Steere, A. C. et al. Lyme borreliosis. Nat. Rev. Dis. Primers 2, 16090 (2016).
Stanek, G. & Strle, F. Lyme borreliosis-from tick bite to diagnosis and treatment. FEMS Microbiol. Rev. 42, 233–258 (2018).
Barbour, A. G. & Gupta, R. S. The family Borreliaceae (Spirochaetales), a diverse group in two genera of tick-borne spirochetes of mammals, birds, and reptiles. J. Med. Entomol. 58, 1513–1524 (2021).
Margos, G. et al. Proposing a subgenus Borreliella. Ticks Tick Borne Dis. 16, 102536 (2025).
Nagarajan, A. et al. The landscape of Lyme borreliosis surveillance in Europe. Vector Borne Zoonotic Dis. 23, 142–155 (2023).
Kugeler, K. J., Earley, A., Mead, P. S. & Hinckley, A. F. Surveillance for Lyme disease after implementation of a revised case definition — United States, 2022. MMWR Morb. Mortal. Wkly Rep. 73, 118–123 (2024).
Gray, J. & Kahl, O. in Lyme borreliosis (eds Hunfeld, K.-P. & Gray, J.) 31–45 (Springer International, 2022).
Marques, A. R., Strle, F. & Wormser, G. P. Comparison of Lyme disease in the United States and Europe. Emerg. Infect. Dis. 27, 2017–2024 (2021).
Becker, N. S. et al. Recurrent evolution of host and vector association in bacteria of the Borrelia burgdorferi sensu lato species complex. BMC Genomics 17, 734 (2016).
Coipan, E. C. et al. Imbalanced presence of Borrelia burgdorferi s.l. multilocus sequence types in clinical manifestations of Lyme borreliosis. Infect. Genet. Evol. 42, 66–76 (2016).
Li, Z. et al. Genomic characterization of clinical Borrelia burgdorferi sensu lato isolates in the Netherlands over a thirty-year period. BMC Genomics 26, 1153 (2025).
Lemieux, J. E. et al. Whole genome sequencing of human Borrelia burgdorferi isolates reveals linked blocks of accessory genome elements located on plasmids and associated with human dissemination. PLoS Pathog. 19, e1011243 (2023).
Kahl, O. & Gray, J. S. The biology of Ixodes ricinus with emphasis on its ecology. Ticks Tick Borne Dis. 14, 102114 (2023).
Foster, E., Maes, S. A., Holcomb, K. M. & Eisen, R. J. Prevalence of five human pathogens in host-seeking Ixodes scapularis and Ixodes pacificus by region, state, and county in the contiguous United States generated through national tick surveillance. Ticks Tick Borne Dis. 14, 102250 (2023).
Gray, J., Kahl, O. & Zintl, A. Pathogens transmitted by Ixodes ricinus. Ticks Tick Borne Dis. 15, 102402 (2024).
Uspensky, I. in Lyme Disease 8–16 (SMGroup, 2016).
Steinbrink, A., Brugger, K., Margos, G., Kraiczy, P. & Klimpel, S. The evolving story of Borrelia burgdorferi sensu lato transmission in Europe. Parasitol. Res. 121, 781–803 (2022).
Public Health Agency of Canada. Lyme disease. Surveillance. Government of Canada https://www.canada.ca/en/public-health/services/diseases/lyme-disease/surveillance-lyme-disease.html (2024).
Arsnoe, I., Tsao, J. I. & Hickling, G. J. Nymphal Ixodes scapularis questing behavior explains geographic variation in Lyme borreliosis risk in the eastern United States. Ticks Tick Borne Dis. 10, 553–563 (2019).
Ginsberg, H. S. et al. Selective host attachment by Ixodes scapularis (Acari: Ixodidae): tick-lizard associations in the southeastern United States. J. Med. Entomol. 59, 267–272 (2022).
Kugeler, K. J., Farley, G. M., Forrester, J. D. & Mead, P. S. Geographic distribution and expansion of human Lyme disease, United States. Emerg. Infect. Dis. 21, 1455–1457 (2015).
Kugeler, K. J., Schwartz, A. M., Delorey, M. J., Mead, P. S. & Hinckley, A. F. Estimating the frequency of Lyme disease diagnoses, United States, 2010-2018. Emerg. Infect. Dis. 27, 616–619 (2021).
Pritt, B. S. et al. Identification of a novel pathogenic Borrelia species causing Lyme borreliosis with unusually high spirochaetaemia: a descriptive study. Lancet Infect. Dis. 16, 556–564 (2016).
Kingry, L. et al. Targeted metagenomics for clinical detection and discovery of bacterial tick-borne pathogens. J. Clin. Microbiol. https://doi.org/10.1128/JCM.00147-20 (2020).
Blanchard, L. et al. Comparison of national surveillance systems for Lyme disease in humans in Europe and North America: a policy review. BMC Public Health 22, 1307 (2022).
Burn, L. et al. Incidence of Lyme borreliosis in Europe from national surveillance systems (2005-2020). Vector Borne Zoonotic Dis. 23, 156–171 (2023).
Vandekerckhove, O., De Buck, E. & Van Wijngaerden, E. Lyme disease in western Europe: an emerging problem? A systematic review. Acta Clin. Belg. 76, 244–252 (2021).
Hofhuis, A. et al. Decrease in tick bite consultations and stabilization of early Lyme borreliosis in the Netherlands in 2014 after 15 years of continuous increase. BMC Public Health 16, 425 (2016).
Sajanti, E. et al. Lyme borreliosis in Finland, 1995-2014. Emerg. Infect. Dis. 23, 1282–1288 (2017).
Geebelen, L. et al. Combining primary care surveillance and a meta-analysis to estimate the incidence of the clinical manifestations of Lyme borreliosis in Belgium, 2015-2017. Ticks Tick Borne Dis. 10, 598–605 (2019).
Petrulioniene, A. et al. Epidemiology of Lyme disease in a highly endemic European zone. Medicina https://doi.org/10.3390/medicina56030115 (2020).
Skufca, J. et al. Incidence of Lyme borreliosis in Germany: exploring observed trends over time using public surveillance data, 2016-2020. Vector Borne Zoonotic Dis. 23, 237–246 (2023).
Ermenlieva, N., Tsankova, G. & Todorova, T. T. Epidemiological study of Lyme disease in Bulgaria. Cent. Eur. J. Public Health 27, 235–238 (2019).
Nuttens, C. et al. Epidemiology of Lyme borreliosis in France in primary care and hospital settings, 2010-2019. Vector Borne Zoonotic Dis. 23, 221–229 (2023).
Cairns, V., Wallenhorst, C., Rietbrock, S. & Martinez, C. Incidence of Lyme disease in the UK: a population-based cohort study. BMJ Open 9, e025916 (2019).
Tulloch, J. S. P. et al. The demographics and geographic distribution of laboratory-confirmed Lyme disease cases in England and Wales (2013-2016): an ecological study. BMJ Open 9, e028064 (2019).
Dedkov, V. G. et al. The burden of tick-borne diseases in the Altai region of Russia. Ticks Tick Borne Dis. 8, 787–794 (2017).
Fang, L. Q. et al. Emerging tick-borne infections in mainland China: an increasing public health threat. Lancet Infect. Dis. 15, 1467–1479 (2015).
Stark, J. H. et al. Systematic review and meta-analysis of Lyme disease data and seropositivity for Borrelia burgdorferi, China, 2005–2020. Emerg. Infect. Dis. 28, 2389–2397 (2022).
Kim, S. Y., Kim, T. K., Kim, T. Y. & Lee, H. I. Geographical distribution of Borrelia burgdorferi sensu lato in ticks collected from wild rodents in the Republic of Korea. Pathogens https://doi.org/10.3390/pathogens9110866 (2020).
Chao, L. L., Chen, Y. J. & Shih, C. M. First detection and molecular identification of Borrelia garinii isolated from human skin in Taiwan. J. Med. Microbiol. 59, 254–257 (2010).
Wang, J., Masuzawa, T. & Yanagihara, Y. Characterization of Borrelia garinii isolated from Lyme disease patients in Hokkaido, Japan, by sequence analysis of OspA and OspB genes. FEMS Microbiol. Lett. 154, 371–375 (1997).
Faccini-MartInez, A. A. Call for caution to consider Lyme neuroborreliosis as a frequent neurological disease in Mexico. Arch. Med. Res. 50, 18 (2019).
Vinayaraj, E. V. et al. Clinical and laboratory evidence of Lyme disease in North India, 2016-2019. Travel. Med. Infect. Dis. 43, 102134 (2021).
Collignon, P. J., Lum, G. D. & Robson, J. M. Does Lyme disease exist in Australia? Med. J. Aust. 205, 413–417 (2016).
Labruna, M. B., Faccini-Martinez, A. A., Munoz-Leal, S., Szabo, M. P. J. & Angerami, R. N. Lyme borreliosis in Brazil: a critical review on the Baggio-Yoshinari syndrome (Brazilian Lyme-like disease). Clin. Microbiol. Rev. 37, e0009724 (2024).
Medlock, J. M. et al. Driving forces for changes in geographical distribution of Ixodes ricinus ticks in Europe. Parasit. Vectors 6, 1 (2013).
Earley, A. R., Schiffman, E. K., Wong, K. K., Hinckley, A. F. & Kugeler, K. J. Epidemiologic and tick exposure characteristics among people with reported Lyme disease — Minnesota, 2011-2019. Zoonoses public. health 71, 779–789 (2024).
Wilking, H. & Stark, K. Trends in surveillance data of human Lyme borreliosis from six federal states in eastern Germany, 2009-2012. Ticks Tick Borne Dis. 5, 219–224 (2014).
Berglund, J. et al. An epidemiologic study of Lyme disease in southern Sweden. N. Engl. J. Med. 333, 1319–1327 (1995).
Eisen, L. Seasonal activity patterns of Ixodes scapularis and Ixodes pacificus in the United States. Ticks Tick Borne Dis. 16, 102433 (2025).
Moon, K. A. et al. Risk factors for Lyme disease stage and manifestation using electronic health records. BMC Infect. Dis. 21, 1269 (2021).
Enkelmann, J. et al. Incidence of notified Lyme borreliosis in Germany, 2013-2017. Sci. Rep. 8, 14976 (2018).
Marques, A., Okpali, G., Liepshutz, K. & Ortega-Villa, A. M. Characteristics and outcome of facial nerve palsy from Lyme neuroborreliosis in the United States. Ann. Clin. Transl. Neurol. 9, 41–49 (2022).
Mead, P. et al. Risk factors for tick exposure in suburban settings in the Northeastern United States. Ticks Tick Borne Dis. 9, 319–324 (2018).
De Keukeleire, M., Robert, A., Luyasu, V., Kabamba, B. & Vanwambeke, S. O. Seroprevalence of Borrelia burgdorferi in Belgian forestry workers and associated risk factors. Parasit. Vectors 11, 277 (2018).
Adam-Poupart, A. et al. Occupations at risk of contracting zoonoses of public health significance in Quebec. Can. Commun. Dis. Rep. 47, 47–58 (2021).
Connally, N. P. et al. Peridomestic Lyme disease prevention: results of a population-based case-control study. Am. J. Prev. Med. 37, 201–206 (2009).
Eisen, L. Pathogen transmission in relation to duration of attachment by Ixodes scapularis ticks. Ticks Tick Borne Dis. 9, 535–542 (2018).
Crippa, M., Rais, O. & Gern, L. Investigations on the mode and dynamics of transmission and infectivity of Borrelia burgdorferi sensu stricto and Borrelia afzelii in Ixodes ricinus ticks. Vector Borne Zoonotic Dis. 2, 3–9 (2002).
Kahl, O. et al. Risk of infection with Borrelia burgdorferi sensu lato for a host in relation to the duration of nymphal Ixodes ricinus feeding and the method of tick removal. Zentralbl Bakteriol. 287, 41–52 (1998).
McQuiston, J. H., Childs, J. E., Chamberland, M. E. & Tabor, E. Transmission of tick-borne agents of disease by blood transfusion: a review of known and potential risks in the United States. Transfusion 40, 274–284 (2000).
Woodrum, J. E. & Oliver, J. H. Jr. Investigation of venereal, transplacental, and contact transmission of the Lyme disease spirochete, Borrelia burgdorferi, in Syrian hamsters. J. Parasitol. 85, 426–430 (1999).
Bockenstedt, L. K., Wooten, R. M. & Baumgarth, N. Immune response to Borrelia: lessons from Lyme disease spirochetes. Curr. Issues Mol. Biol. 42, 145–190 (2021).
Lin, Y. P., Diuk-Wasser, M. A., Stevenson, B. & Kraiczy, P. Complement evasion contributes to Lyme borreliae-host associations. Trends Parasitol. 36, 634–645 (2020).
Coburn, J., Leong, J. & Chaconas, G. Illuminating the roles of the Borrelia burgdorferi adhesins. Trends Microbiol. 21, 372–379 (2013).
Norris, S. J. VLS antigenic variation systems of Lyme disease Borrelia: eluding host immunity through both random, segmental gene conversion and framework heterogeneity. Microbiol. Spectr. https://doi.org/10.1128/microbiolspec.MDNA3-0038-2014 (2014).
Troxell, B. et al. Manganese and zinc regulate virulence determinants in Borrelia burgdorferi. Infect. Immun. 81, 2743–2752 (2013).
Dowdell, A. S. et al. Comprehensive spatial analysis of the Borrelia burgdorferi lipoproteome reveals a compartmentalization bias toward the bacterial surface. J. Bacteriol. https://doi.org/10.1128/JB.00658-16 (2017).
Barbour, A. G. et al. A genome-wide proteome array reveals a limited set of immunogens in natural infections of humans and white-footed mice with Borrelia burgdorferi. Infect. Immun. 76, 3374–3389 (2008).
Oosting, M. et al. TLR1/TLR2 heterodimers play an important role in the recognition of Borrelia spirochetes. PLoS ONE 6, e25998 (2011).
Singh, S. K. & Girschick, H. J. Toll-like receptors in Borrelia burgdorferi-induced inflammation. Clin. Microbiol. Infect. 12, 705–717 (2006).
Cervantes, J. L. et al. Human TLR8 is activated upon recognition of Borrelia burgdorferi RNA in the phagosome of human monocytes. J. Leukoc. Biol. 94, 1231–1241 (2013).
Petzke, M. M., Brooks, A., Krupna, M. A., Mordue, D. & Schwartz, I. Recognition of Borrelia burgdorferi, the Lyme disease spirochete, by TLR7 and TLR9 induces a type I IFN response by human immune cells. J. Immunol. 183, 5279–5292 (2009).
Cervantes, J. L. et al. Phagosomal signaling by Borrelia burgdorferi in human monocytes involves Toll-like receptor (TLR) 2 and TLR8 cooperativity and TLR8-mediated induction of IFN-β. Proc. Natl Acad. Sci. USA 108, 3683–3688 (2011).
Jutras, B. L. et al. Borrelia burgdorferi peptidoglycan is a persistent antigen in patients with Lyme arthritis. Proc. Natl Acad. Sci. USA 116, 13498–13507 (2019).
Lochhead, R. B., Strle, K., Arvikar, S. L., Weis, J. J. & Steere, A. C. Lyme arthritis: linking infection, inflammation and autoimmunity. Nat. Rev. Rheumatol. 17, 449–461 (2021).
Sellati, T. J., Abrescia, L. D., Radolf, J. D. & Furie, M. B. Outer surface lipoproteins of Borrelia burgdorferi activate vascular endothelium in vitro. Infect. Immun. 64, 3180–3187 (1996).
Wooten, R. M., Modur, V. R., McIntyre, T. M. & Weis, J. J. Borrelia burgdorferi outer membrane protein a induces nuclear translocation of nuclear factor-kappa B and inflammatory activation in human endothelial cells. J. Immunol. 157, 4584–4590 (1996).
Schramm, F. et al. Microarray analyses of inflammation response of human dermal fibroblasts to different strains of Borrelia burgdorferi sensu stricto. PLoS ONE 7, e40046 (2012).
Shin, J. J., Glickstein, L. J. & Steere, A. C. High levels of inflammatory chemokines and cytokines in joint fluid and synovial tissue throughout the course of antibiotic-refractory Lyme arthritis. Arthritis Rheum. 56, 1325–1335 (2007).
Codolo, G. et al. Borrelia burgdorferi NapA-driven Th17 cell inflammation in Lyme arthritis. Arthritis Rheum. 58, 3609–3617 (2008).
Oosting, M. et al. Role of interleukin-23 (IL-23) receptor signaling for IL-17 responses in human Lyme disease. Infect. Immun. 79, 4681–4687 (2011).
Strle, K. et al. T-Helper 17 cell cytokine responses in Lyme disease correlate with Borrelia burgdorferi antibodies during early infection and with autoantibodies late in the illness in patients with antibiotic-refractory Lyme arthritis. Clin. Infect. Dis. 64, 930–938 (2017).
Marques, A. et al. Transcriptome assessment of erythema migrans skin lesions in patients with early Lyme disease reveals predominant interferon signaling. J. Infect. Dis. 217, 158–167 (2017).
Branda, J. A. & Steere, A. C. Laboratory diagnosis of Lyme borreliosis. Clin. Microbiol. Rev. https://doi.org/10.1128/CMR.00018-19 (2021).
Blum, L. K. et al. Robust B cell responses predict rapid resolution of Lyme disease. Front. Immunol. 9, 1634 (2018).
Hyde, J. A. Borrelia burgdorferi keeps moving and carries on: a review of borrelial dissemination and invasion. Front. Immunol. 8, 114 (2017).
Rupprecht, T. A., Koedel, U., Fingerle, V. & Pfister, H. W. The pathogenesis of Lyme neuroborreliosis: from infection to inflammation. Mol. Med. 14, 205–212 (2008).
Ogrinc, K. et al. Colocalization of radicular pain and erythema migrans in patients with Bannwarth syndrome suggests a direct spread of Borrelia into the central nervous system. Clin. Infect. Dis. 75, 81–87 (2022).
Gyllemark, P., Sjowall, J., Forsberg, P., Ernerudh, J. & Henningsson, A. J. Intrathecal Th17-driven inflammation is associated with prolonged post-treatment convalescence for patients with Lyme neuroborreliosis. Sci. Rep. 13, 9722 (2023).
Ogrinc, K. et al. Unique clinical, immune, and genetic signature in patients with borrelial meningoradiculoneuritis1. Emerg. Infect. Dis. 28, 766–776 (2022).
Steere, A. C. et al. Treatment of the early manifestations of Lyme disease. Ann. Intern. Med. 99, 22–26 (1983).
Ogrinc, K. et al. Acrodermatitis chronica atrophicans: clinical and microbiological characteristics of a cohort of 693 Slovenian patients. J. Intern. Med. 290, 335–348 (2021).
Aucott, J. N. et al. CCL19 as a chemokine risk factor for posttreatment Lyme disease syndrome: a prospective clinical cohort study. Clin. Vaccine Immunol. 23, 757–766 (2016).
Chandra, A. et al. Anti-neural antibody reactivity in patients with a history of Lyme borreliosis and persistent symptoms. Brain Behav. Immun. 24, 1018–1024 (2010).
Bockenstedt, L. K., Gonzalez, D. G., Haberman, A. M. & Belperron, A. A. Spirochete antigens persist near cartilage after murine Lyme borreliosis therapy. J. Clin. Invest. 122, 2652–2660 (2012).
Gyllemark, P., Forsberg, P., Ernerudh, J. & Henningsson, A. J. Intrathecal Th17- and B cell-associated cytokine and chemokine responses in relation to clinical outcome in Lyme neuroborreliosis: a large retrospective study. J. Neuroinflamm. 14, 27 (2017).
Strle, K., Shin, J. J., Glickstein, L. J. & Steere, A. C. Association of a toll-like receptor 1 polymorphism with heightened Th1 inflammatory responses and antibiotic-refractory Lyme arthritis. Arthritis Rheum. 64, 1497–1507 (2012).
Ehrbar, D. et al. Variants in the late cornified envelope gene locus are associated with elevated T-helper 17 responses in patients with postinfectious Lyme arthritis. J. Infect. Dis. 230, S40–S50 (2024).
Li, X. et al. Burden and viability of Borrelia burgdorferi in skin and joints of patients with erythema migrans or Lyme arthritis. Arthritis Rheum. 63, 2238–2247 (2011).
Stupica, D., Lusa, L., Cerar, T., Ruzic-Sabljic, E. & Strle, F. Comparison of post-Lyme borreliosis symptoms in erythema migrans patients with positive and negative Borrelia burgdorferi sensu lato skin culture. Vector Borne Zoonotic Dis. 11, 883–889 (2011).
Stupica, D., Lusa, L., Ruzic-Sabljic, E., Cerar, T. & Strle, F. Treatment of erythema migrans with doxycycline for 10 days versus 15 days. Clin. Infect. Dis. 55, 343–350 (2012).
Berende, A. et al. Randomized trial of longer-term therapy for symptoms attributed to Lyme disease. N. Engl. J. Med. 374, 1209–1220 (2016).
Fallon, B. A. et al. A randomized, placebo-controlled trial of repeated IV antibiotic therapy for Lyme encephalopathy. Neurology 70, 992–1003 (2008).
Klempner, M. S. et al. Two controlled trials of antibiotic treatment in patients with persistent symptoms and a history of Lyme disease. N. Engl. J. Med. 345, 85–92 (2001).
Krupp, L. B. et al. Study and treatment of post Lyme disease (STOP-LD): a randomized double masked clinical trial. Neurology 60, 1923–1930 (2003).
Sjowall, J., Ledel, A., Ernerudh, J., Ekerfelt, C. & Forsberg, P. Doxycycline-mediated effects on persistent symptoms and systemic cytokine responses post-neuroborreliosis: a randomized, prospective, cross-over study. BMC Infect. Dis. 12, 186 (2012).
Strle, K., Stupica, D., Drouin, E. E., Steere, A. C. & Strle, F. Elevated levels of IL-23 in a subset of patients with post-Lyme disease symptoms following erythema migrans. Clin. Infect. Dis. 58, 372–380 (2014).
Fitzgerald, B. L. et al. Metabolic response in patients with post-treatment Lyme disease symptoms/syndrome. Clin. Infect. Dis. 73, e2342–e2349 (2021).
Hernandez, S. A. et al. Association of persistent symptoms after Lyme neuroborreliosis and increased levels of interferon-alpha in blood. Emerg. Infect. Dis. 29, 1091–1101 (2023).
Jacek, E. et al. Increased IFNα activity and differential antibody response in patients with a history of Lyme disease and persistent cognitive deficits. J. Neuroimmunol. 255, 85–91 (2013).
Wormser, G. P., Nadelman, R. B. & Schwartz, I. The amber theory of Lyme arthritis: initial description and clinical implications. Clin. Rheumatol. 31, 989–994 (2012).
Crowley, J. T. et al. Matrix metalloproteinase-10 is a target of T and B cell responses that correlate with synovial pathology in patients with antibiotic-refractory Lyme arthritis. J. Autoimmunity 69, 24–37 (2016).
Crowley, J. T. et al. A highly expressed human protein, apolipoprotein B-100, serves as an autoantigen in a subgroup of patients with Lyme disease. J. Infect. Dis. 212, 1841–1850 (2015).
Drouin, E. E. et al. A novel human autoantigen, endothelial cell growth factor, is a target of T and B cell responses in patients with Lyme disease. Arthritis Rheum. 65, 186–196 (2013).
Kanjana, K. et al. Autoimmunity to synovial extracellular matrix proteins in patients with postinfectious Lyme arthritis. J. Clin. Invest. https://doi.org/10.1172/JCI161170 (2023).
Londono, D. et al. Antibodies to endothelial cell growth factor and obliterative microvascular lesions in the synovium of patients with antibiotic-refractory Lyme arthritis. Arthritis Rheumatol. 66, 2124–2133 (2014).
Pianta, A., Drouin, E. E., Arvikar, S., Costello, C. E. & Steere, A. C. Identification of annexin A-2 as an autoantigen in rheumatoid arthritis and in Lyme arthritis. Arthritis Rheum. 66, S437 (2014).
Pianta, A. et al. Annexin A2 is a target of autoimmune T and B cell responses associated with synovial fibroblast proliferation in patients with antibiotic-refractory Lyme arthritis. Clin. Immunol. 160, 336–341 (2015).
Shen, S. et al. Treg cell numbers and function in patients with antibiotic-refractory or antibiotic-responsive Lyme arthritis. Arthritis Rheum. 62, 2127–2137 (2010).
Vudattu, N. K., Strle, K., Steere, A. C. & Drouin, E. E. Dysregulation of CD4+CD25high T cells in the synovial fluid of patients with antibiotic-refractory Lyme arthritis. Arthritis Rheum. 65, 1643–1653 (2013).
Ruzic-Sabljic, E. et al. Characterization of Borrelia burgdorferi sensu lato strains isolated from human material in Slovenia. Wien Klin. Wochenschr. 114, 544–550 (2002).
Strle, F., Ruzic-Sabljic, E., Cimperman, J., Lotric-Furlan, S. & Maraspin, V. Comparison of findings for patients with Borrelia garinii and Borrelia afzelii isolated from cerebrospinal fluid. Clin. Infect. Dis. 43, 704–710 (2006).
Ogrinc, K. et al. Proportion of confirmed Lyme neuroborreliosis cases among adult patients with suspected early European Lyme neuroborreliosis. Infection 53, 1403–1412 (2025).
van Dam, A. P. et al. Different genospecies of Borrelia burgdorferi are associated with distinct clinical manifestations of Lyme borreliosis. Clin. Infect. Dis. 17, 708–717 (1993).
Grillon, A. et al. Characteristics and clinical outcomes after treatment of a national cohort of PCR-positive Lyme arthritis. Semin. Arthritis Rheum. 48, 1105–1112 (2019).
Strle, K. et al. Borrelia burgdorferi stimulates macrophages to secrete higher levels of cytokines and chemokines than Borrelia afzelii or Borrelia garinii. J. Infect. Dis. 200, 1936–1943 (2009).
Cerar, T. et al. Differences in genotype, clinical features, and inflammatory potential of Borrelia burgdorferi sensu stricto strains from Europe and the United States. Emerg. Infect. Dis. 22, 818–827 (2016).
Strle, F. et al. Comparison of culture-confirmed erythema migrans caused by Borrelia burgdorferi sensu stricto in New York State and by Borrelia afzelii in Slovenia. Ann. Intern. Med. 130, 32–36 (1999).
Lin, Y. P. et al. Strain-specific variation of the decorin-binding adhesin DbpA influences the tissue tropism of the Lyme disease spirochete. PLoS Pathog. 10, e1004238 (2014).
Lin, Y. P. et al. Strain-specific joint invasion and colonization by Lyme disease spirochetes is promoted by outer surface protein C. PLoS Pathog. 16, e1008516 (2020).
Seshu, J. et al. Inactivation of the fibronectin-binding adhesin gene bbk32 significantly attenuates the infectivity potential of Borrelia burgdorferi. Mol. Microbiol. 59, 1591–1601 (2006).
Hyde, J. A. et al. Bioluminescent imaging of Borrelia burgdorferi in vivo demonstrates that the fibronectin-binding protein BBK32 is required for optimal infectivity. Mol. Microbiol. 82, 99–113 (2011).
Zhi et al. The BBA33 lipoprotein binds collagen and impacts Borrelia burgdorferi pathogenesis. Mol. Microbiol. 96, 68–83 (2015).
Wager, B., Shaw, D. K., Groshong, A. M., Blevins, J. S. & Skare, J. T. BB0744 affects tissue tropism and spatial distribution of Borrelia burgdorferi. Infect. Immun. 83, 3693–3703 (2015).
Kraiczy, P., Skerka, C., Kirschfink, M., Zipfel, P. F. & Brade, V. Mechanism of complement resistance of pathogenic Borrelia burgdorferi isolates. Int. Immunopharmacol. 1, 393–401 (2001).
Sandholm, K. et al. Early cytokine release in response to live Borrelia burgdorferi Sensu Lato Spirochetes is largely complement independent. PLoS ONE 9, e108013 (2014).
Marcinkiewicz, A. L. et al. Eliminating factor H-binding activity of Borrelia burgdorferi CspZ combined with virus-like particle conjugation enhances its efficacy as a Lyme disease vaccine. Front. Immunol. 9, 181 (2018).
Strle, K., Jones, K. L., Drouin, E. E., Li, X. & Steere, A. C. Borrelia burgdorferi RST1 (OspC type A) genotype is associated with greater inflammation and more severe Lyme disease. Am. J. Pathol. 178, 2726–2739 (2011).
Wormser, G. P. et al. Borrelia burgdorferi genotype predicts the capacity for hematogenous dissemination during early Lyme disease. J. Infect. Dis. 198, 1358–1364 (2008).
Schwartz, I., Margos, G., Casjens, S. R., Qiu, W. G. & Eggers, C. H. Multipartite genome of Lyme disease Borrelia: structure, variation and prophages. Curr. Issues Mol. Biol. 42, 409–454 (2021).
Akther, S. et al. Natural selection and recombination at host-interacting lipoprotein loci drive genome diversification of Lyme disease and related bacteria. mBio 15, e0174924 (2024).
Brandt, M. E., Riley, B. S., Radolf, J. D. & Norgard, M. V. Immunogenic integral membrane proteins of Borrelia burgdorferi are lipoproteins. Infect. Immun. 58, 983–991 (1990).
Zhang, J. R., Hardham, J. M., Barbour, A. G. & Norris, S. J. Antigenic variation in Lyme disease Borreliae by promiscuous recombination of VMP-like sequence cassettes. Cell 89, 275–285 (1997).
Casjens, S. R. et al. Primordial origin and diversification of plasmids in Lyme disease agent bacteria. BMC Genomics 19, 218 (2018).
Margos, G. et al. Lost in plasmids: next generation sequencing and the complex genome of the tick-borne pathogen Borrelia burgdorferi. BMC Genomics 18, 422 (2017).
Tyler, S. et al. Whole genome sequencing and phylogenetic analysis of strains of the agent of Lyme disease Borrelia burgdorferi from Canadian emergence zones. Sci. Rep. 8, 10552 (2018).
Steere, A. C., Dwyer, E. & Winchester, R. Association of chronic Lyme arthritis with HLA-DR4 and HLA-DR2 alleles. N. Engl. J. Med. 323, 219–223 (1990).
Steere, A. C. et al. Antibiotic-refractory Lyme arthritis is associated with HLA-DR molecules that bind a Borrelia burgdorferi peptide. J. Exp. Med. 203, 961–971 (2006).
Strausz, S. et al. SCGB1D2 inhibits growth of Borrelia burgdorferi and affects susceptibility to Lyme disease. Nat. Commun. 15, 2041 (2024).
Vrijmoeth, H. D. et al. Genome-wide analyses in Lyme borreliosis: identification of a genetic variant associated with disease susceptibility and its immunological implications. BMC Infect. Dis. 24, 337 (2024).
Bramwell, K. K., Teuscher, C. & Weis, J. J. Forward genetic approaches for elucidation of novel regulators of Lyme arthritis severity. Front. Cell Infect. Microbiol. 4, 76 (2014).
Nadelman, R. B. et al. The clinical spectrum of early Lyme borreliosis in patients with culture-confirmed erythema migrans. Am. J. Med. 100, 502–508 (1996).
Smith, R. P. et al. Clinical characteristics and treatment outcome of early Lyme disease in patients with microbiologically confirmed erythema migrans. Ann. Intern. Med. 136, 421–428 (2002).
Hunt, K. M. et al. Racial differences in the diagnosis of Lyme disease in children. Clin. Infect. Dis. 76, 1129–1131 (2023).
Maraspin, V. et al. Are differences in presentation of early Lyme borreliosis in Europe and North America a consequence of a more frequent spirochetemia in American patients? J. Clin. Med. https://doi.org/10.3390/jcm10071448 (2021).
Maraspin, V. et al. Characteristics of spirochetemic patients with a solitary erythema migrans skin lesion in Europe. PLoS ONE 16, e0250198 (2021).
Strle, F. et al. Comparison of erythema migrans caused by Borrelia burgdorferi and Borrelia garinii. Vector Borne Zoonotic Dis. 11, 1253–1258 (2011).
Logar, M. et al. Comparison of erythema migrans caused by Borrelia afzelii and Borrelia garinii. Infection 32, 15–19 (2004).
Steere, A. C. et al. Systemic symptoms without erythema migrans as the presenting picture of early Lyme disease. Am. J. Med. 114, 58–62 (2003).
Feder, H. M., Gerber, M. A., Krause, P. J., Ryan, R. & Shapiro, E. D. Early Lyme-disease — a flu-like illness without erythema migrans. Pediatrics 91, 456–459 (1993).
Maraspin, V. & Strle, F. Borrelial lymphocytoma. Wien Klin. Wochenschr. https://doi.org/10.1007/s00508-022-02064-5 (2022).
Brestrich, G. et al. Estimation of medically-attended LB incidence in Norway, Finland and Poland using data from national surveillance and published literature. Ticks Tick Borne Dis. 16, 102454 (2025).
Ogrinc, K. et al. Course and outcome of early European Lyme neuroborreliosis (Bannwarth syndrome): clinical and laboratory findings. Clin. Infect. Dis. 63, 346–353 (2016).
Knudtzen, F. C., Andersen, N. S., Jensen, T. G. & Skarphedinsson, S. Characteristics and clinical outcome of Lyme neuroborreliosis in a high endemic area, 1995-2014: a retrospective cohort study in Denmark. Clin. Infect. Dis. 65, 1489–1495 (2017).
Nordberg, C. L. et al. Lyme neuroborreliosis in adults: a nationwide prospective cohort study. Ticks Tick Borne Dis. 11, 101411 (2020).
Solheim, A. M. et al. Clinical and laboratory characteristics during a 1-year follow-up in European Lyme neuroborreliosis: a prospective cohort study. Eur. J. Neurol. 31, e16487 (2024).
Stupica, D. et al. Clinical manifestations and long-term outcome of early Lyme neuroborreliosis according to the European Federation of Neurological Societies diagnostic criteria (definite versus possible) in central Europe. A retrospective cohort study. Eur. J. Neurol. 28, 3155–3166 (2021).
Tetens, M. M. et al. Healthcare-seeking behaviour preceding diagnosis of Lyme neuroborreliosis: population-based nationwide matched nested case-control study. Clin. Microbiol. Infect. 30, 1576–1584 (2024).
Pachner, A. R. & Steere, A. C. The triad of neurologic manifestations of Lyme disease: meningitis, cranial neuritis, and radiculoneuritis. Neurology 35, 47–53 (1985).
Halperin, J. J., Eikeland, R., Branda, J. A. & Dersch, R. Lyme neuroborreliosis: known knowns, known unknowns. Brain 145, 2635–2647 (2022).
Bloch, J. et al. Peripheral facial palsy in children: serum Borrelia antibodies may reduce the need for lumbar puncture. Acta Paediatr. 114, 122–130 (2025).
Sodermark, L., Sigurdsson, V., Nas, W., Wall, P. & Trollfors, B. Neuroborreliosis in Swedish children: a population-based study on incidence and clinical characteristics. Pediatr. Infect. Dis. J. 36, 1052–1056 (2017).
Kaminsky, A.-L. et al. Confirmed cases of Neuroborreliosis with involvement of peripheral nervous system. Medicine 99, e21986 (2020).
Winter, Y. et al. Cerebral vasculitis as clinical manifestation of neuroborreliosis: pattern of vascular pathology and prognostic factors of outcome. Int. J. Stroke 20, 205–214 (2024).
Wittwer, B. et al. Cerebrovascular events in Lyme neuroborreliosis. J. Stroke Cerebrovasc. Dis. 24, 1671–1678 (2015).
Bruinsma, R. A., Zomer, T. P., Skogman, B. H., van Hensbroek, M. B. & Hovius, J. W. Clinical manifestations of Lyme neuroborreliosis in children: a review. Eur. J. Pediatr. 182, 1965–1976 (2023).
Garro, A. et al. Validation of the Rule of 7’s for identifying children at low-risk for Lyme meningitis. Pediatr. Infect. Dis. J. 40, 306–309 (2021).
Shen, R. V., McCarthy, C. A. & Smith, R. P. Lyme carditis in hospitalized children and adults, a case series. Open Forum Infect. Dis. 8, ofab140 (2021).
Steere, A. C. et al. Lyme carditis: cardiac abnormalities of Lyme disease. Ann. Intern. Med. 93, 8–16 (1980).
Tetens, M. M. et al. Risk of heart failure among individuals tested for Borrelia burgdorferi sensu lato antibodies, and serum Borrelia burgdorferi sensu lato seropositive individuals; a nationwide population-based, registry-based matched cohort study. Ticks Tick Borne Dis. 15, 102345 (2024).
Strle, F. et al. Gender disparity between cutaneous and non-cutaneous manifestations of Lyme borreliosis. PLoS ONE 8, e64110 (2013).
Vasiliu, V., Herzer, P., Rossler, D., Lehnert, G. & Wilske, B. Heterogeneity of Borrelia burgdorferi sensu lato demonstrated by an ospA-type-specific PCR in synovial fluid from patients with Lyme arthritis. Med. Microbiol. Immunol. 187, 97–102 (1998).
Jaulhac, B. et al. Direct molecular typing of Borrelia burgdorferi sensu lato species in synovial samples from patients with Lyme arthritis. J. Clin. Microbiol. 38, 1895–1900 (2000).
Corre, C. et al. Lyme arthritis in Western Europe: a multicentre retrospective study. Eur. J. Clin. Microbiol. Infect. Dis. 41, 21–27 (2022).
Steere, A. C. et al. Lyme arthritis: an epidemic of oligoarticular arthritis in children and adults in three Connecticut communities. Arthritis Rheum. 20, 7–17 (1977).
Steere, A. C., Schoen, R. T. & Taylor, E. The clinical evolution of Lyme arthritis. Ann. Intern. Med. 107, 725–731 (1987).
Arvikar, S. L. & Steere, A. C. Lyme arthritis. Infect. Dis. Clin. North Am. 36, 563–577 (2022).
Eckman, E. A., Pacheco-Quinto, J., Herdt, A. R. & Halperin, J. J. Neuroimmunomodulators in neuroborreliosis and Lyme encephalopathy. Clin. Infect. Dis. 67, 80–88 (2018).
Halperin, J. J., Little, B. W., Coyle, P. K. & Dattwyler, R. J. Lyme disease: cause of a treatable peripheral neuropathy. Neurology 37, 1700–1706 (1987).
Logigian, E. L. & Steere, A. C. Clinical and electrophysiologic findings in chronic neuropathy of Lyme disease. Neurology 42, 303–311 (1992).
Wormser, G. P., Strle, F., Shapiro, E. D., Dattwyler, R. J. & Auwaerter, P. G. A critical appraisal of the mild axonal peripheral neuropathy of late neurologic Lyme disease. Diagn. Microbiol. Infect. Dis. 87, 163–167 (2017).
Nelder, M. P. et al. Human pathogens associated with the blacklegged tick Ixodes scapularis: a systematic review. Parasit. Vectors 9, 265 (2016).
Grimaudo, A. T. et al. Geographic variation in risk of blacklegged tick-borne coinfections in the eastern United States. Ticks Tick Borne Dis. 17, 102610 (2026).
Taba, P. et al. EAN consensus review on prevention, diagnosis and management of tick-borne encephalitis. Eur. J. Neurol. 24, 1214–e61 (2017).
Gomer, A., Lang, A., Janshoff, S., Steinmann, J. & Steinmann, E. Epidemiology and global spread of emerging tick-borne Alongshan virus. Emerg. Microbes Infect. 13, 2404271 (2024).
Zhang, M. Z. et al. A series of patients infected with the emerging tick-borne Yezo virus in China: an active surveillance and genomic analysis. Lancet Infect. Dis. 25, 390–398 (2025).
Guan, R. et al. Prevalence of three important tick-borne pathogens in ticks and humans in Shaanxi province, northwest China. Parasitol. Int. 104, 102980 (2025).
Lv, X. et al. Yezo virus infection in tick-bitten patient and ticks, northeastern China. Emerg. Infect. Dis. 29, 797–800 (2023).
Jia, N. et al. Emergence of human infection with Jingmen tick virus in China: a retrospective study. EBioMedicine 43, 317–324 (2019).
Wang, Y. C. et al. A new nairo-like virus associated with human febrile illness in China. Emerg. Microbes Infect. 10, 1200–1208 (2021).
Ma, J. et al. Identification of a new orthonairovirus associated with human febrile illness in China. Nat. Med. 27, 434–439 (2021).
Kodama, F. et al. A novel nairovirus associated with acute febrile illness in Hokkaido, Japan. Nat. Commun. 12, 5539 (2021).
Wang, Z. D. et al. A new segmented virus associated with human febrile illness in China. N. Engl. J. Med. 380, 2116–2125 (2019).
Lindell, K. et al. Epidemiologic and clinical characteristics and outcomes of patients diagnosed with southern tick associated rash illness (STARI) — 2018-2019. Diagn. Microbiol. Infect. Dis. 113, 116928 (2025).
Wormser, G. P. et al. Prospective clinical evaluation of patients from Missouri and New York with erythema migrans-like skin lesions. Clin. Infect. Dis. 41, 958–965 (2005).
Wormser, G. P. et al. Microbiologic evaluation of patients from Missouri with erythema migrans. Clin. Infect. Dis. 40, 423–428 (2005).
Stromdahl, E. Y. & Hickling, G. J. Beyond Lyme: aetiology of tick-borne human diseases with emphasis on the south-eastern United States. Zoonoses Public Health 59 (Suppl. 2), 48–64 (2012).
Molaei, G., Little, E. A. H., Williams, S. C. & Stafford, K. C. Bracing for the worst — range expansion of the lone star tick in the northeastern United States. N. Engl. J. Med. 381, 2189–2192 (2019).
Molaei, G., Eisen, L. M., Price, K. J. & Eisen, R. J. Range expansion of native and invasive ticks: a looming public health threat. J. Infect. Dis. 226, 370–373 (2022).
Raghavan, R. K., Peterson, A. T., Cobos, M. E., Ganta, R. & Foley, D. Current and future distribution of the lone star tick, Amblyomma americanum (L.) (Acari: Ixodidae) in North America. PLoS ONE 14, e0209082 (2019).
Molins, C. R. et al. Metabolic differentiation of early Lyme disease from southern tick-associated rash illness (STARI). Sci. Transl. Med. https://doi.org/10.1126/scitranslmed.aal2717 (2017).
Moriyama, Y. et al. Three cases diagnosed not Lyme disease but “tick-associated rash illness (TARI)” in Japan. J. Infect. Chemother. 27, 650–652 (2021).
Natsuaki, M., Takada, N., Kawabata, H., Ando, S. & Yamanishi, K. Case of tick-associated rash illness caused by Amblyomma testudinarium. J. Dermatol. 41, 834–836 (2014).
Stanek, G. et al. Lyme borreliosis: clinical case definitions for diagnosis and management in Europe. Clin. Microbiol. Infect. 17, 69–79 (2011).
Mygland, A. et al. EFNS guidelines on the diagnosis and management of European Lyme neuroborreliosis. Eur. J. Neurol. 17, 8–16, e11–14. (2010).
Lantos, P. M. et al. Clinical practice guidelines by the Infectious Diseases Society of America (IDSA), American Academy of Neurology (AAN), and American College of Rheumatology (ACR): 2020 guidelines for the prevention, diagnosis and treatment of Lyme disease. Clin. Infect. Dis. 72, 1–8 (2021).
Wormser, G. P. et al. The clinical assessment, treatment, and prevention of Lyme disease, human granulocytic anaplasmosis, and babesiosis: clinical practice guidelines by the Infectious Diseases Society of America. Clin. Infect. Dis. 43, 1089–1134 (2006).
Leeflang, M. M. et al. The diagnostic accuracy of serological tests for Lyme borreliosis in Europe: a systematic review and meta-analysis. BMC Infect. Dis. 16, 140 (2016).
Waddell, L. A. et al. The accuracy of diagnostic tests for Lyme disease in humans, a systematic review and meta-analysis of North American research. PLoS ONE 11, e0168613 (2016).
Dessau, R. B. et al. To test or not to test? Laboratory support for the diagnosis of Lyme borreliosis: a position paper of ESGBOR, the ESCMID study group for Lyme borreliosis. Clin. Microbiol. Infect. 24, 118–124 (2018).
Wilske, B., Fingerle, V. & Schulte-Spechtel, U. Microbiological and serological diagnosis of Lyme borreliosis. FEMS Immunol. Med. Microbiol. 49, 13–21 (2007).
Mead, P., Petersen, J. & Hinckley, A. Updated CDC recommendation for serologic diagnosis of Lyme disease. MMWR Morb. Mortal. Wkly Rep. 68, 703 (2019).
Hauser, U., Lehnert, G. & Wilske, B. Validity of interpretation criteria for standardized Western blots (immunoblots) for serodiagnosis of Lyme borreliosis based on sera collected throughout Europe. J. Clin. Microbiol. 37, 2241–2247 (1999).
Marques, A. R. Revisiting the Lyme disease serodiagnostic algorithm: the momentum gathers. J. Clin. Microbiol. https://doi.org/10.1128/JCM.00749-18 (2018).
Hoeve-Bakker, B. J. A., Kerkhof, K., Heron, M., Thijsen, S. F. T. & van Gorkom, T. Evaluation of different standard and modified two-tier testing strategies for the laboratory diagnosis of Lyme borreliosis in a European setting. Eur. J. Clin. Microbiol. Infect. Dis. 43, 2397–2406 (2024).
Kobayashi, T. & Auwaerter, P. G. Diagnostic testing for Lyme disease. Infect. Dis. Clin. North Am. 36, 605–620 (2022).
Hinckley, A. F. et al. Lyme disease testing by large commercial laboratories in the United States. Clin. Infect. Dis. 59, 676–681 (2014).
Lager, M. et al. Serological diagnostics of Lyme borreliosis: comparison of assays in twelve clinical laboratories in Northern Europe. Eur. J. Clin. Microbiol. Infect. Dis. 38, 1933–1945 (2019).
Kalish, R. A. et al. Persistence of immunoglobulin M or immunoglobulin G antibody responses to Borrelia burgdorferi 10-20 years after active Lyme disease. Clin. Infect. Dis. 33, 780–785 (2001).
Coors, A. et al. Regional seropositivity for Borrelia burgdorferi and associated risk factors: findings from the Rhineland study, Germany. Parasit. Vectors 15, 241 (2022).
Dehnert, M. et al. Seropositivity of Lyme borreliosis and associated risk factors: a population-based study in children and adolescents in Germany (KiGGS). PLoS ONE 7, e41321 (2012).
Garro, A. et al. Positive 2-tiered Lyme disease serology is uncommon in asymptomatic children living in endemic areas of the United States. Pediatr. Infect. Dis. J. 38, e105–e107 (2019).
Brummitt, S. I. et al. Borrelia burgdorferi and Borrelia miyamotoi seroprevalence in California blood donors. PLoS ONE 15, e0243950 (2020).
Sfeir, M. M. et al. Multicenter clinical evaluation of modified two-tiered testing algorithms for Lyme disease using Zeus Scientific commercial assays. J. Clin. Microbiol. 60, e0252821 (2022).
Wormser, G. P. et al. Single-tier testing with the C6 peptide ELISA kit compared with two-tier testing for Lyme disease. Diagn. Microbiol. Infect. Dis. 75, 9–15 (2013).
Wilhelmsson, P. et al. A prospective study on the incidence of Borrelia burgdorferi sensu lato infection after a tick bite in Sweden and on the Aland Islands, Finland (2008-2009). Ticks Tick Borne Dis. 7, 71–79 (2016).
Steere, A. C., Sikand, V. K., Schoen, R. T. & Nowakowski, J. Asymptomatic infection with Borrelia burgdorferi. Clin. Infect. Dis. 37, 528–532 (2003).
Nadelman, R. B. et al. Prophylaxis with single-dose doxycycline for the prevention of Lyme disease after an Ixodes scapularis tick bite. N. Engl. J. Med. 345, 79–84 (2001).
Rojko, T., Ruzic-Sabljic, E., Strle, F. & Lotric-Furlan, S. Prevalence and incidence of Lyme borreliosis among Slovene forestry workers during the period of tick activity. Wien Klin. Wochenschr. 117, 219–225 (2005).
Hansen, K. & Lebech, A. M. Lyme neuroborreliosis: a new sensitive diagnostic assay for intrathecal synthesis of Borrelia burgdorferi-specific immunoglobulin G, A, and M. Ann. Neurol. 30, 197–205 (1991).
Reiber, H. & Peter, J. B. Cerebrospinal fluid analysis: disease-related data patterns and evaluation programs. J. Neurol. Sci. 184, 101–122 (2001).
Cerar, T., Ogrinc, K., Strle, F. & Ruzic-Sabljic, E. Humoral immune responses in patients with Lyme neuroborreliosis. Clin. Vaccine Immunol. 17, 645–650 (2010).
Tetens, M. M. et al. The diagnostic value of serum Borrelia burgdorferi antibodies and seroconversion after Lyme neuroborreliosis, a nationwide observational study. Clin. Microbiol. Infect. 28, 1500.e1–1500.e6 (2022).
Orfanos, I., Chytilova, V. & Eklund, E. A. Clinical characteristics of children with Borrelia-related peripheral facial palsy and utility of cerebrospinal fluid testing. Acta Paediatr. 113, 1630–1636 (2024).
Zomer, T. P. et al. Lyme neuroborreliosis with antibodies in cerebrospinal fluid but not in serum. Eur. J. Neurol. 30, 741–744 (2023).
Naudion, P. et al. Positive intrathecal anti-Borrelia antibody synthesis: what are the implications for clinical practice? Clinical features and outcomes of 138 patients in a French multicenter cohort study. Eur. J. Clin. Microbiol. Infect. Dis. 42, 441–452 (2023).
Hammers-Berggren, S., Hansen, K., Lebech, A. M. & Karlsson, M. Borrelia burgdorferi-specific intrathecal antibody production in neuroborreliosis: a follow-up study. Neurology 43, 169–175 (1993).
Krogen, I., Skarphedinsson, S., Jensen, T. G. & Knudtzen, F. C. No correlation between symptom duration and intrathecal production of IgM and/or IgG antibodies in Lyme neuroborreliosis — a retrospective cohort study in Denmark. J. Infect. 85, 507–512 (2022).
Steere, A. C., Berardi, V. P., Weeks, K. E., Logigian, E. L. & Ackermann, R. Evaluation of the intrathecal antibody response to Borrelia burgdorferi as a diagnostic test for Lyme neuroborreliosis. J. Infect. Dis. 161, 1203–1209 (1990).
Eckman, E. A., Clausen, D. M., Herdt, A. R., Pacheco-Quinto, J. & Halperin, J. J. Specificity and diagnostic utility of cerebrospinal fluid CXCL13 in Lyme neuroborreliosis. Clin. Infect. Dis. 72, 1719–1726 (2021).
Theel, E. S., Aguero-Rosenfeld, M. E., Pritt, B., Adem, P. V. & Wormser, G. P. Limitations and confusing aspects of diagnostic testing for neurologic Lyme disease in the United States. J. Clin. Microbiol. https://doi.org/10.1128/JCM.01406-18 (2019).
Guerin, M. et al. Lyme borreliosis diagnosis: state of the art of improvements and innovations. BMC Microbiology 23, 204 (2023).
Rupprecht, T. A. et al. Diagnostic value of cerebrospinal fluid CXCL13 for acute Lyme neuroborreliosis. A systematic review and meta-analysis. Clin. Microbiol. Infect. 24, 1234–1240 (2018).
Masouris, I., Klein, M. & Kodel, U. The potential for CXCL13 in CSF as a differential diagnostic tool in central nervous system infection. Expert Rev. Anti Infect. Ther. 18, 875–885 (2020).
Eisen, L. Personal protection measures to prevent tick bites in the United States: knowledge gaps, challenges, and opportunities. Ticks Tick Borne Dis. 13, 101944 (2022).
Tarsus Pharmaceuticals. Tarsus Announces Positive Topline Results from Carpo, a Phase 2a Proof-of-Concept “Tick-Kill” Trial Evaluating TP-05 (lotilaner) for the Prevention of Lyme Disease. Tarsus Pharmaceuticals http://ir.tarsusrx.com/news-releases/news-release-details/tarsus-announces-positive-topline-results-carpo-phase-2a-proof (2024).
Harms, M. G. et al. A single dose of doxycycline after an Ixodes ricinus tick bite to prevent Lyme borreliosis: an open-label randomized controlled trial. J. Infect. 82, 98–104 (2021).
Schwan, T. G., Piesman, J., Golde, W. T., Dolan, M. C. & Rosa, P. A. Induction of an outer surface protein on Borrelia burgdorferi during tick feeding. Proc. Natl Acad. Sci. USA 92, 2909–2913 (1995).
Gomes-Solecki, M. et al. Protective immunity and new vaccines for Lyme disease. Clin. Infect. Dis. 70, 1768–1773 (2020).
de Silva, A. M. et al. Borrelia burgdorferi OspA is an arthropod-specific transmission-blocking Lyme disease vaccine. J. Exp. Med. 183, 271–275 (1996).
Van Hoecke, C., Lebacq, E., Beran, J. & Parenti, D. Alternative vaccination schedules (0, 1, and 6 months versus 0, 1, and 12 months) for a recombinant OspA Lyme disease vaccine. Clin. Infect. Dis. 28, 1260–1264 (1999).
Schoen, R. T. et al. Safety and immunogenicity profile of a recombinant outer-surface protein A Lyme disease vaccine: clinical trial of a 3-dose schedule at 0, 1, and 2 months. Clin. Ther. 22, 315–325 (2000).
Wormser, G. P. A brief history of OspA vaccines including their impact on diagnostic testing for Lyme disease. Diagn. Microbiol. Infect. Dis. 102, 115572 (2022).
Bezay, N. et al. Safety and immunogenicity of a novel multivalent OspA-based vaccine candidate against Lyme borreliosis: a randomised, phase 1 study in healthy adults. Lancet Infect. Dis. 23, 1186–1196 (2023).
Bezay, N. et al. Optimisation of dose level and vaccination schedule for the VLA15 Lyme borreliosis vaccine candidate among healthy adults: two randomised, observer-blind, placebo-controlled, multicentre, phase 2 studies. Lancet Infect. Dis. 24, 1045–1058 (2024).
Ghadge, S. K. et al. Immunogenicity and safety of an 18-month booster dose of the VLA15 Lyme borreliosis vaccine candidate after primary immunisation in healthy adults in the USA: results of the booster phase of a randomised, controlled, phase 2 trial. Lancet Infect. Dis. 24, 1275–1286 (2024).
Wagner, L. et al. Immunogenicity and safety of an 18-month booster dose of the VLA15 Lyme borreliosis vaccine candidate after primary immunisation in children, adolescents, and adults in the USA: a randomised, observer-blind, placebo-controlled, phase 2 trial. Lancet Infect. Dis. https://doi.org/10.1016/S1473-3099(25)00541-9 (2025).
Wagner, L. et al. Immunogenicity and safety of different immunisation schedules of the VLA15 Lyme borreliosis vaccine candidate in adults, adolescents, and children: a randomised, observer-blind, placebo-controlled, phase 2 trial. Lancet Infect. Dis. 25, 986–999 (2025).
Simon, R., Lamberth, E., Stark, J. H. & Skinner, J. M. A human Lyme disease vaccine: two steps forward on the path to prevention. Expert Rev. Vaccines 25, 2607482 (2026).
Schiller, Z. A. et al. Blocking Borrelia burgdorferi transmission from infected ticks to nonhuman primates with a human monoclonal antibody. J. Clin. Invest. https://doi.org/10.1172/JCI144843 (2021).
Izac, J. R. & Marconi, R. T. Diversity of the Lyme disease spirochetes and its influence on immune responses to infection and vaccination. Vet. Clin. North Am. Small Anim. Pract. 49, 671–686 (2019).
Asbrink, E., Hovmark, A. & Olsson, I. Clinical manifestations of acrodermatitis chronica atrophicans in 50 Swedish patients. Zentralbl Bakteriol. Mikrobiol. Hyg. A 263, 253–261 (1986).
Strle, F. & Stanek, G. Clinical manifestations and diagnosis of Lyme borreliosis. Curr. Probl. Dermatol. 37, 51–110 (2009).
Hunfeld, K. P., Ruzic-Sabljic, E., Norris, D. E., Kraiczy, P. & Strle, F. In vitro susceptibility testing of Borrelia burgdorferi sensu lato isolates cultured from patients with erythema migrans before and after antimicrobial chemotherapy. Antimicrob. Agents Chemother. 49, 1294–1301 (2005).
Wormser, G. P. et al. Duration of antibiotic therapy for early Lyme disease. A randomized, double-blind, placebo-controlled trial. Ann. Intern. Med. 138, 697–704 (2003).
Cerar, D., Cerar, T., Ruzic-Sabljic, E., Wormser, G. P. & Strle, F. Subjective symptoms after treatment of early Lyme disease. Am. J. Med. 123, 79–86 (2010).
Stupica, D. et al. Treatment of erythema migrans with doxycycline for 7 days versus 14 days in Slovenia: a randomised open-label non-inferiority trial. Lancet Infect. Dis. 23, 371–379 (2023).
Borg, R. et al. Intravenous ceftriaxone compared with oral doxycycline for the treatment of Lyme neuroborreliosis. Scand. J. Infect. Dis. 37, 449–454 (2005).
Ljostad, U. & Henriksen, T. H. Management of neuroborreliosis in European adult patients. Acta Neurol. Scand. Suppl. 188, 22–28 (2008).
Rauer, S. et al. Guidelines for diagnosis and treatment in neurology — Lyme neuroborreliosis. Ger. Med. Sci. 18, Doc03 (2020).
Arnason, S. & Skogman, B. H. Effectiveness of antibiotic treatment in children with Lyme neuroborreliosis — a retrospective study. BMC Pediatr. 22, 332 (2022).
Solheim, A. M. et al. Six versus 2 weeks treatment with doxycycline in European Lyme neuroborreliosis: a multicentre, non-inferiority, double-blinded, randomised and placebo-controlled trial. J. Neurol. Neurosurg. Psychiatry 93, 1222–1228 (2022).
Dersch, R. & Rauer, S. Efficacy and safety of pharmacological treatments for Lyme neuroborreliosis: an updated systematic review. Eur. J. Neurol. 30, 3780–3788 (2023).
Ljostad, U. et al. Oral doxycycline versus intravenous ceftriaxone for European Lyme neuroborreliosis: a multicentre, non-inferiority, double-blind, randomised trial. Lancet Neurol. 7, 690–695 (2008).
Kortela, E. et al. Oral doxycycline compared to intravenous ceftriaxone in the treatment of Lyme neuroborreliosis: a multicenter, equivalence, randomized, open-label trial. Clin. Infect. Dis. 72, 1323–1331 (2021).
Arvikar, S. L. & Steere, A. C. Diagnosis and treatment of Lyme arthritis. Infect. Dis. Clin. North Am. 29, 269–280 (2015).
Figueroa, R. et al. Confirmation of Borrelia burgdorferi spirochetes by polymerase chain reaction in placentas of women with reactive serology for Lyme antibodies. Gynecol. Obstet. Invest. 41, 240–243 (1996).
Markowitz, L. E., Steere, A. C., Benach, J. L., Slade, J. D. & Broome, C. V. Lyme disease during pregnancy. JAMA 255, 3394–3396 (1986).
Walsh, C. A., Mayer, E. W. & Baxi, L. V. Lyme disease in pregnancy: case report and review of the literature. Obstet. Gynecol. Surv. 62, 41–50 (2007).
Maraspin, V. et al. Course and outcome of erythema migrans in pregnant women. J. Clin. Med. https://doi.org/10.3390/jcm9082364 (2020).
Waddell, L. A., Greig, J., Lindsay, L. R., Hinckley, A. F. & Ogden, N. H. A systematic review on the impact of gestational Lyme disease in humans on the fetus and newborn. PLoS ONE 13, e0207067 (2018).
van Samkar, A. et al. Clinical characteristics of Lyme neuroborreliosis in Dutch children and adults. Eur. J. Pediatr. 182, 1183–1189 (2023).
Daikh, B. E., Emerson, F. E., Smith, R. P., Lucas, F. L. & McCarthy, C. A. Lyme arthritis: a comparison of presentation, synovial fluid analysis, and treatment course in children and adults. Arthritis Care Res. 65, 1986–1990 (2013).
Maraspin, V. et al. Erythema migrans in solid-organ transplant recipients. Clin. Infect. Dis. 42, 1751–1754 (2006).
Maraspin, V., Ruzic-Sabljic, E., Lusa, L. & Strle, F. Course and outcome of early Lyme borreliosis in patients with hematological malignancies. Clin. Infect. Dis. 61, 427–431 (2015).
Furst, B., Glatz, M., Kerl, H. & Mullegger, R. R. The impact of immunosuppression on erythema migrans. A retrospective study of clinical presentation, response to treatment and production of Borrelia antibodies in 33 patients. Clin. Exp. Dermatol. 31, 509–514 (2006).
Forrester, J. D. et al. Notes from the field: update on Lyme carditis, groups at high risk, and frequency of associated sudden cardiac death — United States. MMWR Morb. Mortal. Wkly Rep. 63, 982–983 (2014).
Uzomah, U. A. et al. Incidence of carditis and predictors of pacemaker implantation in patients hospitalized with Lyme disease. PLoS ONE 16, e0259123 (2021).
Patel, R., Grogg, K. L., Edwards, W. D., Wright, A. J. & Schwenk, N. M. Death from inappropriate therapy for Lyme disease. Clin. Infect. Dis. 31, 1107–1109 (2000).
Lantos, P. M. et al. Unorthodox alternative therapies marketed to treat Lyme disease. Clin. Infect. Dis. 60, 1776–1782 (2015).
Marzec, N. S. et al. Serious bacterial infections acquired during treatment of patients given a diagnosis of chronic Lyme disease — United States. MMWR Morb. Mortal. Wkly Rep. 66, 607–609 (2017).
Strizova, Z., Patek, O., Vitova, L., Horackova, M. & Bartunkova, J. Internet-based self-diagnosis of Lyme disease caused death in a young woman with systemic lupus erythematosus. Jt Bone Spine 86, 650–651 (2019).
Obel, N. et al. Long term survival, health, social functioning, and education in patients with European Lyme neuroborreliosis: nationwide population based cohort study. BMJ 361, k1998 (2018).
Thortveit, E. T. et al. Subjective health complaints and exposure to tick-borne infections in southern Norway. Acta Neurol. Scand. 142, 260–266 (2020).
Bechtold, K. T., Rebman, A. W., Crowder, L. A., Johnson-Greene, D. & Aucott, J. N. Standardized symptom measurement of individuals with early Lyme disease over time. Arch. Clin. Neuropsychol. 32, 129–141 (2017).
Eliassen, K. E., Hjetland, R., Reiso, H., Lindbaek, M. & Tschudi-Madsen, H. Symptom load and general function among patients with erythema migrans: a prospective study with a 1-year follow-up after antibiotic treatment in Norwegian general practice. Scand. J. Prim. Health Care 35, 75–83 (2017).
Borsic, K., Blagus, R., Cerar, T., Strle, F. & Stupica, D. Clinical course, serologic response, and long-term outcome in elderly patients with early Lyme borreliosis. J. Clin. Med. https://doi.org/10.3390/jcm7120506 (2018).
Stupica, D. et al. Oral doxycycline versus intravenous ceftriaxone for treatment of multiple erythema migrans: an open-label alternate-treatment observational trial. J. Antimicrob. Chemother. 73, 1352–1358 (2018).
Wormser, G. P. et al. Prospective evaluation of the frequency and severity of symptoms in Lyme disease patients with erythema migrans compared with matched controls at baseline, 6 months, and 12 months. Clin. Infect. Dis. 71, 3118–3124 (2020).
Ursinus, J. et al. Prevalence of persistent symptoms after treatment for Lyme borreliosis: a prospective observational cohort study. Lancet Reg. Health Eur. 6, 100142 (2021).
Aucott, J. N. et al. Risk of post-treatment Lyme disease in patients with ideally-treated early Lyme disease: a prospective cohort study. Int. J. Infect. Dis. 116, 230–237 (2022).
Geebelen, L. et al. Non-specific symptoms and post-treatment Lyme disease syndrome in patients with Lyme borreliosis: a prospective cohort study in Belgium (2016-2020). BMC Infect. Dis. 22, 756 (2022).
Oksi, J. et al. Duration of antibiotic treatment in disseminated Lyme borreliosis: a double-blind, randomized, placebo-controlled, multicenter clinical study. Eur. J. Clin. Microbiol. Infect. Dis. 26, 571–581 (2007).
Eikeland, R., Mygland, A., Herlofson, K. & Ljostad, U. European neuroborreliosis: quality of life 30 months after treatment. Acta Neurol. Scand. 124, 349–354 (2011).
Wormser, G. P. et al. Long-term assessment of health-related quality of life in patients with culture-confirmed early Lyme disease. Clin. Infect. Dis. 61, 244–247 (2015).
Wills, A. B. et al. Long-term follow-up of patients with Lyme disease: longitudinal analysis of clinical and quality-of-life measures. Clin. Infect. Dis. 62, 1546–1551 (2016).
Andreassen, S. et al. Assessment of cognitive function, structural brain changes and fatigue 6 months after treatment of neuroborreliosis. J. Neurol. 270, 1430–1438 (2023).
Eikeland, R. et al. Patient-reported outcome after treatment for definite Lyme neuroborreliosis. Brain Behav. 10, e01595 (2020).
Vrijmoeth, H. D. et al. Determinants of persistent symptoms after treatment for Lyme borreliosis: a prospective observational cohort study. EBioMedicine 98, 104825 (2023).
van Middendorp, H. et al. Expectancies as predictors of symptom improvement after antimicrobial therapy for persistent symptoms attributed to Lyme disease. Clin. Rheumatol. 40, 4295–4308 (2021).
Nawrocki, C. C. et al. Nonspecific symptoms attributable to Lyme disease in high-incidence areas, United States, 2017-2021. Emerg. Infect. Dis. 31, 30–37 (2025).
Lantos, P. M. & Wormser, G. P. Chronic coinfections in patients diagnosed with chronic Lyme disease: a systematic review. Am. J. Med. 127, 1105–1110 (2014).
Sigal, L. H. Summary of the first 100 patients seen at a Lyme disease referral center. Am. J. Med. 88, 577–581 (1990).
Steere, A. C., Taylor, E., McHugh, G. L. & Logigian, E. L. The overdiagnosis of Lyme disease. JAMA 269, 1812–1816 (1993).
Rose, C. D., Fawcett, P. T., Gibney, K. M. & Doughty, R. A. The overdiagnosis of Lyme disease in children residing in an endemic area. Clin. Pediatrics 33, 663–668 (1994).
Reid, M. C., Schoen, R. T., Evans, J., Rosenberg, J. C. & Horwitz, R. I. The consequences of overdiagnosis and overtreatment of Lyme disease: an observational study. Ann. Intern. Med. 128, 354–362 (1998).
Aucott, J. N., Seifter, A. & Rebman, A. W. Probable late Lyme disease: a variant manifestation of untreated Borrelia burgdorferi infection. BMC Infect. Dis. 12, 173 (2012).
Kobayashi, T. et al. Misdiagnosis of Lyme disease with unnecessary antimicrobial treatment characterizes patients referred to an academic infectious diseases clinic. Open Forum Infect. Dis. https://doi.org/10.1093/ofid/ofz299 (2019).
Gynthersen, R. M. M. et al. Classification of patients referred under suspicion of tick-borne diseases, Copenhagen, Denmark. Ticks Tick Borne Dis. 12, 101591 (2021).
Kortela, E., Kanerva, M., Kurkela, S., Oksi, J. & Jarvinen, A. Suspicion of Lyme borreliosis in patients referred to an infectious diseases clinic: what did the patients really have? Clin. Microbiol. Infect. 27, 1022–1028 (2021).
Kobayashi, T., Higgins, Y., Melia, M. T. & Auwaerter, P. G. Mistaken identity: many diagnoses are frequently misattributed to Lyme disease. Am. J. Med. 135, 503–511 e505 (2022).
Cottle, L. E., Mekonnen, E., Beadsworth, M. B., Miller, A. R. & Beeching, N. J. Lyme disease in a British referral clinic. QJM 105, 537–543 (2012).
Coumou, J. et al. Ticking the right boxes: classification of patients suspected of Lyme borreliosis at an academic referral center in the Netherlands. Clin. Microbiol. Infect. 21, 368.e11–20 (2015).
Bouiller, K., Klopfenstein, T. & Chirouze, C. Consultation for presumed Lyme borreliosis: the need for multidisciplinary management. Clin. Infect. Dis. 68, 1974 (2019).
Haddad, E. et al. Holistic approach in patients with presumed Lyme borreliosis leads to less than 10% of confirmation and more than 80% of antibiotic failures. Clin. Infect. Dis. 68, 2060–2066 (2019).
Jacquet, C. et al. Multidisciplinary management of patients presenting with Lyme disease suspicion. Med. Mal. Infect. 49, 112–120 (2019).
Djukic, M. et al. The diagnostic spectrum in patients with suspected chronic Lyme neuroborreliosis — the experience from one year of a university hospital’s Lyme neuroborreliosis outpatients clinic. Eur. J. Neurol. 18, 547–555 (2011).
Qureshi, M. Z., New, D., Zulqarni, N. J. & Nachman, S. Overdiagnosis and overtreatment of Lyme disease in children. Pediatr. Infect. Dis. J. 21, 12–14 (2002).
van de Schoor, F. R. et al. Evaluation and 1-year follow-up of patients presenting at a Lyme borreliosis expertise centre: a prospective cohort study with validated questionnaires. Eur. J. Clin. Microbiol. Infect. Dis. 43, 937–946 (2024).
Raffetin, A. et al. Acceptance of diagnosis and management satisfaction of patients with “suspected Lyme borreliosis” after 12 months in a multidisciplinary reference center: a prospective cohort study. BMC Infect. Dis. 23, 380 (2023).
Raffetin, A. et al. Multidisciplinary management of suspected Lyme borreliosis: clinical features of 569 patients, and factors associated with recovery at 3 and 12 months, a prospective cohort study. Microorganisms https://doi.org/10.3390/microorganisms10030607 (2022).
Conlon, C. P., Wormser, G. P. & Strle, F. in Oxford Textbook of Medicine (eds Firth, J., Conlon, C. & Cox, T.) 1182–1187 (Oxford University Press, 2020).
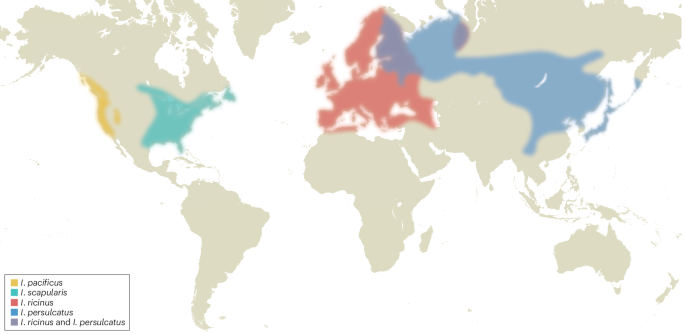
ECDC. Tick Species Distribution Maps: Ixodes ricinus/persultatus — known distribution September 2021. ECDC https://www.ecdc.europa.eu/en/disease-vectors/surveillance-and-disease-data/tick-maps (2024).
Eisen, R. J., Eisen, L. & Beard, C. B. County-scale distribution of Ixodes scapularis and Ixodes pacificus (Acari: Ixodidae) in the continental United States. J. Med. Entomol. 53, 349–386 (2016).
Wang, S. S. et al. Geographical distribution of Ixodes persulcatus and associated pathogens: analysis of integrated data from a China field survey and global published data. One Health 16, 100508 (2023).
Rar, V. et al. Ixodes persulcatus/pavlovskyi natural hybrids in Siberia: occurrence in sympatric areas and infection by a wide range of tick-transmitted agents. Ticks Tick Borne Dis. 10, 101254 (2019).
Mead, P. in Lyme Disease: An Evidence-based Approach (ed. Halperin, J.) 53–73 (CABI, 2025).
Nadelman, R. B. Erythema migrans. Infect. Dis. Clin. North Am. 29, 211–239 (2015).













Leave a Reply