Burrell, C. J., Howard, C. R. & Murphy, F. A. in Fenner and White’s Medical Virology 5th edn (eds Burrell, C. J., Howard, C. R. & Murphy, F. A.) 237–261 (Academic, 2017).
Bhowmik, D. & Zhu, F. Evasion of intracellular DNA sensing by human herpesviruses. Front. Cell. Infect. Microbiol. 11, 647992 (2021).
Berry, R., Watson, G. M., Jonjic, S., Degli-Esposti, M. A. & Rossjohn, J. Modulation of innate and adaptive immunity by cytomegaloviruses. Nat. Rev. Immunol. 20, 113–127 (2020).
Griffin, B. D., Verweij, M. C. & Wiertz, E. J. H. J. Herpesviruses and immunity: the art of evasion. Vet. Microbiol. 143, 89–100 (2010).
Lange, P. T., White, M. C. & Damania, B. Activation and evasion of innate immunity by gammaherpesviruses. J. Mol. Biol. 434, 167214 (2022).
Silva, J.dM., Alves, C. E.dC. & Pontes, G. S. Epstein–Barr virus: the mastermind of immune chaos. Front. Immunol. 15, 1297994 (2024).
Lučin, P., Mahmutefendić, H., Blagojević Zagorac, G. & Ilić Tomaš, M. Cytomegalovirus immune evasion by perturbation of endosomal trafficking. Cell. Mol. Immunol. 12, 154–169 (2015).
Cohen, J. I. Herpesvirus latency. J. Clin. Invest. 130, 3361–3369 (2020).
Vietzen, H. et al. Ineffective control of Epstein–Barr-virus-induced autoimmunity increases the risk for multiple sclerosis. Cell 186, 5705–5718.e13 (2023).
Romanescu, C., Schreiner, T. G. & Mukovozov, I. The role of human herpesvirus 6 infection in Alzheimer’s disease pathogenicity — a theoretical mosaic. J. Clin. Med. 11, 3061 (2022).
Mangold, C. A. & Szpara, M. L. Persistent infection with herpes simplex virus 1 and Alzheimer’s disease — a call to study how variability in both virus and host may impact disease. Viruses 11, 966 (2019).
Brown, Z. A. et al. Effect of serologic status and cesarean delivery on transmission rates of herpes simplex virus from mother to infant. JAMA 289, 203–209 (2003).
Salomè, S. et al. Congenital cytomegalovirus infection: the state of the art and future perspectives. Front. Pediatr. 11, 1276912 (2023).
Shah, R. A. et al. Shingrix for herpes zoster: a review. Skin Therapy Lett. 24, 5–7 (2019).
Creed, R., Satyaprakash, A. & Ravanfar, P. Varicella zoster vaccines. Dermatol. Ther. 22, 143–149 (2009).
Patil, A., Goldust, M. & Wollina, U. Herpes zoster: a review of clinical manifestations and management. Viruses 14, 192 (2022).
Freer, G. & Pistello, M. Varicella-zoster virus infection: natural history, clinical manifestations, immunity and current and future vaccination strategies. New Microbiol. 41, 95–105 (2018).
Mbinta, J. F., Nguyen, B. P., Awuni, P. M. A., Paynter, J. & Simpson, C. R. Post-licensure zoster vaccine effectiveness against herpes zoster and postherpetic neuralgia in older adults: a systematic review and meta-analysis. Lancet Healthy Longev. 3, e263–e275 (2022).
Wutzler, P. et al. Varicella vaccination — the global experience. Expert Rev. Vaccines 16, 833–843 (2017).
Shapiro, E. D. & Marin, M. The effectiveness of varicella vaccine: 25 years of postlicensure experience in the United States. J. Infect. Dis. 226, S425–S430 (2022).
Goldman, G. S. & King, P. G. Review of the United States universal varicella vaccination program: herpes zoster incidence rates, cost-effectiveness, and vaccine efficacy based primarily on the antelope valley varicella active surveillance project data. Vaccine 31, 1680–1694 (2013).
Shu, M., Zhang, D., Ma, R., Yang, T. & Pan, X. Long-term vaccine efficacy of a 2-dose varicella vaccine in China from 2011 to 2021: a retrospective observational study. Front. Public Health 10, 1039537 (2022).
Hardy, I., Gershon, A. A., Steinberg, S. P., LaRussa, P. & Varicella Vaccine Collaborative Study Group. The incidence of zoster after immunization with live attenuated varicella vaccine — a study in children with leukemia. N. Engl. J. Med. 325, 1545–1550 (1991).
Weinmann, S. et al. Incidence and clinical characteristics of herpes zoster among children in the varicella vaccine era, 2005–2009. J. Infect. Dis. 208, 1859–1868 (2013).
Kosinska, A. D., Bauer, T. & Protzer, U. Therapeutic vaccination for chronic hepatitis B. Curr. Opin. Virol. 23, 75–81 (2017).
Wang, L., Zhu, L. & Zhu, H. Efficacy of varicella (VZV) vaccination: an update for the clinician. Ther. Adv. Vaccines 4, 20–31 (2016).
US Centers for Disease Control and Prevention. Shingles vaccination. CDC https://www.cdc.gov/shingles/vaccines/index.html (2025).
Lal, H. et al. Efficacy of an adjuvanted herpes zoster subunit vaccine in older adults. N. Engl. J. Med. 372, 2087–2096 (2015).
Leroux-Roels, I. et al. A phase 1/2 clinical trial evaluating safety and immunogenicity of a varicella zoster glycoprotein E subunit vaccine candidate in young and older adults. J. Infect. Dis. 206, 1280–1290 (2012).
Asada, H. VZV-specific cell-mediated immunity, but not humoral immunity, correlates inversely with the incidence of herpes zoster and the severity of skin symptoms and zoster-associated pain: the SHEZ study. Vaccine 37, 6776–6781 (2019).
Boppana, S. B. et al. Vaccine value profile for cytomegalovirus. Vaccine 41, S53–S75 (2023).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT05085366 (2025).
ACCESS Newswire. Moderna provides business and pipeline updates at 43rd Annual J.P. Morgan Healthcare Conference. ACCESS Newswire https://www.accessnewswire.com/newsroom/en/healthcare-and-pharmaceutical/moderna-provides-business-and-pipeline-updates-at-43rd-annual-j.p.-mor-967378 (2025).
ACCESS Newswire. Moderna announces phase 3 study of investigational cytomegalovirus (CMV) vaccine did not meet primary efficacy endpoint. ACCESS Newswire https://www.accessnewswire.com/newsroom/en/healthcare-and-pharmaceutical/moderna-announces-phase-3-study-of-investigational-cytomegalovirus-cm-1090841 (2025).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT05683457 (2025).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT05164094 (2025).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT06735248 (2025).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04645147 (2025).
Dasari, V. et al. Lymph node targeted multi-epitope subunit vaccine promotes effective immunity to EBV in HLA-expressing mice. Nat. Commun. 14, 4371 (2023).
Bai, L., Xu, J., Zeng, L., Zhang, L. & Zhou, F. A review of HSV pathogenesis, vaccine development, and advanced applications. Mol. Biomed. 5, 35 (2024).
Jarosinski, K. W. Interindividual spread of herpesviruses. Adv. Anat. Embryol. Cell Biol. 223, 195–224 (2017).
Fujiwara, S. Animal models of human gammaherpesvirus infections. Adv. Exp. Med. Biol. 1045, 413–436 (2018).
Kollias, C. M., Huneke, R. B., Wigdahl, B. & Jennings, S. R. Animal models of herpes simplex virus immunity and pathogenesis. J. Neurovirol. 21, 8–23 (2015).
Myers, M. G. & Connelly, B. L. Animal models of varicella. J. Infect. Dis. 166 (Suppl. 1), S48–S50 (1992).
Roark, H. K., Jenks, J. A., Permar, S. R. & Schleiss, M. R. Animal models of congenital cytomegalovirus transmission: implications for vaccine development. J. Infect. Dis. 221, S60–S73 (2020).
Johnson, D. C. & Hill, A. B. Herpesvirus evasion of the immune system. Curr. Top. Microbiol. Immunol. 232, 149–177 (1998).
Sausen, D. G., Reed, K. M., Bhutta, M. S., Gallo, E. S. & Borenstein, R. Evasion of the host immune response by betaherpesviruses. Int. J. Mol. Sci. 22, 7503 (2021).
Koyanagi, N. & Kawaguchi, Y. Evasion of the cell-mediated immune response by alphaherpesviruses. Viruses 12, 1354 (2020).
Yao, Y., Kong, W., Yang, L., Ding, Y. & Cui, H. Immunity and immune evasion mechanisms of Epstein–Barr virus. Viral Immunol. 36, 303–317 (2023).
Wahl, I. & Wardemann, H. Sterilizing immunity: understanding COVID-19. Immunity 55, 2231–2235 (2022).
Dunmire, S. K., Hogquist, K. A. & Balfour, H. H. in Epstein Barr Virus Volume 1 — One Herpes Virus: Many Diseases (ed. Münz, C.) 211–240 (Springer, 2015).
Sacks, S. L. et al. HSV shedding. Antivir. Res. 63, S19–S26 (2004).
Weber, A., Liu, M., Embree, J., Castillo, E. & Poliquin, V. Knowledge of congenital CMV, risk behaviours for CMV acquisition, and acceptance of an educational infographic among postpartum women: a pilot study. J. Obstet. Gynaecol. Can. 44, 200–203 (2022).
Holmgren, J. & Czerkinsky, C. Mucosal immunity and vaccines. Nat. Med. 11, S45–S53 (2005).
Méndez, A. C., Rodríguez-Rojas, C. & Del Val, M. Vaccine vectors: the bright side of cytomegalovirus. Med. Microbiol. Immunol. 208, 349–363 (2019).
Sitki-Green, D., Covington, M. & Raab-Traub, N. Compartmentalization and transmission of multiple Epstein–Barr virus strains in asymptomatic carriers. J. Virol. 77, 1840–1847 (2003).
Goldberg, B. S. & Ackerman, M. E. Antibody-mediated complement activation in pathology and protection. Immunol. Cell Biol. 98, 305–317 (2020).
Jenks, J. A., Goodwin, M. L. & Permar, S. R. The roles of host and viral antibody fc receptors in herpes simplex virus (HSV) and human cytomegalovirus (HCMV) infections and immunity. Front. Immunol. 10, 2110 (2019).
Chandler, T. L., Yang, A., Otero, C. E., Permar, S. R. & Caddy, S. L. Protective mechanisms of nonneutralizing antiviral antibodies. PLoS Pathog. 19, e1011670 (2023).
Roy, V. et al. Epstein–Barr-virus-specific functional antibody signatures in the context of nasopharyngeal carcinoma development. Med 7, 100925 (2026).
Semmes, E. C. et al. Maternal Fc-mediated non-neutralizing antibody responses correlate with protection against congenital human cytomegalovirus infection. J. Clin. Invest. 132, e156827 (2022).
Semmes, E. C. et al. ADCC-activating antibodies correlate with decreased risk of congenital human cytomegalovirus transmission. JCI Insight 8, e167768 (2023).
Backes, I. M., Leib, D. A. & Ackerman, M. E. Monoclonal antibody therapy of herpes simplex virus: an opportunity to decrease congenital and perinatal infections. Front. Immunol. 13, 959603 (2022).
Kohl, S. et al. Neonatal antibody-dependent cellular cytotoxic antibody levels are associated with the clinical presentation of neonatal herpes simplex virus infection. J. Infect. Dis. 160, 770–776 (1989).
Jonjić, S. et al. Antibodies are not essential for the resolution of primary cytomegalovirus infection but limit dissemination of recurrent virus. J. Exp. Med. 179, 1713–1717 (1994).
Polić, B. et al. Hierarchical and redundant lymphocyte subset control precludes cytomegalovirus replication during latent infection. J. Exp. Med. 188, 1047–1054 (1998).
Wirtz, N. et al. Polyclonal cytomegalovirus-specific antibodies not only prevent virus dissemination from the portal of entry but also inhibit focal virus spread within target tissues. Med. Microbiol. Immunol. 197, 151–158 (2008).
Klenovsek, K. et al. Protection from CMV infection in immunodeficient hosts by adoptive transfer of memory B cells. Blood 110, 3472–3479 (2007).
Nelson, C. S. et al. Preexisting antibodies can protect against congenital cytomegalovirus infection in monkeys. JCI Insight 2, e94002 (2017).
Hughes, B. L. et al. A trial of hyperimmune globulin to prevent congenital cytomegalovirus infection. N. Engl. J. Med. 385, 436–444 (2021).
Humar, A. et al. A randomized trial of ganciclovir versus ganciclovir plus immune globulin for prophylaxis against Epstein–Barr virus related posttransplant lymphoproliferative disorder. Transplantation 81, 856–861 (2006).
Kim, J. et al. Epstein–Barr virus (EBV) hyperimmune globulin isolated from donors with high gp350 antibody titers protect humanized mice from challenge with EBV. Virology 561, 80–86 (2021).
Hebeis, B. J. et al. Activation of virus-specific memory B cells in the absence of T cell help. J. Exp. Med. 199, 593–602 (2004).
Cifuentes-Munoz, N., El Najjar, F. & Dutch, R. E. Viral cell-to-cell spread: conventional and non-conventional ways. Adv. Virus Res. 108, 85–125 (2020).
Connolly, S. A., Jardetzky, T. S. & Longnecker, R. The structural basis of herpesvirus entry. Nat. Rev. Microbiol. 19, 110–121 (2021).
Leroy, H. et al. Virus-mediated cell–cell fusion. Int. J. Mol. Sci. 21, 9644 (2020).
Cole, N. L. & Grose, C. Membrane fusion mediated by herpesvirus glycoproteins: the paradigm of varicella-zoster virus. Rev. Med. Virol. 13, 207–222 (2003).
Feutz, E., McLeland-Wieser, H., Ma, J. & Roller, R. J. Functional interactions between herpes simplex virus pUL51, pUL7 and gE reveal cell-specific mechanisms for epithelial cell-to-cell spread. Virology 537, 84–96 (2019).
Bzik, D. J., Fox, B. A., DeLuca, N. A. & Person, S. Nucleotide sequence of a region of the herpes simplex virus type 1 gB glycoprotein gene: mutations affecting rate of virus entry and cell fusion. Virology 137, 185–190 (1984).
Read, G. S., Person, S. & Keller, P. M. Genetic studies of cell fusion induced by herpes simplex virus type 1. J. Virol. 35, 105–113 (1980).
Gerna, G. et al. Human cytomegalovirus replicates abortively in polymorphonuclear leukocytes after transfer from infected endothelial cells via transient microfusion events. J. Virol. 74, 5629–5638 (2000).
Turner, A., Bruun, B., Minson, T. & Browne, H. Glycoproteins gB, gD, and gHgL of herpes simplex virus type 1 are necessary and sufficient to mediate membrane fusion in a Cos cell transfection system. J. Virol. 72, 873–875 (1998).
Avitabile, E., Lombardi, G. & Campadelli-Fiume, G. Herpes simplex virus glycoprotein K, but not its syncytial allele, inhibits cell–cell fusion mediated by the four fusogenic glycoproteins, gD, gB, gH, and gL. J. Virol. 77, 6836–6844 (2003).
Alt, M. et al. Cell-to-cell spread inhibiting antibodies constitute a correlate of protection against herpes simplex virus type 1 reactivations: a retrospective study. Front. Immunol. 14, 1143870 (2023).
Hook, L. M. et al. Vaccine-induced antibodies to herpes simplex virus glycoprotein D epitopes involved in virus entry and cell-to-cell spread correlate with protection against genital disease in guinea pigs. PLoS Pathog. 14, e1007095 (2018).
Criscuolo, E. et al. Cell-to-cell spread blocking activity is extremely limited in the sera of herpes simplex virus 1 (HSV-1)- and HSV-2-infected subjects. J. Virol. 93, e00070-19 (2019).
Cui, X. et al. Impact of antibodies and strain polymorphisms on cytomegalovirus entry and spread in fibroblasts and epithelial cells. J. Virol. 91, e01650-16 (2017).
Falk, J. J. et al. Large-scale screening of HCMV-seropositive blood donors indicates that HCMV effectively escapes from antibodies by cell-associated spread. Viruses 10, 500 (2018).
Revello, M. G. et al. A randomized trial of hyperimmune globulin to prevent congenital cytomegalovirus. N. Engl. J. Med. 370, 1316–1326 (2014).
Kagan, K. O. et al. Prevention of maternal-fetal transmission of cytomegalovirus after primary maternal infection in the first trimester by biweekly hyperimmunoglobulin administration. Ultrasound Obstet. Gynecol. 53, 383–389 (2019).
Petro, C. et al. Herpes simplex type 2 virus deleted in glycoprotein D protects against vaginal, skin and neural disease. eLife 4, e06054 (2015).
Bu, W. et al. Immunization with components of the viral fusion apparatus elicits antibodies that neutralize Epstein–Barr virus in B cells and epithelial cells. Immunity 50, 1305–1316.e6 (2019).
Hilterbrand, A. T., Daly, R. E. & Heldwein, E. E. Contributions of the four essential entry glycoproteins to HSV-1 tropism and the selection of entry routes. mBio 12, e00143-21 (2021).
Cairns, T. M. et al. Dissection of the antibody response against herpes simplex virus glycoproteins in naturally infected humans. J. Virol. 88, 12612–12622 (2014).
Aschner, C. B. & Herold, B. C. Alphaherpesvirus vaccines. Curr. Issues Mol. Biol. 41, 469–508 (2021).
Burn Aschner, C. et al. HVEM signaling promotes protective antibody-dependent cellular cytotoxicity (ADCC) vaccine responses to herpes simplex viruses. Sci. Immunol. 5, eaax2454 (2020).
Ramsey, N. L. M. et al. A single-cycle glycoprotein D deletion viral vaccine candidate, ΔgD-2, elicits polyfunctional antibodies that protect against ocular herpes simplex virus. J. Virol. 94, e00335-20 (2020).
Kuraoka, M. et al. A non-neutralizing glycoprotein B monoclonal antibody protects against herpes simplex virus disease in mice. J. Clin. Invest. 133, e161968 (2023).
Komala Sari, T., Gianopulos, K. A. & Nicola, A. V. Glycoprotein C of herpes simplex virus 1 shields glycoprotein B from antibody neutralization. J. Virol. 94, e01852-19 (2020).
Machiels, B. et al. Antibody evasion by a gammaherpesvirus O-glycan shield. PLoS Pathog. 7, e1002387 (2011).
Jiang, X. J. et al. Human cytomegalovirus glycoprotein polymorphisms and increasing viral load in AIDS patients. PLoS ONE 12, e0176160 (2017).
Day, L. Z. et al. Polymorphisms in human cytomegalovirus glycoprotein O (gO) exert epistatic influences on cell-free and cell-to-cell spread and antibody neutralization on gH epitopes. J. Virol. 94, e02051-19 (2020).
Astronomo, R. D. & Burton, D. R. Carbohydrate vaccines: developing sweet solutions to sticky situations? Nat. Rev. Drug Discov. 9, 308–324 (2010).
Snapper, C. M. & Mond, J. J. A model for induction of T cell-independent humoral immunity in response to polysaccharide antigens. J. Immunol. 157, 2229–2233 (1996).
Vigerust, D. J. & Shepherd, V. L. Virus glycosylation: role in virulence and immune interactions. Trends Microbiol. 15, 211–218 (2007).
Deimel, L. P., Xue, X. & Sattentau, Q. J. Glycans in HIV-1 vaccine design — engaging the shield. Trends Microbiol. 30, 866–881 (2022).
Fukui, A. et al. Dual impacts of a glycan shield on the envelope glycoprotein B of HSV-1: evasion from human antibodies in vivo and neurovirulence. mBio 14, e009923 (2023).
Roark, R. S. et al. Prefusion structure, evasion and neutralization of HSV-1 glycoprotein B. Nat. Microbiol. 10, 2966–2980 (2025).
Wei, X. et al. Antibody neutralization and escape by HIV-1. Nature 422, 307–312 (2003).
Valencia, S. M. et al. Vaccination with a replication-defective cytomegalovirus vaccine elicits a glycoprotein B-specific monoclonal antibody repertoire distinct from natural infection. npj Vaccines 8, 154 (2023).
Gardner, T. J. & Tortorella, D. Virion glycoprotein-mediated immune evasion by human cytomegalovirus: a sticky virus makes a slick getaway. Microbiol. Mol. Biol. Rev. 80, 663–677 (2016).
Kropff, B. et al. Glycoprotein N of human cytomegalovirus protects the virus from neutralizing antibodies. PLoS Pathog. 8, e1002999 (2012).
Burke, H. G. & Heldwein, E. E. Crystal structure of the human cytomegalovirus glycoprotein B. PLoS Pathog. 11, e1005227 (2015).
Hull, M. A., Pritchard, S. M. & Nicola, A. V. Herpes simplex virus 1 envelope glycoprotein C shields glycoprotein D to protect virions from entry-blocking antibodies. J. Virol. 99, e0009025 (2025).
Sodora, D. L., Cohen, G. H. & Eisenberg, R. J. Influence of asparagine-linked oligosaccharides on antigenicity, processing, and cell surface expression of herpes simplex virus type 1 glycoprotein D. J. Virol. 63, 5184–5193 (1989).
Powers, C., DeFilippis, V., Malouli, D. & Früh, K. Cytomegalovirus immune evasion. Curr. Top. Microbiol. Immunol. 325, 333–359 (2008).
Sin, S. H. & Dittmer, D. P. Cytokine homologs of human gammaherpesviruses. J. Interferon Cytokine Res. 32, 53–59 (2012).
McSharry, B. P., Avdic, S. & Slobedman, B. Human cytomegalovirus encoded homologs of cytokines, chemokines and their receptors: roles in immunomodulation. Viruses 4, 2448–2470 (2012).
Kotenko, S. V., Saccani, S., Izotova, L. S., Mirochnitchenko, O. V. & Pestka, S. Human cytomegalovirus harbors its own unique IL-10 homolog (cmvIL-10). Proc. Natl Acad. Sci. USA 97, 1695–1700 (2000).
Spencer, J. V., Cadaoas, J., Castillo, P. R., Saini, V. & Slobedman, B. Stimulation of B lymphocytes by cmvIL-10 but not LAcmvIL-10. Virology 374, 164–169 (2008).
Spencer, J. V. et al. Potent immunosuppressive activities of cytomegalovirus-encoded interleukin-10. J. Virol. 76, 1285–1292 (2002).
Chang, W. L., Baumgarth, N., Yu, D. & Barry, P. A. Human cytomegalovirus-encoded interleukin-10 homolog inhibits maturation of dendritic cells and alters their functionality. J. Virol. 78, 8720–8731 (2004).
Raftery, M. J. et al. Shaping phenotype, function, and survival of dendritic cells by cytomegalovirus-encoded IL-10. J. Immunol. 173, 3383–3391 (2004).
Slobedman, B., Barry, P. A., Spencer, J. V., Avdic, S. & Abendroth, A. Virus-encoded homologs of cellular interleukin-10 and their control of host immune function. J. Virol. 83, 9618–9629 (2009).
Broussard, G. & Damania, B. KSHV: immune modulation and immunotherapy. Front. Immunol. 10, 3084 (2020).
Chen, Y. et al. A homotrimeric GPCR architecture of the human cytomegalovirus revealed by cryo-EM. Cell Discov. 10, 52 (2024).
Paulsen, S. J., Rosenkilde, M. M., Eugen-Olsen, J. & Kledal, T. N. Epstein–Barr virus-encoded BILF1 is a constitutively active G protein-coupled receptor. J. Virol. 79, 536–546 (2005).
Wang, D., Bresnahan, W. & Shenk, T. Human cytomegalovirus encodes a highly specific RANTES decoy receptor. Proc. Natl Acad. Sci. USA 101, 16642–16647 (2004).
Bresnahan, W. A. & Shenk, T. A subset of viral transcripts packaged within human cytomegalovirus particles. Science 288, 2373–2376 (2000).
Vomaske, J., Nelson, J. A. & Streblow, D. N. Human cytomegalovirus US28: a functionally selective chemokine binding receptor. Infect. Disord. Drug Targets 9, 548–556 (2009).
Nakano, K. et al. Human herpesvirus 7 open reading frame U12 encodes a functional β-chemokine receptor. J. Virol. 77, 8108–8115 (2003).
Isegawa, Y., Ping, Z., Nakano, K., Sugimoto, N. & Yamanishi, K. Human herpesvirus 6 open reading frame U12 encodes a functional β-chemokine receptor. J. Virol. 72, 6104–6112 (1998).
Furukawa, T., Hornberger, E., Sakuma, S. & Plotkin, S. A. Demonstration of immunoglobulin G receptors induced by human cytomegalovirus. J. Clin. Microbiol. 2, 332–336 (1975).
Baucke, R. B. & Spear, P. G. Membrane proteins specified by herpes simplex viruses. V. Identification of an Fc-binding glycoprotein. J. Virol. 32, 779–789 (1979).
Ogata, M. & Shigeta, S. Appearance of immunoglobulin G Fc receptor in cultured human cells infected with varicella-zoster virus. Infect. Immun. 26, 770–774 (1979).
Olson, J. K., Santos, R. A. & Grose, C. Varicella-zoster virus glycoprotein gE: endocytosis and trafficking of the Fc receptor. J. Infect. Dis. 178, S2–S6 (1998).
Dubin, G., Socolof, E., Frank, I. & Friedman, H. M. Herpes simplex virus type 1 Fc receptor protects infected cells from antibody-dependent cellular cytotoxicity. J. Virol. 65, 7046–7050 (1991).
Olson, J. K., Bishop, G. A. & Grose, C. Varicella-zoster virus Fc receptor gE glycoprotein: serine/threonine and tyrosine phosphorylation of monomeric and dimeric forms. J. Virol. 71, 110–119 (1997).
Dingwell, K. S. & Johnson, D. C. The herpes simplex virus gE–gI complex facilitates cell-to-cell spread and binds to components of cell junctions. J. Virol. 72, 8933–8942 (1998).
Dingwell, K. S., Doering, L. C. & Johnson, D. C. Glycoproteins E and I facilitate neuron-to-neuron spread of herpes simplex virus. J. Virol. 69, 7087–7098 (1995).
Lubinski, J. M., Lazear, H. M., Awasthi, S., Wang, F. & Friedman, H. M. The herpes simplex virus 1 IgG fc receptor blocks antibody-mediated complement activation and antibody-dependent cellular cytotoxicity in vivo. J. Virol. 85, 3239–3249 (2011).
Watkins, J. F. Adsorption of sensitized sheep erythrocytes to HeLa cells infected with herpes simplex virus. Nature 202, 1364–1365 (1964).
Johnson, D. C., Frame, M. C., Ligas, M. W., Cross, A. M. & Stow, N. D. Herpes simplex virus immunoglobulin G Fc receptor activity depends on a complex of two viral glycoproteins, gE and gI. J. Virol. 62, 1347–1354 (1988).
Chapman, T. L. et al. Characterization of the interaction between the herpes simplex virus type I Fc receptor and immunoglobulin G. J. Biol. Chem. 274, 6911–6919 (1999).
Dubin, G., Frank, I. & Friedman, H. M. Herpes simplex virus type 1 encodes two Fc receptors which have different binding characteristics for monomeric immunoglobulin G (IgG) and IgG complexes. J. Virol. 64, 2725–2731 (1990).
Adler, R., Glorioso, J. C., Cossman, J. & Levine, M. Possible role of Fc receptors on cells infected and transformed by herpesvirus: escape from immune cytolysis. Infect. Immun. 21, 442–447 (1978).
Frank, I. & Friedman, H. M. A novel function of the herpes simplex virus type 1 Fc receptor: participation in bipolar bridging of antiviral immunoglobulin G. J. Virol. 63, 4479–4488 (1989).
Friedman, H. M. Immune evasion by herpes simplex virus type 1, strategies for virus survival. Trans. Am. Clin. Climatol. Assoc. 114, 103–112 (2003).
Manley, K. et al. Human cytomegalovirus escapes a naturally occurring neutralizing antibody by incorporating it into assembling virions. Cell Host Microbe 10, 197–209 (2011).
Sprague, E. R., Wang, C., Baker, D. & Bjorkman, P. J. Crystal structure of the HSV-1 Fc receptor bound to Fc reveals a mechanism for antibody bipolar bridging. PLoS Biol. 4, e148 (2006).
Ndjamen, B., Joshi, D. S., Fraser, S. E. & Bjorkman, P. J. Characterization of antibody bipolar bridging mediated by the human cytomegalovirus Fc receptor gp68. J. Virol. 90, 3262–3267 (2016).
Brideau, A. D., Enquist, L. W. & Tirabassi, R. S. The role of virion membrane protein endocytosis in the herpesvirus life cycle. J. Clin. Virol. 17, 69–82 (2000).
Sprague, E. R., Martin, W. L. & Bjorkman, P. J. pH dependence and stoichiometry of binding to the Fc region of IgG by the herpes simplex virus Fc receptor gE–gI. J. Biol. Chem. 279, 14184–14193 (2004).
Ndjamen, B., Farley, A. H., Lee, T., Fraser, S. E. & Bjorkman, P. J. The herpes virus Fc receptor gE–gI mediates antibody bipolar bridging to clear viral antigens from the cell surface. PLoS Pathog. 10, e1003961 (2014).
Dai, H. S. et al. The Fc domain of immunoglobulin is sufficient to bridge NK cells with virally infected cells. Immunity 47, 159–170.e10 (2017).
Johansson, P. J., Myhre, E. B. & Blomberg, J. Specificity of Fc receptors induced by herpes simplex virus type 1: comparison of immunoglobulin G from different animal species. J. Virol. 56, 489–494 (1985).
Atherton, A., Armour, K. L., Bell, S., Minson, A. C. & Clark, M. R. The herpes simplex virus type 1 Fc receptor discriminates between IgG1 allotypes. Eur. J. Immunol. 30, 2540–2547 (2000).
Johansson, P. J. et al. Studies of protein A and herpes simplex virus-1 induced Fcγ-binding specificities. Different binding patterns for IgG3 from Caucasian and Oriental subjects. Immunology 83, 631–638 (1994).
Pandey, J. P. Immunoglobulin genes and immunity to herpes simplex virus type 1. J. Infect. Dis. 206, 143–144 (2012).
Moraru, M. et al. NK cell and Ig interplay in defense against herpes simplex virus type 1: epistatic interaction of CD16A and IgG1 allotypes of variable affinities modulates antibody-dependent cellular cytotoxicity and susceptibility to clinical reactivation. J. Immunol. 195, 1676–1684 (2015).
Corrales-Aguilar, E., Hoffmann, K. & Hengel, H. CMV-encoded Fcγ receptors: modulators at the interface of innate and adaptive immunity. Semin. Immunopathol. 36, 627–640 (2014).
Vezzani, G. et al. Human immunoglobulins are transported to HCMV viral envelope by viral Fcγ receptors-dependent and independent mechanisms. Front. Microbiol. 13, 1106401 (2022).
Stanton, R. J. et al. Reconstruction of the complete human cytomegalovirus genome in a BAC reveals RL13 to be a potent inhibitor of replication. J. Clin. Invest. 120, 3191–3208 (2010).
Kolb, P. et al. Human cytomegalovirus antagonizes activation of Fcγ receptors by distinct and synergizing modes of IgG manipulation. eLife 10, e63877 (2021).
Sprague, E. R. et al. The human cytomegalovirus Fc receptor gp68 Binds the Fc CH2–CH3 interface of immunoglobulin G. J. Virol. 82, 3490–3499 (2008).
Cramer, P. et al. A viral glycoprotein targets IgG+ memory B cells to mediate humoral immune evasion. EMBO Mol. Med. 18, 795–823 (2026).
Bentley, K. et al. Virion proteomics of genetically intact HCMV reveals a regulator of envelope glycoprotein composition that protects against humoral immunity. Proc. Natl Acad. Sci. USA 122, e2425622122 (2025).
Cortese, M. et al. Recombinant human cytomegalovirus (HCMV) RL13 binds human immunoglobulin G Fc. PLoS One 7, e50166 (2012).
Dolan, A. et al. Genetic content of wild-type human cytomegalovirus. J. Gen. Virol. 85, 1301–1312 (2004).
Thäle, R., Lucin, P., Schneider, K., Eggers, M. & Koszinowski, U. H. Identification and expression of a murine cytomegalovirus early gene coding for an Fc receptor. J. Virol. 68, 7757–7765 (1994).
Crnković-Mertens, I. et al. Virus attenuation after deletion of the cytomegalovirus Fc receptor gene is not due to antibody control. J. Virol. 72, 1377–1382 (1998).
Lenac, T. et al. The herpesviral Fc receptor fcr-1 down-regulates the NKG2D ligands MULT-1 and H60. J. Exp. Med. 203, 1843–1850 (2006).
Mintern, J. D. et al. Viral interference with B7-1 costimulation: a new role for murine cytomegalovirus fc receptor-1. J. Immunol. 177, 8422–8431 (2006).
Arapović, J., Lenac Rovis, T., Reddy, A. B., Krmpotić, A. & Jonjić, S. Promiscuity of MCMV immunoevasin of NKG2D: m138/fcr-1 down-modulates RAE-1epsilon in addition to MULT-1 and H60. Mol. Immunol. 47, 114–122 (2009).
Otero, C. E. et al. Rhesus cytomegalovirus-encoded Fcγ-binding glycoproteins facilitate viral evasion from IgG-mediated humoral immunity. Nat. Commun. 16, 1200 (2025).
Gomes, A. C. et al. The cytomegalovirus gB/MF59 vaccine candidate induces antibodies against an antigenic domain controlling cell-to-cell spread. Nat. Commun. 14, 1041 (2023).
Junker, A. K., Angus, E. & Thomas, E. E. Recurrent varicella-zoster virus infections in apparently immunocompetent children. Pediatric Infect. Dis. J. 10, 569–575 (1991).
Gershon, A. A. Varicella zoster vaccines and their implications for development of HSV vaccines. Virology 435, 29–36 (2013).
Gershon, A. A. et al. Latency of varicella zoster virus in dorsal root, cranial, and enteric ganglia in vaccinated children. Trans. Am. Clin. Climatol. Assoc. 123, 17–33; discussion 33–35 (2012).
Kim, T. & Hearn, C. Vaccinal efficacy of recombinant Marek’s disease vaccine 301B/1 expressing chicken interleukin-15. Avian Dis. 66, 79–84 (2022).
Panther, L. et al. Safety and immunogenicity of mRNA-1647, an mRNA-based cytomegalovirus vaccine in healthy adults: results of a phase 2, randomized, observer-blind, placebo-controlled, dose-finding trial. Open Forum Infect. Dis. 10, ofad500.2475 (2023).
Das, R. et al. Safety, efficacy, and immunogenicity of a replication-defective human cytomegalovirus vaccine, V160, in cytomegalovirus-seronegative women: a double-blind, randomised, placebo-controlled, phase 2b trial. Lancet Infect. Dis. 23, 1383–1394 (2023).
Baraniak, I. et al. Protection from cytomegalovirus viremia following glycoprotein B vaccination is not dependent on neutralizing antibodies. Proc. Natl Acad. Sci. USA 115, 6273–6278 (2018).
Lewis, A. L. et al. in Essentials of Glycobiology 4th edn Ch. 42 (eds Varki, A. et al.) (Cold Spring Harbor Laboratory Press, 2022).
Anish, C., Beurret, M. & Poolman, J. Combined effects of glycan chain length and linkage type on the immunogenicity of glycoconjugate vaccines. npj Vaccines 6, 150 (2021).
Escolano, A. et al. Immunization expands B cells specific to HIV-1 V3 glycan in mice and macaques. Nature 570, 468–473 (2019).
Epperson, M. L., Lee, C. A. & Fremont, D. H. Subversion of cytokine networks by virally encoded decoy receptors. Immunol. Rev. 250, 199–215 (2012).
Deere, J. D. et al. Neutralization of rhesus cytomegalovirus IL-10 reduces horizontal transmission and alters long-term immunity. Proc. Natl Acad. Sci. USA 116, 13036–13041 (2019).
Nelson, J. C. et al. Active postlicensure safety surveillance for recombinant zoster vaccine using electronic health record data. Am. J. Epidemiol. 192, 205–216 (2023).
Warren-Gash, C., Forbes, H. & Breuer, J. Varicella and herpes zoster vaccine development: lessons learned. Expert Rev. Vaccines 16, 1191–1201 (2017).
Pyzik, M., Kozicky, L. K., Gandhi, A. K. & Blumberg, R. S. The therapeutic age of the neonatal Fc receptor. Nat. Rev. Immunol. 23, 415–432 (2023).
Patel, D. D. & Bussel, J. B. Neonatal Fc receptor in human immunity: function and role in therapeutic intervention. J. Allergy Clin. Immunol. 146, 467–478 (2020).
Grevys, A. et al. Fc engineering of human IgG1 for altered binding to the neonatal Fc receptor affects Fc effector functions. J. Immunol. 194, 5497–5508 (2015).
Slein, M. D. et al. Eliminating interactions with the viral Fc receptor improves antibody-mediated protection against neonatal HSV infection in mice. Sci. Transl. Med. 17, eadu8579 (2025).
Levin, M. J. & Weinberg, A. Immune responses to varicella-zoster virus vaccines. Curr. Top. Microbiol. Immunol. 438, 223–246 (2023).
Kenneson, A. & Cannon, M. J. Review and meta-analysis of the epidemiology of congenital cytomegalovirus (CMV) infection. Rev. Med. Virol. 17, 253–276 (2007).
Ssentongo, P. et al. Congenital cytomegalovirus infection burden and epidemiologic risk factors in countries with universal screening: a systematic review and meta-analysis. JAMA Netw. Open 4, e2120736 (2021).
Liu, F. & Zhou, Z. H. in Human Herpesviruses: Biology, Therapy, and Immunoprophylaxis Ch. 3 (eds Arvin, A. et al.) (Cambridge Univ. Press, 2007).
McKendall, R. R. IgG-mediated viral clearance in experimental infection with herpes simplex virus type 1: role for neutralization and Fc-dependent functions but not C′ cytolysis and C5 chemotaxis. J. Infect. Dis. 151, 464–470 (1985).
Balachandran, N., Bacchetti, S. & Rawls, W. E. Protection against lethal challenge of BALB/c mice by passive transfer of monoclonal antibodies to five glycoproteins of herpes simplex virus type 2. Infect. Immun. 37, 1132–1137 (1982).
Ishibashi, K. et al. Strain-specific seroepidemiology and reinfection of cytomegalovirus. Microbes Infect. 10, 1363–1369 (2008).
Landry, M. L. & Zibello, T. A. Ability of herpes simplex virus (HSV) types 1 and 2 to induce clinical disease and establish latency following previous genital infection with the heterologous HSV type. J. Infect. Dis. 158, 1220–1226 (1988).
Schoenfisch, A. L. et al. Cytomegalovirus (CMV) shedding is highly correlated with markers of immunosuppression in CMV-seropositive women. J. Med. Microbiol. 60, 768–774 (2011).
Stowell, J. D. et al. Cross-sectional study of cytomegalovirus shedding and immunological markers among seropositive children and their mothers. BMC Infect. Dis. 14, 568 (2014).
Kotton, C. N. et al. Cytomegalovirus in the transplant setting: where are we now and what happens next? A report from the International CMV Symposium 2021. Transpl. Infect. Dis. 24, e13977 (2022).
Grossi, P. A., Baldanti, F., Andreoni, M. & Perno, C. F. CMV infection management in transplant patients in Italy. J. Clin. Virol. 123, 104211 (2020).
Baraniak, I. et al. Seronegative patients vaccinated with cytomegalovirus gB-MF59 vaccine have evidence of neutralising antibody responses against gB early post-transplantation. EBioMedicine 50, 45–54 (2019).
Baraniak, I. et al. Epitope-specific humoral responses to human cytomegalovirus glycoprotein-B vaccine with MF59: anti-AD2 levels correlate with protection from viremia. J. Infect. Dis. 217, 1907–1917 (2018).
Law, N., Logan, C. & Taplitz, R. EBV reactivation and disease in allogeneic hematopoietic stem cell transplant (HSCT) recipients and its impact on HSCT outcomes. Viruses 16, 1294 (2024).
Yamane, A. et al. Risk factors for developing human herpesvirus 6 (HHV-6) reactivation after allogeneic hematopoietic stem cell transplantation and its association with central nervous system disorders. Biol. Blood Marrow Transpl. 13, 100–106 (2007).
Nakayama, H. et al. Impact of specific antibody level on human herpesvirus 6 reactivation after allogeneic stem cell transplantation. Transplant. Cell. Ther. 27, 174.e1–174.e5 (2021).
Shechter, O., Sausen, D. G., Gallo, E. S., Dahari, H. & Borenstein, R. Epstein–Barr virus (EBV) epithelial associated malignancies: exploring pathologies and current treatments. Int. J. Mol. Sci. 23, 14389 (2022).
Wen, K. W. & Damania, B. Kaposi sarcoma-associated herpesvirus (KSHV): molecular biology and oncogenesis. Cancer Lett. 289, 140–150 (2010).
Posnett, D. N. Herpesviruses and autoimmunity. Curr. Opin. Investig. Drugs 9, 505–514 (2008).
Sokolovska, L., Cistjakovs, M., Matroze, A., Murovska, M. & Sultanova, A. From viral infection to autoimmune reaction: exploring the link between human herpesvirus 6 and autoimmune diseases. Microorganisms 12, 362 (2024).
Vollmer, B. et al. The prefusion structure of herpes simplex virus glycoprotein B. Sci. Adv. 6, eabc1726 (2020).





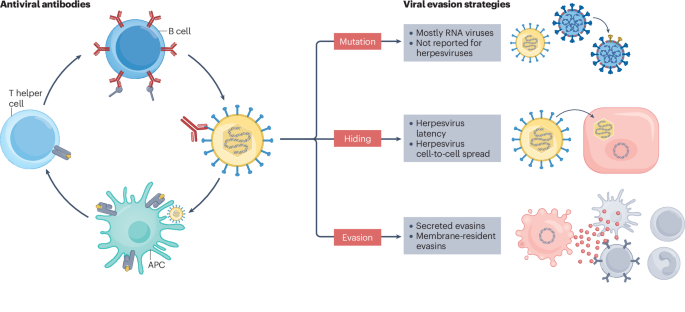










Leave a Reply