Park, M. D., Silvin, A., Ginhoux, F. & Merad, M. Macrophages in health and disease. Cell 185, 4259–4279 (2022).
Italiani, P. & Boraschi, D. From monocytes to M1/M2 macrophages: phenotypical vs. functional differentiation. Front. Immunol. 5, 514 (2014).
Guilliams, M., Mildner, A. & Yona, S. Developmental and functional heterogeneity of monocytes. Immunity 49, 595–613 (2018).
Sommerfeld, S. D. et al. Interleukin-36gamma-producing macrophages drive IL-17-mediated fibrosis. Sci. Immunol. 4, https://doi.org/10.1126/sciimmunol.aax4783 (2019).
Martin, K. E. & Garcia, A. J. Macrophage phenotypes in tissue repair and the foreign body response: Implications for biomaterial-based regenerative medicine strategies. Acta Biomater. 133, 4–16 (2021).
Qian, B. Z. & Pollard, J. W. Macrophage diversity enhances tumor progression and metastasis. Cell 141, 39–51 (2010).
Quail, D. F. & Joyce, J. A. Microenvironmental regulation of tumor progression and metastasis. Nat. Med 19, 1423–1437 (2013).
Kersten, K. et al. Spatiotemporal co-dependency between macrophages and exhausted CD8(+) T cells in cancer. Cancer Cell 40, 624–638.e629 (2022).
Tylek, T., Wong, J., Vaughan, A. E. & Spiller, K. L. Biomaterial-mediated intracellular control of macrophages for cell therapy in pro-inflammatory and pro-fibrotic conditions. Biomaterials 308, 122545 (2024).
Meli, V. S. et al. Biophysical regulation of macrophages in health and disease. J. Leukoc. Biol. 106, 283–299 (2019).
Butenko, S. et al. Hydrogel crosslinking modulates macrophages, fibroblasts, and their communication, during wound healing. Nat. Commun. 15, 6820 (2024).
Chung, L., Maestas, D. R. Jr, Housseau, F. & Elisseeff, J. H. Key players in the immune response to biomaterial scaffolds for regenerative medicine. Adv. Drug Deliv. Rev. 114, 184–192 (2017).
Witherel, C. E., Abebayehu, D., Barker, T. H. & Spiller, K. L. Macrophage and fibroblast interactions in biomaterial-mediated fibrosis. Adv. Health. Mater. 8, e1801451 (2019).
Zarif, J. C. et al. Mannose receptor-positive macrophage infiltration correlates with prostate cancer onset and metastatic castration-resistant disease. Eur. Urol. Oncol. 2, 429–436 (2019).
Jaynes, J. M. et al. Mannose receptor (CD206) activation in tumor-associated macrophages enhances adaptive and innate antitumor immune responses. Sci. Transl. Med. 12, https://doi.org/10.1126/scitranslmed.aax6337 (2020).
Ma, R. Y., Black, A. & Qian, B. Z. Macrophage diversity in cancer revisited in the era of single-cell omics. Trends Immunol. 43, 546–563 (2022).
Strizova, Z. et al. M1/M2 macrophages and their overlaps – myth or reality? Clin. Sci. (Lond.) 137, 1067–1093 (2023).
Adebowale, K. G., Guerriero, J L. & Mitragotri, S. Dynamics of macrophage tumor infiltration. Appl. Phys. Rev. 10, https://doi.org/10.1063/5.0160924 (2023).
Guerriero, J. L. Macrophages: the road less traveled, changing anticancer therapy. Trends Mol. Med 24, 472–489 (2018).
Mulder, K. et al. Cross-tissue single-cell landscape of human monocytes and macrophages in health and disease. Immunity 54, 1883–1900.e1885 (2021).
Ray, A. et al. Targeting CD206+ macrophages disrupts the establishment of a key antitumor immune axis. J. Exp. Med. 222, https://doi.org/10.1084/jem.20240957 (2025).
Cavnar, M. J. et al. Tumor-associated macrophage infiltration in colorectal cancer liver metastases is associated with better outcome. Ann. Surg. Oncol. 24, 1835–1842 (2017).
Zhong, C. et al. PSGL-1 is a phagocytosis checkpoint that enables tumor escape from macrophage clearance. Sci. Immunol. 10, eadn4302 (2025).
Matusiak, M. et al. Spatially segregated macrophage populations predict distinct outcomes in colon cancer. Cancer Discov. 14, 1418–1439 (2024).
Liu, M. et al. Spatially-resolved transcriptomics reveal macrophage heterogeneity and prognostic significance in diffuse large B-cell lymphoma. Nat. Commun. 15, 2113 (2024).
Enfield, K. S. S. et al. Spatial architecture of myeloid and T cells orchestrates immune evasion and clinical outcome in lung cancer. Cancer Discov. 14, 1018–1047 (2024).
Paterson, N. & Lammermann, T. Macrophage network dynamics depend on haptokinesis for optimal local surveillance. Elife 11, https://doi.org/10.7554/eLife.75354 (2022).
Mantovani, A., Allavena, P., Marchesi, F. & Garlanda, C. Macrophages as tools and targets in cancer therapy. Nat. Rev. Drug Discov. 21, 799–820 (2022).
Toledo, B. et al. Deciphering the performance of macrophages in tumour microenvironment: a call for precision immunotherapy. J. Hematol. Oncol. 17, 44 (2024).
Kloosterman, D. J. & Akkari, L. Macrophages at the interface of the co-evolving cancer ecosystem. Cell 186, 1627–1651 (2023).
Cockram, T. O. J., Dundee, J. M., Popescu, A. S. & Brown, G. C. The phagocytic code regulating phagocytosis of mammalian cells. Front. Immunol. 12, 629979 (2021).
Dolasia, K., Bisht, M. K., Pradhan, G., Udgata, A. & Mukhopadhyay, S. TLRs/NLRs: shaping the landscape of host immunity. Int Rev. Immunol. 37, 3–19 (2018).
Kelley, S. M. & Ravichandran, K. S. Putting the brakes on phagocytosis: “don’t-eat-me” signaling in physiology and disease. Embo Rep. 22, e52564 (2021).
Ding, X. et al. Engineering macrophages via nanotechnology and genetic manipulation for cancer therapy. Front. Oncol. 11, 786913 (2021).
Du, H. et al. Tuning immunity through tissue mechanotransduction. Nat. Rev. Immunol. 23, 174–188 (2023).
Zhang, X. et al. Unraveling the mechanobiology of immune cells. Curr. Opin. Biotechnol. 66, 236–245 (2020).
Liu, Z. et al. Viscoelastic synthetic antigen-presenting cells for augmenting the potency of cancer therapies. Nat. Biomed. Eng. 8, 1615–1633 (2024).
Adu-Berchie, K. et al. Generation of functionally distinct T-cell populations by altering the viscoelasticity of their extracellular matrix. Nat. Biomed. Eng. 7, 1374–1391 (2023).
Vining, K. H. et al. Mechanical checkpoint regulates monocyte differentiation in fibrotic niches. Nat. Mater. 21, 939–950 (2022).
Alraies, Z. et al. Cell shape sensing licenses dendritic cells for homeostatic migration to lymph nodes. Nat. Immunol. 25, 1193–1206 (2024).
Settle, A. H. et al. beta2 integrins impose a mechanical checkpoint on macrophage phagocytosis. Nat. Commun. 15, 8182 (2024).
Tello-Lafoz, M. et al. Cytotoxic lymphocytes target characteristic biophysical vulnerabilities in cancer. Immunity 54, 1037–1054.e1037 (2021).
Basu, R. et al. Cytotoxic T cells use mechanical force to potentiate target cell killing. Cell 165, 100–110 (2016).
Huse, M. Mechanical forces in the immune system. Nat. Rev. Immunol. 17, 679–690 (2017).
Lei, K. et al. Cancer-cell stiffening via cholesterol depletion enhances adoptive T-cell immunotherapy. Nat. Biomed. Eng. 5, 1411–1425 (2021).
Zhu, C., Chen, W., Lou, J., Rittase, W. & Li, K. Mechanosensing through immunoreceptors. Nat. Immunol. 20, 1269–1278 (2019).
Li, K. et al. Mechanical force regulates ligand binding and function of PD-1. Nat. Commun. 15, 8339 (2024).
Choi, H. K. et al. Mechanotransduction governs CD40 function and underlies X-linked Hyper IgM syndrome. Preprint at https://doi.org/10.1101/2023.07.23.550231 (2023).
Zhong, Z. et al. Human immune organoids to decode B cell response in healthy donors and patients with lymphoma. Nat. Mater. 24, 297–311 (2025).
Assen, F. P. et al. Multitier mechanics control stromal adaptations in the swelling lymph node. Nat. Immunol. 23, 1246–1255 (2022).
Horsnell, H. L. et al. Lymph node homeostasis and adaptation to immune challenge resolved by fibroblast network mechanics. Nat. Immunol. 23, 1169–1182 (2022).
Narain, R., Muncie-Vasic, J. M. & Weaver, V. M. Forcing the code: tension modulates signaling to drive morphogenesis and malignancy. Genes Dev. 39, 163–181 (2025).
Tharp, K. M. et al. Tumor-associated macrophages restrict CD8(+) T cell function through collagen deposition and metabolic reprogramming of the breast cancer microenvironment. Nat. Cancer 5, 1045–1062 (2024).
Jeffreys, N., Brockman, J. M., Zhai, Y., Ingber, D. E. & Mooney, D. J. Mechanical forces amplify TCR mechanotransduction in T cell activation and function. Appl. Phys. Rev. 11, 011304 (2024).
Ma, S. et al. A role of PIEZO1 in iron metabolism in mice and humans. Cell 184, 969–982.e913 (2021).
Reis-Rodrigues, P. et al. Migrating immune cells globally coordinate protrusive forces. Nat. Immunol. 26, 1258–1266 (2025).
Fritzsche, M. & Kruse, K. Mechanical force matters in early T cell activation. Proc. Natl. Acad. Sci. USA 121, e2404748121 (2024).
Dustin, M. L. & Kam, L. C. Tapping out a mechanical code for T cell triggering. J. Cell Biol. 213, 501–503 (2016).
Zhang, J. et al. Osr2 functions as a biomechanical checkpoint to aggravate CD8(+) T cell exhaustion in tumor. Cell 187, 3409–3426.e3424 (2024).
Meizlish, M. L. et al. Mechanosensing regulates tissue repair program in macrophages. Sci. Adv. 10, eadk6906 (2024).
Lewis, J. S. et al. The effect of cyclic mechanical strain on activation of dendritic cells cultured on adhesive substrates. Biomaterials 34, 9063–9070 (2013).
Golo, M., Newman, P. L. H., Kempe, D. & Biro, M. Mechanoimmunology in the solid tumor microenvironment. Biochem Soc. Trans. 52, 1489–1502 (2024).
Jain, N., Lord, J. M. & Vogel, V. Mechanoimmunology: Are inflammatory epigenetic states of macrophages tuned by biophysical factors? APL Bioeng. 6, 031502 (2022).
Zhao, L., Gui, Y. & Deng, X. Focus on mechano-immunology: new direction in cancer treatment. Int J. Surg. 111, 2590–2602 (2025).
Wang, Z. et al. Gut mechanoimmunology: shaping immune response through physical cues. Phys. Life Rev. 50, 13–26 (2024).
Pageon, S. V., Govendir, M. A., Kempe, D. & Biro, M. Mechanoimmunology: molecular-scale forces govern immune cell functions. Mol. Biol. Cell 29, 1919–1926 (2018).
He, Y. et al. Nanoscale acoustic oscillator for mechanoimmunology: NAOMI. Sci. Adv. 11, eadx3851 (2025).
Mukhopadhyay, M. Immunomechanics. Nat. Methods 20, 35 (2023).
Janssen, E., van den Dries, K., Ventre, M. & Cambi, A. Mechanobiology of myeloid cells. Curr. Opin. Cell Biol. 86, 102311 (2024).
Lee, M., Du, H., Winer, D. A., Clemente-Casares, X. & Tsai, S. Mechanosensing in macrophages and dendritic cells in steady-state and disease. Front. Cell Dev. Biol. 10, 1044729 (2022).
Hong, J. et al. A TCR mechanotransduction signaling loop induces negative selection in the thymus. Nat. Immunol. 19, 1379–1390 (2018).
Comrie, W. A., Babich, A. & Burkhardt, J. K. F-actin flow drives affinity maturation and spatial organization of LFA-1 at the immunological synapse. J. Cell Biol. 208, 475–491 (2015).
Luo, B. H., Carman, C. V. & Springer, T. A. Structural basis of integrin regulation and signaling. Annu. Rev. Immunol. 25, 619–647 (2007).
Choi, H. K. & Zhu, C. Catch bonds in immunology. Annu. Rev. Immunol. 43, 641–666 (2025).
Ezzo, M. et al. Acute contact with profibrotic macrophages mechanically activates fibroblasts via alphavbeta3 integrin-mediated engagement of Piezo1. Sci. Adv. 10, eadp4726 (2024).
Atcha, H. et al. Mechanically activated ion channel Piezo1 modulates macrophage polarization and stiffness sensing. Nat. Commun. 12, 3256 (2021).
Sreejit, G., Fleetwood, A. J., Murphy, A. J. & Nagareddy, P. R. Origins and diversity of macrophages in health and disease. Clin. Transl. Immunol. 9, e1222 (2020).
Ni, Y. et al. Macrophages modulate stiffness-related foreign body responses through plasma membrane deformation. Proc. Natl. Acad. Sci. USA 120, e2213837120 (2023).
Ranade, S. S., Syeda, R. & Patapoutian, A. Mechanically activated ion channels. Neuron 87, 1162–1179 (2015).
Yang, S. et al. Membrane curvature governs the distribution of Piezo1 in live cells. Nat. Commun. 13, 7467 (2022).
Wang, J. et al. Piezo1: structural pharmacology and mechanotransduction mechanisms. Trends Pharm. Sci. 46, 752–770 (2025).
Solis, A. G. et al. Mechanosensation of cyclical force by PIEZO1 is essential for innate immunity. Nature 573, 69–74 (2019).
Wang, Y. et al. Stiffness sensing via Piezo1 enhances macrophage efferocytosis and promotes the resolution of liver fibrosis. Sci. Adv. 10, eadj3289 (2024).
Lee, J. et al. Determining the Young’s modulus of the bacterial cell envelope. ACS Biomater. Sci. Eng. 10, 2956–2966 (2024).
Islamzada, E. et al. Red blood cells with reduced deformability are selectively cleared from circulation in a mouse model. Blood Adv. 9, 2988–2996 (2025).
Sosale, N. G. et al. Cell rigidity and shape override CD47’s “self”-signaling in phagocytosis by hyperactivating myosin-II. Blood 125, 542–552 (2015).
Liu, Z. et al. Cancer cells display increased migration and deformability in pace with metastatic progression. Faseb J. 34, 9307–9315 (2020).
Joshi, H. et al. Mechanosensitivity of macrophage polarization: comparing small molecule leukadherin-1 to substrate stiffness. Front. Immunol. 16, 1420325 (2025).
Meli, V. S. et al. YAP-mediated mechanotransduction tunes the macrophage inflammatory response. Sci. Adv. 6, https://doi.org/10.1126/sciadv.abb8471 (2020).
Sridharan, R., Cavanagh, B., Cameron, A. R., Kelly, D. J. & O’Brien, F. J. Material stiffness influences the polarization state, function and migration mode of macrophages. Acta Biomater. 89, 47–59 (2019).
Mei, F. et al. Matrix stiffness regulates macrophage polarisation via the Piezo1-YAP signalling axis. Cell Prolif. 57, e13640 (2024).
Adlerz, K. M., Aranda-Espinoza, H. & Hayenga, H. N. Substrate elasticity regulates the behavior of human monocyte-derived macrophages. Eur. Biophys. 45, 301–309 (2016).
Neupane, A. S. et al. Patrolling alveolar macrophages conceal bacteria from the immune system to maintain homeostasis. Cell 183, 110–125.e111 (2020).
Hu, J. et al. Microglial Piezo1 senses Abeta fibril stiffness to restrict Alzheimer’s disease. Neuron 111, 15–29.e18 (2023).
Dooling, L. J. et al. Cooperative phagocytosis of solid tumours by macrophages triggers durable anti-tumour responses. Nat. Biomed. Eng. 7, 1081–1096 (2023).
Morva, A., Arroyo, A. B., Andreeva, L., Tapia-Abellan, A. & Luengo-Gil, G. Unleashing the power of CAR-M therapy in solid tumors: a comprehensive review. Front. Immunol. 16, 1615760 (2025).
Chao, M. P., Weissman, I. L. & Majeti, R. The CD47-SIRPalpha pathway in cancer immune evasion and potential therapeutic implications. Curr. Opin. Immunol. 24, 225–232 (2012).
Tang, Z. et al. CD47 masks pro-phagocytic ligands in cis on tumor cells to suppress antitumor immunity. Nat. Immunol. 24, 2032–2041 (2023).
Zhang, H. et al. Silencing of SIRPalpha enhances the antitumor efficacy of CAR-M in solid tumors. Cell Mol. Immunol. 21, 1335–1349 (2024).
Liu, Y. et al. Emerging phagocytosis checkpoints in cancer immunotherapy. Signal Transduct. Target Ther. 8, 104 (2023).
Yang, H., Xun, Y. & You, H. The landscape overview of CD47-based immunotherapy for hematological malignancies. Biomark. Res. 11, 15 (2023).
Jiang, C. et al. Targeting the CD47/SIRPalpha pathway in malignancies: recent progress, difficulties and future perspectives. Front. Oncol. 14, 1378647 (2024).
ClinicalTrials.gov. CD47 Clinical Trials. https://clinicaltrials.gov/search?cond=cd47 (2025).
Muntjewerff, E. M., Meesters, L. D. & van den Bogaart, G. Antigen cross-presentation by macrophages. Front. Immunol. 11, 1276 (2020).
Arnold, C. E., Gordon, P., Barker, R. N. & Wilson, H. M. The activation status of human macrophages presenting antigen determines the efficiency of Th17 responses. Immunobiology 220, 10–19 (2015).
Silva, M. et al. A particulate saponin/TLR agonist vaccine adjuvant alters lymph flow and modulates adaptive immunity. Sci. Immunol. 6, eabf1152 (2021).
Liu, H. et al. Structure-based programming of lymph-node targeting in molecular vaccines. Nature 507, 519–522 (2014).
Irvine, D. J., Aung, A. & Silva, M. Controlling timing and location in vaccines. Adv. Drug Deliv. Rev. 158, 91–115 (2020).
Dash, S. P., Gupta, S. & Sarangi, P. P. Monocytes and macrophages: origin, homing, differentiation, and functionality during inflammation. Heliyon 10, e29686 (2024).
Hoffmann, E. J. & Ponik, S. M. Biomechanical contributions to macrophage activation in the tumor microenvironment. Front. Oncol. 10, 787 (2020).
McNally, A. K. & Anderson, J. M. Beta1 and beta2 integrins mediate adhesion during macrophage fusion and multinucleated foreign body giant cell formation. Am. J. Pathol. 160, 621–630 (2002).
Gui, P. et al. The protease-dependent mesenchymal migration of tumor-associated macrophages as a target in cancer immunotherapy. Cancer Immunol. Res 6, 1337–1351 (2018).
Friedl, P. & Wolf, K. Plasticity of cell migration: a multiscale tuning model. J. Cell Biol. 188, 11–19 (2010).
Friedl, P. & Wolf, K. Proteolytic and non-proteolytic migration of tumour cells and leucocytes. Biochem. Soc. Symp. 277–285. https://doi.org/10.1042/bss0700277 (2003).
Travnickova, J. et al. Macrophage morphological plasticity and migration is Rac signalling and MMP9 dependant. Sci. Rep. 11, 10123 (2021).
Sanz-Moreno, V. Tumour invasion: a new twist on Rac-driven mesenchymal migration. Curr. Biol. 22, R449–R451 (2012).
Friedl, P. & Weigelin, B. Interstitial leukocyte migration and immune function. Nat. Immunol. 9, 960–969 (2008).
Adebowale, K. et al. Monocytes use protrusive forces to generate migration paths in viscoelastic collagen-based extracellular matrices. Proc. Natl. Acad. Sci. USA 122, e2309772122 (2025).
Argyle, D. & Kitamura, T. Targeting macrophage-recruiting chemokines as a novel therapeutic strategy to prevent the progression of solid tumors. Front. Immunol. 9, 2629 (2018).
Sica, A. & Mantovani, A. Macrophage plasticity and polarization: in vivo veritas. J. Clin. Invest. 122, 787–795 (2012).
Son, B., Lee, W., Kim, H., Shin, H. & Park, H. H. Targeted therapy of cancer stem cells: inhibition of mTOR in pre-clinical and clinical research. Cell Death Dis. 15, 696 (2024).
Panwar, V. et al. Multifaceted role of mTOR (mammalian target of rapamycin) signaling pathway in human health and disease. Signal Transduct. Target Ther. 8, 375 (2023).
Lee, H. W., Choi, H. J., Ha, S. J., Lee, K. T. & Kwon, Y. G. Recruitment of monocytes/macrophages in different tumor microenvironments. Biochim. Biophys. Acta 1835, 170–179 (2013).
Du, W. et al. High-motility pro-tumorigenic monocytes drive macrophage enrichment in the tumor microenvironment. Preprint at https://doi.org/10.1101/2024.07.16.603739 (2024).
Khurana, A. et al. Visualization of macrophage recruitment in head and neck carcinoma model using fluorine-19 magnetic resonance imaging. Magn. Reson Med 79, 1972–1980 (2018).
Yang, H. et al. Magnetic Resonance Imaging of Macrophage Response to Radiation Therapy. Cancers 15, https://doi.org/10.3390/cancers15245874 (2023).
Sugimoto, M. A., Sousa, L. P., Pinho, V., Perretti, M. & Teixeira, M. M. Resolution of inflammation: What Controls Its Onset? Front. Immunol. 7, 160 (2016).
Serhan, C. N. & Savill, J. Resolution of inflammation: the beginning programs the end. Nat. Immunol. 6, 1191–1197 (2005).
Zaveri, T. D., Lewis, J. S., Dolgova, N. V., Clare-Salzler, M. J. & Keselowsky, B. G. Integrin-directed modulation of macrophage responses to biomaterials. Biomaterials 35, 3504–3515 (2014).
Zaveri, T. D. et al. Macrophage integrins modulate response to ultra-high molecular weight polyethylene particles and direct particle-induced osteolysis. Biomaterials 115, 128–140 (2017).
Haskill, S., Yurochko, A. D. & Isaacs, K. L. Regulation of macrophage infiltration and activation in sites of chronic inflammation. Ann. N. Y Acad. Sci. 664, 93–102 (1992).
McCarthy, J. B., Vachhani, B. V., Wahl, S. M., Finbloom, D. S. & Feldman, G. M. Human monocyte binding to fibronectin enhances IFN-gamma-induced early signaling events. J. Immunol. 159, 2424–2430 (1997).
Schmid, M. C. et al. Integrin CD11b activation drives anti-tumor innate immunity. Nat. Commun. 9, 5379 (2018).
Atcha, H. et al. Crosstalk between CD11b and Piezo1 mediates macrophage responses to mechanical cues. Front. Immunol. 12, 689397 (2021).
Yao, M. et al. Force- and cell state-dependent recruitment of Piezo1 drives focal adhesion dynamics and calcium entry. Sci. Adv. 8, eabo1461 (2022).
Lei, M. et al. Cell-cell and cell-matrix adhesion regulated by Piezo1 is critical for stiffness-dependent DRG neuron aggregation. Cell Rep. 42, 113522 (2023).
Wang, Y. et al. Macrophage-derived extracellular vesicles: diverse mediators of pathology and therapeutics in multiple diseases. Cell Death Dis. 11, 924 (2020).
Kumar, M. A. et al. Extracellular vesicles as tools and targets in therapy for diseases. Signal Transduct. Target Ther. 9, 27 (2024).
Cianciaruso, C. et al. Molecular profiling and functional analysis of macrophage-derived tumor extracellular vesicles. Cell Rep. 27, 3062–3080.e3011 (2019).
Zhou, J. et al. Exosomes released from tumor-associated macrophages transfer miRNAs that induce a Treg/Th17 cell imbalance in epithelial ovarian cancer. Cancer Immunol. Res. 6, 1578–1592 (2018).
Prestwich, R. J. et al. The immune system-is it relevant to cancer development, progression and treatment? Clin. Oncol. 20, 101–112 (2008).
Virchow, R. Cellular pathology. As based upon physiological and pathological histology. Lecture XVI-Atheromatous affection of arteries. 1858. Nutr. Rev. 47, 23–25 (1989).
Zhang, Y. & Zhang, Z. The history and advances in cancer immunotherapy: understanding the characteristics of tumor-infiltrating immune cells and their therapeutic implications. Cell Mol. Immunol. 17, 807–821 (2020).
Coley, W. B. I. I. Contribution to the Knowledge of Sarcoma. Ann. Surg. 14, 199–220 (1891).
McCarthy, E. F. The toxins of William B. Coley and the treatment of bone and soft-tissue sarcomas. Iowa Orthop. J. 26, 154–158 (2006).
Idos, G. E. et al. The prognostic implications of tumor infiltrating lymphocytes in colorectal cancer: a systematic review and meta-analysis. Sci. Rep. 10, 3360 (2020).
Bida, M., Miya, T. V., Hull, R. & Dlamini, Z. Tumor-infiltrating lymphocytes in melanoma: from prognostic assessment to therapeutic applications. Front. Immunol. 15, 1497522 (2024).
Ozcan, D. et al. Prognostic value of tumor-infiltrating lymphocytes in irradiated node-positive breast cancer patients. Breast 83, 104525 (2025).
Du, M., Cai, Y. M., Yin, Y. L., Xiao, L. & Ji, Y. Evaluating tumor-infiltrating lymphocytes in hepatocellular carcinoma using hematoxylin and eosin-stained tumor sections. World J. Clin. Cases 10, 856–869 (2022).
Fidler, I. J. Therapy of cancer metastasis by systemic activation of macrophages. Adv. Pharm. 30, 271–326 (1994).
Fidler, I. J. & Kleinerman, E. S. Therapy of cancer metastasis by systemic activation of macrophages: from the bench to the clinic. Res Immunol. 144, 284–287 (1993).
Gentles, A. J. et al. The prognostic landscape of genes and infiltrating immune cells across human cancers. Nat. Med. 21, 938–945 (2015).
Canella, A. & Rajappa, P. Therapeutic utility of engineered myeloid cells in the tumor microenvironment. Cancer Gene Ther. 30, 964–972 (2023).
Clevenger, A. J., Jha, A., Moore, E. & Raghavan, S. A. Manipulating immune activity of macrophages: a materials and mechanics perspective. Trends Biotechnol. 43, 131–144 (2025).
Noy, R. & Pollard, J. W. Tumor-associated macrophages: from mechanisms to therapy. Immunity 41, 49–61 (2014).
Pittet, M. J., Michielin, O. & Migliorini, D. Clinical relevance of tumour-associated macrophages. Nat. Rev. Clin. Oncol. 19, 402–421 (2022).
Hochstadt, J., Martinez Pacheco, S. & Casanova-Acebes, M. Embracing diversity: macrophage complexity in cancer. Trends Cancer 11, 351–364 (2025).
Brownlie, D. et al. Metastasis-associated macrophages constrain antitumor capability of natural killer cells in the metastatic site at least partially by membrane bound transforming growth factor beta. J. Immunother. Cancer 9, https://doi.org/10.1136/jitc-2020-001740 (2021).
Timperi, E. et al. Lipid-associated macrophages are induced by cancer-associated fibroblasts and mediate immune suppression in breast cancer. Cancer Res. 82, 3291–3306 (2022).
Guzman, G., Reed, M. R., Bielamowicz, K., Koss, B. & Rodriguez, A. CAR-T therapies in solid tumors: opportunities and challenges. Curr. Oncol. Rep. 25, 479–489 (2023).
Peng, L., Sferruzza, G., Yang, L., Zhou, L. & Chen, S. CAR-T and CAR-NK as cellular cancer immunotherapy for solid tumors. Cell Mol. Immunol. 21, 1089–1108 (2024).
Yan, T., Zhu, L. & Chen, J. Current advances and challenges in CAR T-Cell therapy for solid tumors: tumor-associated antigens and the tumor microenvironment. Exp. Hematol. Oncol. 12, 14 (2023).
Anderson, N. R., Minutolo, N. G., Gill, S. & Klichinsky, M. Macrophage-based approaches for cancer immunotherapy. Cancer Res. 81, 1201–1208 (2021).
Lu, J. et al. CAR Macrophages: a promising novel immunotherapy for solid tumors and beyond. Biomark. Res. 12, 86 (2024).
Klichinsky, M. et al. Human chimeric antigen receptor macrophages for cancer immunotherapy. Nat. Biotechnol. 38, 947–953 (2020).
Sunseri, N., O’Brien, M., Bhardwaj, N. & Landau, N. R. Human immunodeficiency virus type 1 modified to package Simian immunodeficiency virus Vpx efficiently infects macrophages and dendritic cells. J. Virol. 85, 6263–6274 (2011).
Bobadilla, S., Sunseri, N. & Landau, N. R. Efficient transduction of myeloid cells by an HIV-1-derived lentiviral vector that packages the Vpx accessory protein. Gene Ther. 20, 514–520 (2013).
Reiss, K. A. et al. CAR-macrophage therapy for HER2-overexpressing advanced solid tumors: a phase 1 trial. Nat. Med. 31, 1171–1182 (2025).
Chen, Y. et al. CAR-macrophage: a new immunotherapy candidate against solid tumors. Biomed. Pharmacother. 139, 111605 (2021).
Morrissey, M. A. et al. Chimeric antigen receptors that trigger phagocytosis. Elife 7, https://doi.org/10.7554/eLife.36688 (2018).
Schlam, D. et al. Phosphoinositide 3-kinase enables phagocytosis of large particles by terminating actin assembly through Rac/Cdc42 GTPase-activating proteins. Nat. Commun. 6, 8623 (2015).
Dooling, L. J. et al. Clustered macrophages cooperate to eliminate tumors via coordinated intrudopodia. Proc. Natl. Acad. Sci. USA 122, e2425452122 (2025).
Chen, S. et al. CAR macrophages with built-In CD47 blocker combat tumor antigen heterogeneity and activate T cells via cross-presentation. Nat. Commun. 16, 4069 (2025).
Polak, R. et al. Liposome-loaded cell backpacks. Adv. Health. Mater. 4, 2832–2841 (2015).
Xu, L., Zolotarskaya, O. Y., Yeudall, W. A. & Yang, H. Click hybridization of immune cells and polyamidoamine dendrimers. Adv. Health. Mater. 3, 1430–1438 (2014).
Ayer, M. & Klok, H. A. Cell-mediated delivery of synthetic nano- and microparticles. J. Control Release 259, 92–104 (2017).
Shields, C. W. T. et al. Cellular backpacks for macrophage immunotherapy. Sci. Adv. 6, eaaz6579 (2020).
Zhao, Z. et al. Engineering of living cells with polyphenol-functionalized biologically active nanocomplexes. Adv. Mater. 32, e2003492 (2020).
Kwan, M. M. C. et al. Particle Shape Modulates the Function of Adoptive Macrophage Transfers. Adv. Healthc. Mater. e01348, https://doi.org/10.1002/adhm.202501348 (2025).
Rodell, C. B. et al. TLR7/8-agonist-loaded nanoparticles promote the polarization of tumour-associated macrophages to enhance cancer immunotherapy. Nat. Biomed. Eng. 2, 578–588 (2018).
Griffin, K. V., Saunders, M. N., Lyssiotis, C. A. & Shea, L. D. Engineering immunity using metabolically active polymeric nanoparticles. Trends Biotechnol. 43, 1371–1384 (2025).
Bender, E. C., Sircar, A. J., Taubenfeld, E. K. & Suggs, L. J. Modulating lipid-polymer nanoparticles’ physicochemical properties to alter macrophage uptake. ACS Biomater. Sci. Eng. 10, 2911–2924 (2024).
Kraynak, C. A., Yan, D. J. & Suggs, L. J. Modulating inflammatory macrophages with an apoptotic body-inspired nanoparticle. Acta Biomater. 108, 250–260 (2020).
Li, S. et al. Nanomedicine engulfed by macrophages for targeted tumor therapy. Int. J. Nanomed. 11, 4107–4124 (2016).
Nguyen, V. D. et al. Macrophage-mediated delivery of multifunctional nanotherapeutics for synergistic chemo-photothermal therapy of solid tumors. ACS Appl. Mater. Interfaces 12, 10130–10141 (2020).
Ramesh, A., Kumar, S., Nandi, D. & Kulkarni, A. CSF1R- and SHP2-inhibitor-loaded nanoparticles enhance cytotoxic activity and phagocytosis in tumor-associated macrophages. Adv. Mater. 31, e1904364 (2019).
Wesolowski, R. et al. Phase Ib study of the combination of pexidartinib (PLX3397), a CSF-1R inhibitor, and paclitaxel in patients with advanced solid tumors. Ther. Adv. Med Oncol. 11, 1758835919854238 (2019).
Mitchell, M. J. & King, M. R. Leukocytes as carriers for targeted cancer drug delivery. Expert Opin. Drug Deliv. 12, 375–392 (2015).
Wang, L. L. et al. Preclinical characterization of macrophage-adhering gadolinium micropatches for MRI contrast after traumatic brain injury in pigs. Sci. Transl. Med. 16, eadk5413 (2024).
Lin, F., Luo, H., Wang, J., Li, Q. & Zha, L. Macrophage-derived extracellular vesicles as new players in chronic non-communicable diseases. Front. Immunol. 15, 1479330 (2024).
Zou, Y., Zhou, Y., Li, G., Dong, Y. & Hu, S. Clinical applications of extracellular vesicles: recent advances and emerging trends. Front. Bioeng. Biotechnol. 13, 1671963 (2025).
Anfray, C., Ummarino, A., Andon, F. T. & Allavena, P. Current strategies to target tumor-associated-macrophages to improve anti-tumor immune responses. Cells 9, https://doi.org/10.3390/cells9010046 (2019).
Sun, M. et al. Adoptive cell therapy with macrophage-drug conjugates facilitates cytotoxic drug transfer and immune activation in glioblastoma models. Sci. Transl. Med. 17, eadr4058 (2025).
Huang, W. et al. Clinical pharmacology perspectives for adoptive cell therapies in oncology. Clin. Pharm. Ther. 112, 968–981 (2022).
Li, R., Grosskopf, A. K., Joslyn, L. R., Stefanich, E. G. & Shivva, V. Cellular kinetics and biodistribution of adoptive T cell therapies: from biological principles to effects on patient outcomes. AAPS J. 27, 55 (2025).
Veerasubramanian, P. K. et al. A Src-H3 acetylation signaling axis integrates macrophage mechanosensation with inflammatory response. Biomaterials 279, 121236 (2021).
Jain, N., Moeller, J. & Vogel, V. Mechanobiology of macrophages: how physical factors coregulate macrophage plasticity and phagocytosis. Annu Rev. Biomed. Eng. 21, 267–297 (2019).
Sapudom, J. et al. 3D in vitro M2 macrophage model to mimic modulation of tissue repair. NPJ Regen. Med 6, 83 (2021).
Burchett, A., Siri, S., Li, J., Lu, X. & Datta, M. Novel 3-D macrophage spheroid model reveals reciprocal regulation of immunomechanical stress and mechano-immunological response. Cell Mol. Bioeng. 17, 329–344 (2024).
Burchett, A., Chen, H., Najera, J., Howard, S. & Datta, M. Chronic compression drives macrophages toward a pathological pro-tumor state. Preprint at https://doi.org/10.1101/2025.05.13.653793 (2025).
Liu, Y. & Segura, T. Biomaterials-mediated regulation of macrophage cell fate. Front. Bioeng. Biotechnol. 8, 609297 (2020).
Graf, J. et al. Macrophage variance: investigating how macrophage origin influences responses to soluble and physical cues with immortalized vs. primary cells in 2D and 3D culture. Front. Biomater. Sci. 3, https://doi.org/10.3389/fbiom.2024.1399448 (2024).
Guillot, A. & Tacke, F. Liver macrophages revisited: The expanding universe of versatile responses in a spatiotemporal context. Hepatol. Commun. 8, https://doi.org/10.1097/HC9.0000000000000491 (2024).
Stark, C. J S. E. N, M. T. -Omics approaches to study and model cell-cell interactions in engineered tissues. Front. Chem. Eng. 7, https://doi.org/10.3389/fceng.2025.1629455.
Ma, C. et al. Bioengineered immunocompetent preclinical trial-on-chip tool enables screening of CAR T cell therapy for leukaemia. Nat. Biomed. Eng. https://doi.org/10.1038/s41551-025-01428-2 (2025).
Knight, H. R. et al. Bioengineering approaches to trained immunity: Physiologic targets and therapeutic strategies. Elife 14, https://doi.org/10.7554/eLife.106339 (2025).
Xiang, Y. et al. Multifaceted cancer alleviation by cowpea mosaic virus in a bioprinted ovarian cancer peritoneal spheroid model. Biomaterials 311, 122663 (2024).
Tang, M. et al. Integration of 3D bioprinting and multi-algorithm machine learning identified glioma susceptibilities and microenvironment characteristics. Cell Discov. 10, 39 (2024).
Staros, R. et al. Perspectives for 3D-bioprinting in modeling of tumor immune evasion. Cancers 14, https://doi.org/10.3390/cancers14133126 (2022).
Wang, X., Luo, Y., Ma, Y., Wang, P. & Yao, R. Converging bioprinting and organoids to better recapitulate the tumor microenvironment. Trends Biotechnol. 42, 648–663 (2024).
Jung, M., Ghamrawi, S., Du, E. Y., Gooding, J. J. & Kavallaris, M. Advances in 3D bioprinting for cancer biology and precision medicine: from matrix design to application. Adv. Health. Mater. 11, e2200690 (2022).
Li, Y. C, H.; Cui, H. Precision Spatial Control of Tumor-Stroma Interactions in Cancer Models via 3D Bioprinting for Advanced Research and Therapy. Adv. Funct. Mater. https://doi.org/10.1002/adfm.202503391 (2025).
Datta, P., Dey, M., Ataie, Z., Unutmaz, D. & Ozbolat, I. T. 3D bioprinting for reconstituting the cancer microenvironment. NPJ Precis Oncol. 4, 18 (2020).
Zhang, Z., Chen, X., Gao, S., Fang, X. & Ren, S. 3D bioprinted tumor model: a prompt and convenient platform for overcoming immunotherapy resistance by recapitulating the tumor microenvironment. Cell Oncol. 47, 1113–1126 (2024).
Visalakshan, R. M. et al. Opportunities and challenges to engineer 3D models of tumor-adaptive immune interactions. Front. Immunol. 14, 1162905 (2023).
Mu, P. et al. Newly developed 3D in vitro models to study tumor-immune interaction. J. Exp. Clin. Cancer Res. 42, 81 (2023).
Ahmed, T. Biomaterial-based in vitro 3D modeling of glioblastoma multiforme. Cancer Pathog. Ther. 1, 177–194 (2023).
Huang, M. S. C, F.; Roth, J. G., Heilshorn, S. C. Organoid bioprinting: from cells to functional tissues. Nat. Rev. Bioeng. https://doi.org/10.1038/s44222-024-00268-0 (2024).
Zhang, D., Huerta-Lopez, C. & Heilshorn, S. C. Organoid bioprinting to pattern the matrix microenvironment. Curr. Opin. Biomed. Eng. 35, https://doi.org/10.1016/j.cobme.2025.100607 (2025).
Hanif, F., Muzaffar, K., Perveen, K., Malhi, S. M. & Simjee Sh, U. Glioblastoma multiforme: a review of its epidemiology and pathogenesis through clinical presentation and treatment. Asian Pac. J. Cancer Prev. 18, 3–9 (2017).
Pierini, S. et al. Chimeric antigen receptor macrophages (CAR-M) sensitize HER2+ solid tumors to PD1 blockade in pre-clinical models. Nat. Commun. 16, 706 (2025).
Bhatia, D., Dolcetti, R. & Mazzieri, R. Are monocytes a preferable option to develop myeloid cell-based therapies for solid tumors? J. Exp. Clin. Cancer Res 44, 98 (2025).
Billingsley, M. M. et al. In vivo mRNA CAR T cell engineering via targeted ionizable lipid nanoparticles with extrahepatic tropism. Small 20, e2304378 (2024).
Yang, S. et al. Advances in engineered macrophages: a new frontier in cancer immunotherapy. Cell Death Dis. 15, 238 (2024).
Na, Y. R., Kim, S. W. & Seok, S. H. A new era of macrophage-based cell therapy. Exp. Mol. Med. 55, 1945–1954 (2023).





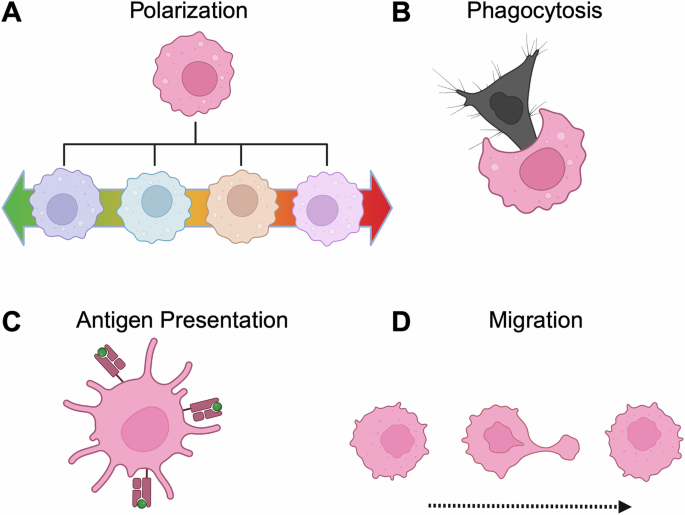












Leave a Reply