Marit G, Boiron JM, Reiffers J. Autologous blood stem cell transplantation in high risk myeloma. Bone Marrow Transplant. 1990;5:55.
Hideshima T, Chauhan D, Shima Y, Raje N, Davies FE, Tai YT, et al. Thalidomide and its analogs overcome drug resistance of human multiple myeloma cells to conventional therapy. Blood. 2000;96:2943–50.
Richardson PG, Barlogie B, Berenson J, Singhal S, Jagannath S, Irwin D, et al. A phase 2 study of bortezomib in relapsed, refractory myeloma. N Engl J Med. 2003;348:2609–17.
Rajkumar SV, Hayman SR, Lacy MQ, Dispenzieri A, Geyer SM, Kabat B, et al. Combination therapy with lenalidomide plus dexamethasone (Rev/Dex) for newly diagnosed myeloma. Blood. 2005;106:4050–3.
Stewart AK, Rajkumar SV, Dimopoulos MA, Masszi T, Špička I, Oriol A, et al. Carfilzomib, lenalidomide, and dexamethasone for relapsed multiple myeloma. N Engl J Med. 2015;372:142–52.
Miguel JS, Weisel K, Moreau P, Lacy M, Song K, Delforge M, et al. Pomalidomide plus low-dose dexamethasone versus high-dose dexamethasone alone for patients with relapsed and refractory multiple myeloma (MM-003): a randomised, open-label, phase 3 trial. Lancet Oncol. 2013;14:1055–66.
Dimopoulos MA, Oriol A, Nahi H, San-Miguel J, Bahlis NJ, Usmani SZ, et al. Daratumumab, lenalidomide, and dexamethasone for multiple myeloma. N Engl J Med. 2016;375:1319–31.
Palumbo A, Chanan-Khan A, Weisel K, Nooka AK, Masszi T, Beksac M, et al. Daratumumab, bortezomib, and dexamethasone for multiple myeloma. N Engl J Med. 2016;375:754–66.
Attal M, Richardson PG, Rajkumar SV, San-Miguel J, Beksac M, Spicka I, et al. Isatuximab plus pomalidomide and low-dose dexamethasone versus pomalidomide and low-dose dexamethasone in patients with relapsed and refractory multiple myeloma (ICARIA-MM): a randomised, multicentre, open-label, phase 3 study. Lancet. 2019;394:2096–107.
SEER*Explorer: An interactive website for SEER cancer statistics [Internet]. Surveillance Research Program, National Cancer Institute. https://seer.cancer.gov/explorer/. Accessed 23 Feb 2023.
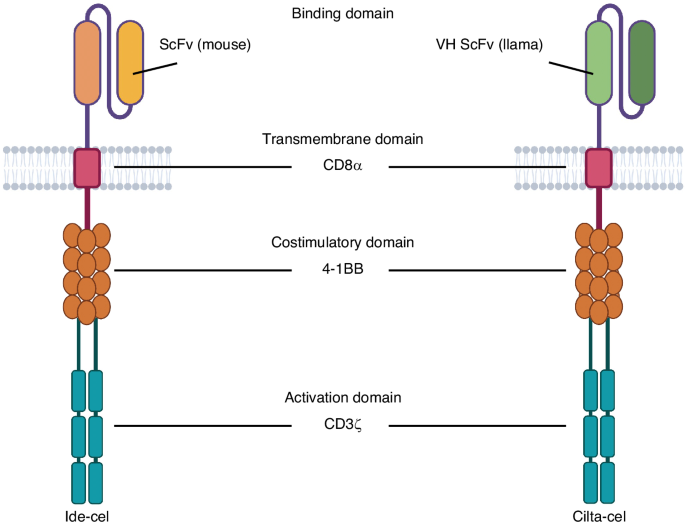
Munshi NC, Anderson LD Jr, Shah N, Madduri D, Berdeja J, Lonial S, et al. Idecabtagene vicleucel in relapsed and refractory multiple myeloma. N Engl J Med. 2021;384:705–16.
Berdeja JG, Madduri D, Usmani SZ, Jakubowiak A, Agha M, Cohen AD, et al. Ciltacabtagene autoleucel, a B-cell maturation antigen-directed chimeric antigen receptor T-cell therapy in patients with relapsed or refractory multiple myeloma (CARTITUDE-1): a phase 1b/2 open-label study. Lancet. 2021;398:314–24.
Rodriguez-Otero P, Ailawadhi S, Arnulf B, Patel K, Cavo M, Nooka AK, et al. Ide-cel or standard regimens in relapsed and refractory multiple myeloma. N Engl J Med. 2023;388:1002–14.
San-Miguel J, Dhakal B, Yong K, Spencer A, Anguille S, Mateos MV, et al. Cilta-cel or standard care in lenalidomide-refractory multiple myeloma. N Engl J Med. 2023;389:335–47.
Martin T, Usmani SZ, Berdeja JG, Agha M, Cohen AD, Hari P, et al. Ciltacabtagene autoleucel, an anti-B-cell maturation antigen chimeric antigen receptor T-cell therapy, for relapsed/refractory multiple myeloma: CARTITUDE-1 2-year follow-up. J Clin Oncol. 2023;41:1265–74.
Chekol Abebe E, Yibeltal Shiferaw M, Tadele Admasu F, Asmamaw Dejenie T. Ciltacabtagene autoleucel: the second anti-BCMA CAR T-cell therapeutic armamentarium of relapsed or refractory multiple myeloma. Front Immunol. 2022;13:991092.
Mann H, Comenzo RL. Evaluating the therapeutic potential of idecabtagene vicleucel in the treatment of multiple myeloma: evidence to date. OncoTargets Ther. 2022;15:799–813.
Fandrei D, Rade M, Kreuz M, Fischer L, Born P, Seiffert S, et al. The Differences between ide-cel and cilta-cel in relapsed myeloma at single cell resolution. Blood. 2024;144:1877.
Atanackovic D, Luetkens T, Schneider D, Hu P, Wang X, Shetty AC, et al. Immune correlates of anti-BCMA CAR-T products idecabtagene vicleucel and ciltacabtagene autoleucel in a real-world cohort of patients with multiple myeloma. Nat Commun. 2025;16:6154.
Raje N, Berdeja J, Lin Y, Siegel D, Jagannath S, Madduri D, et al. Anti-BCMA CAR T-cell therapy bb2121 in relapsed or refractory multiple myeloma. N Engl J Med. 2019;380:1726–37.
Zhao WH, Liu J, Wang BY, Chen YX, Cao XM, Yang Y, et al. A phase 1, open-label study of LCAR-B38M, a chimeric antigen receptor T cell therapy directed against B cell maturation antigen, in patients with relapsed or refractory multiple myeloma. J Hematol Oncol. 2018;11:141.
Anderson L, San-Miguel J, Martin T, Jagannath S, Berdeja JG, Lonial S, et al. Idecabtagene vicleucel (ide-cel) in patients (pts) with triple-class–exposed (TCE) relapsed and refractory multiple myeloma (RRMM): the 5-year follow-up analysis from KarMMa. 21st International Myeloma Society Annual Meeting (September 2024).
Jagannath S, Martin T, Usmani S, Berdeja J, Jakubowiak A, Agha M, et al. Cartitude-1 final results: phase 1b/2 study of ciltacabtagene autoleucel in heavily pretreated patients with relapsed/refractory multiple myeloma. (Abstract release date: 05/25/23) EHA Library Munshi N 06/08/2023; 387902; S202.
Reshef R, Jensen A, Gordillo C, Reid B, Dima D, Peres L, et al. Association between cilta-cel dose and efficacy and toxicity outcomes for patients with Relapsed/Refractory multiple myeloma (RRMM): A real-world analysis from the US multiple myeloma immunotherapy consortium. Blood. 2025;146:134.
Usmani SZ, Quach H, Mateos MV, Landgren O, Leleu X, Siegel D, et al. Carfilzomib, dexamethasone, and daratumumab versus carfilzomib and dexamethasone for patients with relapsed or refractory multiple myeloma (CANDOR): updated outcomes from a randomised, multicentre, open-label, phase 3 study. Lancet Oncol. 2022;23:65–76.
Martin T, Dimopoulos MA, Mikhael J, Yong K, Capra M, Facon T, et al. Isatuximab, carfilzomib, and dexamethasone in patients with relapsed multiple myeloma: updated results from IKEMA, a randomized Phase 3 study. Blood Cancer J. 2023;13:72.
Sonneveld P, Zweegman S, Cavo M, Nasserinejad K, Broijl A, Troia R, et al. Carfilzomib, pomalidomide, and dexamethasone as second-line therapy for lenalidomide-refractory multiple myeloma. Hemasphere. 2022;6:e786.
Ailawadhi S, Arnulf B, Patel K, Cavo M, Nooka AK, Manier S, et al. Ide-cel vs standard regimens in triple-class-exposed relapsed and refractory multiple myeloma: updated KarMMa-3 analyses. Blood. 2024;144:2389–401.
Patel K, Rodríguez-Otero P, Manier S, Baz R, Raab MS, Cavo M, et al. Idecabtagene vicleucel (ide-cel) vs standard regimens in patients with triple-class–exposed (tce) relapsed and refractory multiple myeloma (rrmm): a karmma-3 analysis in high-risk subgroups. (Abstract release date: 05/11/23) EHA Library Patel K 06/08/2023; 387895; S195.
Weisel K, Costa LJ, van de Donk NWCJ, Sidana S, Cohen Y, Purtill D, et al. Ciltacabtagene autoleucel vs standard of care in patients with functional high-risk multiple myeloma: cartitude-4 subgroup analysis. EHA Library Weisel K 06/13/2024; 421023; P959.
Mina R, Dhakal B, San Miguel J, Kwon M, Purtill D, Magen H, et al. Ciltacabtagene autoleucel vs standard of care in lenalidomide-refractory multiple myeloma: phase 3 cartitude-4 subgroup analysis by cytogenetic risk. EHA Library Mina R 06/13/2024; 421042; P978.
Popat R, Oriol A, Cavo M, Karlin L, Avivi Mazza I, Roeloffzen W, et al. Ciltacabtagene autoleucel (Cilta-cel) Vs standard of care (SoC) in patients with lenalidomide (Len)-refractory multiple myeloma (MM) after 1-3 lines of therapy: minimal residual disease (MRD) negativity in the phase 3 cartitude-4 trial. Blood. 2024;144:1032.
Dimopoulos M, Quach H, Mateos MV, Landgren O, Leleu X, Siegel D, et al. Carfilzomib, dexamethasone, and daratumumab versus carfilzomib and dexamethasone for patients with relapsed or refractory multiple myeloma (CANDOR): results from a randomised, multicentre, open-label, phase 3 study. Lancet. 2020;396:186–97.
Moreau P, Dimopoulos MA, Mikhael J, Yong K, Capra M, Facon T, et al. Isatuximab, carfilzomib, and dexamethasone in relapsed multiple myeloma (IKEMA): a multicentre, open-label, randomised phase 3 trial. Lancet. 2021;397:2361–71.
Oncologic Drugs Advisory Committee Meeting. FDA. 2024. https://www.fda.gov/media/177060/download. Accessed 15 Mar 2024.
Oncologic Drugs Advisory Committee meeting introductory comments. FDA. 2024. https://www.fda.gov/media/177057/download. Accessed 15 Mar 2024.
Mohyuddin GR, Prasad V. Detecting selection bias in observational studies-when interventions work too fast. JAMA Intern Med. 2023;183:897–8.
FDA approves idecabtagene vicleucel for multiple myeloma. FDA. 2021. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-idecabtagene-vicleucel-multiple-myeloma. Accessed 15 Mar 2024.
FDA approves ciltacabtagene autoleucel for relapsed or refractory multiple myeloma. FDA. 2022. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-ciltacabtagene-autoleucel-relapsed-or-refractory-multiple-myeloma. Accessed 15 Mar 2024.
Hansen DK, Sidana S, Peres LC, Colin Leitzinger C, Shune L, Shrewsbury A, et al. Idecabtagene vicleucel for relapsed/refractory multiple myeloma: real-world experience from the myeloma CAR T consortium. J Clin Oncol. 2023;41:2087–97.
Sidana S, Ahmed N, Akhtar OS, Brazauskas R, Oloyede T, Bye M, et al. Standard-of-care idecabtagene vicleucel for relapsed/refractory multiple myeloma. Blood. 2025;146:167–77.
Sidana S, Patel KK, Peres LC, Bansal R, Kocoglu MH, Shune L, et al. Safety and efficacy of standard-of-care ciltacabtagene autoleucel for relapsed/refractory multiple myeloma. Blood. 2025;145:85–97.
Hansen DK, Peres LC, Dima D, Richards A, Shune L, Afrough A, et al. Comparison of standard-of-care idecabtagene vicleucel and ciltacabtagene autoleucel in relapsed/refractory multiple myeloma. J Clin Oncol. 2025;43:1597–1609.
Merz M, Albici AM, von Tresckow B, Rathje K, Fenk R, Holderried T, et al. Idecabtagene vicleucel or ciltacabtagene autoleucel for relapsed or refractory multiple myeloma: An international multicenter study. Hemasphere. 2025;9:e70070.
Ailawadhi S, Anderson LD, Jr, Dhakal B, Shune L, Sborov DW, Hansen DK. Optimizing selection of bridging therapies prior to CAR-T therapy administration for multiple myeloma: clinical pearls from an expert roundtable. Clin Lymphoma Myeloma Leuk. 2025. https://doi.org/10.1016/j.clml.2025.08.005 Online ahead of print.
Afrough A, Hashmi H, Hansen DK, Sidana S, Ahn C, Peres LC, et al. Real-world impact of bridging therapy on outcomes of ide-cel for myeloma in the U.S. myeloma immunotherapy consortium. Blood Cancer J. 2024;14:63.
Frenking JH, Zhou X, Rejeski K, Wagner V, Costello P, Hielscher T, et al. Bridging intensity is associated with impaired hematopoietic recovery after BCMA CAR-T therapy for multiple myeloma. Blood Adv. 2025;9:4151–66.
Zafar A, Huang CY, Lo M, Arora S, Chung A, Wong SW, et al. Intensity of cyclophosphamide-based bridging therapy before chimeric antigen receptor T cell therapy in myeloma. Transplant Cell Ther. 2023;29:504 e1–7.
Fandrei D, Seiffert S, Rade M, Rieprecht S, Gagelmann N, Born P, et al. Bispecific antibodies as bridging to BCMA CAR-T cell therapy for relapsed/refractory multiple myeloma. Blood Cancer Discov. 2025;6:38–54.
Dhakal B, Akhtar OS, Fandrei D, Jensen A, Banerjee R, Pan D, et al. Sequential targeting in multiple myeloma: talquetamab, a GPRC5D bispecific antibody, as a Bridge to BCMA CAR-T cell therapy. Blood. 2025;146:2063–72.
Pan D, Mouhieddine TH, Sheng T, Fu W, Moshier E, Richter J, et al. Extramedullary disease but not paraskeletal disease portends inferior outcomes after CAR T cell therapy in multiple myeloma. Bone Marrow Transplant. 2025;60:1114–9.
Dima D, Abdallah AO, Davis JA, Awada H, Goel U, Rashid A, et al. Impact of extraosseous extramedullary disease on outcomes of patients with relapsed-refractory multiple myeloma receiving standard-of-care chimeric antigen receptor T-cell therapy. Blood Cancer J. 2024;14:90.
Zanwar S, Sidana S, Shune L, Puglianini OC, Pasvolsky O, Gonzalez R, et al. Impact of extramedullary multiple myeloma on outcomes with idecabtagene vicleucel. J Hematol Oncol. 2024;17:42.
Steinhardt MJ, Schaefers C, Leypoldt LB, Blau IW, Harzer M, Zhou X, et al. Activity of CAR-T cells and bispecific antibodies in multiple myeloma with extramedullary involvement. Blood Cancer J. 2025;15:126.
Cohen YJM-L, Khan AM, Oriol A, Spencer A, Dhakal B, Touzeau C, et al. Ciltacabtagene autoleucel versus standard of care in patients with relapsed/refractory multiple myeloma: cartitude-4 survival subgroup analyses. (Abstract release date: 05/22/25) EHA Library Cohen Y 06/14/2025; 4160798; PS1723.
Yao H, Ren SH, Wang LH, Ren MQ, Cai J, Chen D, et al. BCMA/GPRC5D bispecific CAR T-cell therapy for relapsed/refractory multiple myeloma with extramedullary disease: a single-center, single-arm, phase 1 trial. J Hematol Oncol. 2025;18:56.
Portuguese AJ, Liang EC, Huang JJ, Jeon Y, Dima D, Banerjee R, et al. Extramedullary disease is associated with severe toxicities following B-cell maturation antigen CAR T-cell therapy in multiple myeloma. Haematologica. 2025;110:3065–77.
Knudsen LM, Hippe E, Hjorth M, Holmberg E, Westin J. Renal function in newly diagnosed multiple myeloma-a demographic study of 1353 patients. The Nordic Myeloma Study Group. Eur J Haematol. 1994;53:207–12.
Sidana S, Peres LC, Hashmi H, Hosoya H, Ferreri C, Khouri J, et al. Idecabtagene vicleucel chimeric antigen receptor T-cell therapy for relapsed/refractory multiple myeloma with renal impairment. Haematologica. 2024;109:777–86.
Sidana S, Peres LC, Shune L, Richards A, Kocoglu MH, Ferreri CJ, et al. Ciltacabtagene vicluecel (Cilta-cel) chimeric antigen receptor T-cell therapy for relapsed/refractory multiple myeloma (RRMM) with renal impairment: real world experience. Transplant Cell Therapy 2025;31:S403.
Habib A, Ahmed N, Khan AM, Chang D, Paul B, Shaikh H, et al. A real-world analysis of the safety and efficacy of BCMA-directed CAR T-cell therapy for patients with relapsed/refractory multiple myeloma and baseline renal impairment. Blood. 2024;144:2009.
Cohen AD, Mateos MV, Cohen YC, Rodriguez-Otero P, Paiva B, van de Donk N, et al. Efficacy and safety of cilta-cel in patients with progressive multiple myeloma after exposure to other BCMA-targeting agents. Blood. 2023;141:219–30.
Ferreri CJ, Hildebrandt MAT, Hashmi H, Shune LO, McGuirk JP, Sborov DW, et al. Real-world experience of patients with multiple myeloma receiving ide-cel after a prior BCMA-targeted therapy. Blood Cancer J. 2023;13:117.
Mohan Lal B, Alzubi M, Alrawabdeh J, Shaughnessy J, John D, Zhan F, et al. Prior Exposure to belantamab mafodotin influences outcomes with idecabtagene vicluecel in patients with multiple myeloma. Blood. 2024;144:3789.
Richardson T, Holtick U, Frenking JH, Tharmaseelan H, Balke-Want H, Flumann R, et al. Sequential BCMA CAR T-cell therapy in refractory multiple myeloma. Blood Adv. 2025;9:4624–30.
Dima D, Vazquez-Martinez MA, Davis JA, Goel U, Afrough A, Sannareddy A, et al. Outcomes of teclistamab in patients with relapsed/refractory multiple myeloma with prior exposure to BCMA-directed therapy: a multicenter study from the U.S. Multiple Myeloma Immunotherapy Consortium. Blood Cancer J. 2025;15:111.
Nooka AK, Lesokhin AM, Mohty M, Niesvizky R, Maisel C, Arnulf B, et al. Efficacy and safety of elranatamab in patients with relapsed/refractory multiple myeloma (RRMM) and prior B-cell maturation antigen (BCMA)-directed therapies: a pooled analysis from MagnetisMM studies. J Clin Oncol. 2023;41:8008.
Patel U, Oluwole OO, Kassim A, Jayani R, Belliveau P, Savani B, et al. Sequencing bispecific antibodies and CAR T cell therapy in multiple myeloma with prior exposure to BCMA-targeted therapies. J Clin Oncol. 2023;41:e20049–e.
Lee H, Ahn S, Maity R, Leblay N, Ziccheddu B, Truger M, et al. Mechanisms of antigen escape from BCMA- or GPRC5D-targeted immunotherapies in multiple myeloma. Nat Med. 2023;29:2295–306.
Lee H, Durante M, Skerget S, Vishwamitra D, Benaoudia S, Ahn S, et al. Impact of soluble BCMA and non-T-cell factors on refractoriness to BCMA-targeting T-cell engagers in multiple myeloma. Blood. 2024;144:2637–51.
Worel N, Mooyaart JE, Hoogenboom JD, Daskalakis M, Tudesq JJ, Ram R, et al. CAR-T cell manufacturing failures and out-of-specification products in the real-world setting: A survey from the EBMT cellular therapy and immunobiology working party. Bone Marrow Transplant. 2025;60:1184–6.
Grischke TK, Cooperrider JH, Pula A, Sneider A, Yates S, Jakubowiak AJ, et al. Brain-to-vein and vein-to-vein times and outcomes in CAR T-cell therapy in myeloma. Blood Cancer J. 2025;15:47.
Perez A, Al Sagheer T, Nahas GR, Linhares YPL. Outpatient administration of CAR T-cell therapy: a focused review with recommendations for implementation in community based centers. Front Immunol. 2024;15:1412002.
Rejeski K, Jain MD, Shah NN, Perales MA, Subklewe M. Immune effector cell-associated haematotoxicity after CAR T-cell therapy: from mechanism to management. Lancet Haematol. 2024;11:e459–e70.
Beyar-Katz O, Rejeski K, Shouval R. Immune effector cell-associated hematotoxicity: mechanisms, clinical manifestations, and management strategies. Haematologica. 2025;110:1254–68.
Yang Y, Peng H, Wang J, Li F. New insights into CAR T-cell hematological toxicities: manifestations, mechanisms, and effective management strategies. Exp Hematol Oncol. 2024;13:110.
Rejeski K, Hansen DK, Bansal R, Sesques P, Ailawadhi S, Logue JM, et al. The CAR-HEMATOTOX score as a prognostic model of toxicity and response in patients receiving BCMA-directed CAR-T for relapsed/refractory multiple myeloma. J Hematol Oncol. 2023;16:88.
Dima D, Logue JM, Waqar SHB, Peres LC, Colin-Leitzinger CM, De Avila G, et al. Cytopenias and infections following ciltacabtagene autoleucel in heavily-pretreated relapsed or refractory multiple myeloma. Haematologica. 2025. https://doi.org/10.3324/haematol.2025.287783. Online ahead of print.
Palacios-Berraquero ML, Rodriguez-Marquez P, Calleja-Cervantes ME, Berastegui N, Zabaleta A, Burgos L, et al. Molecular mechanisms promoting long-term cytopenia after BCMA CAR-T therapy in multiple myeloma. Blood Adv. 2024;8:5479–92.
Thiruvengadam SK, Sheng Y, Huang CY, Bylsma S, Lo M, Kennedy V, et al. Infectious complications in patients with relapsed refractory multiple myeloma after BCMA CAR T-cell therapy. Blood Adv. 2022;6:2045–54.
Logue JM, Peres LC, Hashmi H, Colin-Leitzinger CM, Shrewsbury AM, Hosoya H, et al. Early cytopenias and infections after standard of care idecabtagene vicleucel in relapsed or refractory multiple myeloma. Blood Adv. 2022;6:6109–19.
Wang Y, Li C, Xia J, Li P, Cao J, Pan B, et al. Humoral immune reconstitution after anti-BCMA CAR T-cell therapy in relapsed/refractory multiple myeloma. Blood Adv. 2021;5:5290–9.
Kumar AD, Atallah-Yunes SA, Rajeeve S, Abdelhak A, Hashmi H, Corraes A, et al. Delayed neurotoxicity after CAR-T in multiple myeloma: results from a global IMWG registry. Blood. 2024;144:4758.
Cohen AD, Parekh S, Santomasso BD, Gallego Perez-Larraya J, van de Donk N, Arnulf B, et al. Incidence and management of CAR-T neurotoxicity in patients with multiple myeloma treated with ciltacabtagene autoleucel in CARTITUDE studies. Blood Cancer J. 2022;12:32.
Ellithi M, Elsallab M, Lunning MA, Holstein SA, Sharma S, Trinh JQ, et al. Neurotoxicity and rare adverse events in BCMA-directed CAR T cell therapy: a comprehensive analysis of real-world FAERS data. Transplant Cell Ther. 2025;31:71 e1–14.
Couturier A, Escoffre M, Leh F, Villoteau AS, Palard X, Le Jeune F, et al. Parkinson-like neurotoxicity in female patients treated with idecabtagene-vicleucel. Hemasphere. 2024;8:e131.
Graham CE, Lee WH, Wiggin HR, Supper VM, Leick MB, Birocchi F, et al. Chemotherapy-induced reversal of ciltacabtagene autoleucel-associated movement and neurocognitive toxicity. Blood. 2023;142:1248–52.
Kelly K, Cooperrider JH, Bishop MR, Kosuri S, Jakubowiak A, Derman BA. Intrathecal chemotherapy for ciltacabtagene autoleucel-associated movement and neurocognitive toxicity. Blood Adv. 2025;9:3613–6.
Van Oekelen O, Aleman A, Upadhyaya B, Schnakenberg S, Madduri D, Gavane S, et al. Neurocognitive and hypokinetic movement disorder with features of Parkinsonism after BCMA-targeting CAR-T cell therapy. Nat Med. 2021;27:2099–103.
Kathari YK, Ahmad H, Kallen ME, Koka R, Omili D, Iraguha T, et al. Immune-mediated facial nerve paralysis in a myeloma patient post B-cell maturation antigen-targeted chimeric antigen receptor T cells. Haematologica. 2024;109:682–8.
Fortuna GG, Banerjee R, Savid-Frontera C, Song J, Morán-Segura CM, Nguyen JV, et al. Immune effector cell-associated enterocolitis following chimeric antigen receptor T-cell therapy in multiple myeloma. Blood Cancer J. 2024;14:180.
Banerjee R, Hosoya H, Mikkilineni L, Hansen DK, Wolf JL, Lin Y. Managing IEC-associated enterocolitis following CAR-T therapy in multiple myeloma. Blood Cancer J. 2025;15:112.
Ozdemirli M, Loughney TM, Deniz E, Chahine JJ, Albitar M, Pittaluga S, et al. Indolent CD4+ CAR T-cell lymphoma after cilta-cel CAR T-cell therapy. N Engl J Med. 2024;390:2074–82.
Elsallab M, Ellithi M, Lunning MA, D’Angelo C, Ma J, Perales M-A, et al. Second primary malignancies after commercial CAR T-cell therapy: analysis of the FDA adverse events reporting system. Blood. 2024;143:2099–105.
Harrison SJ, Nguyen T, Rahman M, Er J, Li J, Li K, et al. CAR+ T-cell lymphoma post ciltacabtagene autoleucel therapy for relapsed refractory multiple myeloma. Blood. 2023;142:6939.
Braun T, Rade M, Merz M, Klepzig H, Große F, Fandrei D, et al. Multiomic profiling of T cell lymphoma after therapy with anti-BCMA CAR T cells and GPRC5D-directed bispecific antibody. Nat Med. 2025;31:1145–53.
Cordas Dos Santos DM, Tix T, Shouval R, Gafter-Gvili A, Alberge JB, Cliff ERS, et al. A systematic review and meta-analysis of nonrelapse mortality after CAR T cell therapy. Nat Med. 2024;30:2667–78.
Wesson W, Dima D, Suleman N, Saif MSI, Tabak C, Logan E, et al. Timing of toxicities and non-relapse mortality following CAR T therapy in myeloma. Transplant Cell Ther. 2024;30:876–84.
Rejeski K, Hansen DK, Cordas Dos Santos DM, De Menezes Silva Corraes A, Castaneda O, Dillard CM, et al. Pre-apheresis prediction of toxicity and response in patients receiving BCMA-directed CAR-T for relapsed/refractory multiple myeloma. Blood. 2024;144:895.
Akhtar OS, Modi K, Kim J, Skelson L, Smith E, Al-Jumayli MA, et al. Simple score of albumin and CRP predicts high-grade toxicity in patients with multiple myeloma receiving CAR-T therapy. Transplant Cell Ther. 2024;30:283 e1–10.
Mohan M, Schinke C. Stem cell boost for immune effector cell associated hematotoxicity in multiple myeloma: minimizing long-term complications and expanding post CAR T-cell therapy relapse options. Expert Rev Hematol. 2025:1–7. https://doi.org/10.1080/17474086.2025.2570335. Online ahead of print.
Gagelmann N, Dima D, Merz M, Hashmi H, Ahmed N, Tovar N, et al. Development and validation of a prediction model of outcome after B-cell maturation antigen-directed chimeric antigen receptor T-cell therapy in relapsed/refractory multiple myeloma. J Clin Oncol. 2024;42:1665–75.
Ramamurthy A, Tommasi A, Saha K. Advances in manufacturing chimeric antigen receptor immune cell therapies. Semin Immunopathol. 2024;46:12.
Bicudo E, Brass I. Institutional and infrastructure challenges for hospitals producing advanced therapies in the UK: the concept of ‘point-of-care manufacturing readiness. Regen Med. 2022;17:719–37.
Hort S, Herbst L, Bäckel N, Erkens F, Niessing B, Frye M, et al. Toward rapid, widely available autologous CAR-T cell therapy – artificial intelligence and automation enabling the smart manufacturing hospital. Front Med. 2022;9:913287.
Trainor N, Purpura KA, Middleton K, Fargo K, Hails L, Vicentini-Hogan M, et al. Automated production of gene-modified chimeric antigen receptor T cells using the Cocoon Platform. Cytotherapy. 2023;25:1349–60.
Yang J, He J, Zhang X, Li J, Wang Z, Zhang Y, et al. Next-day manufacture of a novel anti-CD19 CAR-T therapy for B-cell acute lymphoblastic leukemia: first-in-human clinical study. Blood Cancer J. 2022;12:104.
Ghassemi S, Durgin JS, Nunez-Cruz S, Patel J, Leferovich J, Pinzone M, et al. Rapid manufacturing of non-activated potent CAR T cells. Nat Biomed Eng. 2022;6:118–28.
Mueller KP, Piscopo NJ, Forsberg MH, Saraspe LA, Das A, Russell B, et al. Production and characterization of virus-free, CRISPR-CAR T cells capable of inducing solid tumor regression. J Immunother Cancer. 2022;10:e004446.
Kitte R, Rabel M, Geczy R, Park S, Fricke S, Koehl U, et al. Lipid nanoparticles outperform electroporation in mRNA-based CAR T cell engineering. Mol Ther Methods Clin Dev. 2023;31:101139.
Tipanee J, Samara-Kuko E, Gevaert T, Chuah MK, VandenDriessche T. Universal allogeneic CAR T cells engineered with Sleeping Beauty transposons and CRISPR-CAS9 for cancer immunotherapy. Mol Ther. 2022;30:3155–75.
Boretti A. Improving chimeric antigen receptor T-cell therapies by using artificial intelligence and internet of things technologies: a narrative review. Eur J Pharmacol. 2024;974:176618.
Zhao J, Lin Q, Song Y, Liu D. Universal CARs, universal T cells, and universal CAR T cells. J Hematol Oncol. 2018;11:132.
Bal S, Berdeja J, Htut M, Kocoglu M, Gregory T, Anderson LD, et al. Bms-986393 (cc-95266), a g protein–coupled receptor class c group 5 member d (gprc5d)–targeted car t-cell therapy for relapsed/refractory multiple myeloma (rrmm): results from a phase 1 study. (Abstract release date: 05/11/23) EHA Library Bal S 06/08/2023; 387893; S193.
Bal S, Anderson LD Jr, Nadeem O, Berdeja JG, Rossi A, Gregory T, et al. Efficacy and safety with extended follow-up in a phase 1 study of BMS-986393, a G protein-coupled receptor class C group 5 member D (GPRC5D)-targeted CAR T cell therapy, in patients (pts) with heavily pretreated relapsed/refractory (RR) multiple myeloma (MM). Blood. 2024;144:922.
Bal S, Htut M, Berdeja JG, Anderson LD Jr, Rossi A, Gregory T, et al. BMS-986393, a G protein-coupled receptor class C group 5 member D (GPRC5D)-targeted CAR T cell therapy, in patients (pts) with relapsed/refractory (RR) multiple myeloma (MM) and 1-3 prior regimens: updated phase 1 safety and efficacy results. Blood. 2024;144:2069.
Zhang M, Wei G, Zhou L, Zhou J, Chen S, Zhang W, et al. GPRC5D CAR T cells (OriCAR-017) in patients with relapsed or refractory multiple myeloma (POLARIS): a first-in-human, single-centre, single-arm, phase 1 trial. Lancet Haematol. 2023;10:e107–16.
Du J, Fu W, Lu J, Qiang W, He H, Liu J, et al. Phase I open-label single-arm study of BCMA/CD19 dual-targeting FasTCAR-T Cells (GC012F) as first-line therapy for transplant-eligible newly diagnosed high-risk multiple myeloma. Blood. 2022;140:889–90.
Jiang H, Li L, Chen K, Yang C, Ma R, Xie L, et al. A prospective investigator-initiated phase 1/2 study of BCMA/CD19 dual-targeting CAR T therapy in patients with relapsed/refractory multiple myeloma including those with extramedullary disease. Blood. 2024;144:923.
Derrien J, Gastineau S, Frigout A, Giordano N, Cherkaoui M, Gaborit V, et al. Acquired resistance to a GPRC5D-directed T-cell engager in multiple myeloma is mediated by genetic or epigenetic target inactivation. Nat Cancer. 2023;4:1536–43.
Mi X, Penson A, Abdel-Wahab O, Mailankody S. Genetic basis of relapse after GPRC5D-targeted CAR T cells. N Engl J Med. 2023;389:1435–7.
Zhou J, Luo X, Lei B, Wu K, Shi F, Zhang Y, et al. Armored multivalent CAR-T Cells targeting BCMA, GPRC5D, and CD38 eradicate antigen-heterogeneous multiple myeloma, address antigen escape and enhance expansion. Blood. 2024;144:2038.
Khan AN, Goda JS, Asija S, Karulkar A, Palkar P, Chowdury A, et al. Development of a novel humanized BCMA-directed CAR for multiple myeloma: robust activity and low toxicity in preclinical studies. Blood. 2024;144:4809.
Miljkovic MD, Asch AS, Orloff G, Boccia R, Berdeja JG, Altuntas F, et al. Safety and tolerability of BCMA-directed mRNA CAR T-cell therapy in multiple myeloma and autoimmune disease. Blood. 2024;144:2080.
Bishop MR, Rosenblatt J, Dhakal B, Raje N, Cook D, Gaballa MR, et al. Phase 1 study of anitocabtagene autoleucel for the treatment of patients with relapsed and/or refractory multiple myeloma (RRMM): efficacy and safety with 34-month median follow-up. Blood. 2024;144:4825.
Freeman CL, Dhakal B, Kaur G, Maziarz RT, Callander N, Sperling AS, et al. Phase 2 registrational study of anitocabtagene autoleucel for the treatment of patients with relapsed and/or refractory multiple myeloma: preliminary results from the IMMagine-1 trial. Blood. 2024;144:1031.
Fu C, Yan L, Yao W, Shang J, Jin S, Yan S, et al. A first-in-human study of CT0590, a triple knock-out, allogeneic CAR T-cell therapy targeting BCMA and NKG2A, in subjects with relapsed/refractory multiple myeloma. Blood. 2024;144:4843.
Dholaria DB, Kocoglu MH, Kin A, Shune L, Ganguly SMD, Cruz JCC, et al. A phase 1 study of P-BCMA-ALLO1, a non-viral, allogeneic BCMA directed CAR-T in relapsed/refractory multiple myeloma (RRMM): results from optimized lymphodepletion cohort (Arm C). Transplant Cell Therapy. 2025;31:S38.
Harrison S, Ho PJ, Lim S-L, Talam S, Pahl H, Dingar D, et al. Minimal residual disease (MRD)-negative outcomes following a novel, in vivo gene therapy generating anti–B-cell maturation antigen (BCMA) chimeric antigen receptor (CAR)-T cells in patients with relapsed and refractory multiple myeloma: Preliminary results from inMMyCAR, the first-in-human phase 1 study of KLN-1010. Blood. 2025;146:LBA-1.
Dytfeld D, Dhakal B, Agha M, Manier S, Delforge M, Kuppens S, et al. Bortezomib, lenalidomide and dexamethasone (VRd) followed by ciltacabtagene autoleucel versus Vrd followed by lenalidomide and dexamethasone (Rd) maintenance in patients with newly diagnosed multiple myeloma not intended for transplant: a randomized, phase 3 study (CARTITUDE-5). Blood. 2021;138:1835.
Boccadoro M, San-Miguel J, Suzuki K, Van De Donk NWCJ, Cook G, Jakubowiak A, et al. DVRd followed by ciltacabtagene autoleucel versus DVRd followed by ASCT in patients with newly diagnosed multiple myeloma who are transplant eligible: a randomized phase 3 study (EMagine/CARTITUDE-6). Blood. 2022;140:4630–2.
Usmani S, Patel K, Hari P, Berdeja J, Alsina M, Vij R, et al. KarMMa-2 cohort 2a: efficacy and safety of idecabtagene vicleucel in clinical high-risk multiple myeloma patients with early relapse after frontline autologous stem cell transplantation. Blood. 2022;140:875–7.
Donk NWCJVD, Agha ME, Cohen AD, Cohen YC, Anguille S, Kerre T, et al. Biological correlative analyses and updated clinical data of ciltacabtagene autoleucel (cilta-cel), a BCMA-directed CAR-T cell therapy, in patients with multiple myeloma (MM) and early relapse after initial therapy: CARTITUDE-2, cohort B. J Clin Oncol. 2022;40:8029.
Xu J, Wang BY, Yu SH, Chen SJ, Yang SS, Liu R, et al. Long-term remission and survival in patients with relapsed or refractory multiple myeloma after treatment with LCAR-B38M CAR T cells: 5-year follow-up of the LEGEND-2 trial. J Hematol Oncol. 2024;17:23.
















Leave a Reply