Animal details
All animal procedures were approved and carried out in accordance with the University of Oxford Ethical Committee and the UK Home Office Animals (Scientific Procedures) Act 1986 and conformed with Directive 2010/63/EU of the European Parliament. We used Nos2−/− (Nos2tm1Lau) (iNOS-KO) and WT C57BL6/J mice purchased from The Jackson Laboratory (stock no. 002609). A conditional KO (floxed) of the Gch1 allele using the Cre/loxP strategy was generated as previously described, producing Gch1 WT (Gch1fl/fl; containing the gene encoding BH4) and Gch1-KO (Gch1fl/fl; Tie2-Cre; with excision of the gene encoding BH4) mice. Experiments were performed using bone marrow isolated from 10–16-week-old adult male and female mice for all genotypes1,10,45.
Isolation and stimulation of BMDMs
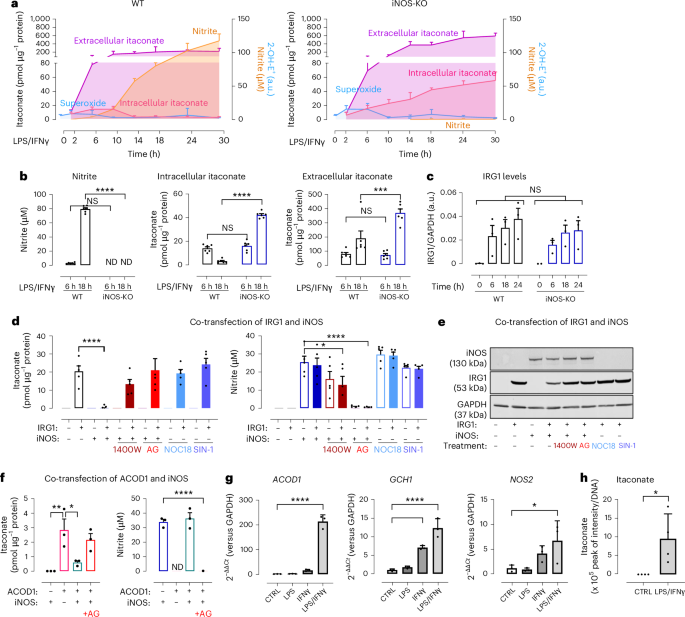
BMDMs were obtained and cultured as described in our published protocol13,46. In brief, after bone marrow cell isolation, cells were plated in non-tissue-culture-treated plastic at 1 × 106 cells per well of a six-well plate for time course experiments or 3 × 106 cells per 10 cm dish for IP experiments using DMEM:F12 (ThermoFisher Scientific) supplemented with penicillin (100 U ml−1), streptomycin (100 ng ml−1; Sigma-Aldrich), ultra-low-endotoxin FBS (5% (v/v); Biowest) and l-glutamine (5 mmol l−1; Sigma-Aldrich). Recombinant macrophage colony-stimulating factor (M-CSF) protein (Peprotech) and recombinant granulocyte M-CSF (GM-CSF) protein (Peprotech) were added at day 0, day 5, day 6 and day 7 to ensure macrophage differentiation (see published protocol for concentration). Cells were then stimulated with IFNγ (10 ng ml−1; Peprotech) and LPS (100 ng ml−1; Sigma-Aldrich) in 2% (v/v) in low-endotoxin Biowest FBS-supplemented DMEM:F12. If specified, cells were treated with 1 mM aminoguanidine hydrochloride (Sigma-Aldrich, 396494) at the same time as LPS and IFNγ. Cells were then collected at different time points.
Culturing HEK-293T cells
HEK-293T (HEK) cells were obtained from European Collection of Authenticated Cell Cultures (ECACC; Merck, cat. no. 12022001) and passaged routinely in DMEM supplemented with penicillin, streptomycin and 10% (v/v) FBS (Sigma-Aldrich, F7524).
Culturing hMDMs
PBMCs were obtained from anonymous healthy donors (sex unknown). Informed consent and ethical approvals were obtained from the NHS Blood and Transplant service (UK). PBMCs were isolated from leucocyte cones after gently layering blood over Histopaque (Sigma-Aldrich). Following density centrifugation, the PBMC layer was carefully collected and washed repeatedly with PBS and centrifuged. CD14+ monocytes were then isolated by positive selection using magnetic beads conjugated to anti-CD14 antibody (EasySep Human Monocyte Isolation Kit, Stemcell Technologies). Once isolated, 1 x 106 cells were plated in a six-well plate in RPMI 1640 medium supplemented with penicillin (100 U ml−1), streptomycin (100 ng ml−1; Sigma-Aldrich), FBS (10% (v/v); Sigma-Aldrich), l-glutamine (5 mmol l−1; Sigma-Aldrich) and 100 ng ml−1 of human recombinant M-CSF (Peprotech) (day 0). At day 5, 50 ng ml−1 of human recombinant GM-CSF (Peprotech) was added, followed by 100 ng ml−1 on day 6. On day 7, cells were stimulated with IFNγ (20 ng ml−1; Peprotech) and LPS (100 ng ml−1; Sigma-Aldrich) in 2% (v/v) FBS-supplemented RPMI with 100 ng ml−1 of GM-CSF.
Human bone marrow organoids culture and stimulation
KOLF2.1J human iPS cells were kindly offered by the Induced Pluripotent Stem Cell Facility from the NDM–Centre for Human Genetics at the University of Oxford. The iPS cells were differentiated into bone marrow organoids following a previously published protocol21 using a variety of cytokines, chemokines and growth factors21,47. In brief, cells were first allowed to form iPS cell aggregates and undergo mesodermal induction (days 0–3) before committing to vascular and haematopoietic lineages (days 3–5). These aggregates were then embedded into a mixture of collagen-Matrigel hydrogels, allowing vascular sprouting (days 5–12). At day 12, each sprout was collected and added one by one into 96-well ultra-low attachment plates. At day 16, bone marrow organoids were formed containing haematopoietic, endothelial and stromal cells, organized in 3D and able to generate myeloid cell types. These newly formed organoids were then stimulated for 24 h with 20 ng ml−1 of IFNγ, 100 ng ml−1 LPS or LPS and IFNγ. To assess gene expression and intracellular itaconate levels, four organoids (=four wells) were pooled together, counting as a representative of n = 1 biological replicate. Organoids were then centrifuged gently at 1,000g for 5 min and digested with 2.5 mg ml−1 of Collagenase II (17101-015, Gibco) and 2% BSA in DMEM for 15 min at 37 °C. This incubation was repeated until organoids formed a single-cell suspension. Cells were then spun at 500g for 5 min and the pellet obtained was directly processed for real-time qPCR with reverse transcription analysis or itaconate measurement by mass spectrometry.
Real-time qPCR
Total RNA was extracted from cells using the Qiagen RNeasy Mini Kit. RNA purity and quantity were then determined using an ND-1000 Nanodrop spectrophotometer and converted to cDNA using the Qiagen QuantiTect Reverse Transcription Kit. Using the QuantStudio 6 Flex Real-Time PCR System, 5 ng of cDNA was used for each qPCR reaction in a 384-well plate alongside TaqMan Fast Advanced Master Mix and TaqMan assay primers specific of our genes of interest (respectively; Acod1 (Hs00985781_m1), Nos2 (Hs01075529_m1), Gch1 (Hs00609198_m1)) or internal control (either 18S (4333760T) or GAPDH(4326317E)). Gene expression was then assessed using the 2−ΔΔCt method, where ΔΔCt is the fold change for each condition relative to unstimulated cells following normalization using an internal control.
Fugene-HD transfection and stimulation
HEK cells were plated in a six-well plate at 1 × 106 cells in 2 ml. At 60–70% confluence, cells were transfected at a ratio of 1:3 of plasmid DNA:Fugene-HD. In brief, 3 µl of Fugene-HD was added to 100 µl serum-free DMEM, mixed well and left for 10 min. Then, 1 µg of plasmid DNA (either Nos2 (NM_010927) mouse tagged ORF clone or WT or mutated Acod1 (NM_008392) mouse tagged ORF clone) was then added to the Fugene-HD solution. Following incubation for 10 min at room temperature (20°C to 22°C), 100 µl of transfection mix was added to the cell media. The cells were collected 24 h later.
Transfected cells were treated 1 h following transient transfection with 50 µM of NOC12 or NOC18 (EMD Millipore, 487955 or Sigma-Aldrich, 487957, respectively), 10 µM of 1400W dihydrochloride (Sigma-Aldrich, W4262), 1 mM aminoguanidine hydrochloride or 100 µM of SIN-1 chloride (Tocris, 5245).
Mutagenesis of IRG1
Acod1 (NM_008392) mouse tagged ORF clone was subjected to different mutations, using the QuickChange Lightning Site-Directed Mutagenesis kit if only one mutation was required (C184A, C340A, C387A, C432A, C452A, T319F) and the QuickChange Lightning Multi Site-Directed Mutagenesis kit if more than one mutation was required (for example, ALA5). Primers used were:
C184A(MF)cys184ala-IRG15′-CTCAGCTTGACAAAGGCCCGCGAGGCATTGGCT-3′
(MR)cys184ala-IRG15′-AGCCAATGCCTCGCGGGCCTTTGTCAAGCTGAG-3′
C340A (MF)cys340ala-IRG15′-TTCCAGTATGTGGCCGCTGCCTCGCTGCTCGAC-3′
(MR)cys340ala-IRG15′-GTCGAGCAGCGAGGCAGCGGCCACATACTGGAA-3′
C387A(MF)cys387ala-IRG15′-TTCGACACGCTATACGCTGAAATAAGCATCACT-3′
(MR)cys387ala-IRG15′-AGTGATGCTTATTTCAGCGTATAGCGTGTCGAA-3′
C432A (MF)cys432ala-IRG15′-GCCTCAAAGATGCTAGCCAGGGACACGGTGGAA-3′
(MR)cys432ala-IRG15′-TTCCACCGTGTCCCTGGCTAGCATCTTTGAGGC-3′
C452A (MF)cys452ala-IRG15′-GAAGACCTAGAAGACGCCTCTGTGCTAACCAGA-3′
(MR)cys452ala-IRG15′-TCTGGTTAGCACAGAGGCGTCTTCTAGGTCTTC-3′
Validation of mutants was performed using the Source BioScience sequencing platform (see Extended Data Fig. 3g).
Mutagenesis of iNOS
The Nos2 (NM_010927) mouse tagged ORF clone was subjected to different mutations (W457A and W457F) using the QuickChange Lightning Site-Directed Mutagenesis kit. Primers used were:
W457A (MF)-iNOS: 5′-GAGGGACCAGCGCAATCCAGTCTGCCGGG-3′
(MR)-iNOS: 5′-CCCGGCAGACTGGATTGCGCTGGTCCCTC-3′
W457F (MF)-iNOS: 5′-GGAGGGACCAGGAAAATCCAGTCTGCCGGGC-3′
(MR)-iNOS: 5′-GCCCGGCAGACTGGATTTTCCTGGTCCCTCC-3′
Validation of mutants was performed using the Source BioScience sequencing platform.
Dihydroethidium HPLC
Cultured macrophages were incubated with 25 µM dihydroethidium (Invitrogen) for 15 min at 37 °C and were protected from light before collection. Cell pellets were lysed in ice-cold methanol, and protein was removed by acid precipitation with 0.1 M HCl. Separation of dihydroethidium and its oxidized products, 2-hydroxyethidium and ethidium, was performed using a gradient HPLC system (Jasco) with an ODS3 reverse phase column (250 mm, 4.5 mm; Hichrom) and quantified using a fluorescence detector set at 510 nm (excitation) and 595 nm (emission). A linear gradient was applied from mobile phase A (0.1% (w/v) trifluoroacetic acid) to mobile phase B (0.085% (w/v) trifluoroacetic acid in acetonitrile), over 23 min (30–50% (v/v) acetonitrile).
H2O2 measurement
Media from transfected HEK cells, seeded at 1 x 106 cells in six-well plates, were centrifuged to remove debris at 500g for 5 min at 4 °C and frozen directly. The H2O2 concentration was then measured by inserting an amperometric H2O2 microsensor electrode connected to a free radical analyser system (WPI) into a PBS solution in which 100 µl of thawed media was added. The output current corresponding to the H2O2 concentration detected was recorded and analysed using LabTrax 2 software. Sample concentrations were determined using a known H2O2 standard curve.
Western blotting using SDS–PAGE
Cell lysates were prepared by homogenization in ice-cold CellLytic M buffer (Sigma-Aldrich) containing protease inhibitor cocktail (Roche Applied Science). Lysates were centrifuged at 17,000g for 10 min at 4 °C, and samples were prepared using NuPAGE LDS sample buffer (Invitrogen) with the addition of 10% dithiothreitol (DTT). Then, 5–10 µg of protein was loaded per well in a 4–12% Bis-Tris NuPAGE gel (Thermofisher) and transferred to a nitrocellulose membrane. Anti-iNOS (Abcam, ab49999; diluted at 1:1,000), anti-IRG1 (Abcam, ab222411; diluted at 1:1,000) and anti-GAPDH (Merck, MAB374; diluted at 1:1,000) were then coupled with Licor antibodies (IRDye680RD goat anti-mouse and IRDye488RD goat anti-rabbit; both diluted at 1:25,000). Anti-iNOS (BD Biosciences, 610431; diluted at 1:1,000) was coupled to anti-mouse IgG (H + L) and HRP conjugate (Promega, W4028; diluted at 1:25,000). Proteins were detected using the ChemiDoc Imaging system (Bio-Rad) following Amersham ECL Select western blotting detection reagent (Cytiva, RPN2235).
Nitrite measurement
Nitrite accumulation was measured in the cell culture medium of the samples using the Griess assay with colourimetric detection in 96-well plates. Cell culture supernatants were mixed 1:1 with the Griess reagent (Sigma-Aldrich) and quantified by comparison to a sodium nitrite (Sigma-Aldrich) standard curve produced in tissue culture media at 550 nm.
Itaconate measurement by HPLC
Intracellular itaconic acid levels were measured using HPLC48,49,50,51. Pellets from 1 x 106 macrophage cells were resuspended in PBS at pH 7.4 and lysed by three freeze–thaw cycles. After centrifugation at 17,000g for 15 min at 4 °C, debris-free lysates were transferred to new tubes. Proteins were then precipitated by the addition of 10% (v/v) of 2 M perchloric acid and centrifuged at 17,000g for 15 min at 4 °C. Cell media was centrifuged at 500g for 5 min at 4 °C before the addition of 10% (v/v) of 2 M perchloric acid and centrifuged at 17,000g for 15 min at 4 °C. Samples were injected onto a 250 mm ACE C18 column (Hichrom), and itaconate was quantified using UV detection at 210 nm. HPLC separation was performed using a mobile phase comprising of 2.5% (v/v) acetonitrile and 0.1% (v/v) phosphoric acid (all ultrapure electrochemical HPLC grade), at a flow rate of 1.0 ml min−1. Quantification of itaconic acid was made by comparison with pure itaconic acid (Sigma-Aldrich, I29204) standard range from 0.50 μM to 500 μM in PBS or cell media. Final results were normalized using each sample’s protein concentration, determined by the bicinchoninic acid protein assay. Extracellular itaconate levels were calculated by comparing the itaconate concentration in cell media samples to a standard prepared in the same media, then multiplying by the total volume exposed to the cells and normalizing to the protein content.
Itaconate measurement by ion chromatography coupled to mass spectrometry
Human iPS cell pellets were lysed in ice-cold methanol and then spun at 17,000g for 30 min. The supernatant containing intracellular itaconate was then measured using ion chromatography coupled to mass spectrometry as detailed previously52. DNA was further assessed for normalization.
Purification of mouse IRG1
The IRG1 sequence from Acod1 (NM_008392) mouse tagged ORF clone was expressed in the pOPINEneo-3C-Strep2-His8 vector in which IRG1 was tagged at the carboxy terminus with a Strep-Tag II. The plasmid was transiently expressed in HEK cells (expi293, Thermofisher Scientific) as previously described53. Cells were then lysed in 50 mM Tris, pH 7.5, 500 mM NaCl, 30 mM imidazole, 0.2% Tween, protease inhibitors and DNAse I, using a cell disruptor. Debris were removed by centrifugation at 30,000g for 30 min at 4 °C. The transfected protein was then purified from cleared lysates using the StrepTactin purification protocol and reagents from IBA Lifesciences. Protein was concentrated in GF buffer (10 mM HEPES, 150 mM NaCl, 0.1 mM TCEP, 10% (v/v) glycerol) as previously proposed28. A western blot and Coomassie gel were performed after each purification to assess the quality of the purification process.
IRG1 enzymatic activity
A 2.5 µg aliquot of purified IRG1 in GF buffer was added to 150 µl of 0.2 M sodium phosphate buffer at pH 6.5 containing 15 µM cis-aconitate for 4 h and incubated at 37 °C. Drugs (1 mM of sodium nitrite, 1 mM SIN-1, 1 mM NOC12 and NOC18, 1 mM GSNO (+10 mM of GSH or GSSG), 1 mM H2O2, 1 mM DTT, 50 or 500 µM of citrulline and 50 or 500 µM of arginine) were added at the same time as purified IRG1. Itaconate was then determined by boiling samples at 95 °C, centrifuging at 17,000g for 5 min and injecting 100 µl onto the HPLC column.
To assess interaction with iNOS, 0.5 µg of purified IRG1 was added to 200 µl of HEPES pH 7.4 with 25 µM of cis-aconitate overnight and incubated at 37 °C with different concentrations of mouse recombinant active iNOS from Cayman Chemical (60864; purified in 50 mM HEPES, pH 7.4, with 10% glycerol, 8 µM BH4, 2% protease inhibitor cocktail (EDTA free) and 0.05% nuclease), ranging from 0.05–50 µg. Inactivated iNOS was obtained by boiling the protein at 95 °C for 10 min. Respective protein concentrations of iNOS and IRG1 were obtained using the bicinchoninic acid assay.
Co-IP of IRG1 and analysis by mass spectrometry
BMDMs were grown at a density of 1 x 107 cells in a T75 non-treated flask. Cell pellets were then lysed in NP-40 buffer (50 mM Tris base, 0.5% (v/v) NP-40, 150 mM NaCl, 20 mM MgCl2 in distilled water) containing phosphoSTOP and protease inhibitor cocktail. Lysates were incubated overnight at 4 °C with Protein A-dynabeads (Invitrogen), pre-washed and pre-incubated with target (anti-IRG1 (ab222411)) and control antibodies (rabbit IgG isotype control; Cell Signaling Technology, 3900S). After washing beads from the cell lysate, the target protein was either eluted in 2.5× Laemmli loading buffer at 95 °C for western blot analysis or directly sent to the mass spectrometry facility. Samples were processed by on-bead digestion (SMART digestion using SMART Digest Kit Soluble Trypsin). Peptides obtained were then resuspended in 5% (v/v) formic acid and 5% (v/v) dimethylsulfoxide and then trapped on an Acclaim PepMap 100 C18 HPLC Column (Thermo Fisher, PepMapC18; 300 µm × 5 mm, 5 µm particle size) using solvent A (0.1% (v/v) formic acid in water) at a pressure of 60 bar and separated on an Ultimate 3000 UHPLC system (Thermo Fisher Scientific) coupled to a QExactive mass spectrometer (Thermo Fisher Scientific). The peptides were separated on an EASY-Spray PepMap RSLC column (75 µm i.d. × 2 µm × 50 mm, 100 Å; Thermo Fisher) and then electrosprayed directly into a QExactive mass spectrometer (Thermo Fisher Scientific) through an EASY-Spray nano-electrospray ion source (Thermo Fisher Scientific) using a linear gradient (length, 60 min; 5% to 35% solvent B (0.1% (v/v) formic acid in acetonitrile), with a flow rate of 250 nl min−1). The raw data were acquired on the mass spectrometer in data-dependent mode. Full scan MS spectra were acquired in the Orbitrap (scan range, 380–1,800 m/z; resolution, 70,000; AGC target, 3 × 106; maximum injection time, 100 ms). After the mass spectrometry scans, the 15 most intense peaks were selected for HCD fragmentation at 28% of normalized collision energy. HCD spectra were also acquired in the Orbitrap (resolution, 17,500; AGC target, 1 × 105; maximum injection time, 128 ms) with a first fixed mass at 100 m/z.
The raw data files generated were processed using MaxQuant (v.1.6.17.0) and integrated with the Andromeda search engine. For protein group identification, peak lists were searched against the human database (UPR_MusMusculus_UP00000589_10090_.fasta) as well as a list of common contaminants by Andromeda. Trypsin with a maximum number of missed cleavages of two was chosen. Oxidation and deamidation were used as variable modifications, while carbamidomethylation was set as a fixed modification. Protein and PSM false discovery rate were set at 0.01. Match between runs was applied. Data were uploaded to PRIDE (project accession, PXD048712). Results obtained by mass spectrometry were analysed by median normalization using Perseus and are displayed as log2 values.
Isolation of mitochondria
Murine BMDMs were grown at a density of 750,000 cells in 10 cm Petri dishes and stimulated for 18 h with 100 ng ml−1 LPS and 10 ng ml−1 IFNγ, in the presence or absence of 1 mM AG. Mitochondria and cytosol were then isolated using the standard procedure of the Qproteome Mitochondria Isolation Kit (QIAGEN). In brief, cells were collected and washed using a 0.9% sodium chloride solution before being lysed and mechanically disrupted using a blunt needle and syringe. Following a series of centrifugation and washes, the mitochondria and cytosol fraction were separated, and 7 µg of protein from each compartment was analysed using SDS–PAGE with GAPDH as a cytosolic control (Merck, MAB374; diluted at 1:1,000) and VDAC as a mitochondria control (Cell Signaling Technology, 4661S; diluted at 1:1,000); proteins of interest were studied using anti-iNOS (Abcam, ab49999; diluted at 1:1,000) and anti-IRG1 (Abcam; ab222411; diluted at 1:1,000). Each primary antibody was then coupled with Licor antibodies (IRDye680RD goat anti-mouse and IRDye488RD goat anti-rabbit; both diluted at 1:25,000). The same threshold (brightness, contrast, opacity and filters) was applied during analysis between the target and its respective loading control using Licor to ensure accurate comparison of protein levels.
Immunofluorescence staining and Airyscan imaging
Following cell culture of BMDMs in 10 cm dishes, cells were gently detached at day 6 and plated at 300,000 cells on coverslips in a 12-well plate in DMEM:F12 supplemented with 2% (v/v) FBS. After cell adherence, BMDMs were stimulated with LPS/IFNγ overnight. The following day, cells were washed with PBS, fixed with 4% (v/v) PFA for 5 min and washed again with PBS before permeabilization with 0.05% (v/v) Triton in PBS for 5 min. Following three washes, cells were incubated in 5% (v/v) donkey serum in PBS used as a blocking buffer. Coverslips were then incubated overnight at 4 °C with anti-iNOS (Abcam, ab49999; diluted at 1:500), anti-Hsp60 (Abcam, ab46798; diluted at 1:200), anti-mouse IgG (Cell Signaling Technology, 5415S; diluted at 1:1,000) or anti-rabbit IgG (Cell Signaling Technology, 3900S; diluted at 1:500) used as controls. The next day, coverslips were washed and incubated with donkey AF488 anti-mouse (ThermoFisher, 32766TR) or donkey AF680 anti-rabbit (ThermoFisher; A32788), both diluted at 1:500. After PBS washes, coverslips were attached to slides using 20 µl of Fluoromount-G mounting medium containing DAPI. Images of labelled fixed cells were captured using AiryScan microscopy under the ×60 oil objective at high resolution. Colocalization analyses were performed on the obtained images using the ZEN Blue software colocalization tool with the same threshold determined from the control condition (WT M1) and applied to each image and condition on the same z-stack.
Structural modelling
Structural predictions of the (IRG1)2–(iNOS)2 heterotetramer were carried out using AlphaFold Multimer (v.2.3)44. Following the observation that predictions using the full UniProt sequences exhibited a large proportion of disordered regions (defined as long structured loops with predicted local distance difference test scores of <60) that exhibited notable variability across replicates, the terminal ends of both proteins were trimmed manually using PyMOL, leading to stable predictions. The constructs used in the UniProt numbering were IRG1_MOUSE 1-460 and NOS2_MOUSE 77-1,058, IRG1_HUMAN 1-460 and NOS2_HUMAN 84-1145. In the constructs involving calmodulin, the full sequence of CALM1_MOUSE or CALM1_HUMAN, respectively, was used. Predictions were run using the protocol described in the original publication54, including sequence information from UniRef as well as BFD and Mgnify, and templates available in the Protein Data Bank. All predictions were run on a NVIDIA A100 80 GB GPU. While this article was under review, an improved version of AlphaFold, AlphaFold 3 (ref. 54), was released to the public. Our predictions were subsequently compared with the more recent AlphaFold 3 server, and the results were found to be nearly identical.
Molecular dynamics simulations
Molecular dynamics simulations were carried out starting from predicted AlphaFold Multimer (v.2.3) structures generated as described above, where iNOS is predicted to be in the input state30. Cis-peptide bonds and chiral centres with incorrect stereochemistry were identified and corrected using the Cispeptide and Chirality plugins of VMD55. Complexes were then solvated in orthorhombic boxes with a minimum of 1.4 nm between protein atoms and the box edge, and the protonation states were adjusted to pH 7.2 using the H++ server56. Counterions of Na+ and Cl− were added to match an ionic force of 0.1 M using the SPLIT method57. All simulations were carried out using the AMBER ff94SB force field for protein atoms58, the TIP3P model for rigid water molecules59, the parameters for the haem group reported previously60, the parameters for flavin mononucleotide and flavin adenine dinucleotide reported previously61 and the General Amber Force Field (GAFF) for tetrahydrobiopterin ligands, with atomic charges derived from the minimized AM1 structures using ANTECHAMBER62. Pressure was fixed to 1 bar using a Monte Carlo barostat, and temperature was set using the Langevin thermostat. Before production simulations were carried out, complexes were minimized and then heated to 298 K at constant volume, with protein atoms restrained. These restraints were relaxed over a set of five 100 ps simulations at constant pressure, culminating with 100 ps of unrestrained equilibration. The production simulations used to produce the results shown in this paper used three simulation replicates of 300 ns that were run using OpenMM 7 on NVIDIA Quadro RTX 6000 GPUs63.
Free energy calculations
Free energy calculations were carried out using the molecular mechanics with generalized born and surface area solvation (MM/GBSA) method64. Each calculation used 30 replicates of 5 ns equilibrium trajectories, started from a relaxed and equilibrated structure extracted from long molecular dynamics simulations. Each of the 30 replicates was subject to the same minimization, heating and equilibration procedure described in the section above. Free energies were calculated using the MMGBSA.py programme in AmberTools65, and 100 frames were collected every 40 ps from the final 4 ns of each 5 ns trajectory (that is, 3,000 frames per complex). Per-residue energy contributions to the binding energy were computed using the MMPBSA.py DECOMP functionality.
Surface plasma resonance
The surface plasma resonance experiments were performed using a Biacore X100 equipped with a CM5 sensor (Cytiva, BR100012) chip. The CM5 sensor chips were immobilized with the IRG1 or iNOS using an amine-coupling kit (Cytiva, BR100050). The process was performed using both human and mouse proteins, resulting in four immobilized chips. The flow cell surfaces were activated for 7 min with 0.1 M NHS (N-hydroxysuccinimide) and 0.4 M EDC (1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide 3) at a flow rate of 10 μl min−1. IRG1 and iNOS were diluted in 10 mM sodium acetate, pH 4.0 and pH 5.0, respectively. IRG1 and iNOS were immobilized onto different chips at a density of 3,423 RU and 200 RU, respectively, onto flow cell 2. Flow cell 1 was left blank to enable a blank correction of the sensograms. Both surfaces were blocked with a 7 min injection of 1 M ethanolamine-HCl, pH 8.5. All analyte proteins were dialysed into 1× HBS P+ buffer (Cytiva, BR100671) to match the running buffer used for analysis. A multi-cycle kinetics analysis was performed in duplicate for each ligand and respective analyte using twofold dilutions ranging from 25 nM to 200 nM. The association and dissociation time for each injection was 120 s and 600 s, respectively, followed by a regeneration using a 20 s injection of glycine 3 and a 30 s stabilization period. The blank-subtracted data were fit to a 1:1 interaction model using the Biacore Evaluation software. An eNOS chip was prepared and tested with IRG1 following the same protocol as the iNOS chip. Moreover, to confirm binding specificity, BSA (10 µg ml−1) was included as a negative control. Mouse iNOS was acquired from Cayman chemicals (60864), human iNOS from Origene (TP311819) and both human and mouse IRG1 were purified as previously described (see ‘Purification of Mouse Irg1’).
Blue native gels
Cell lysates were prepared by homogenization in ice-cold non-denaturing cell lysis buffer (Cell Signaling Technology) containing fresh 1 mM PMSF or in CellLytic M buffer. Lysates were centrifuged at 17,000g for 10 min at 4 °C, and samples were prepared using Native PAGE buffer with 1% digitonin (Invitrogen). Then, 5 µg of protein was loaded by well in a 4–12% NuPAGE gel (Thermofisher) and run at 4 °C using Native PAGE running buffer and Native Blue for 30 min at 150 V, followed by 60 min at 200 V. Proteins were then transferred to a nitrocellulose membrane, which was then blocked with 5% (w/v) milk in PBS with 0.5% (v/v) Tween. Membranes were then exposed sequentially to anti-iNOS (BD Biosciences, 610431; diluted at 1:1,000), followed by anti-mouse IgG (H + L) and HRP conjugate (Promega, W4028; diluted at 1:25,000). Proteins were detected using the ChemiDoc Imaging system (Bio-Rad) following Amersham ECL Select western blotting detection reagent (Cytiva, RPN2235).
Statistical analysis
All statistical analyses were carried out using Microsoft Excel (Microsoft) and GraphPad Prism 8 (GraphPad) software. Data are expressed as mean; error bars, s.e.m. or s.d. One-way ANOVA was used to compare multiple data groups affected by one single variable, with Dunnett’s test to compare each group with each other. Two-way ANOVA was used to compare multiple data groups affected by two independent variables, with Tukey’s post hoc test used to compare groups with each other. Statistical significance was indicated as ****P < 0.0001; ***P < 0.001; **P < 0.005; *P < 0.05; NS, not significant.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.















Leave a Reply