Eaden, J. A., Abrams, K. R. & Mayberry, J. F. The risk of colorectal cancer in ulcerative colitis: a meta-analysis. Gut 48, 526–535 (2001).
Ekbom, A., Helmick, C., Zack, M. & Adami, H. O. Ulcerative colitis and colorectal cancer. A population-based study. N. Engl. J. Med. 323, 1228–1233 (1990).
Söderlund, S. et al. Decreasing time-trends of colorectal cancer in a large cohort of patients with inflammatory bowel disease. Gastroenterology 136, 1561–1567 (2009).
Hracs, L. et al. Global evolution of inflammatory bowel disease across epidemiologic stages. Nature 642, 458–466 (2025).
Olén, O. et al. Colorectal cancer in ulcerative colitis: a Scandinavian population-based cohort study. Lancet 395, 123–131 (2020).
Olén, O. et al. Colorectal cancer in Crohn’s disease: a Scandinavian population-based cohort study. Lancet. Gastroenterol. Hepatol. 5, 475–484 (2020).
Kuenzig, M. E. et al. Twenty-first century trends in the global epidemiology of pediatric-onset inflammatory bowel disease: systematic review. Gastroenterology 162, 1147–1159.e4 (2022).
Ng, S. C. et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet 390, 2769–2778 (2017).
Choi, C. R. et al. Cumulative burden of inflammation predicts colorectal neoplasia risk in ulcerative colitis: a large single-centre study. Gut 68, 414–422 (2019).
Gordon, H. et al. ECCO guidelines on inflammatory bowel disease and malignancies. J. Crohns Colitis 17, 827–854 (2022).
Askling, J. et al. Family history as a risk factor for colorectal cancer in inflammatory bowel disease. Gastroenterology 120, 1356–1362 (2001).
Fumery, M. et al. Detection of dysplasia or cancer in 3.5% of patients with inflammatory bowel disease and colonic strictures. Clin. Gastroenterol. Hepatol. 13, 1770–1775 (2015).
Trivedi, P. J. et al. Effects of primary sclerosing cholangitis on risks of cancer and death in people with inflammatory bowel disease, based on sex, race, and age. Gastroenterology 159, 915–928 (2020).
Wijnands, A. M. et al. Prognostic factors for advanced colorectal neoplasia in inflammatory bowel disease: systematic review and meta-analysis. Gastroenterology 160, 1584–1598 (2021).
Zhou, R. W., Harpaz, N., Itzkowitz, S. H. & Parsons, R. E. Molecular mechanisms in colitis-associated colorectal cancer. Oncogenesis 12, 48 (2023).
Chatila, W. K. et al. Integrated clinical and genomic analysis identifies driver events and molecular evolution of colitis-associated cancers. Nat. Commun. 14, 110 (2023).
Murthy, S. K., Feuerstein, J. D., Nguyen, G. C. & Velayos, F. S. AGA clinical practice update on endoscopic surveillance and management of colorectal dysplasia in inflammatory bowel diseases: expert review. Gastroenterology 161, 1043–1051.e4 (2021).
East, J. E. et al. British Society of Gastroenterology guidelines on colorectal surveillance in inflammatory bowel disease. Gut 75, 442–475 (2026).
Stjärngrim, J., Ekbom, A., Hammar, U., Hultcrantz, R. & Forsberg, A. M. Rates and characteristics of postcolonoscopy colorectal cancer in the Swedish IBD population: what are the differences from a non-IBD population. Gut 68, 1588–1596 (2019).
van Schaik, F. D. M. et al. Endoscopic and pathological aspects of colitis-associated dysplasia. Nat. Rev. Gastroenterol. Hepatol. 6, 671–678 (2009).
Uchino, M. et al. Histological differentiation between sporadic and colitis-associated intestinal cancer in a nationwide study: a propensity-score-matched analysis. J. Gastroenterol. Hepatol. 39, 893–901 (2024).
Lu, C. et al. Survival outcomes and clinicopathological features in inflammatory bowel disease-associated colorectal cancer: a systematic review and meta-analysis. Ann. Surg. 276, e319–e330 (2022).
Velayos, F. S., Terdiman, J. P. & Walsh, J. M. Effect of 5-aminosalicylate use on colorectal cancer and dysplasia risk: a systematic review and metaanalysis of observational studies. Am. J. Gastroenterol. 100, 1345–1353 (2005).
Ullman, T. et al. Progression to colorectal neoplasia in ulcerative colitis: effect of mesalamine. Clin. Gastroenterol. Hepatol. 6, 1225–1230 (2008).
Lopez, A. & Peyrin-Biroulet, L. 5-Aminosalicylic acid and chemoprevention: does it work. Digestive Dis. 31, 248–253 (2013).
Bak, M. T. J. et al. Endoscopic management of patients with high-risk colorectal colitis-associated neoplasia: a Delphi study. Gastrointest. Endosc. 97, 767–779.e6 (2023).
Kabir, M. et al. DECIDE: Delphi expert consensus statement on inflammatory bowel disease dysplasia shared management decision-making. J. Crohns Colitis 17, 1652–1671 (2023).
Mohan, B. P. et al. Endoscopic resection of colon dysplasia in patients with inflammatory bowel disease: a systematic review and meta-analysis. Gastrointest. Endoscopy 93, 59–67.e10 (2021).
Yaeger, R. et al. Systemic chemotherapy for metastatic colitis-associated cancer has a worse outcome than sporadic colorectal cancer: matched case cohort analysis. Clin. Colorectal Cancer 19, e151–e156 (2020).
Abu-Sbeih, H. et al. Immune checkpoint inhibitor therapy in patients with preexisting inflammatory bowel disease. J. Clin. Oncol. 38, 576–583 (2020).
Neurath, M. F., Artis, D. & Becker, C. The intestinal barrier: a pivotal role in health, inflammation, and cancer. Lancet Gastroenterol. Hepatol. 10, 573–592 (2025).
Choi, J. & Augenlicht, L. H. Intestinal stem cells: guardians of homeostasis in health and aging amid environmental challenges. Exp. Mol. Med. 56, 495–500 (2024).
Greenstein, A. J., Heimann, T. M., Sachar, D. B., Slater, G. & Aufses, A. H. J. A comparison of multiple synchronous colorectal cancer in ulcerative colitis, familial polyposis coli, and de novo cancer. Ann. Surg. 203, 123–128 (1986).
Baker, A. M. et al. Evolutionary history of human colitis-associated colorectal cancer. Gut 68, 985–995 (2019).
Galandiuk, S. et al. Field cancerization in the intestinal epithelium of patients with Crohn’s ileocolitis. Gastroenterology 142, 855–864.e8 (2012).
Leedham, S. J. et al. Clonality, founder mutations, and field cancerization in human ulcerative colitis-associated neoplasia. Gastroenterology 136, 542–550.e6 (2009).
Kakiuchi, N. et al. Frequent mutations that converge on the NFKBIZ pathway in ulcerative colitis. Nature 577, 260–265 (2020).
Nanki, K. et al. Somatic inflammatory gene mutations in human ulcerative colitis epithelium. Nature 577, 254–259 (2020).
van Neerven, S. M. & Vermeulen, L. Cell competition in development, homeostasis and cancer. Nat. Rev. Mol. Cell Biol. 24, 221–236 (2023).
Olafsson, S. et al. Somatic evolution in non-neoplastic IBD-affected colon. Cell 182, 672–684.e11 (2020).
Jee, J. et al. Automated real-world data integration improves cancer outcome prediction. Nature 636, 728–736 (2024).
Brentnall, T. A. et al. Mutations in the p53 gene: an early marker of neoplastic progression in ulcerative colitis. Gastroenterology 107, 369–378 (1994).
Beaugerie, L. & Itzkowitz, S. H. Cancers complicating inflammatory bowel disease. N. Engl. J. Med. 372, 1441–1452 (2015).
Eischen, C. M. Genome stability requires p53. Cold Spring Harb. Perspect. Med. 6, a026096 (2016).
Verhagen, M. P. et al. Non-stem cell lineages as an alternative origin of intestinal tumorigenesis in the context of inflammation. Nat. Genet. 56, 1456–1467 (2024).
Dekker, E., Tanis, P. J., Vleugels, J. L. A., Kasi, P. M. & Wallace, M. B. Colorectal cancer. Lancet 394, 1467–1480 (2019).
Vogelstein, B. et al. Genetic alterations during colorectal-tumor development. N. Engl. J. Med. 319, 525–532 (1988).
Fearon, E. R. & Vogelstein, B. A genetic model for colorectal tumorigenesis. Cell 61, 759–767 (1990).
van Neerven, S. M. et al. Apc-mutant cells act as supercompetitors in intestinal tumour initiation. Nature 594, 436–441 (2021).
Flanagan, D. J. et al. NOTUM from Apc-mutant cells biases clonal competition to initiate cancer. Nature 594, 430–435 (2021).
Kern, S. E. et al. Molecular genetic profiles of colitis-associated neoplasms. Gastroenterology 107, 420–428 (1994).
Tarmin, L. et al. Adenomatous polyposis coli gene mutations in ulcerative colitis-associated dysplasias and cancers versus sporadic colon neoplasms. Cancer Res. 55, 2035–2038 (1995).
Robles, A. I. et al. Whole-exome sequencing analyses of inflammatory bowel disease-associated colorectal cancers. Gastroenterology 150, 931–943 (2016).
Beggs, A. D. et al. Validation of epigenetic markers to identify colitis associated cancer: results of module 1 of the ENDCAP-C study. EBioMedicine 39, 265–271 (2019).
Glória, L. et al. DNA hypomethylation and proliferative activity are increased in the rectal mucosa of patients with long-standing ulcerative colitis. Cancer 78, 2300–2306 (1996).
Rajamäki, K. et al. Genetic and epigenetic characteristics of inflammatory bowel disease-associated colorectal cancer. Gastroenterology 161, 592–607 (2021).
Sottoriva, A. et al. A Big Bang model of human colorectal tumor growth. Nat. Genet. 47, 209–216 (2015).
Gao, J. et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci. Signal. 6, pl1 (2013).
Fujita, M. et al. Genomic landscape of colitis-associated cancer indicates the impact of chronic inflammation and its stratification by mutations in the Wnt signaling. Oncotarget 9, 969–981 (2017).
Konishi, K. et al. Rare CpG island methylator phenotype in ulcerative colitis-associated neoplasias. Gastroenterology 132, 1254–1260 (2007).
Sanchez, J. A., DeJulius, K. L., Bronner, M., Church, J. M. & Kalady, M. F. Relative role of methylator and tumor suppressor pathways in ulcerative colitis-associated colon cancer. Inflamm. Bowel Dis. 17, 1966–1970 (2011).
Rheinbay, E. et al. Analyses of non-coding somatic drivers in 2,658 cancer whole genomes. Nature 578, 102–111 (2020).
Shah, S. C. & Itzkowitz, S. H. Colorectal cancer in Inflammatory bowel disease: mechanisms and management. Gastroenterology 162, 715–730.e3 (2022).
Din, S. et al. Mutational analysis identifies therapeutic biomarkers in inflammatory bowel disease-associated colorectal cancers. Clin. Cancer Res. 24, 5133–5142 (2018).
Rubin, C. E. et al. DNA aneuploidy in colonic biopsies predicts future development of dysplasia in ulcerative colitis. Gastroenterology 103, 1611–1620 (1992).
Yaeger, R. et al. Genomic alterations observed in colitis-associated cancers are distinct from those found in sporadic colorectal cancers and vary by type of inflammatory bowel disease. Gastroenterology 151, 278–287.e276 (2016).
Wanders, L. K. et al. IBD-associated dysplastic lesions show more chromosomal instability than sporadic adenomas. Inflamm. Bowel Dis. 26, 167–180 (2020).
Tsai, J.-H. et al. Association of aneuploidy and flat dysplasia with development of high-grade dysplasia or colorectal cancer in patients with inflammatory bowel disease. Gastroenterology 153, 1492–1495.e4 (2017).
Heiser, C. N. et al. Molecular cartography uncovers evolutionary and microenvironmental dynamics in sporadic colorectal tumors. Cell 186, 5620–5637.e16 (2023).
Degirmenci, B., Valenta, T., Dimitrieva, S., Hausmann, G. & Basler, K. GLI1-expressing mesenchymal cells form the essential Wnt-secreting niche for colon stem cells. Nature 558, 449–453 (2018).
Kraiczy, J. et al. Graded BMP signaling within intestinal crypt architecture directs self-organization of the Wnt-secreting stem cell niche. Cell Stem Cell 30, 433–449.e8 (2023).
Kinchen, J. et al. Structural remodeling of the human colonic mesenchyme in inflammatory bowel disease. Cell 175, 372–386.e17 (2018).
Smillie, C. S. et al. Intra- and inter-cellular rewiring of the human colon during ulcerative colitis. Cell 178, 714–730.e22 (2019).
Friedrich, M. et al. IL-1-driven stromal-neutrophil interactions define a subset of patients with inflammatory bowel disease that does not respond to therapies. Nat. Med. 27, 1970–1981 (2021).
Guinney, J. et al. The consensus molecular subtypes of colorectal cancer. Nat. Med. 21, 1350–1356 (2015).
Joanito, I. et al. Single-cell and bulk transcriptome sequencing identifies two epithelial tumor cell states and refines the consensus molecular classification of colorectal cancer. Nat. Genet. 54, 963–975 (2022).
Cornish, A. J. et al. The genomic landscape of 2,023 colorectal cancers. Nature 633, 127–136 (2024).
Priestley, P. et al. Pan-cancer whole-genome analyses of metastatic solid tumours. Nature 575, 210–216 (2019).
Nunes, L. et al. Prognostic genome and transcriptome signatures in colorectal cancers. Nature 633, 137–146 (2024).
Lovisa, S., Genovese, G. & Danese, S. Role of epithelial-to-mesenchymal transition in inflammatory bowel disease. J. Crohns Colitis 13, 659–668 (2019).
Kobayashi, H. et al. The origin and contribution of cancer-associated fibroblasts in colorectal carcinogenesis. Gastroenterology 162, 890–906 (2022).
Sahai, E. et al. A framework for advancing our understanding of cancer-associated fibroblasts. Nat. Rev. Cancer 20, 174–186 (2020).
Koliaraki, V., Prados, A., Armaka, M. & Kollias, G. The mesenchymal context in inflammation, immunity and cancer. Nat. Immunol. 21, 974–982 (2020).
Neufert, C. et al. Tumor fibroblast-derived epiregulin promotes growth of colitis-associated neoplasms through ERK. J. Clin. Invest. 123, 1428–1443 (2013).
Lenos, K. J. et al. Stem cell functionality is microenvironmentally defined during tumour expansion and therapy response in colon cancer. Nat. Cell Biol. 20, 1193–1202 (2018).
Heichler, C. et al. STAT3 activation through IL-6/IL-11 in cancer-associated fibroblasts promotes colorectal tumour development and correlates with poor prognosis. Gut 69, 1269–1282 (2020).
Tauriello, D. V. F. et al. TGFβ drives immune evasion in genetically reconstituted colon cancer metastasis. Nature 554, 538–543 (2018).
Calon, A. et al. Dependency of colorectal cancer on a TGF-β-driven program in stromal cells for metastasis initiation. Cancer Cell 22, 571–584 (2012).
Nishina, T. et al. Interleukin-11-expressing fibroblasts have a unique gene signature correlated with poor prognosis of colorectal cancer. Nat. Commun. 12, 2281 (2021).
Putoczki, T. L. et al. Interleukin-11 is the dominant IL-6 family cytokine during gastrointestinal tumorigenesis and can be targeted therapeutically. Cancer Cell 24, 257–271 (2013).
Salama, P. et al. Tumor-infiltrating FOXP3+ T regulatory cells show strong prognostic significance in colorectal cancer. J. Clin. Oncol. 27, 186–192 (2009).
Hu, B. et al. Multifocal epithelial tumors and field cancerization from loss of mesenchymal CSL signaling. Cell 149, 1207–1220 (2012).
Nicolas, A. M. et al. Inflammatory fibroblasts mediate resistance to neoadjuvant therapy in rectal cancer. Cancer Cell 40, 168–184.e13 (2022).
Lavie, D., Ben-Shmuel, A., Erez, N. & Scherz-Shouval, R. Cancer-associated fibroblasts in the single-cell era. Nat. Cancer 3, 793–807 (2022).
Buechler, M. B. et al. Cross-tissue organization of the fibroblast lineage. Nature 593, 575–579 (2021).
Yang, Y. et al. Cross-talk between the gut microbiota and monocyte-like macrophages mediates an inflammatory response to promote colitis-associated tumourigenesis. Gut 70, 1495–1506 (2020).
König, J., Rokavec, M., Öner-Ziegler, M. G., Fei, Y. & Hermeking, H. Myeloid Mir34a suppresses colitis-associated colon cancer: characterization of mediators by single-cell RNA sequencing. Cell Death Differ. 32, 225–241 (2025).
Canli, Ö et al. Myeloid cell-derived reactive oxygen species induce epithelial mutagenesis. Cancer Cell 32, 869–883.e5 (2017).
Bollrath, J. et al. gp130-mediated Stat3 activation in enterocytes regulates cell survival and cell-cycle progression during colitis-associated tumorigenesis. Cancer Cell 15, 91–102 (2009).
Shin, A. E. et al. F4/80+Ly6Chigh macrophages lead to cell plasticity and cancer initiation in colitis. Gastroenterology 164, 593–609.e13 (2023).
Katoh, H. et al. CXCR2-expressing myeloid-derived suppressor cells are essential to promote colitis-associated tumorigenesis. Cancer Cell 24, 631–644 (2013).
Bui, T. M. et al. Tissue-specific reprogramming leads to angiogenic neutrophil specialization and tumor vascularization in colorectal cancer. J. Clin. Invest. 134, e174545 (2024).
Qi, J. et al. Single-cell and spatial analysis reveal interaction of FAP+ fibroblasts and SPP1+ macrophages in colorectal cancer. Nat. Commun. 13, 1742 (2022).
Germann, M. et al. Neutrophils suppress tumor-infiltrating T cells in colon cancer via matrix metalloproteinase-mediated activation of TGFβ. EMBO Mol. Med. 12, e10681 (2020).
Pelka, K. et al. Spatially organized multicellular immune hubs in human colorectal cancer. Cell 184, 4734–4752.e20 (2021).
Väyrynen, J. P. et al. Prognostic significance of myeloid immune cells and their spatial distribution in the colorectal cancer microenvironment. J. Immunother. Cancer 9, e002297 (2021).
Gungabeesoon, J. et al. A neutrophil response linked to tumor control in immunotherapy. Cell 186, 1448–1464.e20 (2023).
Pagès, F. et al. International validation of the consensus immunoscore for the classification of colon cancer: a prognostic and accuracy study. Lancet 391, 2128–2139 (2018).
Keerthivasan, S. et al. β-Catenin promotes colitis and colon cancer through imprinting of proinflammatory properties in T cells. Sci. Transl. Med. 6, 225ra28 (2014).
Quandt, J. et al. Wnt-β-catenin activation epigenetically reprograms Treg cells in inflammatory bowel disease and dysplastic progression. Nat. Immunol. 22, 471–484 (2021).
Shaw, D. G. et al. Antigen-driven colonic inflammation is associated with development of dysplasia in primary sclerosing cholangitis. Nat. Med. 29, 1520–1529 (2023).
Genua, F., Raghunathan, V., Jenab, M., Gallagher, W. M. & Hughes, D. J. The role of gut barrier dysfunction and microbiome dysbiosis in colorectal cancer development. Front. Oncol. 11, 626349 (2021).
Grivennikov, S. I. et al. Adenoma-linked barrier defects and microbial products drive IL-23/IL-17-mediated tumour growth. Nature 491, 254–258 (2012).
Landy, J. et al. Tight junctions in inflammatory bowel diseases and inflammatory bowel disease associated colorectal cancer. World J. Gastroenterol. 22, 3117–3126 (2016).
Neurath, M. F. IL-23 in inflammatory bowel diseases and colon cancer. Cytokine Growth Factor. Rev. 45, 1–8 (2019).
Tosolini, M. et al. Clinical impact of different classes of infiltrating T cytotoxic and helper cells (Th1, Th2, Treg, Th17) in patients with colorectal cancer. Cancer Res. 71, 1263–1271 (2011).
Jobin, C. Colorectal cancer: CRC — all about microbial products and barrier function? Nat. Rev. Gastroenterol. Hepatol. 9, 694–696 (2012).
Wu, J. et al. Crosstalk between gut microbiota and metastasis in colorectal cancer: implication of neutrophil extracellular traps. Front. Immunol. 14, 1296783 (2023).
Zhao, H. et al. Effect of junctional adhesion molecule-2 expression on cell growth, invasion and migration in human colorectal cancer. Int. J. Oncol. 48, 929–936 (2016).
Langlois, M. J. et al. The PTEN phosphatase controls intestinal epithelial cell polarity and barrier function: role in colorectal cancer progression. PLoS ONE 5, e15742 (2010).
Okumura, S. et al. Gut bacteria identified in colorectal cancer patients promote tumourigenesis via butyrate secretion. Nat. Commun. 12, 5674 (2021).
Ahmad Kendong, S. M., Raja Ali, R. A., Nawawi, K. N. M., Ahmad, H. F. & Mokhtar, N. M. Gut dysbiosis and intestinal barrier dysfunction: potential explanation for early-onset colorectal cancer. Front. Cell Infect. Microbiol. 11, 744606 (2021).
Van Hul, M. et al. What defines a healthy gut microbiome. Gut 73, 1893–1908 (2024).
Qin, J. et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464, 59–65 (2010).
Zuo, T. et al. Gut mucosal virome alterations in ulcerative colitis. Gut 68, 1169–1179 (2019).
Norman, J. M. et al. Disease-specific alterations in the enteric virome in inflammatory bowel disease. Cell 160, 447–460 (2015).
Sokol, H. et al. Fungal microbiota dysbiosis in IBD. Gut 66, 1039–1048 (2017).
Krawczyk, A., Gosiewski, T., Zapała, B., Kowalska-Duplaga, K. & Salamon, D. Alterations in intestinal Archaea composition in pediatric patients with Crohn’s disease based on next-generation sequencing — a pilot study. Gut Microbes 15, 2276806 (2023).
Lee, M. & Chang, E. B. Inflammatory bowel diseases (IBD) and the microbiome — searching the crime scene for clues. Gastroenterology 160, 524–537 (2021).
Kumar, P. et al. Intestinal Interleukin-17 receptor signaling mediates reciprocal control of the gut microbiota and autoimmune inflammation. Immunity 44, 659–671 (2016).
Lin, H. et al. Polymeric immunoglobulin receptor deficiency exacerbates autoimmune hepatitis by inducing intestinal dysbiosis and barrier dysfunction. Cell Death Dis. 14, 68 (2023).
Federici, S. et al. Targeted suppression of human IBD-associated gut microbiota commensals by phage consortia for treatment of intestinal inflammation. Cell 185, 2879–2898.e24 (2022).
Wirbel, J. et al. Meta-analysis of fecal metagenomes reveals global microbial signatures that are specific for colorectal cancer. Nat. Med. 25, 679–689 (2019).
Arthur, J. C. et al. Intestinal inflammation targets cancer-inducing activity of the microbiota. Science 338, 120–123 (2012).
Chen, B. et al. Contribution of pks+ E. coli mutations to colorectal carcinogenesis. Nat. Commun. 14, 7827 (2023).
Cao, Y. et al. Enterotoxigenic Bacteroides fragilis promotes intestinal inflammation and malignancy by inhibiting exosome-packaged miR-149-3p. Gastroenterology 161, 1552–1566.e12 (2021).
Brennan, C. A. & Garrett, W. S. Fusobacterium nucleatum — symbiont, opportunist and oncobacterium. Nat. Rev. Microbiol. 17, 156–166 (2019).
Dregelies, T., Haumaier, F., Sterlacci, W., Backert, S. & Vieth, M. Detection of Fusobacterium nucleatum in patients with colitis-associated colorectal cancer. Curr. Microbiol. 80, 293 (2023).
Tsoi, H. et al. Peptostreptococcus anaerobius induces intracellular cholesterol biosynthesis in colon cells to induce proliferation and causes dysplasia in mice. Gastroenterology 152, 1419–1433.e5 (2017).
Kwong, T. N. Y. et al. Association between bacteremia from specific microbes and subsequent diagnosis of colorectal cancer. Gastroenterology 155, 383–390.e8 (2018).
Cao, Y. et al. Commensal microbiota from patients with inflammatory bowel disease produce genotoxic metabolites. Science 378, eabm3233 (2022).
Díaz-Gay, M. et al. Geographic and age variations in mutational processes in colorectal cancer. Nature 643, 230–240 (2025).
Kadosh, E. et al. The gut microbiome switches mutant p53 from tumour-suppressive to oncogenic. Nature 586, 133–138 (2020).
Rubinstein, M. R. et al. Fusobacterium nucleatum promotes colorectal carcinogenesis by modulating E-cadherin/β-catenin signaling via its FadA adhesin. Cell Host Microbe 14, 195–206 (2013).
Rubinstein, M. R. et al. Fusobacterium nucleatum promotes colorectal cancer by inducing Wnt/β-catenin modulator annexin A1. EMBO Rep. 20, e47638 (2019).
Ternes, D. et al. The gut microbial metabolite formate exacerbates colorectal cancer progression. Nat. Metab. 4, 458–475 (2022).
Gur, C. et al. Binding of the Fap2 protein of Fusobacterium nucleatum to human inhibitory receptor TIGIT protects tumors from immune cell attack. Immunity 42, 344–355 (2015).
Tanoue, T. et al. A defined commensal consortium elicits CD8 T cells and anti-cancer immunity. Nature 565, 600–605 (2019).
Wang, L. et al. A purified membrane protein from Akkermansia muciniphila or the pasteurised bacterium blunts colitis associated tumourigenesis by modulation of CD8+ T cells in mice. Gut 69, 1988–1997 (2020).
Ralser, A. et al. Helicobacter pylori promotes colorectal carcinogenesis by deregulating intestinal immunity and inducing a mucus-degrading microbiota signature. Gut 72, 1258–1270 (2023).
Galeano Niño, J. L. et al. Effect of the intratumoral microbiota on spatial and cellular heterogeneity in cancer. Nature 611, 810–817 (2022).
Bullman, S. et al. Analysis of Fusobacterium persistence and antibiotic response in colorectal cancer. Science 358, 1443–1448 (2017).
Bertocchi, A. et al. Gut vascular barrier impairment leads to intestinal bacteria dissemination and colorectal cancer metastasis to liver. Cancer Cell 39, 708–724.e11 (2021).
Coker, O. O., Wu, W. K. K., Wong, S. H., Sung, J. J. Y. & Yu, J. Altered gut archaea composition and interaction with bacteria are associated with colorectal cancer. Gastroenterology 159, 1459–1470.e5 (2020).
Richard, M. L. et al. Mucosa-associated microbiota dysbiosis in colitis associated cancer. Gut Microbes 9, 131–142 (2018).
Zhang, Y. et al. Discovery of bioactive microbial gene products in inflammatory bowel disease. Nature 606, 754–760 (2022).
Tol, J., Nagtegaal, I. D. & Punt, C. J. A. BRAF mutation in metastatic colorectal cancer. N. Engl. J. Med. 361, 98–99 (2009).
Claessen, M. M. et al. More right-sided IBD-associated colorectal cancer in patients with primary sclerosing cholangitis. Inflamm. Bowel Dis. 15, 1331–1336 (2009).
Söderlund, S. et al. Inflammatory bowel disease confers a lower risk of colorectal cancer to females than to males. Gastroenterology 138, 1697–1703 (2010).
Amos-Landgraf, J. M. et al. Sex disparity in colonic adenomagenesis involves promotion by male hormones, not protection by female hormones. Proc. Natl Acad. Sci. USA. 111, 16514–16519 (2014).
Neurath, M. F., Sands, B. E. & Rieder, F. Cellular immunotherapies and immune cell depleting therapies in inflammatory bowel diseases: the next magic bullet. Gut 74, 9–14 (2025).
Wang, L. et al. Gut microbiome in tumorigenesis and therapy of colorectal cancer. J. Cell. Physiol. 238, 94–108 (2023).
Itzkowitz, S. et al. Assessment of stool DNA markers to detect colorectal neoplasia in patients with inflammatory bowel disease: a multi-site case-control study. J. Crohns Colitis https://doi.org/10.1093/ecco-jcc/jjad069 (2023).
Al Bakir, I. et al. Low-coverage whole genome sequencing of low-grade dysplasia strongly predicts advanced neoplasia risk in ulcerative colitis. Gut 74, 740–751 (2025).
Lee, J. H. & Wang, T. D. Molecular endoscopy for targeted imaging in the digestive tract. Lancet Gastroenterol. Hepatol. 1, 147–155 (2016).
Chadebecq, F., Lovat, L. B. & Stoyanov, D. Artificial intelligence and automation in endoscopy and surgery. Nat. Rev. Gastroenterol. Hepatol. 20, 171–182 (2023).
Wijnands, A. M. et al. Dynamic prediction of advanced colorectal neoplasia in inflammatory bowel disease. Clin. Gastroenterol. Hepatol. 22, 1697–1708 (2024).
Ten Hoorn, S., de Back, T. R., Sommeijer, D. W. & Vermeulen, L. Clinical value of consensus molecular subtypes in colorectal cancer: a systematic review and meta-analysis. J. Natl Cancer Inst. 114, 503–516 (2022).
Courneya, K. S. et al. Structured exercise after adjuvant chemotherapy for colon cancer. N. Engl. J. Med. 393, 13–25 (2025).
Tsimberidou, A. M. et al. Molecular tumour boards — current and future considerations for precision oncology. Nat. Rev. Clin. Oncol. 20, 843–863 (2023).
Desai, K., Pereira, K., Jain, A., Iqbal, S. & Thirumaran, R. Ivosidenib as a novel treatment for patients with isocitrate dehydrogenase-1 positive advanced colon cancer [abstract]. J. Natl Compr. Cancer Netw. 21, CLO23-047 (2023).
Abou-Alfa, G. K. et al. Ivosidenib in IDH1-mutant, chemotherapy-refractory cholangiocarcinoma (ClarIDHy): a multicentre, randomised, double-blind, placebo-controlled, phase 3 study. Lancet Oncol. 21, 796–807 (2020).
Martling, A. et al. Low-dose aspirin for PI3K-altered localized colorectal cancer. N. Engl. J. Med. 393, 1051–1064 (2025).
Patel, P. et al. Daily aspirin use does not impact clinical outcomes in patients with inflammatory bowel disease. Inflamm. Bowel Dis. 27, 236–241 (2021).
Awad, M. M. et al. Acquired resistance to KRASG12C inhibition in cancer. N. Engl. J. Med. 384, 2382–2393 (2021).
Esposito, A. et al. Colorectal cancer patients-derived immunity-organoid platform unveils cancer-specific tissue markers associated with immunotherapy resistance. Cell Death Dis. 15, 878 (2024).
Günther, C., Winner, B., Neurath, M. F. & Stappenbeck, T. S. Organoids in gastrointestinal diseases: from experimental models to clinical translation. Gut 71, 1892–1908 (2022).
Bullock, A. J. et al. Botensilimab plus balstilimab in relapsed/refractory microsatellite stable metastatic colorectal cancer: a phase 1 trial. Nat. Med. 30, 2558–2567 (2024).
Wu, B., Zhang, B., Li, B., Wu, H. & Jiang, M. Cold and hot tumors: from molecular mechanisms to targeted therapy. Signal Transduct. Target. Ther. 9, 274 (2024).
Lang, F., Schrörs, B., Löwer, M., Türeci, Ö & Sahin, U. Identification of neoantigens for individualized therapeutic cancer vaccines. Nat. Rev. Drug Discov. 21, 261–282 (2022).
Mackensen, A. et al. CLDN6-specific CAR-T cells plus amplifying RNA vaccine in relapsed or refractory solid tumors: the phase 1 BNT211-01 trial. Nat. Med. https://doi.org/10.1038/s41591-023-02612-0 (2023).
Vincent, R. L. et al. Probiotic-guided CAR-T cells for solid tumor targeting. Science 382, 211–218 (2023).
Wehrli, M. et al. Mesothelin CAR T cells secreting anti-FAP/anti-CD3 molecules efficiently target pancreatic adenocarcinoma and its stroma. Clin. Cancer Res. 30, 1859–1877 (2024).
Farin, H. F. et al. Colorectal cancer organoid–stroma biobank allows subtype-specific assessment of individualized therapy responses. Cancer Discov. 13, 2192–2211 (2023).
Garrett, W. S. The gut microbiota and colon cancer. Science 364, 1133–1135 (2019).
Sung, H. et al. Colorectal cancer incidence trends in younger versus older adults: an analysis of population-based cancer registry data. Lancet Oncol. 26, 51–63 (2025).
Vuik, F. E. et al. Increasing incidence of colorectal cancer in young adults in Europe over the last 25 years. Gut 68, 1820–1826 (2019).
Sinicrope, F. A. Increasing incidence of early-onset colorectal cancer. N. Engl. J. Med. 386, 1547–1558 (2022).
Rubin, D. T., Ananthakrishnan, A. N., Siegel, C. A., Barnes, E. L. & Long, M. D. ACG clinical guideline update: ulcerative colitis in adults. Am. J. Gastroenterol. 120, 1187–1224 (2025).
Raine, T. et al. ECCO guidelines on therapeutics in ulcerative colitis: medical treatment. J. Crohns Colitis 16, 2–17 (2021).





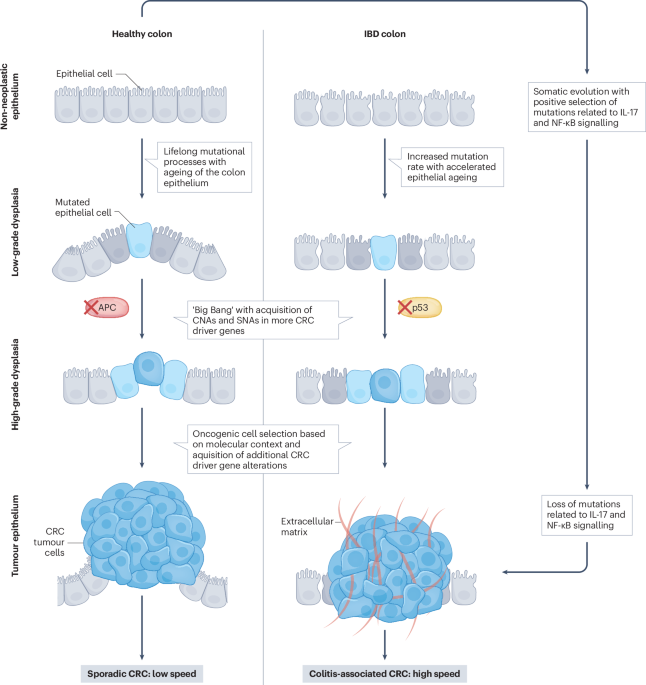








Leave a Reply