Huang, Y., Xu, W. & Zhou, R. NLRP3 inflammasome activation and cell death. Cell. Mol. Immunol. 18, 2114–2127 (2021).
Xu, J. & Nunez, G. The NLRP3 inflammasome: activation and regulation. Trends Biochem. Sci. 48, 331–344 (2023).
Swanson, K. V., Deng, M. & Ting, J. P. The NLRP3 inflammasome: molecular activation and regulation to therapeutics. Nat. Rev. Immunol. 19, 477–489 (2019).
Fu, J. & Wu, H. Structural mechanisms of NLRP3 inflammasome assembly and activation. Annu. Rev. Immunol. 41, 301–316 (2023).
O’Keefe, M. E., Dubyak, G. R. & Abbott, D. W. Post-translational control of NLRP3 inflammasome signaling. J. Biol. Chem. 300, 107386 (2024).
Sharma, B. R. & Kanneganti, T. D. NLRP3 inflammasome in cancer and metabolic diseases. Nat. Immunol. 22, 550–559 (2021).
Chen, Y. et al. The NLRP3 inflammasome: contributions to inflammation-related diseases. Cell. Mol. Biol. Lett. 28, 51 (2023).
Zhou, J. et al. Pyroptosis and degenerative diseases of the elderly. Cell Death Dis. 14, 94 (2023).
Schroder, K. & Tschopp, J. The inflammasomes. Cell 140, 821–832 (2010).
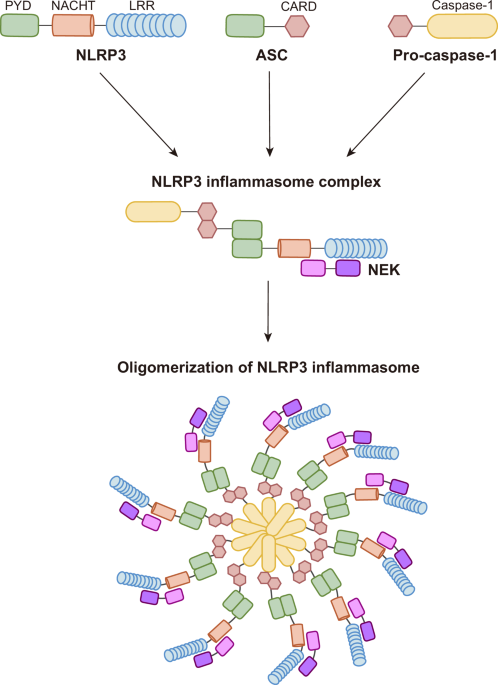
Sharif, H. et al. Structural mechanism for NEK7-licensed activation of NLRP3 inflammasome. Nature 570, 338–343 (2019).
Cai, X. et al. Prion-like polymerization underlies signal transduction in antiviral immune defense and inflammasome activation. Cell 156, 1207–1222 (2014).
Xiao, L., Magupalli, V. G. & Wu, H. Cryo-EM structures of the active NLRP3 inflammasome disc. Nature 613, 595–600 (2023).
He, Y., Zeng, M. Y., Yang, D., Motro, B. & Nunez, G. NEK7 is an essential mediator of NLRP3 activation downstream of potassium efflux. Nature 530, 354–357 (2016).
McDonald, B. et al. Intravascular danger signals guide neutrophils to sites of sterile inflammation. Science 330, 362–366 (2010).
Mariathasan, S. et al. Cryopyrin activates the inflammasome in response to toxins and ATP. Nature 440, 228–232 (2006).
Wang, Y. et al. Cellular localization of NLRP3 inflammasome. Protein Cell 4, 425–431 (2013).
Kanneganti, T. D. et al. Pannexin-1-mediated recognition of bacterial molecules activates the cryopyrin inflammasome independent of Toll-like receptor signaling. Immunity 26, 433–443 (2007).
Sutterwala, F. S. et al. Critical role for NALP3/CIAS1/Cryopyrin in innate and adaptive immunity through its regulation of caspase-1. Immunity 24, 317–327 (2006).
Hornung, V. et al. Silica crystals and aluminum salts activate the NALP3 inflammasome through phagosomal destabilization. Nat. Immunol. 9, 847–856 (2008).
Kuroda, E. et al. Silica crystals and aluminum salts regulate the production of prostaglandin in macrophages via NALP3 inflammasome-independent mechanisms. Immunity 34, 514–526 (2011).
Giamarellos-Bourboulis, E. J. et al. Crystals of monosodium urate monohydrate enhance lipopolysaccharide-induced release of interleukin 1β by mononuclear cells through a caspase 1-mediated process. Ann. Rheum. Dis. 68, 273–278 (2009).
Krantz, M., Eklund, D., Sarndahl, E. & Hedbrant, A. A detailed molecular network map and model of the NLRP3 inflammasome. Front. Immunol. 14, 1233680 (2023).
Munoz-Planillo, R. et al. K+ efflux is the common trigger of NLRP3 inflammasome activation by bacterial toxins and particulate matter. Immunity 38, 1142–1153 (2013).
Di, A. et al. The TWIK2 potassium efflux channel in macrophages mediates NLRP3 inflammasome-induced inflammation. Immunity 49, 56–65 (2018).
Huang, L. S. et al. Endosomal trafficking of two-pore K+ efflux channel TWIK2 to plasmalemma mediates NLRP3 inflammasome activation and inflammatory injury. eLife https://doi.org/10.7554/eLife.83842 (2023).
Gross, C. J. et al. K+ efflux-independent NLRP3 inflammasome activation by small molecules targeting mitochondria. Immunity 45, 761–773 (2016).
Gaidt, M. M. et al. Human monocytes engage an alternative inflammasome pathway. Immunity 44, 833–846 (2016).
Schorn, C. et al. Sodium overload and water influx activate the NALP3 inflammasome. J. Biol. Chem. 286, 35–41 (2011).
Wang, J. et al. Hepatitis B virus-mediated sodium influx contributes to hepatic inflammation via synergism with intrahepatic danger signals. iScience 27, 108723 (2024).
Tang, T. et al. CLICs-dependent chloride efflux is an essential and proximal upstream event for NLRP3 inflammasome activation. Nat. Commun. 8, 202 (2017).
Domingo-Fernandez, R., Coll, R. C., Kearney, J., Breit, S. & O’Neill, L. A. J. The intracellular chloride channel proteins CLIC1 and CLIC4 induce IL-1β transcription and activate the NLRP3 inflammasome. J. Biol. Chem. 292, 12077–12087 (2017).
Scambler, T. et al. ENaC-mediated sodium influx exacerbates NLRP3-dependent inflammation in cystic fibrosis. eLife https://doi.org/10.7554/eLife.49248 (2019).
Mayes-Hopfinger, L. et al. Chloride sensing by WNK1 regulates NLRP3 inflammasome activation and pyroptosis. Nat. Commun. 12, 4546 (2021).
Murakami, T. et al. Critical role for calcium mobilization in activation of the NLRP3 inflammasome. Proc. Natl Acad. Sci. USA 109, 11282–11287 (2012).
Lee, G. S. et al. The calcium-sensing receptor regulates the NLRP3 inflammasome through Ca2+ and cAMP. Nature 492, 123–127 (2012).
Zhou, R., Yazdi, A. S., Menu, P. & Tschopp, J. A role for mitochondria in NLRP3 inflammasome activation. Nature 469, 221–225 (2011).
Nakahira, K. et al. Autophagy proteins regulate innate immune responses by inhibiting the release of mitochondrial DNA mediated by the NALP3 inflammasome. Nat. Immunol. 12, 222–230 (2011).
Shimada, K. et al. Oxidized mitochondrial DNA activates the NLRP3 inflammasome during apoptosis. Immunity 36, 401–414 (2012).
Zhong, Z. et al. New mitochondrial DNA synthesis enables NLRP3 inflammasome activation. Nature 560, 198–203 (2018).
Xian, H. et al. Metformin inhibition of mitochondrial ATP and DNA synthesis abrogates NLRP3 inflammasome activation and pulmonary inflammation. Immunity 54, 1463–1477 (2021).
Billingham, L. K. et al. Mitochondrial electron transport chain is necessary for NLRP3 inflammasome activation. Nat. Immunol. 23, 692–704 (2022).
Xiao, Y. et al. Mitochondrial dysfunction by FADDosome promotes gastric mucosal injury in portal hypertensive gastropathy. Int. J. Biol. Sci. 20, 2658–2685 (2024).
Subramanian, N., Natarajan, K., Clatworthy, M. R., Wang, Z. & Germain, R. N. The adaptor MAVS promotes NLRP3 mitochondrial localization and inflammasome activation. Cell 153, 348–361 (2013).
Iyer, S. S. et al. Mitochondrial cardiolipin is required for Nlrp3 inflammasome activation. Immunity 39, 311–323 (2013).
Ichinohe, T., Yamazaki, T., Koshiba, T. & Yanagi, Y. Mitochondrial protein mitofusin 2 is required for NLRP3 inflammasome activation after RNA virus infection. Proc. Natl Acad. Sci. USA 110, 17963–17968 (2013).
Martinon, F., Petrilli, V., Mayor, A., Tardivel, A. & Tschopp, J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440, 237–241 (2006).
Dostert, C. et al. Innate immune activation through Nalp3 inflammasome sensing of asbestos and silica. Science 320, 674–677 (2008).
Halle, A. et al. The NALP3 inflammasome is involved in the innate immune response to amyloid-β. Nat. Immunol. 9, 857–865 (2008).
Duewell, P. et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature 464, 1357–1361 (2010).
Hou, Y., He, H., Ma, M. & Zhou, R. Apilimod activates the NLRP3 inflammasome through lysosome-mediated mitochondrial damage. Front. Immunol. 14, 1128700 (2023).
McCarthy, A. E., Yoshioka, C. & Mansoor, S. E. Full-Length P2X(7) structures reveal how palmitoylation prevents channel desensitization. Cell 179, 659–670 (2019).
Browne, L. E., Compan, V., Bragg, L. & North, R. A. P2X7 receptor channels allow direct permeation of nanometer-sized dyes. J. Neurosci. 33, 3557–3566 (2013).
Wolf, A. J. et al. Hexokinase is an innate immune receptor for the detection of bacterial peptidoglycan. Cell 166, 624–636 (2016).
Xie, M. et al. PKM2-dependent glycolysis promotes NLRP3 and AIM2 inflammasome activation. Nat. Commun. 7, 13280 (2016).
Tannahill, G. M. et al. Succinate is an inflammatory signal that induces IL-1β through HIF-1α. Nature 496, 238–242 (2013).
Lampropoulou, V. et al. Itaconate links inhibition of succinate dehydrogenase with macrophage metabolic remodeling and regulation of inflammation. Cell Metab. 24, 158–166 (2016).
Humphries, F. et al. Succination inactivates gasdermin D and blocks pyroptosis. Science 369, 1633–1637 (2020).
Wen, H. et al. Fatty acid-induced NLRP3–ASC inflammasome activation interferes with insulin signaling. Nat. Immunol. 12, 408–415 (2011).
Xing, J. H. et al. NLRP3 inflammasome mediate palmitate-induced endothelial dysfunction. Life Sci. 239, 116882 (2019).
Youm, Y. H. et al. The ketone metabolite beta-hydroxybutyrate blocks NLRP3 inflammasome-mediated inflammatory disease. Nat. Med. 21, 263–269 (2015).
Yan, H. et al. Hexokinase 2 senses fructose in tumor-associated macrophages to promote colorectal cancer growth. Cell Metab. 36, 2449–2467 (2024).
Misawa, T. et al. Microtubule-driven spatial arrangement of mitochondria promotes activation of the NLRP3 inflammasome. Nat. Immunol. 14, 454–460 (2013).
Magupalli, V. G. et al. HDAC6 mediates an aggresome-like mechanism for NLRP3 and pyrin inflammasome activation. Science https://doi.org/10.1126/science.aas8995 (2020).
Baldrighi, M. et al. PLK1 inhibition dampens NLRP3 inflammasome-elicited response in inflammatory disease models. J. Clin. Invest. https://doi.org/10.1172/JCI162129 (2023).
Guo, C. et al. Cholesterol homeostatic regulator SCAP–SREBP2 integrates NLRP3 inflammasome activation and cholesterol biosynthetic signaling in macrophages. Immunity 49, 842–856 (2018).
Chen, J. & Chen, Z. J. PtdIns4P on dispersed trans-Golgi network mediates NLRP3 inflammasome activation. Nature 564, 71–76 (2018).
Nie, L. et al. Consecutive palmitoylation and phosphorylation orchestrates NLRP3 membrane trafficking and inflammasome activation. Mol. Cell 84, 3336–3353 (2024).
Yu, X. et al. Structural basis for the oligomerization-facilitated NLRP3 activation. Nat. Commun. 15, 1164 (2024).
Schmacke, N. A. et al. IKKβ primes inflammasome formation by recruiting NLRP3 to the trans-Golgi network. Immunity 55, 2271–2284 (2022).
Williams, D. M. & Peden, A. A. S-acylation of NLRP3 provides a nigericin sensitive gating mechanism that controls access to the Golgi. eLife https://doi.org/10.7554/eLife.94302 (2024).
Ni, J. et al. Ubc13 Promotes K63-Linked Polyubiquitination of NLRP3 to Activate Inflammasome. J. Immunol. 206, 2376–2385 (2021).
Tang, J. et al. Sequential ubiquitination of NLRP3 by RNF125 and Cbl-b limits inflammasome activation and endotoxemia. J. Exp. Med. https://doi.org/10.1084/jem.20182091 (2020).
Song, H. et al. The E3 ubiquitin ligase TRIM31 attenuates NLRP3 inflammasome activation by promoting proteasomal degradation of NLRP3. Nat. Commun. 7, 13727 (2016).
Li, Y. T. et al. USP13 stabilizes NLRP3 to facilitate inflammasome activation by preventing TRIM31-mediated NLRP3 ubiquitination and degradation. Sci. Adv. 11, eadx3827 (2025).
Yan, Y. et al. Dopamine controls systemic inflammation through inhibition of NLRP3 inflammasome. Cell 160, 62–73 (2015).
Wan, P. et al. Cullin1 binds and promotes NLRP3 ubiquitination to repress systematic inflammasome activation. FASEB J. 33, 5793–5807 (2019).
Han, S. et al. Lipopolysaccharide primes the NALP3 inflammasome by inhibiting its ubiquitination and degradation mediated by the SCFFBXL2 E3 ligase. J. Biol. Chem. 290, 18124–18133 (2015).
Kawashima, A. et al. ARIH2 ubiquitinates NLRP3 and negatively regulates NLRP3 inflammasome activation in macrophages. J. Immunol. 199, 3614–3622 (2017).
Xu, T. et al. Ubiquitination of NLRP3 by gp78/Insig-1 restrains NLRP3 inflammasome activation. Cell Death Differ. 29, 1582–1595 (2022).
Ge, X. et al. PARK2 attenuates house dust mite-induced inflammatory reaction, pyroptosis and barrier dysfunction in BEAS-2B cells by ubiquitinating NLRP3. Am. J. Transl. Res. 13, 326–335 (2021).
Park, Y. J. et al. MARCH5-dependent NLRP3 ubiquitination is required for mitochondrial NLRP3–NEK7 complex formation and NLRP3 inflammasome activation. EMBO J. 42, e113481 (2023).
Tang, T. et al. The E3 ubiquitin ligase TRIM65 negatively regulates inflammasome activation through promoting ubiquitination of NLRP3. Front. Immunol. 12, 741839 (2021).
Guo, Y. et al. HUWE1 mediates inflammasome activation and promotes host defense against bacterial infection. J. Clin. Invest. 130, 6301–6316 (2020).
Wang, D. et al. YAP promotes the activation of NLRP3 inflammasome via blocking K27-linked polyubiquitination of NLRP3. Nat. Commun. 12, 2674 (2021).
Humphries, F. et al. The E3 ubiquitin ligase Pellino2 mediates priming of the NLRP3 inflammasome. Nat. Commun. 9, 1560 (2018).
Hang, Y., Tan, L., Chen, Q., Liu, Q. & Jin, Y. E3 ubiquitin ligase TRIM24 deficiency promotes NLRP3/caspase-1/IL-1β-mediated pyroptosis in endometriosis. Cell Biol. Int. 45, 1561–1570 (2021).
Shen, J. et al. TRIM40 inhibits IgA1-induced proliferation of glomerular mesangial cells by inactivating NLRP3 inflammasome through ubiquitination. Mol. Immunol. 140, 225–232 (2021).
Lin, Y. et al. TRIM50 promotes NLRP3 inflammasome activation by directly inducing NLRP3 oligomerization. EMBO Rep. 23, e54569 (2022).
Song, H. et al. UAF1 deubiquitinase complexes facilitate NLRP3 inflammasome activation by promoting NLRP3 expression. Nat. Commun. 11, 6042 (2020).
Li, X. et al. MF-094, a potent and selective USP30 inhibitor, accelerates diabetic wound healing by inhibiting the NLRP3 inflammasome. Exp. Cell Res. 410, 112967 (2022).
Zhao, C. et al. USP50 regulates NLRP3 inflammasome activation in duodenogastric reflux-induced gastric tumorigenesis. Front. Immunol. 15, 1326137 (2024).
Xiang, Y., Li, X., Cai, M. & Cai, D. USP9X promotes lipopolysaccharide-stimulated acute lung injury by deubiquitination of NLRP3. Cell Biol. Int. 47, 394–405 (2023).
Lu, H. et al. The E3 ubiquitin ligase MARCH9 alleviates pyroptosis by regulating NLPR3 ubiquitination following myocardial ischemia reperfusion. Cell. Mol. Life Sci. 82, 348 (2025).
Wang, W. et al. STING promotes NLRP3 localization in ER and facilitates NLRP3 deubiquitination to activate the inflammasome upon HSV-1 infection. PLoS Pathog. 16, e1008335 (2020).
Zhang, W. et al. Cytosolic escape of mitochondrial DNA triggers cGAS–STING–NLRP3 axis-dependent nucleus pulposus cell pyroptosis. Exp. Mol. Med. 54, 129–142 (2022).
Py, B. F., Kim, M. S., Vakifahmetoglu-Norberg, H. & Yuan, J. Deubiquitination of NLRP3 by BRCC3 critically regulates inflammasome activity. Mol. Cell 49, 331–338 (2013).
Ren, G. et al. ABRO1 promotes NLRP3 inflammasome activation through regulation of NLRP3 deubiquitination. EMBO J. https://doi.org/10.15252/embj.2018100376 (2019).
Yalcinkaya, M. et al. BRCC3-mediated NLRP3 deubiquitylation promotes inflammasome activation and atherosclerosis in Tet2 clonal hematopoiesis. Circulation 148, 1764–1777 (2023).
Liu, C. et al. YOD1 protects against MRSA sepsis-induced DIC through Lys33-linked deubiquitination of NLRP3. Cell Death Dis. 15, 360 (2024).
Palazon-Riquelme, P. et al. USP7 and USP47 deubiquitinases regulate NLRP3 inflammasome activation. EMBO Rep. https://doi.org/10.15252/embr.201744766 (2018).
Rao, Z. et al. Vitamin D receptor inhibits NLRP3 activation by impeding its BRCC3-mediated deubiquitination. Front. Immunol. 10, 2783 (2019).
Bednash, J. S. et al. The deubiquitinase STAMBP modulates cytokine secretion through the NLRP3 inflammasome. Cell. Signal. 79, 109859 (2021).
Zhang, Z. et al. Protein kinase D at the Golgi controls NLRP3 inflammasome activation. J. Exp. Med. 214, 2671–2693 (2017).
Mortimer, L., Moreau, F., MacDonald, J. A. & Chadee, K. NLRP3 inflammasome inhibition is disrupted in a group of auto-inflammatory disease CAPS mutations. Nat. Immunol. 17, 1176–1186 (2016).
Guo, C. et al. Bile acids control inflammation and metabolic disorder through inhibition of NLRP3 inflammasome. Immunity 45, 802–816 (2016).
Guo, X. et al. AKT controls NLRP3 inflammasome activation by inducing DDX3X phosphorylation. FEBS Lett. 595, 2447–2462 (2021).
Zhu, K. et al. Priming of NLRP3 inflammasome activation by Msn kinase MINK1 in macrophages. Cell. Mol. Immunol. 18, 2372–2382 (2021).
Chung, I. C. et al. Pyk2 activates the NLRP3 inflammasome by directly phosphorylating ASC and contributes to inflammasome-dependent peritonitis. Sci. Rep. 6, 36214 (2016).
Niu, T. et al. NLRP3 phosphorylation in its LRR domain critically regulates inflammasome assembly. Nat. Commun. 12, 5862 (2021).
Ma, Z. et al. PGK1 phosphorylates NLRP3 and mediates inflammasome activation independent of its glycolytic activity. Cell Rep. 44, 115785 (2025).
Kim, H. J. et al. The Src family kinase, Lyn, suppresses osteoclastogenesis in vitro and in vivo. Proc. Natl Acad. Sci. USA 106, 2325–2330 (2009).
Bittner, Z. A. et al. BTK operates a phospho-tyrosine switch to regulate NLRP3 inflammasome activity. J. Exp. Med. https://doi.org/10.1084/jem.20201656 (2021).
Zhang, A. et al. EphA2 phosphorylates NLRP3 and inhibits inflammasomes in airway epithelial cells. EMBO Rep. 21, e49666 (2020).
Spalinger, M. R. et al. PTPN22 regulates NLRP3-mediated IL1B secretion in an autophagy-dependent manner. Autophagy 13, 1590–1601 (2017).
Spalinger, M. R. et al. NLRP3 tyrosine phosphorylation is controlled by protein tyrosine phosphatase PTPN22. J. Clin. Invest. https://doi.org/10.1172/JCI169304 (2023).
Zhao, W. et al. AKT regulates NLRP3 inflammasome activation by phosphorylating NLRP3 serine 5. J. Immunol. 205, 2255–2264 (2020).
Fischer, F. A. et al. TBK1 and IKKepsilon act like an OFF switch to limit NLRP3 inflammasome pathway activation. Proc. Natl Acad. Sci. USA https://doi.org/10.1073/pnas.2009309118 (2021).
Zhang, Y. et al. Acetylation is required for full activation of the NLRP3 inflammasome. Nat. Commun. 14, 8396 (2023).
He, M. et al. An acetylation switch of the NLRP3 inflammasome regulates aging-associated chronic inflammation and insulin resistance. Cell Metab. 31, 580–591 (2020).
Zhang, L. et al. Tau induces inflammasome activation and microgliosis through acetylating NLRP3. Clin. Transl. Med. 14, e1623 (2024).
Guan, X. et al. Inhibition of HDAC2 sensitises antitumour therapy by promoting NLRP3/GSDMD-mediated pyroptosis in colorectal cancer. Clin. Transl. Med. 14, e1692 (2024).
Lin, Q. C. et al. Hippocampal HDAC6 promotes POCD by regulating NLRP3-induced microglia pyroptosis via HSP90/HSP70 in aged mice. Biochim. Biophys. Acta Mol. Basis Dis. 1870, 167137 (2024).
Yang, M. et al. HDAC10 switches NLRP3 modification from acetylation to ubiquitination and attenuates acute inflammatory diseases. Cell Commun. Signal. 22, 615 (2024).
Shao, L. et al. SUMO1 SUMOylates and SENP3 deSUMOylates NLRP3 to orchestrate the inflammasome activation. FASEB J. 34, 1497–1515 (2020).
Barry, R. et al. SUMO-mediated regulation of NLRP3 modulates inflammasome activity. Nat. Commun. 9, 3001 (2018).
Chen, J. et al. SENP3 attenuates foam cell formation by deSUMOylating NLRP3 in macrophages stimulated with ox-LDL. Cell. Signal. 117, 111092 (2024).
Jing, F. et al. Zmiz1-mediated SUMOylation of NLRP3 inflammasome regulates satellite glial cell activation and neuronal autophagy in trigeminal neuralgia. Inflammation 48, 4342–4363 (2025).
Qin, Y. et al. TRIM28 SUMOylates and stabilizes NLRP3 to facilitate inflammasome activation. Nat. Commun. 12, 4794 (2021).
Iarmonenko, S. P. Radiomodifiers and the progress of radiation oncology]. Vopr. Onkol. 41, 93–94 (1995).
Shim, D. W. et al. BOT-4-one attenuates NLRP3 inflammasome activation: NLRP3 alkylation leading to the regulation of its ATPase activity and ubiquitination. Sci. Rep. 7, 15020 (2017).
Juliana, C. et al. Anti-inflammatory compounds parthenolide and Bay 11-7082 are direct inhibitors of the inflammasome. J. Biol. Chem. 285, 9792–9802 (2010).
He, Y. et al. 3,4-Methylenedioxy-β-nitrostyrene inhibits NLRP3 inflammasome activation by blocking assembly of the inflammasome. J. Biol. Chem. 289, 1142–1150 (2014).
Yang, S. et al. ABHD8 antagonizes inflammation by facilitating chaperone-mediated autophagy-mediated degradation of NLRP3. Autophagy 21, 338–351 (2025).
Zheng, S. et al. ZDHHC5-mediated NLRP3 palmitoylation promotes NLRP3–NEK7 interaction and inflammasome activation. Mol. Cell 83, 4570–4585.e4577 (2023).
Hu, D. et al. Palmitoylation of NLRP3 modulates inflammasome activation and inflammatory bowel disease development. J. Immunol. 213, 481–493 (2024).
Zou, G. et al. Signal-induced NLRP3 phase separation initiates inflammasome activation. Cell Res. 35, 437–452 (2025).
Chiu, L. Y., Huang, D. Y. & Lin, W. W. PARP-1 regulates inflammasome activity by poly-ADP-ribosylation of NLRP3 and interaction with TXNIP in primary macrophages. Cell. Mol. Life. Sci. 79, 108 (2022).
Bose, S. et al. ADP-ribosylation of NLRP3 by Mycoplasma pneumoniae CARDS toxin regulates inflammasome activity. mBio https://doi.org/10.1128/mBio.02186-14 (2014).
Luo, L. et al. STAT3 promotes NLRP3 inflammasome activation by mediating NLRP3 mitochondrial translocation. Exp. Mol. Med. 56, 1980–1990 (2024).
Liang, Z. et al. Proximity proteomics reveals UCH-L1 as an essential regulator of NLRP3-mediated IL-1β production in human macrophages and microglia. Cell Rep. 43, 114152 (2024).
Ha, J. et al. SERTAD1 initiates NLRP3-mediated inflammasome activation through restricting NLRP3 polyubiquitination. Cell Rep. 43, 113752 (2024).
Yu, J. et al. Inhibition of NLRP3 inflammasome activation by A20 through modulation of NEK7. Proc. Natl Acad. Sci. USA 121, e2316551121 (2024).
Guo, X. B. et al. HAX-1 interferes in assembly of NLRP3–ASC to block microglial pyroptosis in cerebral I/R injury. Cell Death Discov. 10, 264 (2024).
Liu, S. et al. The E3 ubiquitin ligase MARCH2 protects against myocardial ischemia–reperfusion injury through inhibiting pyroptosis via negative regulation of PGAM5/MAVS/NLRP3 axis. Cell Discov. 10, 24 (2024).
Huang, J. et al. The human disease-associated gene ZNFX1 controls inflammation through inhibition of the NLRP3 inflammasome. EMBO J. 43, 5469–5493 (2024).
Yang, S. et al. ABHD8 antagonizes inflammation by facilitating chaperone-mediated autophagy-mediated degradation of NLRP3. Autophagy 21, 1–14 (2024).
Cheng, Z. et al. HECTD3 inhibits NLRP3 inflammasome assembly and activation by blocking NLRP3–NEK7 interaction. Cell Death Dis. 15, 86 (2024).













Leave a Reply