Grattoni, A. et al. Harnessing cellular therapeutics for type 1 diabetes mellitus: progress, challenges, and the road ahead. Nat. Rev. Endocrinol. 21, 14–30 (2025).
Bluestone, J. A., Buckner, J. H. & Herold, K. C. Immunotherapy: building a bridge to a cure for type 1 diabetes. Science 373, 510–516 (2021).
Herold, K. C. et al. An anti-CD3 antibody, teplizumab, in relatives at risk for type 1 diabetes. N. Engl. J. Med. 381, 603–613 (2019).
Warshauer, J. T., Bluestone, J. A. & Anderson, M. S. New frontiers in the treatment of type 1 diabetes. Cell Metab. 31, 46–61 (2020).
Xiao, Y. et al. Fatty acid binding protein 4 promotes autoimmune diabetes by recruitment and activation of pancreatic islet macrophages. JCI Insight 6, e141814 (2021).
Gur, C. et al. The activating receptor NKp46 is essential for the development of type 1 diabetes. Nat. Immunol. 11, 121–128 (2010).
Gomez-Muñoz, L. et al. NK cell subsets changes in partial remission and early stages of pediatric type 1 diabetes. Front. Immunol. 11, 611522 (2020).
Dean, J. W. et al. Innate inflammation drives NK cell activation to impair Treg activity. J. Autoimmun. 108, 102417 (2020).
Xhonneux, L. P. et al. Transcriptional networks in at-risk individuals identify signatures of type 1 diabetes progression. Sci. Transl. Med. 13, eabd5666 (2021).
Ogasawara, K. et al. NKG2D blockade prevents autoimmune diabetes in NOD mice. Immunity 20, 757–767 (2004).
Beilke, J. N. et al. NK cells are not required for spontaneous autoimmune diabetes in NOD mice. PLoS ONE 7, e36011 (2012).
Herold, K. C. et al. The immunology of type 1 diabetes. Nat. Rev. Immunol. 24, 435–451 (2024).
Rebuffet, L. et al. High-dimensional single-cell analysis of human natural killer cell heterogeneity. Nat. Immunol. 25, 1474–1488 (2024).
Tang, F. et al. A pan-cancer single-cell panorama of human natural killer cells. Cell 186, 4235–4251.e20 (2023).
Zhong, T. et al. The remission phase in type 1 diabetes: changing epidemiology, definitions, and emerging immuno-metabolic mechanisms. Diabetes Metab. Res. Rev. 36, e3207 (2020).
Gomez-Muñoz, L., Dominguez-Bendala, J., Pastori, R. L. & Vives-Pi, M. Immunometabolic biomarkers for partial remission in type 1 diabetes mellitus. Trends Endocrinol. Metab. 35, 151–163 (2024).
Li, X. et al. PD-1 and PD-L1 Expression in peripheral CD4/CD8+ T cells is restored in the partial remission phase in type 1 diabetes. J. Clin. Endocrinol. Metab. 105, dgaa130 (2020).
Zhong, T. et al. CXCL12–CXCR4 mediates CD57+ CD8+ T cell responses in the progression of type 1 diabetes. J. Autoimmun. 143, 103171 (2024).
Zhong, T. et al. TGF-β-mediated crosstalk between TIGIT+ Tregs and CD226+CD8+ T cells in the progression and remission of type 1 diabetes. Nat. Commun. 15, 8894 (2024).
Crinier, A. et al. High-dimensional single-cell analysis identifies organ-specific signatures and conserved NK cell subsets in humans and mice. Immunity 49, 971–986.e5 (2018).
Di Vito, C., Mikulak, J. & Mavilio, D. On the way to become a natural killer cell. Front. Immunol. 10, 1812 (2019).
Gulati, G. S. et al. Single-cell transcriptional diversity is a hallmark of developmental potential. Science 367, 405–411 (2020).
Thacker, G. et al. Immature natural killer cells promote progression of triple-negative breast cancer. Sci. Transl. Med. 15, eabl4414 (2023).
Zakharov, P. N., Hu, H., Wan, X. & Unanue, E. R. Single-cell RNA sequencing of murine islets shows high cellular complexity at all stages of autoimmune diabetes. J. Exp. Med. 217, e20192362 (2020).
Jiang, P. et al. Systematic investigation of cytokine signaling activity at the tissue and single-cell levels. Nat. Methods 18, 1181–1191 (2021).
Martinet, L. & Smyth, M. J. Balancing natural killer cell activation through paired receptors. Nat. Rev. Immunol. 15, 243–254 (2015).
Briukhovetska, D. et al. T cell-derived interleukin-22 drives the expression of CD155 by cancer cells to suppress NK cell function and promote metastasis. Immunity 56, 143–161.e11 (2023).
Hsieh, W. C. et al. NK cell receptor and ligand composition influences the clearance of SARS-CoV-2. J. Clin. Invest. 131, e146408 (2021).
Martinet, L. et al. DNAM-1 expression marks an alternative program of NK cell maturation. Cell Rep. 11, 85–97 (2015).
Stannard, K. A. et al. Human peripheral blood DNAM-1neg NK cells are a terminally differentiated subset with limited effector functions. Blood Adv. 3, 1681–1694 (2019).
Crinier, A., Narni-Mancinelli, E., Ugolini, S. & Vivier, E. SnapShot: natural killer cells. Cell 180, 1280–1280.e1 (2020).
Glaviano, A. et al. PI3K/AKT/mTOR signaling transduction pathway and targeted therapies in cancer. Mol. Cancer 22, 138 (2023).
Chou, W. C., Rampanelli, E., Li, X. & Ting, J. P. Impact of intracellular innate immune receptors on immunometabolism. Cell. Mol. Immunol. 19, 337–351 (2022).
Chi, H. Regulation and function of mTOR signalling in T cell fate decisions. Nat. Rev. Immunol. 12, 325–338 (2012).
Jeong, W. I., Park, O., Radaeva, S. & Gao, B. STAT1 inhibits liver fibrosis in mice by inhibiting stellate cell proliferation and stimulating NK cell cytotoxicity. Hepatology 44, 1441–1451 (2006).
Gotthardt, D. et al. Loss of STAT3 in murine NK cells enhances NK cell-dependent tumor surveillance. Blood 124, 2370–2379 (2014).
Sutherland, A. P. et al. Interleukin-21 is required for the development of type 1 diabetes in NOD mice. Diabetes 58, 1144–1155 (2009).
Ferreira, R. C. et al. IL-21 production by CD4+ effector T cells and frequency of circulating follicular helper T cells are increased in type 1 diabetes patients. Diabetologia 58, 781–790 (2015).
Rydén, A. K. et al. Anti-IL-21 monoclonal antibody combined with liraglutide effectively reverses established hyperglycemia in mouse models of type 1 diabetes. J. Autoimmun. 84, 65–74 (2017).
von Herrath, M. et al. Anti-interleukin-21 antibody and liraglutide for the preservation of β-cell function in adults with recent-onset type 1 diabetes: a randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Diabetes Endocrinol. 9, 212–224 (2021).
Van Belle, T. L., Nierkens, S., Arens, R. & von Herrath, M. G. Interleukin-21 receptor-mediated signals control autoreactive T cell infiltration in pancreatic islets. Immunity 36, 1060–1072 (2012).
Duurland, C. L. et al. CD161 expression and regulation defines rapidly responding effector CD4+ T cells associated with improved survival in HPV16-associated tumors. J. Immunother. Cancer 10, e003995 (2022).
Kleinschek, M. A. et al. Circulating and gut-resident human Th17 cells express CD161 and promote intestinal inflammation. J. Exp. Med. 206, 525–534 (2009).
Liu, R. et al. Autoreactive T cells mediate NK cell degeneration in autoimmune disease. J. Immunol. 176, 5247–5254 (2006).
Parrish-Novak, J. et al. Interleukin 21 and its receptor are involved in NK cell expansion and regulation of lymphocyte function. Nature 408, 57–63 (2000).
Seyedsadr, M. et al. A pathologically expanded, clonal lineage of IL-21-producing CD4+ T cells drives inflammatory neuropathy. J. Clin. Invest. 134, e178602 (2024).
Suto, A. et al. Development and characterization of IL-21-producing CD4+ T cells. J. Exp. Med. 205, 1369–1379 (2008).
Braun, M. et al. CD155 on tumor cells drives resistance to immunotherapy by inducing the degradation of the activating receptor CD226 in CD8+ T cells. Immunity 53, 805–823.e15 (2020).
Brown, M. E. et al. Human stem cell-derived β cells expressing an optimized CD155 reduce cytotoxic immune cell function for application in type 1 diabetes. Sci. Adv. 11, eadx9755 (2025).
American Diabetes Association Professional Practice Committee. Diagnosis and classification of diabetes: standards of care in diabetes—2024. Diabetes Care 47, S20–S42 (2024).
Mortensen, H. B. et al. New definition for the partial remission period in children and adolescents with type 1 diabetes. Diabetes Care 32, 1384–1390 (2009).
Butler, A., Hoffman, P., Smibert, P., Papalexi, E. & Satija, R. Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat. Biotechnol. 36, 411–420 (2018).
Korsunsky, I. et al. Fast, sensitive and accurate integration of single-cell data with Harmony. Nat. Methods 16, 1289–1296 (2019).
Aibar, S. et al. SCENIC: single-cell regulatory network inference and clustering. Nat. Methods 14, 1083–1086 (2017).
Jin, S. et al. Inference and analysis of cell–cell communication using CellChat. Nat. Commun. 12, 1088 (2021).
Linderman, G. C. et al. Zero-preserving imputation of single-cell RNA-seq data. Nat. Commun. 13, 192 (2022).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
McGuire, H. M. et al. Interleukin-21 is critically required in autoimmune and allogeneic responses to islet tissue in murine models. Diabetes 60, 867–875 (2011).





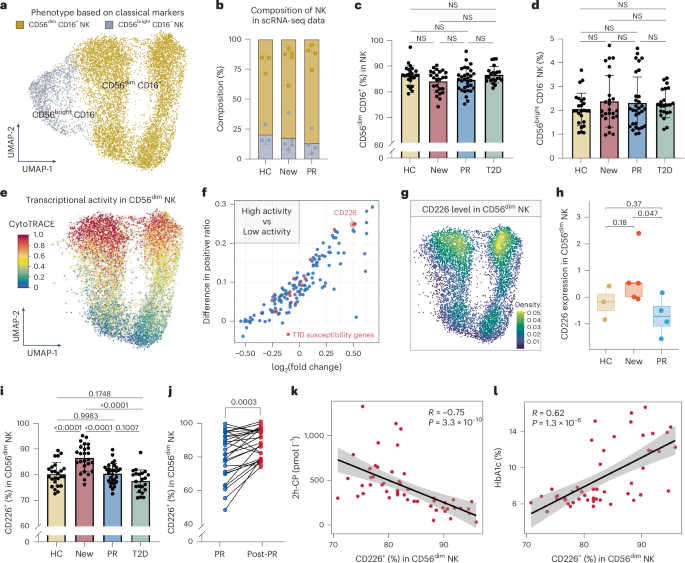









Leave a Reply