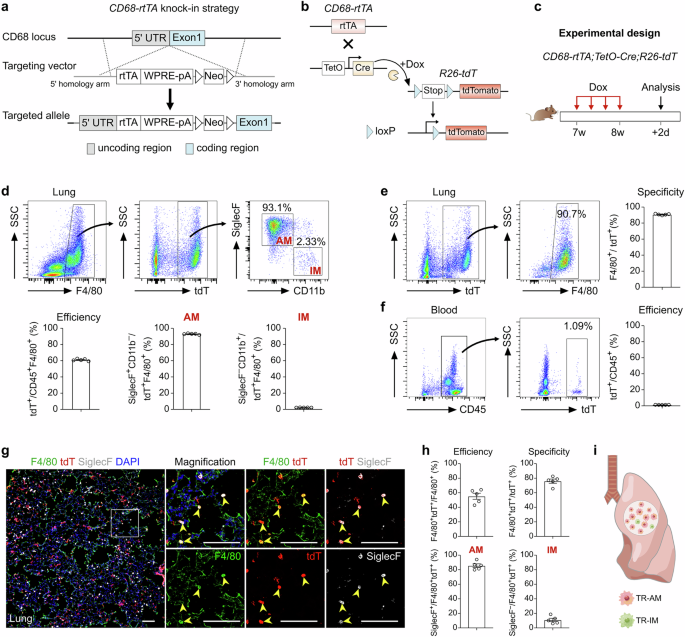
Generation of a CD68-rtTA mouse line for lineage tracing of TRMs
To overcome the limitations of previous approaches in distinguishing between TRMs and MDMs after lung injury8,29,30, we generated a CD68-rtTA mouse line for efficient lineage tracing of TRMs. Earlier studies relied on CD45.1/CD45.2 bone marrow chimeras, predominantly used in cell transplantation13,23, which cannot specifically target TRMs during lung homeostasis. To address this, we initially employed the CX3CR1-CreER system31,32 to label TRMs through tamoxifen (Tam) induction at the embryonic and neonatal stages. This method resulted in low labeling efficiency and poor survival rates, particularly when Tam was administered during embryogenesis (Supplementary Fig. S1a–f). Thus, we sought to improve TRM labeling by combining the tetracycline-on (Tet-On) and Cre-loxP systems to increase labeling efficiency in adult mice.
Analysis of single-cell RNA sequencing (scRNA-seq) data from lung tissue revealed high expression of CD68 across macrophage populations, with the majority of AMs exhibiting strong CD68 expression (Supplementary Fig. S2a). Based on this expression pattern, we generated CD68-rtTA mice, in which rtTA expression is controlled by the CD68 promoter in macrophages (Fig. 1a). In this system, TetO-Cre induces Cre recombinase expression under the control of a minimal promoter containing the tet operator (tetO). Upon doxycycline (Dox) administration, rtTA binds to TetO-Cre, driving Cre expression and subsequently activating a reporter gene in CD68+ macrophages (Fig. 1b). In the absence of Dox, rtTA does not bind to the tetO promoter, preventing the continuous or uncontrolled activation of Cre.
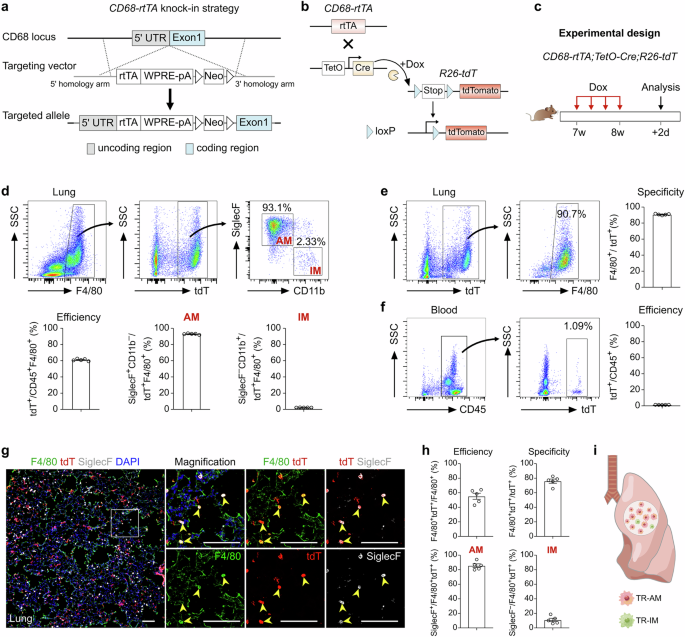
a Schematic diagram showing the knock-in strategy of the CD68-rtTA line by homologous recombination. b Genetic lineage tracing strategy by Cre-loxP recombination in CD68+ cells after doxycycline (Dox) treatment. c Schematic showing the experimental design. d Flow cytometric and quantification analyses of the percentage of tdT+ cells among CD45+F4/80+ macrophages (efficiency) and the percentage of SiglecF+ alveolar macrophages (AMs, left) and CD11b+ interstitial macrophages (IMs, right) among tdT+ macrophages from the lungs. Data are presented as the mean ± SEM; n = 5 mice per group. e Flow cytometric and quantification analyses of the percentage of tdT+ cells expressing F4/80 (specificity) in the lungs. Data are presented as the mean ± SEM; n = 5 mice per group. f Flow cytometric and quantification analyses of the percentage of CD45+ monocytes expressing tdT from blood. Data are presented as the mean ± SEM; n = 5 mice per group. g Immunostaining for F4/80, tdT, and SiglecF in lung tissue sections. The boxed region is magnified. Yellow arrowheads indicate labeled macrophages. h Quantitative analysis of the percentage of tdT+ cells among F4/80+ macrophages (efficiency), the percentage of F4/80+ cells among tdT+ cells (specificity), and the percentage of AMs and IMs among labeled tdT+ macrophages from images of immunostained lung sections. Data are presented as the mean ± SEM; n = 5 mice per group. i Cartoon image showing the effective labeling of tissue-resident macrophages (TRMs) in CD68-rtTA;TetO-Cre;R26-tdT mice. Scale bars, 100 µm. Each image is representative of five individual samples.
To validate this system, we crossed CD68-rtTA, TetO-Cre, and R26-tdT mice to generate a triple-positive mouse line and analyzed macrophage labeling after 1 week of Dox treatment in adult mice (Fig. 1c). FACS analysis revealed that more than 60% of lung macrophages expressing F4/80 were labeled with tdT, while nearly all CD45+ immune cells from blood remained tdT-negative (Fig. 1d, f). Furthermore, 90.75 ± 0.35% of the tdT+ cells in the lungs expressed F4/80, confirming the high specificity of the CD68-rtTA;TetO-Cre;R26-tdT system for labeling macrophages (Fig. 1e). Among the labeled tdT+ macrophages, the majority were AMs (92.94 ± 0.33%), while IMs were minimally labeled (2.31 ± 0.05%, Fig. 1d). Notably, more than 70% of all AMs were tdT-positive, compared with only approximately 7.58% of IMs, which is consistent with the known predominance of AMs within homeostatic TRM populations (Supplementary Fig. S2b). Immunostaining of lung sections for tdT, F4/80, and SiglecF further demonstrated greater labeling efficiency and specificity in CD68-rtTA;TetO-Cre;R26-tdT mice than in CX3CR1-CreER;R26-tdT mice (Fig. 1g, h). In addition, by employing CD64 and MerTK as specific markers to accurately identify TRMs in the lung, FACS analysis demonstrated that more than 40% of the TRMs were tdT+, with the vast majority being AMs (96.07 ± 1.54%), whereas IMs showed minimal labeling (3.79 ± 1.12%). Furthermore, 95.01 ± 2.02% of all the tdT+ cells coexpressed CD64 and MerTK, confirming the high specificity of this system for macrophage labeling (Supplementary Fig. S2c, d). Immunostaining further confirmed the specificity of the system, demonstrating that lung tdT+ cells coexpressed CD68 and MerTK and were positive for the AM marker SiglecF, while no detectable tdT or CD68 signal was detected in blood Ly6C+ monocytes after Dox treatment (Supplementary Fig. S2e). Importantly, negligible tdT signals were detected in CD68-rtTA;TetO-Cre;R26-tdT mice without Dox treatment (Supplementary Fig. S3). Whole-mount fluorescence and immunofluorescence imaging further confirmed that sporadic tdT+ cells were not F4/80+ macrophages (Supplementary Fig. S3b, c). Additionally, FACS analysis revealed that fewer than 0.01% of F4/80+ macrophages and Ly6C+ monocytes expressed tdT in the absence of Dox (Supplementary Fig. S3d). In summary, the CD68-rtTA;TetO-Cre;R26-tdT mouse line offers a highly specific and efficient tool for the genetic targeting and lineage tracing of endogenous TRMs in the lungs (Fig. 1i). This model provides a valuable resource for studying the roles of TRMs in pulmonary homeostasis and disease.
TRMs decrease significantly after injury and are maintained at normal levels during recovery
To investigate the role and dynamics of TRMs in lung injury and repair, we induced acute lung inflammation and fibrosis through bleomycin induction. In this model, the levels of proinflammatory cytokines rapidly increase during the first 3 days, peak at approximately day 7, and remain elevated until day 10, followed by the fibrotic phase of lung recovery28. To assess the behavior of TRMs during these stages, we treated CD68-rtTA;TetO-Cre;R26-tdT mice with Dox to label TRMs, followed by a 3-week Dox washout period before administering bleomycin. The lungs were then harvested at different time points: 3, 7, 14, and 28 days postinjury (Fig. 2a). Whole-mount fluorescence images of the lungs of CD68-rtTA;TetO-Cre;R26-tdT mice revealed a significant decrease in the number of tdT-labeled TRMs over time after injury (Fig. 2b). Sirius red staining of lung sections revealed a sharp increase in fibrosis starting on day 7 postinjury, with the severity of fibrosis worsening at days 14 and 28 compared with that of the sham control (Fig. 2b).
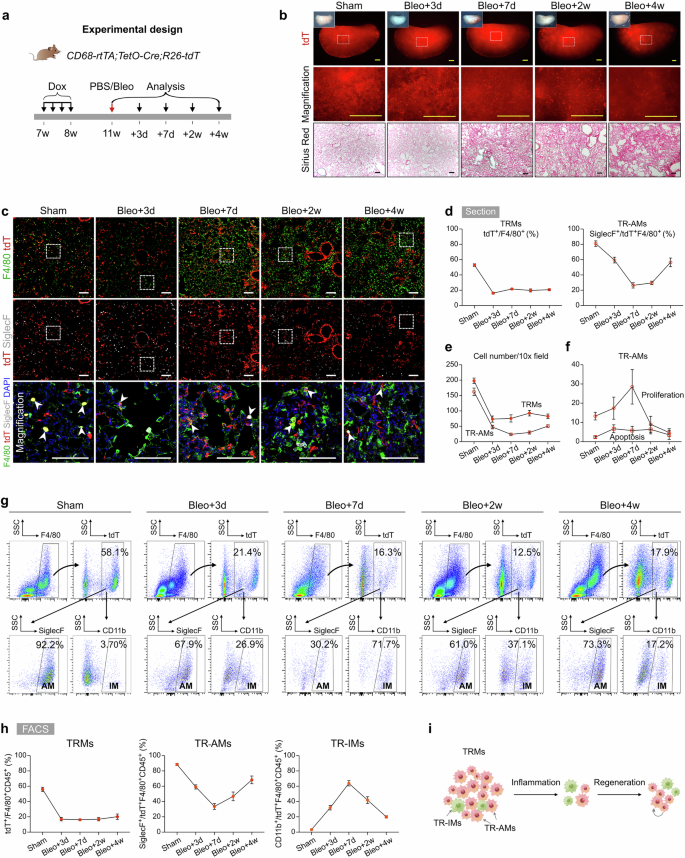
a Schematic diagram showing the experimental design. Dox, doxycycline. Bleo, bleomycin. b Whole-mount bright field, epifluorescence, and Sirius red images of lungs after bleomycin treatment. The boxed region is magnified. c Immunostaining for tdT, F4/80, and SiglecF in lung tissue sections. The boxed region is magnified. White arrowheads indicate TRMs. d Quantitative analysis of the percentage of F4/80+ macrophages expressing tdT (left) and the percentage of SiglecF+ alveolar macrophages among labeled tdT+ macrophages (right) in immunostained lung sections. Data are presented as the mean ± SEM; n = 5 mice per group. e Quantification of the number of TRMs (tdT+F4/80+) and TR-AMs (tdT+SiglecF+) in each 10× field from immunostained lung sections. Data are presented as the mean ± SEM; n = 5 mice per group. f Quantification of the percentage of Ki67+ (proliferative) and γ-H2AX+ (apoptotic) cells among labeled TR-AMs (tdT+SiglecF+) in each 10× field from immunostained lung sections. Data are presented as the mean ± SEM; n = 5 mice per group. g, h Flow cytometric analysis and quantification of the percentage of tdT+ cells among CD45+F4/80+ macrophages (TRMs), the percentage of SiglecF+ alveolar macrophages among tdT+ macrophages (TR-AMs), and the percentage of CD11b+ interstitial macrophages among tdT+ macrophages (TR-IMs) from the lungs. Data are presented as the mean ± SEM; n = 5 mice per group. i Cartoon image showing the dynamic changes in TRMs in the lungs after bleomycin-induced injury. TRMs tissue-resident macrophages, TR-AMs tissue-resident alveolar macrophages, TR-IMs tissue-resident interstitial macrophages. Scale bars, yellow, 1 mm; white and black, 100 µm. Each image is representative of five individual samples.
Next, we performed immunofluorescence staining of lung sections for tdT, F4/80, and SiglecF and found that the percentage of TRMs among total macrophages was significantly decreased by day 3 postinjury and remained relatively constant throughout the later stages of injury and recovery (Fig. 2c, d; Supplementary Fig. S4). Concurrently, we observed a marked increase in the number of tdT–F4/80+ macrophages from day 7 to day 28, suggesting infiltration of macrophages from other sources, particularly during the fibrotic phase (Fig. 2c; Supplementary Fig. S4). The percentage of TR-AMs among the tdT+ TRMs initially decreased on days 3 and 7 but then gradually increased to 56.44 ± 5.71% by day 28 (Fig. 2d). During the fibrotic phase, especially on day 28, there was a notable increase in the number of TR-AMs compared with that in the earlier inflammatory phase, whereas the overall number of TRMs remained stable (Fig. 2e). The proliferation of TR-AMs peaked on day 7 and returned to baseline levels by day 14, with minimal changes in apoptosis over time (Fig. 2f; Supplementary Fig. S5), suggesting that TR-AM self-renewal partially contributed to their increased numbers during the fibrotic phase.
We next performed a FACS analysis of lung macrophages and found that the percentage of tdT+ TRMs sharply decreased by day 3 but remained largely unchanged thereafter (Fig. 2g, h). Among the labeled TRMs, the percentage of TR-AMs decreased to its lowest point (33.58 ± 3.80%) during the early injury phase and then increased to 68.33 ± 5.08% by day 28, although this value remained below the steady-state level (Fig. 2g, h). In contrast, the proportion of tissue-resident IMs (TR-IMs) peaked on day 7 and subsequently decreased to 19.93 ± 1.50% on day 28, which was still higher than the steady-state level (Fig. 2g, h). Quantification of apoptosis in TRMs (including both TR-AMs and TR-IMs) revealed no significant differences from day 7 to day 28, further indicating that TRM numbers remained relatively constant during this period (Fig. 2f; Supplementary Fig. S5d–f). These findings were corroborated in CX3CR1-CreER;R26-tdT mice, in which a similar pattern of TRM reduction at days 3 and 7 followed by a stage of stability thereafter was observed (Supplementary Fig. S1g–l). Taken together, these results highlight that TRMs undergo significant depletion during the acute inflammatory phase of lung injury but remain stable throughout the repair and fibrotic phases, with TR-AM self-renewal contributing to the replenishment of the TRM population in the lungs (Fig. 2i).
Infiltrating macrophages accumulate during pulmonary fibrosis
To investigate the role of monocytes and MDMs in lung inflammation and fibrosis, we utilized the Ms4a3-CreER mouse model33, which allows the specific labeling and tracing of monocytes. Ms4a3-CreER mice were crossed with R26-tdT reporter mice, and following Tam administration, we examined the monocytes labeling efficiency and specificity (Supplementary Fig. S6a, b). There was no detectable leakiness in the labeling of monocytes in the lungs, blood, and BM without Tam treatment (Supplementary Fig. S6c, d, g, i). Additionally, immunostaining of blood samples for CD68, tdT, and Ly6C revealed that nearly all Ly6C+ monocytes were tdT-positive but CD68-negative (Supplementary Fig. S6e). FACS analysis of blood and BM further confirmed the high efficiency and specificity of targeting monocytes in this mouse line (Supplementary Fig. S6f). During homeostasis, a small fraction of lung macrophages (2.63 ± 0.38%) originated from circulating monocytes, demonstrating the specificity of Ms4a3-CreER for targeting monocytes and MDMs (Supplementary Fig. S6h). Thus, the Ms4a3-CreER mouse line is an effective tool for investigating MDMs in the lungs (Supplementary Fig. S6j).
We collected lung samples from Tam-treated Ms4a3-CreER;R26-tdT mice on days 3, 7, 14, and 28 following bleomycin-induced lung injury to track their infiltration and fate during inflammation and fibrosis (Fig. 3a). Whole-mount fluorescence images revealed a low baseline presence of tdT+ cells in healthy lungs but significantly increased tdT+ signals in injured lungs, which correlated with the extent of tissue damage (Fig. 3b). Sirius red staining of lung sections revealed marked fibrosis, especially at 7 and 14 days postinjury (Fig. 3b). Immunostaining for tdT, F4/80, and SiglecF revealed that tdT+ cells were sparsely distributed in the sham group and were mainly Mo-IMs (Fig. 3c, d). During the early stage of lung inflammation, a large influx of tdT+ monocytes was observed at injury sites, where these cells began to differentiate into macrophages. By day 7 postinjury, a significant number of tdT+ cells accumulated in fibrotic areas, and many of these cells differentiated into macrophages, with a small subset becoming monocyte-derived AMs (Mo-AMs), marked by tdT+SiglecF+F4/80+, and the majority becoming monocyte-derived IMs (Mo-IMs), marked by tdT+SiglecF–F4/80+ (Fig. 3c, e).
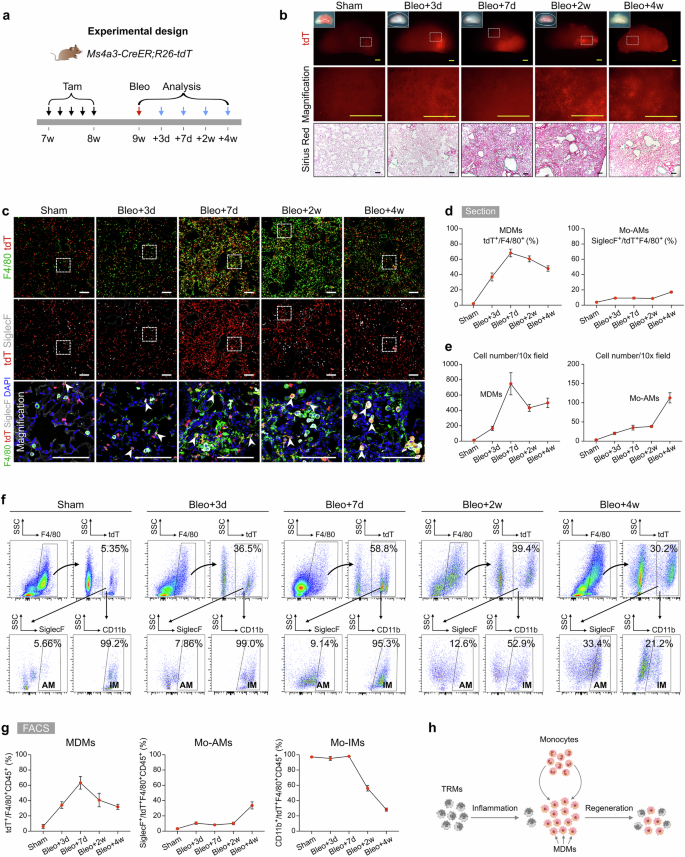
a Schematic diagram showing the experimental design. Tam tamoxifen, Bleo bleomycin. b Whole-mount bright field, epifluorescence, and Sirius red images of lungs after bleomycin treatment. The boxed region is magnified. c Immunostaining for tdT, F4/80, and SiglecF in lung tissue sections. The boxed region is magnified. White arrowheads indicate MDMs. d Quantitative analysis of the percentage of F4/80+ macrophages expressing tdT (left) and the percentage of alveolar macrophages among labeled tdT+ macrophages (right) in immunostained lung sections. Data are presented as the mean ± SEM; n = 5 mice per group. e Quantification of the numbers of MDMs (tdT+F4/80+) and Mo-AMs (tdT+SiglecF+) in each 10× field from immunostained lung sections. Data are presented as the mean ± SEM; n = 5 mice per group. f, g Flow cytometric and quantification analyses of the percentage of tdT+ cells among CD45+F4/80+ macrophages (MDMs), the percentage of SiglecF+ alveolar macrophages among tdT+ macrophages (Mo-AMs), and the percentage of CD11b+ interstitial macrophages among tdT+ macrophages (Mo-IMs) from the lungs. Data are presented as the mean ± SEM; n = 5 mice per group. h Cartoon image showing the dynamic changes in the number of recruited macrophages in the lungs after bleomycin-induced injury. MDMs monocyte-derived macrophages, Mo-AMs monocyte-derived alveolar macrophages, Mo-IMs monocyte-derived interstitial macrophages. Scale bars, yellow, 1 mm; white and black, 100 µm. Each image is representative of five individual samples.
As the injury progressed, particularly on day 14, the number of MDMs decreased, whereas the proportion of Mo-AMs in the injury areas increased. By day 28 postinjury, almost half of the macrophages in the lungs were MDMs, with a notable increase in the number of Mo-AMs (Fig. 3c–e). FACS analysis further supported these findings, as the number of MDMs increased during the inflammatory phase but decreased during the lung regeneration phase (Fig. 3f, g). Specifically, the percentage of Mo-IMs sharply decreased from 97.96 ± 0.33% at the peak of inflammation to 28.23 ± 2.04% during recovery, whereas the proportion of Mo-AMs significantly increased, suggesting that some Mo-IMs may convert into Mo-AMs as the lung heals (Fig. 3f, g). Overall, these results reveal that circulating monocytes rapidly infiltrate sites of lung injury, where they primarily differentiate into interstitial macrophages during the inflammatory phase and contribute to alveolar macrophages during the fibrotic phase. This process highlights the dynamic roles of MDMs in both lung inflammation and repair, with the potential conversion of Mo-IMs to Mo-AMs as fibrosis progresses and lung regeneration occurs (Fig. 3h).
Macrophage heterogeneity during lung injury and repair
To comprehensively understand the macrophage dynamics in bleomycin-injured lungs, we performed a time series of scRNA-seq analyses. Immune cells were isolated from PBS-treated and bleomycin-injured lungs at 3, 7, 14, and 28 days posttreatment (Fig. 4a, b). A total of 40,006 cells (sham, 8416; bleomycin group: day 3, 7646; day 7, 7956; day 14, 6762; day 28, 9226) passed quality control and were categorized into 11 clusters based on the expression of representative marker genes (Supplementary Fig. S7a). The major immune cell clusters included macrophages (Itgax), monocytes (Csf1r, Itgam), neutrophils (Clec4e), T cells (Il7r), B cells (Cd79a), dendritic cells (Bst2), and NK cells (Klrb1c) (Supplementary Fig. S7b). To further explore the dynamic changes within each cluster, we examined single-cell transcriptional profiles at individual time points (Supplementary Fig. S7c). Among the immune cell clusters, macrophages accounted for the greatest proportion, and their transcriptional profiles changed significantly during the course of lung injury and repair (Supplementary Fig. S7d, e). The proportion of macrophages decreased after injury but recovered during the fibrotic stage, which was consistent with the lineage tracing results (Fig. 2). Additionally, the percentage of monocytes increased markedly during the early inflammatory phase but gradually decreased during lung repair (Supplementary Fig. S7e).
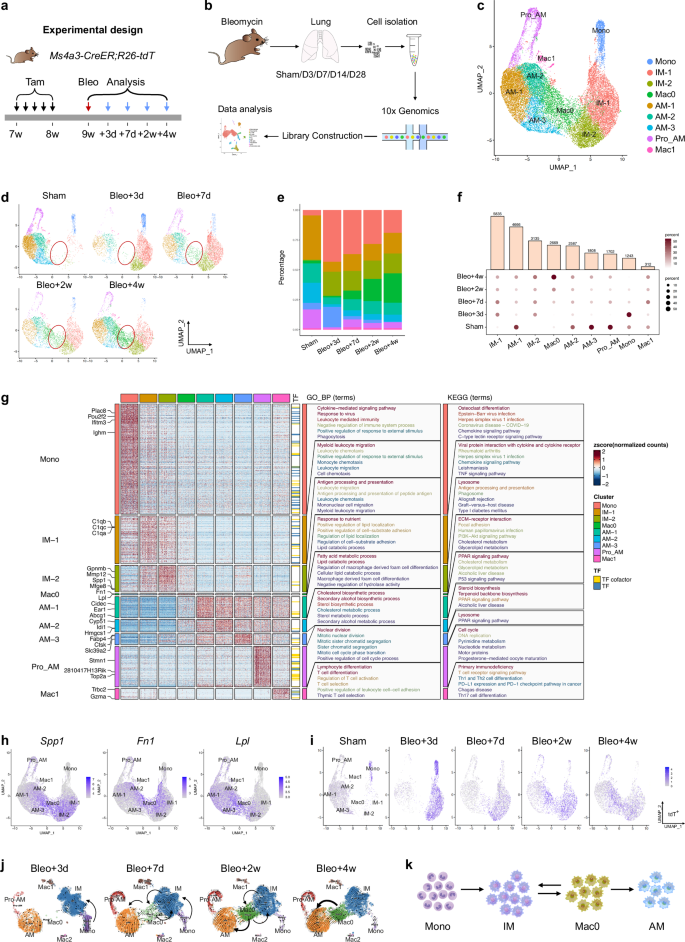
a Schematic diagram showing the experimental design. Tam tamoxifen, Bleo bleomycin. b Flow chart showing the experimental process. c UMAP visualization of macrophage-related cell populations in the mouse lung. d UMAP plots indicating the appearance of Mac0 cells on day 7 postinjury. e Bar plot representation of the relative frequency of different macrophage subclusters at different time points. f Dot plot showing the percentage of each macrophage-related cluster contributing at different time points. The total percentage of one cluster is 100%. g Heatmap showing the results of the GO and KEGG pathway analyses of macrophage-related cell populations. h FeaturePlot showing the highly expressed genes characteristic of the Mac0 cluster. i UMAP projection of labeled monocytes (tdT+) visualized separately at different time points. j UMAP colored by identified clusters with trajectories inferred from RNA velocity. The black arrows represent the calculated velocity trajectories. The Mac2 cluster resulted from a separate reclustering based on spliced and unspliced RNA counts required for RNA velocity analysis, which differed from the primary analysis using total gene expression. k Cartoon image showing the cell fate transition from monocytes to alveolar macrophages. Mono monocytes, IMs interstitial macrophages, AMs alveolar macrophages.
To gain a more detailed understanding of the transition from homeostasis to the postbleomycin response in macrophages, we aggregated and subclustered macrophages and monocytes from both steady-state and injured samples. Nine subclusters were identified, which could be classified into three macrophage subtypes: AM, IM, and a novel population termed Mac0 (Fig. 4c). In steady-state lungs, the majority of the macrophages were AMs, with low proportions of IMs and a negligible Mac0 population. By day 3 postinjury, a large influx of circulating monocytes infiltrated the lungs, leading to a significant increase in IMs and a decrease in AMs. Simultaneously, the Mac0 cluster began to emerge (Fig. 4d). One week later, Mac0 cells continued to expand and became increasingly involved in lung repair during the fibrotic phase. By day 28 postinjury, the percentage of AMs had partially recovered, while the percentage of IMs had decreased. The number of monocytes remained low during the lung recovery stage (Fig. 4d–f).
Further bioinformatic analysis revealed that the Mac0 cluster exhibited a unique transcriptional profile marked by the upregulation of lysosomal and antigen presentation genes (e.g., Ctsb, H2-Aa, and Cd74), along with the upregulation of genes related to lipid metabolism (e.g., Lpl and Cyp51) and extracellular matrix interaction (e.g., Fn1 and Cd44). Compared with AMs, Mac0 cells showed reduced alveolar identity but increased injury-responsive functions; compared with monocytes, Mac0 cells presented downregulated expression of inflammatory and migratory genes, indicating a functional transition toward tissue repair. These features positioned Mac0 cells as a transitional, injury-specific subset that supported debris clearance, immunomodulation, and matrix remodeling during lung injury (Fig. 4g; Supplementary Fig. S8a). FeaturePlot and Violin plot analyses revealed that cells within the Mac0 cluster highly expressed the genes Spp1, Fn1, and Lpl (Fig. 4h; Supplementary Fig. S8b).
To better understand the relationships among monocytes, IMs, Mac0 cells, and AMs during lung injury and repair, we performed an RNA velocity analysis on the genetically labeled tdT+ cells from Ms4a3-CreER;R26-tdT mice (Fig. 4i). This analysis revealed that a subset of tdT+ monocytes differentiated into IMs by day 3 postinjury, followed by a clear developmental progression from IMs to Mac0 cells by day 7. Two weeks after injury, labeled tdT+ cells were distributed among the IM, Mac0, and AM cell clusters. Four weeks later, the tdT+ cells were more concentrated in the AM cluster. These findings suggest that a subset of newly generated AMs arises from IMs via Mac0 cells. To investigate and model this potential cell trajectory, we analyzed the UMAP projection of monocytes and macrophages at various time points postinjury, which revealed a differentiation pathway from monocytes to IMs, then to Mac0 cells and finally toward AMs (Fig. 4j). By day 7 postinjury, Mac0 cells contributed to both the IM and AM populations, but by week 4, Mac0 cells contributed only to AMs.
Pathway overrepresentation analysis using the Kyoto Encyclopedia of Genes and Genomes (KEGG) database indicated that Notch and Wnt/β-catenin signaling were highly enriched in the Mac0 cluster, which is consistent with their activity in AMs (Supplementary Fig. S8c). In terms of transcription, compared with both AMs and IMs, Mac0 cells represented a unique transitional state with a moderate inflammatory signature (Supplementary Fig. S8d–f). Mac0 cells displayed low CD11b (Itgam), intermediate SiglecF, and uniform CD11c (Itgax) expression, distinguishing them from previously described populations (Supplementary Fig. S8g). Moreover, comparative transcriptomic profiling revealed that the IM1 subcluster exhibited a pronounced proinflammatory and chemotactic phenotype compared with that of the IM2 cluster (Supplementary Fig. S8h).
In summary, we utilized scRNA-seq to characterize the heterogeneity of macrophages after bleomycin-induced lung injury over time and identified a novel transitional macrophage subcluster, Mac0 cells, which originate from monocytes and differentiate into AMs (Fig. 4k).
Genetic ablation of infiltrating macrophages alleviates pulmonary fibrosis
Monocyte-derived macrophages (MDMs) have been shown to drive fibrogenesis in various organs, partly through their interactions with fibroblasts7,28,34. To specifically investigate the reparative role of MDMs at different stages following lung injury, we crossed Ms4a3-CreER;R26-tdT mice with the inducible diphtheria toxin receptor (DTR) mouse line R26-iDTR, allowing for the ablation of monocytes via diphtheria toxin (DT) treatment (Fig. 5a). First, we treated the Ms4a3-CreER;R26-tdT/iDTR and Ms4a3-CreER;R26-tdT control mice with Tam to efficiently label monocytes with tdT, followed by DT injection, followed by the induction of bleomycin injury (Fig. 5b). FACS analysis revealed a significant reduction in the number of circulating tdT+ monocytes in Ms4a3-CreER;R26-tdT/iDTR mice, indicating that the majority of tdT+ monocytes were successfully ablated (Fig. 5c). Whole-mount fluorescence imaging of the lungs of these mice revealed reduced tdT+ signals in fibrotic regions (Supplementary Fig. S9a). Additionally, Sirius red staining of lung sections revealed a reduced fibrotic response in the monocyte-depleted groups compared with the control groups (Supplementary Fig. S9b).
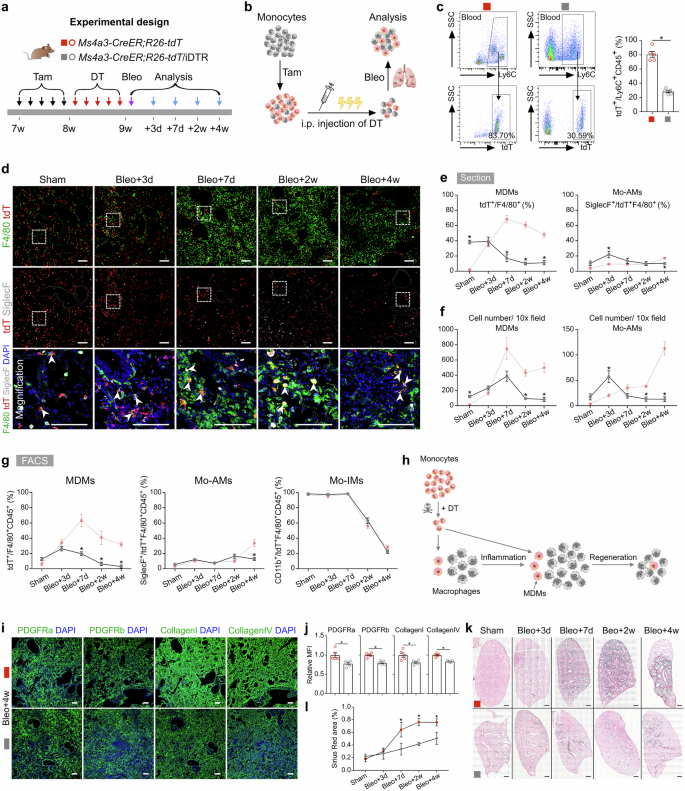
a Schematic diagram showing the experimental design. Tam, tamoxifen; DT, diphtheria toxin; Bleo, bleomycin. b Schematic diagram illustrating the working process. c Flow cytometric and quantification analysis of the percentage of tdT+ cells among CD45+Ly6C+ monocytes from blood. Data are presented as the mean ± SEM; n = 5 mice per group. d Immunostaining for tdT, F4/80 and SiglecF in tissue sections of Ms4a3-CreER;R26-tdT/iDTR lungs. The boxed region is magnified. White arrowheads indicate MDMs. e Quantitative analysis of the percentage of F4/80+ macrophages expressing tdT (MDMs) and the percentage of Mo-AMs in labeled tdT+ macrophages from immunostained lung sections of Ms4a3-CreER;R26-tdT/iDTR mice. The results of the control experiment were the same as those in Fig. 3d. Data are presented as the mean ± SEM; n = 5 mice per group. f Quantification of the numbers of MDMs (tdT+F4/80+) and Mo-AMs (tdT+SiglecF+) in each 10× field from immunostained lung sections of Ms4a3-CreER;R26-tdT/iDTR mice. The results of the control experiment were the same as those in Fig. 3e. Data are presented as the mean ± SEM; n = 5 mice per group. g Flow cytometric and quantification analyses of the percentage of tdT+ cells among CD45+F4/80+ macrophages (MDMs), the percentage of SiglecF+ alveolar macrophages among tdT+ macrophages (Mo-AMs), and the percentage of CD11b+ interstitial macrophages among tdT+ macrophages (Mo-IMs) from the lungs. The results of the control experiment were the same as those in Fig. 3g. Data are presented as the mean ± SEM; n = 5 mice per group. h Cartoon image showing the dynamic changes in the number of recruited macrophages in Ms4a3-CreER;R26-tdT/iDTR lungs after treatment with bleomycin. MDMs, monocyte-derived macrophages; Mo-AMs, monocyte-derived alveolar macrophages; Mo-IMs, monocyte-derived interstitial macrophages. i Immunostaining for PDGFRa, PDGFRb, collagen I, and collagen IV in lung tissue sections. j Quantitative analysis of the relative mean fluorescence intensity (MFI) of PDGFRa, PDGFRb, collagen I, and collagen IV per 10× field of lung sections. The data are presented as the mean ± SEM; n = 5 mice per group. k, l Sirius red images and quantification of pulmonary fibrosis after bleomycin treatment. Data are presented as the mean ± SEM; n = 5 mice per group. Scale bars, white, 100 µm; black, 1 mm. Each image is representative of five individual samples.
Immunostaining for tdT, F4/80, and SiglecF in lung sections revealed a markedly greater number of tdT+ macrophages at homeostasis in Ms4a3-CreER;R26-tdT/iDTR mice than in Ms4a3-CreER;R26-tdT controls after DT treatment (Fig. 5d). Quantitative analysis further revealed a significantly increased proportion of MDMs in monocyte-ablated mice without injury, indicating that monocyte depletion during homeostasis triggered a strong immune response (Fig. 5e, g). Following bleomycin injury, the decrease in the number of circulating monocytes led to a significant decrease in the number of MDMs, particularly Mo-AMs, on day 28 (Fig. 5e–g). By day 7 postinjury, only 19.75 ± 2.57% of the MDMs were present at the injury site, whereas this percentage was 63.29 ± 8.25% in the control lungs (Fig. 5e compared with Fig. 3d). During the fibrotic phase, very few MDMs were detected in the injured area (Fig. 5e, g). Interestingly, the ablation of monocytes had little effect on the percentage of Mo-IMs throughout the injury and recovery process (Fig. 5g). Overall, these results demonstrated that monocytes and MDMs could be specifically and effectively depleted in Ms4a3-CreER;R26-tdT/iDTR mice during the inflammatory and fibrotic phases (Fig. 5h).
Next, we explored the role of monocytes in pulmonary inflammation and fibrosis. Quantitative real-time PCR (qRT-PCR) revealed that the deletion of circulating monocytes led to a reduction in the expression of anti-inflammatory genes such as Arg1 and Chil3, while the expression of the proinflammatory genes Il6 and Il1β was significantly upregulated, indicating that monocyte ablation aggravated inflammation in the early stage after injury (Supplementary Fig. S9c). Furthermore, the expression of profibrotic and fibroblast markers, including Fn1, Col3a1, Pdgfra, and Pdgfrb, was markedly reduced in the monocyte-depleted groups during the recovery phase, suggesting that bleomycin-induced fibrosis was more severe in control lungs (Supplementary Fig. S9c). Immunostaining for PDGFRa, PDGFRb, collagen I, and collagen IV in lung sections revealed reduced protein expression levels in monocyte-depleted mice after bleomycin treatment (Fig. 5i, j). Histological studies further confirmed that monocyte ablation significantly reduced the extent of pulmonary fibrosis during the progressive fibrotic phase (Fig. 5k, l).
Notably, neutrophils were also affected by DT treatment in Ms4a3-CreER;R26-tdT/iDTR mice. However, in our model, neutrophils, which were primarily associated with acute inflammation rather than fibrosis, exhibited early infiltration that peaked within the first week after injury but decreased sharply during the fibrotic phase (Supplementary Fig. S9d). Temporal analysis revealed no significant difference in lung neutrophil levels during key inflammatory and fibrotic stages following depletion. These kinetic patterns suggested that the reduced fibrosis was more likely due to the depletion of MDMs and Mo-AMs than to the loss of neutrophils.
Taken together, these results suggest that the depletion of circulating monocytes affects lung macrophage populations in both physiological and pathological states, reducing fibroblast accumulation and alleviating fibrosis following lung injury.
Monocyte-specific Notch knockout attenuates pulmonary fibrosis
RBP-J-mediated Notch signaling has been demonstrated to be a crucial regulator of monocyte differentiation and function. RBP-J, known as the master nuclear mediator of canonical Notch signaling35, also plays a pivotal role in monocyte cell fate decisions36,37,38. Our scRNA-seq analysis indicated that Notch signaling was markedly activated in the AM and Mac0 clusters during lung repair and regeneration (Fig. 6a; Supplementary Fig. S8c). We generated monocyte-specific Rbpj knockout mice by crossing Rbpjflox/flox mice with Ms4a3-CreER;R26-tdT mice (Fig. 6b). Bleomycin injury was induced 1 week after Tam treatment (Fig. 6b). qRT-PCR analysis confirmed the efficient knockout of Rbpj in sorted tdT+ cells from both blood and lungs (Fig. 6c). FACS analysis revealed that the conditional knockout of Rbpj in monocytes resulted in a reduction in the number of circulating monocytes (Fig. 6d). Immunostaining for tdT, F4/80, and SiglecF in lung sections revealed that on day 3 postinjury, Rbpj deficiency resulted in an increase in the number of MDMs, which subsequently decreased by day 7 (Fig. 6e–g). Although the number of Mo-AMs was significantly increased on day 3 after injury, it sharply decreased thereafter (Fig. 6g). This reduction in the number of Mo-AMs during the fibrotic phase suggested defective differentiation of Mo-IMs into Mo-AMs (Fig. 6f, g). FACS analysis further confirmed that in the absence of injury, the percentage of MDMs increased to 18.55 ± 1.06% in Rbpj knockout mice compared with 6.18 ± 2.64% in wild-type mice (Fig. 6h). Additionally, the elevated proportion of Mo-IMs on day 28 in Rbpj knockout mice suggested that Notch signaling may promote the differentiation of Mo-IMs into Mo-AMs (Fig. 6h).
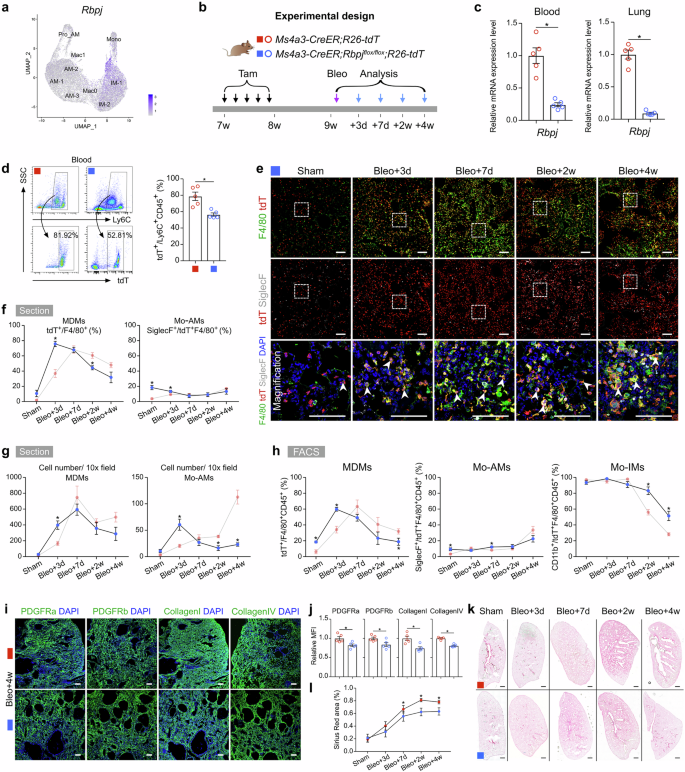
a Feature plots illustrating Rbpj expression in macrophage-related clusters. b Schematic diagram showing the experimental design. Tam tamoxifen, Bleo bleomycin. c qRT-PCR analysis showing the knockout efficiency of Rbpj in tdT+ cells from the blood and lungs. Data are presented as the mean ± SEM; n = 5 mice per group. d Flow cytometric and quantification analysis of the percentage of tdT+ cells among CD45+Ly6C+ monocytes from blood. Data are presented as the mean ± SEM; n = 5 mice per group. e Immunostaining for tdT, F4/80 and SiglecF in tissue sections of Ms4a3-CreER;Rbpjflox/flox;R26-tdT lungs. The boxed region is magnified. White arrowheads indicate MDMs. f Quantitative analysis of the percentage of F4/80+ macrophages expressing tdT (MDMs) and the percentage of Mo-AMs in labeled tdT+ macrophages from immunostained lung sections from Ms4a3-CreER;Rbpjflox/flox;R26-tdT mice. The results of the control experiment were the same as those in Fig. 3d. Data are presented as the mean ± SEM; n = 5 mice per group. g Quantification of the numbers of MDMs (tdT+F4/80+) and Mo-AMs (tdT+SiglecF+) in each 10× field of immunostained lung sections from Ms4a3-CreER;Rbpjflox/flox;R26-tdT mice. The results of the control experiment were the same as those in Fig. 3e. Data are presented as the mean ± SEM; n = 5 mice per group. h Flow cytometric and quantification analyses of the percentage of tdT+ cells among CD45+F4/80+ macrophages (MDMs), the percentage of SiglecF+ alveolar macrophages among tdT+ macrophages (Mo-AMs), and the percentage of CD11b+ interstitial macrophages among tdT+ macrophages (Mo-IMs) from the lungs. The results of the control experiment were the same as those in Fig. 3g. Data are presented as the mean ± SEM; n = 5 mice per group. i Immunostaining for PDGFRa, PDGFRb, collagen I, and collagen IV in lung tissue sections. j Quantitative analysis of the relative mean fluorescence intensity (MFI) of PDGFRa, PDGFRb, collagen I, and collagen IV per 10× field of lung sections. Data are presented as the mean ± SEM; n = 5 mice per group. k, l Sirius red images and quantification of pulmonary fibrosis after bleomycin treatment. Data are presented as the mean ± SEM; n = 5 mice per group. Scale bars, white, 100 µm; black, 1 mm. Each image is representative of five individual samples.
The expression of signature genes associated with the M2 phenotype, such as Arg1 and Chil3, was significantly upregulated in Rbpj knockout mice. In contrast, the gene expression of proinflammatory cytokines and chemokines, including Il6 and Il1β, was unchanged or downregulated (Supplementary Fig. S10). Immunostaining revealed reduced expression of PDGFRa, PDGFRb, collagen I, and collagen IV and a decreased degree of fibrosis, as assessed by Sirius red staining, in the lungs of Rbpj knockout mice following bleomycin treatment (Fig. 6i–l). qRT-PCR analysis further validated the reduced mRNA levels of profibrotic and fibrosis-related genes, including Fn1, Pdgfra, and Pdgfrb, in Rbpj knockout mice after injury (Supplementary Fig. S10). To determine whether the conditional knockout of Rbpj in monocytes affects the function of Mo-AMs and Mo-IMs, we isolated tdT+ Mo-AMs and Mo-IMs from the lung tissues of knockout mice and assessed their phagocytic capacity and polarization status (Supplementary Fig. S11). In vitro functional analysis revealed significantly impaired phagocytosis in both Mo-AMs and Mo-IMs. Additionally, the loss of Rbpj promoted a shift toward M2-like polarization in both subsets.
Collectively, these findings demonstrate that monocyte-specific Rbpj deficiency not only impairs the differentiation of monocytes into AMs but also disrupts key functional properties of MDMs, ultimately attenuating bleomycin-induced lung inflammation and fibrosis.
Loss of Wnt/β-catenin signaling in monocytes exacerbates pulmonary fibrosis
Previous studies have indicated that Wnt/β-catenin signaling plays a critical role in the differentiation of monocytes into macrophages39,40,41. Our scRNA-seq data revealed that Wnt/β-catenin signaling was active in AM and Mac0 clusters (Fig. 7a). β-Catenin, encoded by the Ctnnb1 gene, is a key effector downstream of canonical Wnt signaling42. To further investigate the specific roles of Wnt/β-catenin in the recruitment of macrophages during lung repair and regeneration, we crossed Ctnnb1flox/flox mice with Ms4a3-CreER;R26-tdT mice to specifically disrupt Wnt/β-catenin signaling in monocytes (Fig. 7b). The knockout efficiency in blood and lung tissues exceeded 95% (Fig. 7c). We observed a significant reduction in the number of monocytes in Ctnnb1 knockout mice in the absence of injury (Fig. 7d). Immunostaining for tdT, F4/80, and SiglecF in lung sections, along with quantification analysis, revealed a greater percentage of MDMs at homeostasis in Ctnnb1 knockout mice (Fig. 7e–g).
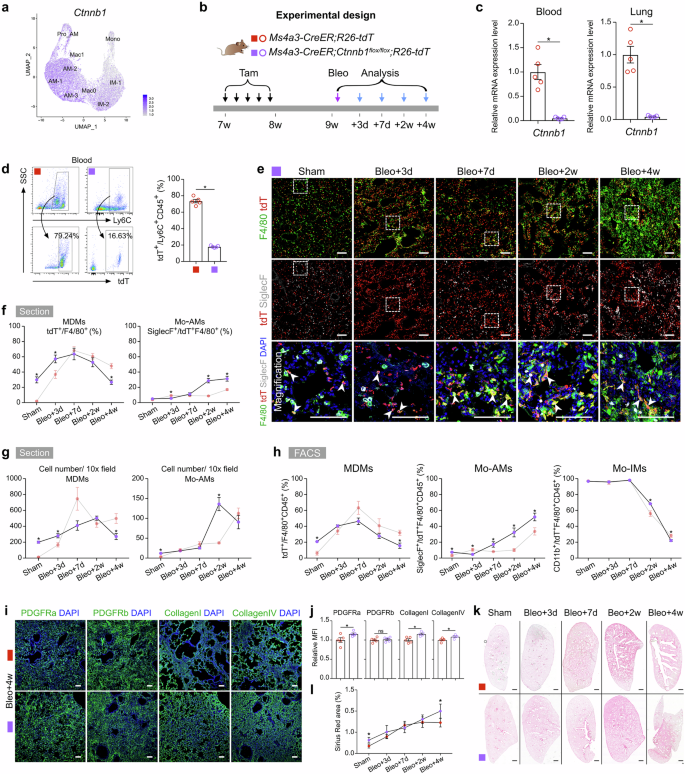
a Feature plots illustrating Ctnnb1 expression in macrophage-related clusters. b Schematic showing the experimental design. Tam tamoxifen, Bleo bleomycin. c qRT-PCR analysis showing the knockout efficiency of Ctnnb1 in tdT+ cells from the blood and lungs. Data are presented as the mean ± SEM; n = 5 mice per group. d Flow cytometric and quantification analyses of the percentage of tdT+ cells among CD45+Ly6C+ monocytes from blood. Data are presented as the mean ± SEM; n = 5 mice per group. e Immunostaining for tdT, F4/80 and SiglecF in tissue sections of Ms4a3-CreER;Ctnnb1flox/flox;R26-tdT lungs. The boxed region is magnified. White arrowheads indicate monocyte-derived macrophages (MDMs). f Quantitative analysis of the percentage of F4/80+ macrophages expressing tdT (MDMs) and the percentage of Mo-AMs among labeled tdT+ macrophages in immunostained lung sections from Ms4a3-CreER;Ctnnb1flox/flox;R26-tdT mice. The results of the control experiment were the same as those in Fig. 3d. Data are presented as the mean ± SEM; n = 5 mice per group. g Quantification of the numbers of MDMs (tdT+F4/80+) and Mo-AMs (tdT+SiglecF+) in each 10× field of immunostained lung sections from Ms4a3-CreER;Ctnnb1flox/flox;R26-tdT mice. The results of the control experiment were the same as those in Fig. 3e. Data are presented as the mean ± SEM; n = 5 mice per group. h Flow cytometric and quantification analyses of the percentage of tdT+ cells among CD45+F4/80+ macrophages (MDMs), the percentage of SiglecF+ alveolar macrophages among tdT+ macrophages (Mo-AMs), and the percentage of CD11b+ interstitial macrophages among tdT+ macrophages (Mo-IMs) from the lungs. The results of the control experiment were the same as those in Fig. 3g. Data are presented as the mean ± SEM; n = 5 mice per group. i Immunostaining for PDGFRa, PDGFRb, collagen I, and collagen IV in lung tissue sections. The boxed region is magnified. j Quantitative analysis of the relative mean fluorescence intensity (MFI) of PDGFRa, PDGFRb, collagen I, and collagen IV per 10× field of lung sections. Data are presented as the mean ± SEM; n = 5 mice per group. k, l Sirius red images and quantification analyses of pulmonary fibrosis after bleomycin treatment. Data are presented as the mean ± SEM; n = 5 mice per group. Scale bars, white, 100 µm; black, 1 mm. Each image is representative of five individual samples.
Following injury, the percentage of MDMs increased to 63.65 ± 7.61% by day 7 in Ctnnb1 knockout mice, similar to that in wild-type controls. However, by day 28, this percentage decreased to 26.78 ± 2.85%, which was significantly lower than that in wild-type mice (Fig. 7e–g). During the progression of fibrosis, a significant increase in the number of recruited Mo-AMs was observed in Ctnnb1-knockout mice (Fig. 7e–g). FACS analysis also revealed an increase in the percentage of MDMs in uninjured lungs, and a high level of Mo-AMs was maintained until day 28 after bleomycin treatment. However, absolute cell quantification indicated that the apparent increase in the proportion of Mo-AMs on day 28 was primarily due to a reduction in total MDMs rather than a substantial increase in absolute Mo-AM numbers (Supplementary Fig. S12). Notably, the percentage of Mo-IMs remained unaffected throughout the entire process in Ctnnb1-knockout mice (Fig. 7h).
The expression of M1 macrophage cytokines, such as Il6 and Il1β, which are associated with proinflammatory responses, was significantly greater on days 3 and 7 postinjury in Ctnnb1 knockout mice than in wild-type control mice. Moreover, the expression of M2 macrophage-related genes, such as Arg1 and Chil3, did not significantly change, suggesting that Wnt/β-catenin blockade promoted M1 macrophage polarization under inflammatory stimulation (Supplementary Fig. S10). Moreover, 4 weeks after bleomycin treatment, there was a marked increase in lung fibrosis in Ctnnb1-knockout mice, as assessed by anti-PDGFRa, anti-PDGFRb, anti-collagen I, and anti-collagen IV immunofluorescence staining and Sirius red staining of lung sections (Fig. 7i–l). Consistently, qRT-PCR analysis revealed elevated mRNA expression of profibrotic genes, such as Col3a1, in β-catenin-deficient mice (Supplementary Fig. S10). Furthermore, isolated tdT+ Mo-AMs and Mo-IMs from Ctnnb1 monocyte-specific knockout mice exhibited impaired phagocytic ability along with distinct alterations in polarization, specifically, reduced M1 markers in Mo-AMs and increased M1 coupled with decreased M2 markers in Mo-IMs (Supplementary Fig. S11). These results indicate that the conditional knockout of Ctnnb1 disrupted both the phagocytic function and the polarization of MDMs.
Overall, the loss of Wnt/β-catenin signaling in monocytes significantly increased the number of MDMs at homeostasis and promoted the differentiation of Mo-IMs into Mo-AMs during recovery. This dysregulated differentiation likely contributed to severe pulmonary fibrosis.













Leave a Reply