The aging brain experiences profound immunological and vascular changes that increase its vulnerability to both neurodegenerative pathologies and neoplastic processes. One of the most distinctive features of this deterioration is the phenomenon known as inflammaging, a state of chronic low-level inflammation that contributes to synaptic loss, sustained glial activation and immune system dysfunction1. A central axis in these processes is the progressive disruption of the blood-brain barrier (BBB), a key event that allows the entry of peripheral immune cells into the central nervous system (CNS). Once inside, these cells can adopt maladaptive phenotypes that directly impact the onset and progression of multiple diseases. This phenomenon particularly affects the brain’s resident myeloid cells, which perform fundamental homeostatic functions. When their balance is disrupted, a generalized disorder characterized by an imbalance between proinflammatory and immunosuppressive mechanisms is generated.
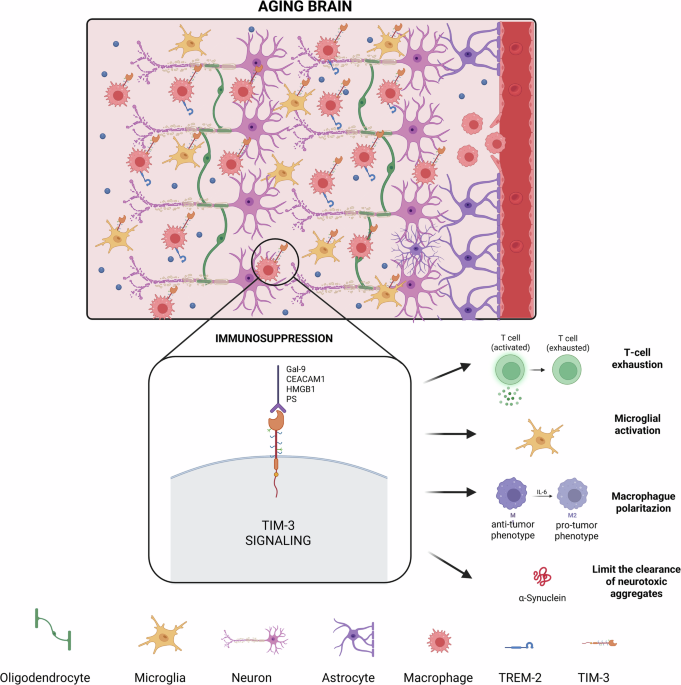
In this context, the role of immunosuppressive pathways, such as those mediated by TIM-3, is becoming increasingly relevant. TIM-3, which is encoded by the HAVCR2 gene, is a co-inhibitory receptor expressed on multiple immune cell population, including T cells and myeloid cells. Its presence on IFN-γ–producing effector T cells, FoxP3⁺ regulatory T cells, exhausted T cells, and tumor-infiltrating lymphocytes underscores its role in limiting T-cell activity. In parallel, its expression on myeloid population, including microglia, dendritic cells and macrophages derived from monocytes, reflects its functional relevance both peripheral tissues and the central nervous system. The interaction of TIM-3 with its known ligands such as galectin-9, CEACAM1, HMGB1 and phosphatidylserine, contributes to immunosuppression by promoting T-cell exhaustion and modulating the suppressive activity of myeloid cells (Fig. 1)2. Notably, its expression has been detected not only in aggressive brain tumors such as glioblastoma, but also in neurodegenerative diseases associated with aging, such as Alzheimer’s and Parkinson’s, has gained relevance. This pathological convergence in immune modulation of the aging brain points to a common mechanism that transcends traditional clinical categories.
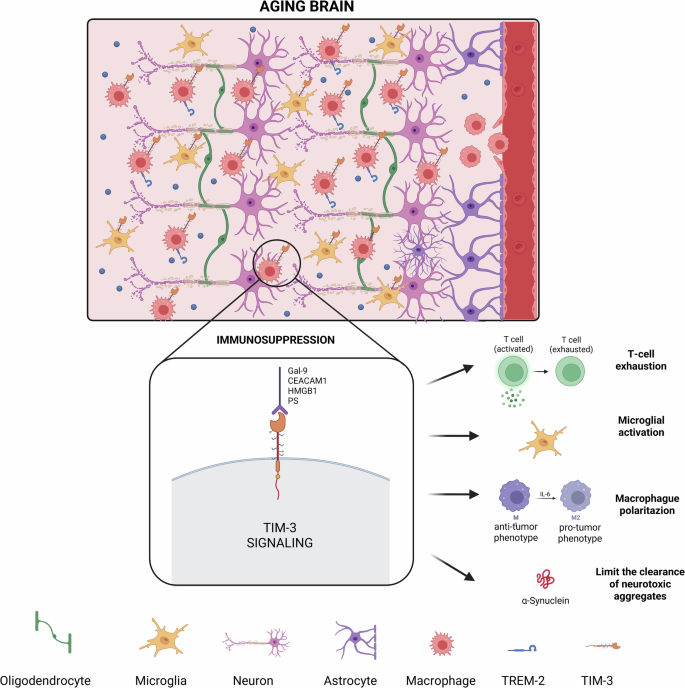
Schematic representation of the principal biological processes connecting age-related inflammation with blood-brain barrier dysfunction, peripheral immune cell infiltration, and the activation of TIM-3 dependent immunosuppressive mechanisms in both resident and infiltrating myeloid populations. Created in BioRender. Jareño, C. (2025).
Understanding the mechanisms of immunosuppression in the CNS, both under physiological and pathological conditions, is increasingly recognized as a critical scientific and clinical need. Advancing the functional and molecular characterization of these immune circuits is essential—not only to explain treatment resistance and disease progression in neurological disorders, but also to inform the development of more precise and effective therapeutic strategies. This research will deepen our knowledge of brain immunobiology across the lifespan and pave the way for interventions tailored to the unique immunological landscape of the CNS.
















Leave a Reply