpx:GFP)13 (where Tg is transgenic, mpx is myeloid-specific peroxidase and GFP is green fluorescent protein) and Tg(mpeg:GFP)14 (where mpeg is macrophage expressed 1 (tandem duplicate 1; mpeg1.1)), which label neutrophils and macrophages, respectively. While no mCherrybright cells expressed GFP in the neutrophil line (Fig. 2a, top), they all expressed GFP in the macrophage line (Fig. 2a, bottom), indicating a macrophage identity. mCherrybright cells were also detected using combinations of alternative endothelial specific (that is, kdrl and flt4)15,16 and macrophage (mpeg)17 reporter line (Extended Data Fig. 2a–g). We thus identified a population of phagocytic macrophages expressing endothelial markers and confined to the vasculature, which we termed blood vessel-resident macrophages (bMΦs).
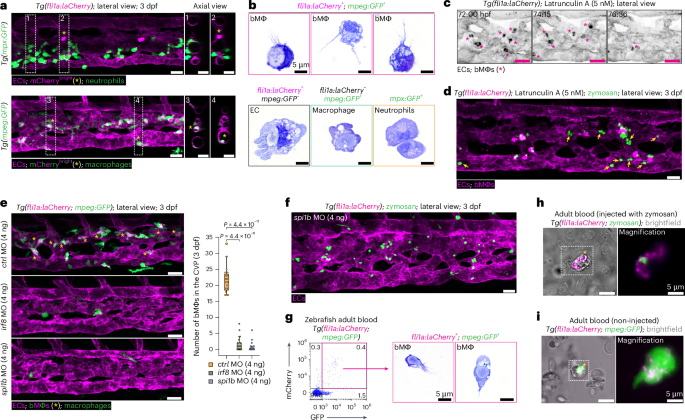
a, Images of the CVP of 3-dpf Tg(fli1a:laCherry; mpx:GFP) (top) and Tg(fli1a:laCherry; mpeg:GFP) (bottom) embryos, where neutrophils and macrophages are marked by GFP (green), respectively. All ECs are marked by mCherry (magenta). Asterisks depict mCherrybright cells. b, Images of bMΦs, ECs, tissue macrophage and neutrophils sorted by flow cytometry based on the indicated fluorophores after May–Grünwald Giemsa staining. c, Still images of Supplementary Video 5 in which a 3-dpf Tg(fli1a:laCherry) embryo was treated with Latrunculin A. Asterisks indicate bMΦs. d, Image of the CVP of a 3-dpf Tg(fli1a:laCherry) embryo pretreated for 1 h with Latrunculin A, followed by an intravascular injection of zymosan particles (green). Arrows indicate nonphagocytosed zymosan. e, Images and quantification of bMΦs in the CVP of 3-dpf Tg(fli1a:laCherry; mpeg:GFP) embryos injected with a control, irf8 or spi1b MO (4 ng). f, Still image of Supplementary Video 6 in which zymosan particles were intravascularly injected in a 3-dpf spi1b morphant (Tg(fli1a:laCherry)). g, Representative flow cytometry plot and gating strategy for sorting bMΦs from adult Tg(fli1a:laCherry; mpeg:GFP) zebrafish blood. Sorted cells were cytocentrifuged (cytospin) and stained by May–Grünwald Giemsa before imaging. Representative pictures of bMΦs. h, Representative images of a blood smear of adult Tg(fli1a:laCherry; mpeg:GFP) zebrafish. i, Representative images of a blood smear of adult Tg(fli1a:laCherry) zebrafish retro-orbitally injected with zymosan. Images are representative of n = 3 independent experiments (30 embryos analyzed in total) (a), n = 4 independent sorts and cytospins (b), n = 2 independent time-lapse experiments (c, d and f) and n = 2 (20 embryos) (e). The flow cytometry plot is a representative of n = 3 (4 adults/n) independent experiments (g), and images are representative of n = 2 (4 adults/n) independent experiments (h and i). Percentages of populations are indicated in the quadrants and gates of each plot. Scale bars, 20 µm (a and c–f) and 5 µm (b). The box plot shows the median (center), first and third quartiles (bounds) and 1.5 times the interquartile range (whiskers) (e). Statistical analysis was performed using a Kruskal–Wallis test followed by Dunn’s multiple comparisons test versus WT (e).
Source data
We then sorted bMΦs (fli1a+mpeg+), macrophages residing within the tissue (fli1a−mpeg+, referred to as tissue macrophages), ECs (fli1a+mpeg−) and neutrophils (mpx+) on the basis of fluorescent reporter expression to compare their morphology. bMΦs were distinguishable by filopodia-like protrusions on their surface (Fig. 2b and Extended Data Fig. 2h,i). Such actin-rich protrusions are typically used for attachment, migration, and as cellular tentacles to sense, catch and pull particles18,19. Inhibition of actin polymerization by a low dose of Latrunculin A impaired bMΦ attachment (Fig. 2c and Supplementary Video 5) and reduced clearance of intravascularly injected zymosan, leaving most particles circulating freely (Fig. 2d, arrows). These protrusions are therefore essential for endothelial adhesion and phagocytosis. Intravascular injection of fluorescently labeled peanut agglutinin (PNA) did not bind to bMΦs, but only to CVP venous ECs (Extended Data Fig. 2j), demonstrating that bMΦs are not dendritic cells (DCs), which also display similar filopodia-like protrusions or dendrites20,21. Altogether, we identified a previously unrecognized population of intravascular macrophages, marked by endothelial (fli1a, kdrl and flt4) and macrophage (mpeg) reporters, that actively phagocytoses foreign particles and cells within the bloodstream.
Macrophage development depends on spi-1 proto-oncogene b (spi1b) (PU.1)22 and downstream-acting interferon regulatory factor 8 (irf8)23. While spi1b is required for the differentiation of EMPs into bipotential neutrophil–macrophage progenitors22, irf8 skews these progenitors toward macrophage lineage at the expense of neutrophils24,25. Injection of well-characterized morpholino oligonucleotides (MOs) to knock down irf825 or spi1b22 eliminated nearly all bMΦs and tissue macrophages (Fig. 2e), indicating that bMΦ formation relies on classical myeloid transcription factors. Accordingly, zymosan injected into the circulation of spi1b morphants was not phagocytosed (Fig. 2f and Supplementary Video 6), confirming that bMΦs are solely responsible for clearing foreign particles from the blood. We next analyzed blood from Tg(fli1a:laCherry; mpeg:GFP) (where laCherry is lifeactCherry) adult zebrafish and identified laCherry+GFP+ cells resembling embryonic bMΦs (Fig. 2g,h), which phagocytosed retro-orbitally injected zymosan into Tg(fli1a:laCherry) adults (Fig. 2i). Thus, bMΦs are also present and functionally active in adult zebrafish.
We then asked whether bMΦs contribute to blood homeostasis, for example by monitoring endogenous circulating cells. bMΦs can be uniquely identified by their distinct mCherrybright appearance, allowing the use of GFP reporter lines to mark erythrocytes or hematopoietic stem and progenitor cells (HSPCs). High-temporal single z-plane imaging of the CVP region showed bMΦs (mCherrybright) catching circulating erythrocytes (GFP+) and, after prolonged surface interactions, either releasing them (Fig. 3a and Supplementary Video 7) or phagocytosing them (Fig. 3b and Supplementary Videos 8 and 9). Phagocytosed erythrocytes were often unusually large, suggesting that bMΦs eliminate abnormal or unfit cells from the circulation. Importantly, this surveillance behavior was not restricted to erythrocytes but extended also to HSPCs (Fig. 3c and Supplementary Video 10), with occasional uptake of a small portion of fluorescent cytoplasmic material through trogocytosis (Fig. 3d), release or phagocytosis (Fig. 3e).
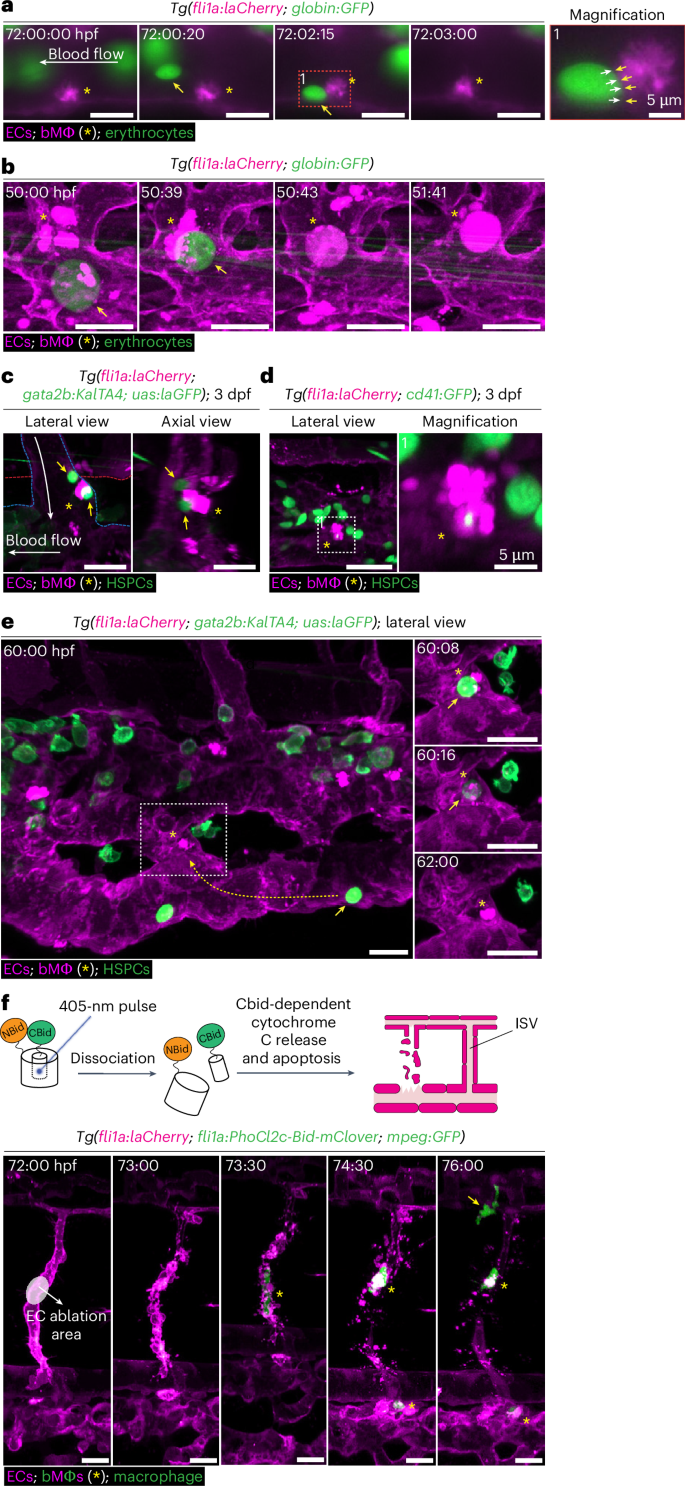
a,b, Still images of Supplementary Videos 7 (a) and 8 (b), illustrating the catching, assessment and release (a) or phagocytosis (b) of erythrocytes by bMΦs, respectively, in the CVP of Tg(fli1a:laCherry; globin:GFP) embryos in which erythrocytes are GFP+ (globin, arrow) and bMΦs are mCherrybright (fli1a, asterisk). Arrows in the magnification image show the intimate contact between the erythrocyte and bMΦ. c, Still images of Supplementary Video 10, showing the interactions between HSPCs that are marked by GFP (gata2b, arrows) and bMΦs that are mCherrybright (fli1a, asterisk). d, Images of the CVP region of a 3-dpf Tg(fli1a:laCherry; cd41:GFP) embryo, where HSPCs are GFP+ (cd41) and bMΦs are mCherrybright (fli1a). The asterisk indicates a bMΦ that engulfed cytoplasmic material of a GFP+ HSPC. The dashed square is shown enlarged in the right image. e, Time-lapse still images illustrating the phagocytosis of a HSPC by a bMΦ. In the CVP of 3-dpf Tg(fli1a:laCherry; gata2b:KalTA4; uas:laGFP), HSPCs are GFP+ (gata2b, arrow) and bMΦs are mCherrybright (fli1a, asterisk). f, Top: schematic representation of the optogenetic tool PhoCl2c-Bid. Upon a short UV light pulse of 405 nm, Bid is photocleaved and activated, which ultimately leads to the release of cytochrome c from the mitochondria and apoptosis of the cell. Bottom: still images of Supplementary Video 11, showing the optogenetic ablation of targeted ECs (translucent circle) in Tg(fli1a:laCherry; fli1a:PhoCl2c-Bid-mClover; mpeg:GFP) embryos in which tissue macrophages are GFP+laCherry− (arrow), ECs are GFP−laCherry+ and bMΦs are GFP+laCherry+ (asterisk). Images are representative of n = 3 independent time-lapse experiments (a–f). Scale bars, 20 µm, except for a (5 µm).
To further explore bMΦ function in tissue homeostasis, we adapted an ultraviolet (UV) light-responsive optogenetic tool26 to induce EC apoptosis (Fig. 3f, top). We ablated ECs in a single intersegmental vessel (ISV) in embryos expressing the transgene PhoCl2c-Bid-mClover (where Bid is BH3 interacting-domain death agonist), either stably in all ECs or mosaically. In these embryos, bMΦs (fli1a+mpeg+) rapidly arrived at the injury site, efficiently clearing EC debris (fli1a+mpeg−) while neighboring tissue macrophages (fli1a−mpeg+) dwelled only shortly around the injury site and occasionally phagocytosed EC debris (Fig. 3f, bottom, asterisks, Extended Data Fig. 3a and Supplementary Videos 11 and 12). Neutrophils are known for their rapid response to damage. However, like tissue macrophages, neutrophils were only occasionally recruited, had short dwell times around the injury and showed little to no phagocytosis of EC debris (Extended Data Fig. 3b, arrows, and Supplementary Video 13). Altogether, these data suggest that bMΦs contribute to blood and vascular homeostasis by surveying the circulation for foreign particles and unfit endogenous cells and by serving as first responders to EC damage.
To study the spatiotemporal origin of bMΦs, we imaged the entire trunk region starting from ~20 hpf. Just after the initiation of blood flow (~25 hpf), bMΦs (visualized as mCherrybright) emerged from both the roof and floor of the dorsal aorta (DA) and from the roof of the posterior cardinal vein (PCV) (Fig. 4a). Although bMΦs were also produced from the PCV floor, their emergence was difficult to image owing to delayed mCherry expression at this time point27. After emergence, bMΦs entered the bloodstream; some appeared in the common cardinal vein (CCV), which runs bilaterally over the yolk ball, while others traveled to the caudal vein (Fig. 4a, Extended Data Fig. 4a,b and Supplementary Video 14). Consistently, venous ECs photoconverted in the PCV at 24 hpf could be traced into the caudal vein (CV) 1 day later (Extended Data Fig. 4c). While the DA and PCV stopped producing bMΦs after ~30 hpf, venous ECs in the CVP continued producing bMΦs up to 3 dpf (Fig. 4b,c, Extended Data Fig. 4d and Supplementary Videos 15 and 16). bMΦ production involved rapid morphological changes in ECs, enabling them to emerge directly as bMΦs from the vessel wall into the circulation, with mCherry adopting its characteristic appearance. We termed this process endothelial-to-macrophage transition (EMacT). Thus, bMΦ production starts just after the initiation of circulation (~25 hpf) from both arterial and venous ECs of the DA and PCV and becomes restricted to the CVP around 30 hpf.
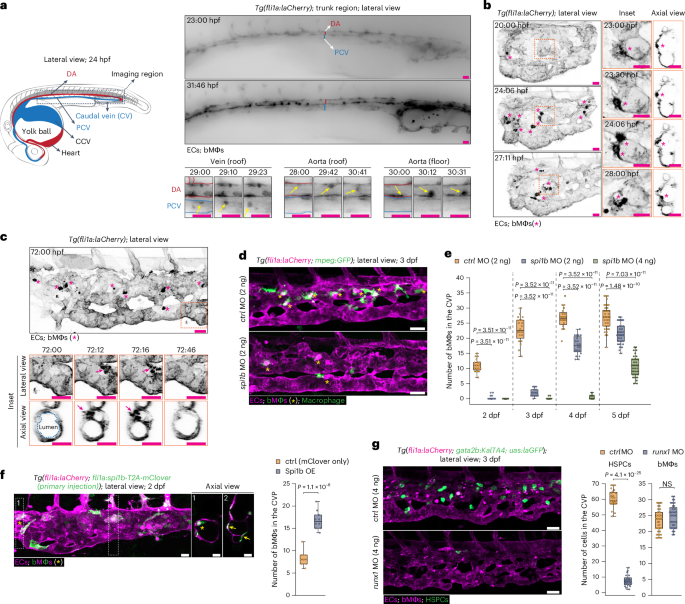
a, Left: schematic representation of a 24-hpf zebrafish embryo. The dashed line indicates the imaging region (trunk). Right: still images of Supplementary Video 14 at two time points. All ECs are marked by laCherry (black). Bottom: insets from different locations and time to illustrate the emergence of bMΦs (arrows). b, Still images of Supplementary Video 15. CVP region of a Tg(fli1a:laCherry) embryo. Orange boxes represent the insets shown on the left. Asterisks show venous ECs undergoing EMacT toward the luminal of the CV. c, Time-lapse still images, showing the transition of a venous EC into a bMΦ through EMacT (arrows). Asterisks: bMΦs already present in the CVP. Insets are a magnification of the dashed orange box in the top panel, shown in lateral and axial views. d,e, Images (d) and quantification (e) of bMΦs in the CVP of Tg(fli1a:laCherry; mpeg:GFP) embryos after control (ctrl) MO, or low (2 ng) or high (4 ng) doses of spi1b MO injection. Asterisks indicate de novo generated bMΦs (mCherrybrightGFP+). f, Images and quantification of bMΦs in the CVP of 2-dpf Tg(fli1a:laCherry; fli1a:spi1b-T2A-mClover) embryos, showing ectopically produced bMΦs by mosaic OE of Spi1b-T2A-laClover. The asterisk depicts a bMΦ. Arrows depict ECs expressing Spi1b that did not undergo EMacT. g, Images and quantification of HSPCs and bMΦs in the CVP of Tg(fli1a:laCherry; gata2b:KalTA4; uas:laGFP) embryos after ctrl MO and runx1 MO injection. HSPCs are GFP+ (gata2b), and bMΦs are mCherrybright (fli1a). Images are representative of n = 3 independent time-lapse experiments (a–c), n = 2 (2, 3 and 4 dpf, 20 embryos per condition; 5 dpf, 41 embryos per condition) (d and e), n = 2 (10 embryos) (f), n = 2 (ctrl MO HSPCs, 24 embryos; bMΦs, 42 embryos; runx1 MO HSPCs, 24 embryos; bMΦs, 40 embryos) (g). Scale bars, 20 µm. OE, overexpression; NS, not significant. Box plots show the median (center), first and third quartiles (bounds) and 1.5 times the interquartile range (whiskers) (e–g). Statistical analysis was performed using a Kruskal–Wallis test followed by Dunn’s multiple comparisons test versus WT (e) and a two-tailed Mann–Whitney U test for comparison of two conditions (f and g).
Source data
To further explore the capacity of the CVP to produce bMΦs, we prevented their production by injecting a low dose of spi1b MO (2 ng), which interfered with spi1b mRNA splicing up to 3 dpf (Extended Data Fig. 4e). In these embryos, the CVP started to produce bMΦs de novo from 3 dpf onward (Fig. 4d,e and Extended Data Fig. 4f). A higher spi1b MO dose (4 ng) caused an additional 1-day delay in bMΦ production (Fig. 4e). These de novo produced bMΦs were functional, phagocytosing intravascularly injected zymosan (Extended Data Fig. 4g). Mosaic overexpression of spi1b in ECs increased the number of ECs undergoing EMacT, and consequently the number of bMΦs, but this occurred at the expense of vascular morphology and integrity. (Fig. 4f, asterisk, and Extended Data Fig. 4h, magenta arrow). Importantly, some venous spi1b-expressing ECs (mClover+) did not undergo EMacT (Fig. 4f, yellow arrows, and Extended Data Fig. 4h, yellow arrows), suggesting that not all ECs are competent for this transition.
Arterial ECs in the floor of the DA (in the aorta–gonad–mesonephros (AGM) region) produce HSPCs via a Runx1-dependent endothelial-to-hematopoietic transition28,29,30,31,32. In runx1 morphants, the number of bMΦs remained normal, while the number of HSPCs was significantly reduced (Fig. 4g and Extended Data Fig. 4i). Furthermore, bMΦs lacked co-expression of the HSPC markers cd41 or gata2b (Extended Data Fig. 4j), confirming that bMΦs do not originate from HSPCs but instead arise through a novel runx1-independent EMacT that persists in the CVP until at least 5 dpf.
To gain further insight into the cellular and molecular difference between bMΦs, ECs and tissue macrophages, we sorted ~5,000 cells of each population and performed mass spectrometry (Extended Data Fig. 5a). We identified 55 unique proteins in bMΦs, 95 shared with tissue macrophages and 33 shared with ECs (Fig. 5a,b and Extended Data Fig. 5b). Filtering for proteins with known function in myelopoiesis, endothelial biology and adhesion (Fig. 5c) revealed a notable lack of detection of colony-stimulating factor 1 receptor (Csf1r or M-csfr) in bMΦs (Fig. 5c). Animals deficient in Csf1r are largely macrophage deficient33,34. To test whether the lack of csf1r affected bMΦ production and/or function, we intercrossed csfr1a+/−csfr1b+/− zebrafish35 and found a reduction in tissue macrophage numbers but normal numbers and distribution of bMΦs in double mutants (csf1ra−/−b−/−) (Fig. 5d,e). These bMΦs were functional, phagocytosing intravascularly injected zymosan (Extended Data Fig. 5c) and responding to endothelial damage (Extended Data Fig. 5d and Supplementary Video 17). HSPC numbers colonizing the CHT in csf1r double mutants at 3 dpf were unchanged (Extended Data Fig. 5e), suggesting that bMΦs alone are sufficient for normal HSPC production in the AGM and expansion in the CHT. However, in the irf8 and spi1b morphants, where all macrophages are absent, HSPC numbers were similarly unaffected (Extended Data Fig. 5f). This probably reflects an accumulation of stressed HSPCs, which would typically be eliminated, coupled with a decrease in proliferating HSPCs36, both processes probably regulated by bMΦs, ultimately resulting in no net change in HSPC numbers at 2 and 3 dpf.
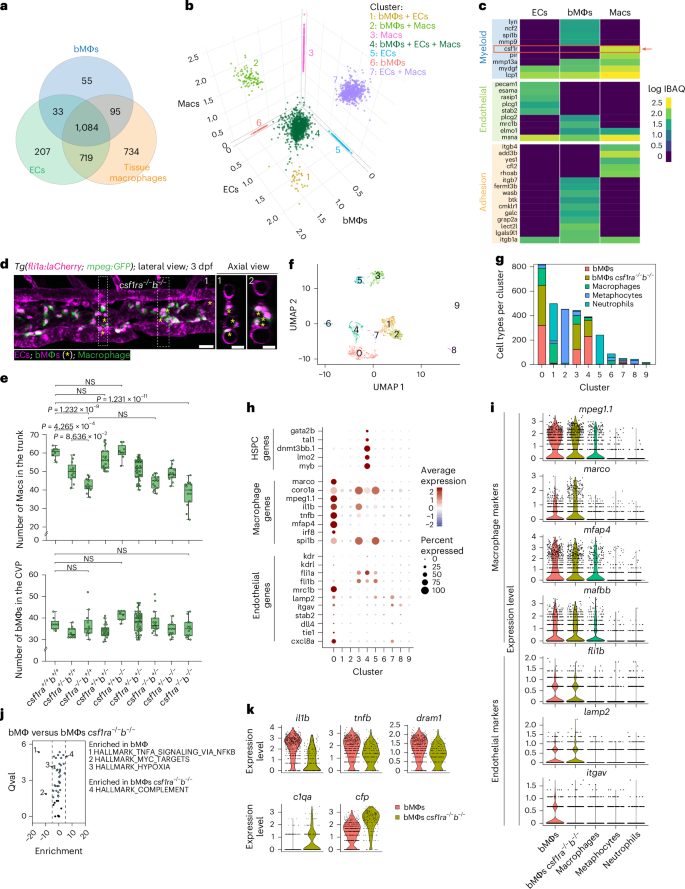
a, Venn diagram showing shared and distinct proteins per cell population. b, Three-dimensional plot of clustered proteins by K-means clustering. c, Heatmap of proteins involved in myeloid differentiation, EC biology or cell adhesion. d, Image of the CVP of 3-dpf Tg(fli1a:laCherry; mpeg:GFP) csf1ra−/−b−/− (double mutant). Asterisks depict bMΦs. e, Top: quantification of tissue macrophages (Macs) in the trunk region of the indicated offspring of an incross of Tg(fli1a:laCherry; mpeg:GFP) csf1ra+/−csf1rb+/− (referred to as csf1ra+/−b+/−) zebrafish. Bottom: quantification of bMΦs in the CVP of the offspring of an incross of Tg(fli1a:laCherry; mpeg:GFP) csf1ra+/−b+/− zebrafish. f, UMAP scatter plot after unsupervised clustering of scRNA-seq data. g, Distribution of the sorted cell types in the different clusters shown in f. h, Dot plot representing the average expression and percent of cells expressing a selection of HSPC, macrophage and endothelial genes, expressed in the different clusters shown in f. i, Violin plots of gene expression levels of mpeg1.1, marco, mfap4, mfapbb, fli1b, lamp2 and itgav, comparing bMΦs (WT), bMΦs (csf1ra−/−b−/−), tissue macrophages, metaphocytes and neutrophils. j, Hallmark pathway enrichment in bMΦs from WT or csf1ra−/−b−/− embryos. k, Violin plots of gene expression levels of il1b, tnfb, dram1, c1qa and cfp, comparing bMΦs from WT and csf1ra−/−b−/− embryos. Images are representative of n = 5 independent experiments (5 embryos) (d), and quantification is from n = 1 experiment (138 embryos) (e). Box plots show the median (center), first and third quartiles (bounds) and 1.5 times the interquartile range (whiskers) (e). Statistical analysis was performed using a Kruskal–Wallis test followed by Dunn’s multiple comparisons test versus WT (e). log IBAQ, log Intensity-Based Absolute Quantification.
Source data
In addition, we performed single-cell RNA sequencing (scRNA-seq) on bMΦs, tissue macrophages and neutrophils sorted from wild-type (WT) zebrafish, and on bMΦs and metaphocytes from csf1ra−/−b−/− mutants35. bMΦs and tissue macrophages clustered together (in clusters 0, 3 and 4), separately from neutrophils (clusters 1 and 5) and metaphocytes (cluster 2), supporting the notion that bMΦs are molecularly distinct from these lineages (Fig. 5f,g and Extended Data Fig. 6a,b). Clusters 0 and 3 were enriched for classical macrophage markers such as marco, coro1a, mpeg1.1, spi1b and/or mfap4 (Fig. 5h). Most bMΦs (80% from both WT and csf1ra−/−b−/− mutants) resided in cluster 0, which also included a small fraction (17%) of tissue macrophages (Fig. 5g). Consistently, bMΦs sorted from zymosan injected embryos (bMΦs zymosan+; cluster 10) clustered adjacent to cluster 0 and additionally expressed inflammatory/activation-associated genes (tnfa, il1b and dram1) (Extended Data Fig. 6c,d). Thus, cluster 0 is enriched for functional bMΦs, which exhibit high expression of macrophage markers as well as some endothelial markers (fli1b, mrc1b, lamp2, cxcl8a and itgav) (Fig. 5h). Because cluster 4 was characterized by the expression of dnmt3bb1, myb and gata2b37,38,39 —representing HSPCs/myeloid progenitors that are also marked by the fli1a:laCherry;mpeg:GFP transgenes (Fig. 5h)—we excluded cluster 4 from further analysis and show that bMΦs do express endothelial and macrophage markers (Fig. 5i). Aside from slightly lower expression of some pro-inflammatory markers (Fig. 5j,k), bMΦs from WT and csf1ra−/−b−/− mutants were transcriptionally nearly identical. This reinforces that bMΦs are generated and function independently of csf1ra and csf1rb, despite low-level transcript expression in some bMΦs (Extended Data Fig. 6e). Together, these data support our functional and imaging data that bMΦs represent a distinct subset of bona fide macrophages, characterized by the co-expression of macrophage and endothelial markers. Although molecularly similar to tissue macrophages, bMΦs are distinguished by their morphology, function and intravascular localization.
Using a similar approach as in zebrafish, we analyzed blood from adult and neonate mice 1 h after intravenous zymosan injection by multicolor flow cytometry. Zymosan particles were phagocytosed by cells with the following phenotype: CSF1R−/(low) CX3CR1⁻ CD11b⁺ Ly6Chigh Ly6G⁻ CD43⁺ CD31⁺ F4/80− (Extended Data Fig. 7a,b (adult blood); Extended Data Fig. 7c,d (neonatal blood); Extended Data Fig. 8a,b (controls)). This signature differs from known circulating myeloid populations, including patrolling/nonclassical monocytes (NCMs) (CSF1R⁺ CX3CR1high CD11b+ Ly6CLow/− CD43+ CD31⁻)11,40 (Extended Data Fig. 8c,d), classical monocytes (CSF1R⁺ CX3CR1low CD11b+ Ly6Chigh CD43low) and neutrophils (Ly6G⁺). NCMs were nearly depleted from the blood following zymosan injection (Extended Data Fig. 8e), whereas bMΦs remained in circulation (Extended Data Fig. 7a–d). In addition to sharing phagocytic capacity and phenotypic features (CSF1R− CD31+ (endothelial marker)) with zebrafish bMΦs, their mouse counterparts also displayed a similar morphology with pseudopod-like extensions (Extended Data Figs. 7d and 8f), strongly suggesting that bMΦs are evolutionarily conserved.
Here, we identify a specialized and conserved population of macrophages that reside and function within the vasculature of zebrafish and mice, termed bMΦs. These cells maintain blood homeostasis by catching, assessing and either releasing or phagocytosing circulating foreign particles and abnormal endogenous cells, a process recently dubbed ‘grooming’ and ‘dooming’36. bMΦs also act as primary responders to endothelial damage, clearing endothelial debris, whereas tissue macrophages and neutrophils contribute minimally. Whether bMΦs are required for blood vessel maintenance and function, analogous to perivascular macrophages in skin capillaries41 or microglia in the brain42,43, remains unknown and will require tissue-specific or conditional knockout strategies.
bMΦs persist beyond development into adulthood with similar phenotype and function. Whether these adult bMΦs have an embryonic origin and are maintained locally through self-renewal, or are produced de novo through EmacT, remains unresolved. Notably, bMΦ formation and function are preserved in csf1r-deficient embryos that lack nearly all other tissue macrophages35,44,45,46. Because Csf1r is broadly expressed across tissue macrophages, the Csf1r-EGFP mouse line (MacGreen) is widely used to detect and visualize macrophages47. The lack of Csf1r detection in bMΦs probably explains why they have remained unnoticed thus far.
Macrophages in zebrafish have three established origins: the rostral blood island (primitive macrophages), the posterior blood island (via EMPs) and the ventral endothelial wall of the DA (via HSPCs). We now introduce a fourth origin generating intravascular bMΦs dedicated to blood and vessel homeostasis. bMΦs arise directly through an unexpected EMacT, depending on irf8 and spi1b but not runx1. Ectopic spi1b expression forces some venous ECs to undergo EMacT, while others do not, supporting the heterogeneity of PCV and caudal vein ECs, which can adopt arterial, venous, lymphatic48 or, as shown here, myeloid fates.
We also identified bMΦ-like cells in neonatal and adult mouse blood, which are phenotypically distinct from circulating myeloid populations (patrolling monocytes/NCMs, classical monocytes and neutrophils). bMΦs also differ from macrophages derived from CX3CR1+CSF1R+ endothelial–macrophage progenitors, which do not circulate and are confined to the aortic adventitia49. Murine bMΦ-like cells exhibit phagocytic capacity and share phenotypic and morphological similarities with zebrafish bMΦs, suggesting that they represent true and evolutionarily conserved bMΦs. These findings challenge current views of blood and vascular system maintenance and might pave the way for using bMΦs or bioengineered versions to target blood-related diseases, as well as cardiovascular diseases involving endothelial damage.












Leave a Reply