Bohm, M. et al. Vitiligo — a disease: a position paper on stigmatization, life quality impairment and psychosocial comorbidity. J. Dtsch Dermatol. Ges. 22, 1327–1335 (2024).
Ezzedine, K. et al. Psychosocial effects of vitiligo: a systematic literature review. Am. J. Clin. Dermatol. 22, 757–774 (2021).
Akl, J. et al. Estimating the burden of vitiligo: a systematic review and modelling study. Lancet Public Health 9, e386–e396 (2024).
Sheth, V. M., Guo, Y. & Qureshi, A. A. Comorbidities associated with vitiligo: a ten-year retrospective study. Dermatology 227, 311–315 (2013).
Alkhateeb, A., Fain, P. R., Thody, A., Bennett, D. C. & Spritz, R. A. Epidemiology of vitiligo and associated autoimmune diseases in caucasian probands and their families. Pigment Cell Res. 16, 208–214 (2003).
Lee, J. H. et al. Comorbidities in patients with vitiligo: a systematic review and meta-analysis. J. Invest. Dermatol. 143, 777–789.e776 (2023).
van Geel, N. & Speeckaert, R. Segmental vitiligo. Dermatol. Clin. 35, 145–150 (2017).
Liu, J. B. et al. Association of vitiligo with HLA-A2: a meta-analysis. J. Eur. Acad. Dermatol. Venereol. 21, 205–213 (2007).
Fain, P. R., Babu, S. R., Bennett, D. C. & Spritz, R. A. HLA class II haplotype DRB1*04-DQB1*0301 contributes to risk of familial generalized vitiligo and early disease onset. Pigment Cell Res. 19, 51–57 (2006).
Jin, Y. et al. Genome-wide association analyses identify 13 new susceptibility loci for generalized vitiligo. Nat. Genet. 44, 676–680 (2012).
Jin, Y. et al. Genome-wide association studies of autoimmune vitiligo identify 23 new risk loci and highlight key pathways and regulatory variants. Nat. Genet. 48, 1418–1424 (2016).
Okamura, K. & Suzuki, T. Genetics and epigenetics in vitiligo. J. Dermatol. Sci. 117, 45–51 (2025).
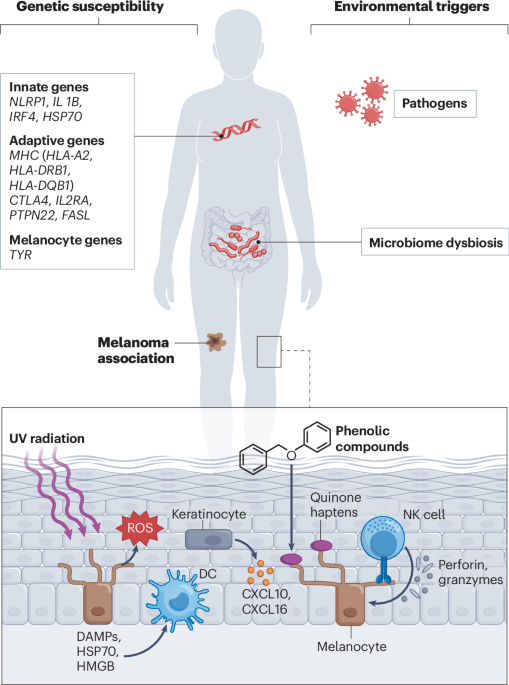
Jin, Y. et al. Variant of TYR and autoimmunity susceptibility loci in generalized vitiligo. N. Engl. J. Med. 362, 1686–1697 (2010). This genome-wide association study shows that human polymorphisms in tyrosinase and MHC class I genes are associated with predisposition to vitiligo, underscoring a link between genetic determinants and melanocyte-targeted autoimmunity.
Puri, N., Mojamdar, M. & Ramaiah, A. In vitro growth characteristics of melanocytes obtained from adult normal and vitiligo subjects. J. Invest. Dermatol. 88, 434–438 (1987).
Schallreuter, K. U. et al. In vivo and in vitro evidence for hydrogen peroxide (H2O2) accumulation in the epidermis of patients with vitiligo and its successful removal by a UVB-activated pseudocatalase. J. Investig. Dermatol. Symp. Proc. 4, 91–96 (1999).
Maresca, V. et al. Increased sensitivity to peroxidative agents as a possible pathogenic factor of melanocyte damage in vitiligo. J. Invest. Dermatol. 109, 310–313 (1997).
Passi, S., Grandinetti, M., Maggio, F., Stancato, A. & De Luca, C. Epidermal oxidative stress in vitiligo. Pigment Cell Res. 11, 81–85 (1998).
Sravani, P. V. et al. Determination of oxidative stress in vitiligo by measuring superoxide dismutase and catalase levels in vitiliginous and non-vitiliginous skin. Indian J. Dermatol. Venereol. Leprol. 75, 268–271 (2009).
Ozel Turkcu, U., Solak Tekin, N., Gokdogan Edgunlu, T., Karakas Celik, S. & Oner, S. The association of FOXO3A gene polymorphisms with serum FOXO3A levels and oxidative stress markers in vitiligo patients. Gene 536, 129–134 (2014).
Yildirim, M., Baysal, V., Inaloz, H. S. & Can, M. The role of oxidants and antioxidants in generalized vitiligo at tissue level. J. Eur. Acad. Dermatol. Venereol. 18, 683–686 (2004).
Khalid-Meften, A. et al. The effect of monobenzone cream on oxidative stress and its relationship with serum levels of IL-1β and IL-18 in vitiligo patients. J. Cosmet. Dermatol. 23, 4085–4093 (2024).
Manini, P., Napolitano, A., Westerhof, W., Riley, P. A. & d’Ischia, M. A reactive ortho-quinone generated by tyrosinase-catalyzed oxidation of the skin depigmenting agent monobenzone: self-coupling and thiol-conjugation reactions and possible implications for melanocyte toxicity. Chem. Res. Toxicol. 22, 1398–1405 (2009).
Jin, Y., Santorico, S. A. & Spritz, R. A. Pediatric to adult shift in vitiligo onset suggests altered environmental triggering. J. Invest. Dermatol. 140, 241–243.e244 (2020).
Ganju, P. et al. Microbial community profiling shows dysbiosis in the lesional skin of vitiligo subjects. Sci. Rep. 6, 18761 (2016).
Bzioueche, H. et al. Analysis of matched skin and gut microbiome of patients with vitiligo reveals deep skin dysbiosis: link with mitochondrial and immune changes. J. Invest. Dermatol. 141, 2280–2290 (2021).
Luan, M. et al. Metagenomic sequencing reveals altered gut microbial compositions and gene functions in patients with non-segmental vitiligo. BMC Microbiol. 23, 265 (2023).
Dellacecca, E. R. et al. Antibiotics drive microbial imbalance and vitiligo development in mice. J. Invest. Dermatol. 140, 676–687.e676 (2020).
Touni, A. A. et al. Topical antibiotics limit depigmentation in a mouse model of vitiligo. Pigment Cell Melanoma Res. 37, 583–596 (2024).
Li, J. X., Yu, T. S., Hsu, S. B., Lin, H. J. & Tsai, F. J. Association of herpes simplex virus infection and vitiligo: a nationwide retrospective cohort study. Arch. Dermatol. Res. 317, 90 (2024).
Erf, G. F., Bersi, T. K., Wang, X., Sreekumar, G. P. & Smyth, J. R. Jr. Herpesvirus connection in the expression of autoimmune vitiligo in Smyth line chickens. Pigment Cell Res. 14, 40–46 (2001).
Zhuang, T. et al. Intracellular virus sensor MDA5 exacerbates vitiligo by inducing the secretion of chemokines in keratinocytes under virus invasion. Cell Death Dis. 11, 453 (2020).
Yu, H., Cen, J., Lin, X., Cheng, H. & Seifert, O. Imiquimod induced vitiligo-like lesions — a consequence of modified melanocyte function. Immun. Inflamm. Dis. 10, 70–77 (2022).
Alatabani, M., Ghobara, Y. & Alissa, A. Vitiligo-like depigmentation following treatment with imiquimod 5% cream for condylomata acuminata. Case Rep. Dermatol. 13, 36–41 (2021).
Burdick, K. H. & Hawk, W. A. Vitiligo in a case of vaccinia virus-treated melanoma. Cancer 17, 708–712 (1964).
Quaglino, P. et al. Vitiligo is an independent favourable prognostic factor in stage III and IV metastatic melanoma patients: results from a single-institution hospital-based observational cohort study. Ann. Oncol. 21, 409–414 (2010).
Hua, C. et al. Association of vitiligo with tumor response in patients with metastatic melanoma treated with pembrolizumab. JAMA Dermatol. 152, 45–51 (2016).
Nishitani, N., Bito, T., Ikeda, T., Tokura, Y. & Nishigori, C. Complete remission of metastatic malignant melanoma after surgery in association with development of systemic vitiligo. J. Dermatol. 37, 770–772 (2010).
Teulings, H. E. et al. Vitiligo-like depigmentation in patients with stage III–IV melanoma receiving immunotherapy and its association with survival: a systematic review and meta-analysis. J. Clin. Oncol. 33, 773–781 (2015).
Nardin, C. et al. Vitiligo under anti-programmed cell death-1 therapy is associated with increased survival in melanoma patients. J. Am. Acad. Dermatol. 82, 770–772 (2020).
Freeman-Keller, M. et al. Nivolumab in resected and unresectable metastatic melanoma: characteristics of immune-related adverse events and association with outcomes. Clin. Cancer Res. 22, 886–894 (2016).
Wu, W. et al. Inverse relationship between vitiligo-related genes and skin cancer risk. J. Invest. Dermatol. 138, 2072–2075 (2018).
Lindelof, B., Hedblad, M. A. & Sigurgeirsson, B. On the association between vitiligo and malignant melanoma. Acta Derm. Venereol. 78, 483–484 (1998).
Teulings, H. E. et al. Decreased risk of melanoma and nonmelanoma skin cancer in patients with vitiligo: a survey among 1307 patients and their partners. Br. J. Dermatol. 168, 162–171 (2013). This retrospective cohort study showed that individuals with vitiligo have a threefold decreased probability of developing melanoma, underscoring a strong link between autoimmunity and antitumour immunity.
Lommerts, J. E. et al. Melanoma-associated leukoderma and vitiligo cannot be differentiated based on blinded assessment by experts in the field. J. Am. Acad. Dermatol. 75, 1198–1204 (2016).
Marchioro, H. Z. et al. Update on the pathogenesis of vitiligo. Bras. Dermatol. 97, 478–490 (2022).
Xu, X., Lu, X., Zheng, Y., Xie, Y. & Lai, W. Cytosolic mtDNA–cGAS–STING axis mediates melanocytes pyroptosis to promote CD8+ T-cell activation in vitiligo. J. Dermatol. Sci. 117, 61–70 (2025).
Wu, X., Yang, Y., Xiang, L. & Zhang, C. The fate of melanocyte: mechanisms of cell death in vitiligo. Pigment Cell Melanoma Res. 34, 256–267 (2021).
Toosi, S., Orlow, S. J. & Manga, P. Vitiligo-inducing phenols activate the unfolded protein response in melanocytes resulting in upregulation of IL6 and IL8. J. Invest. Dermatol. 132, 2601–2609 (2012).
Li, S. et al. Oxidative stress drives CD8+ T-cell skin trafficking in patients with vitiligo through CXCL16 upregulation by activating the unfolded protein response in keratinocytes. J. Allergy Clin. Immunol. 140, 177–189.e179 (2017).
Birol, A. et al. Increased tumor necrosis factor alpha (TNF-α) and interleukin 1 alpha (IL1-α) levels in the lesional skin of patients with nonsegmental vitiligo. Int. J. Dermatol. 45, 992–993 (2006).
Yang, L. et al. Role of chemokines and the corresponding receptors in vitiligo: a pilot study. J. Dermatol. 45, 31–38 (2018).
Speeckaert, R. et al. Critical appraisal of the oxidative stress pathway in vitiligo: a systematic review and meta-analysis. J. Eur. Acad. Dermatol. Venereol. 32, 1089–1098 (2018).
Green, D. R., Ferguson, T., Zitvogel, L. & Kroemer, G. Immunogenic and tolerogenic cell death. Nat. Rev. Immunol. 9, 353–363 (2009).
Kuppner, M. C. et al. The role of heat shock protein (hsp70) in dendritic cell maturation: hsp70 induces the maturation of immature dendritic cells but reduces DC differentiation from monocyte precursors. Eur. J. Immunol. 31, 1602–1609 (2001).
Kroll, T. M. et al. 4-Tertiary butyl phenol exposure sensitizes human melanocytes to dendritic cell-mediated killing: relevance to vitiligo. J. Invest. Dermatol. 124, 798–806 (2005).
Mosenson, J. A. et al. HSP70i is a critical component of the immune response leading to vitiligo. Pigment Cell Melanoma Res. 25, 88–98 (2012).
Denman, C. J. et al. HSP70i accelerates depigmentation in a mouse model of autoimmune vitiligo. J. Invest. Dermatol. 128, 2041–2048 (2008).
Mosenson, J. A. et al. Mutant HSP70 reverses autoimmune depigmentation in vitiligo. Sci. Transl. Med. 5, 174ra128 (2013). This study revealed the DAMP molecule HSP70 as a link between stress and vitiligo pathogenesis, and demonstrated that mutant HSP70 effectively promotes repigmentation in a mouse model.
Cui, T. et al. Oxidative stress-induced HMGB1 release from melanocytes: a paracrine mechanism underlying the cutaneous inflammation in vitiligo. J. Invest. Dermatol. 139, 2174–2184.e2174 (2019).
Tulic, M. K. et al. Innate lymphocyte-induced CXCR3B-mediated melanocyte apoptosis is a potential initiator of T-cell autoreactivity in vitiligo. Nat. Commun. 10, 2178 (2019).
Yu, R. et al. Transcriptome analysis reveals markers of aberrantly activated innate immunity in vitiligo lesional and non-lesional skin. PLoS ONE 7, e51040 (2012).
van den Boorn, J. G. et al. Inflammasome-dependent induction of adaptive NK cell memory. Immunity 44, 1406–1421 (2016). Demonstrating a link between innate immunity and melanocyte killing, this study shows that memory-like NK cells recognize haptens produced by the depigmenting agent monobenzone, leading to NK cell recognition and killing of melanocytes.
Rashighi, M. et al. CXCL10 is critical for the progression and maintenance of depigmentation in a mouse model of vitiligo. Sci. Transl. Med. 6, 223ra223 (2014).
Palermo, B. et al. Specific cytotoxic T lymphocyte responses against Melan-A/MART1, tyrosinase and gp100 in vitiligo by the use of major histocompatibility complex/peptide tetramers: the role of cellular immunity in the etiopathogenesis of vitiligo. J. Invest. Dermatol. 117, 326–332 (2001).
van den Boorn, J. G. et al. Autoimmune destruction of skin melanocytes by perilesional T cells from vitiligo patients. J. Invest. Dermatol. 129, 2220–2232 (2009). This human study provided compelling mechanistic evidence that CD8+ T cells mediate vitiligo disease pathogenesis.
Ogg, G. S., Rod Dunbar, P., Romero, P., Chen, J. L. & Cerundolo, V. High frequency of skin-homing melanocyte-specific cytotoxic T lymphocytes in autoimmune vitiligo. J. Exp. Med. 188, 1203–1208 (1998).
Richmond, J. M. et al. Antibody blockade of IL-15 signaling has the potential to durably reverse vitiligo. Sci. Transl. Med. 10, eaam7710 (2018). Studies here showed a critical role for IL-15 and memory T cells in sustaining vitiligo pathogenesis, and further established IL-15 blocking antibodies as a promising therapeutic tool.
Boniface, K. et al. Vitiligo skin is imprinted with resident memory CD8 T cells expressing CXCR3. J. Invest. Dermatol. 138, 355–364 (2018).
Richmond, J. M., Frisoli, M. L. & Harris, J. E. Innate immune mechanisms in vitiligo: danger from within. Curr. Opin. Immunol. 25, 676–682 (2013).
Cheuk, S. et al. CD49a expression defines tissue-resident CD8+ T cells poised for cytotoxic function in human skin. Immunity 46, 287–300 (2017). This was the first study to implicate skin CD8+ TRM cells in human vitiligo by revealing TRM cell presence in patient skin and the propensity to produce IFNγ.
Gellatly, K. J. et al. scRNA-seq of human vitiligo reveals complex networks of subclinical immune activation and a role for CCR5 in Treg function. Sci. Transl. Med. 13, eabd8995 (2021). These studies show the importance of Treg cells in suppressing vitiligo through the CCR5–CCL5 axis.
Zloza, A. et al. Engagement of NK receptor NKG2D, but not 2B4, results in self-reactive CD8+ T cells and autoimmune vitiligo. Autoimmunity 44, 599–606 (2011).
Plaza-Rojas, L. & Guevara-Patino, J. A. The role of the NKG2D in vitiligo. Front. Immunol. 12, 624131 (2021).
Wang, C. Q. et al. Th17 cells and activated dendritic cells are increased in vitiligo lesions. PLoS ONE 6, e18907 (2011).
Srivastava, N. et al. Dendritic cells sub-sets are associated with inflammatory cytokine production in progressive vitiligo disease. Arch. Dermatol. Res. 313, 759–767 (2021).
Frisoli, M. L., Richmond, J. M. & Harris, J. E. IL-12/IL-23-independent function of BATF3-dependent dendritic cells is required for initiation of disease in a mouse model of vitiligo. J. Invest. Dermatol. 144, 2574–2577.e2572 (2024).
Ramirez, D. E. et al. Depletion of conventional CD4+ T cells is required for robust priming and dissemination of tumor antigen-specific CD8+ T cells in the setting of anti-CD4 therapy. J. Immunother. Cancer 12, e010170 (2024).
Pittet, M. J. et al. High frequencies of naive melan-A/MART-1-specific CD8+ T cells in a large proportion of human histocompatibility leukocyte antigen (HLA)-A2 individuals. J. Exp. Med. 190, 705–715 (1999).
Colella, T. A. et al. Self-tolerance to the murine homologue of a tyrosinase-derived melanoma antigen: implications for tumor immunotherapy. J. Exp. Med. 191, 1221–1232 (2000). This study showed how an altered peptide ligand from tyrosinase can overcome self tolerance, leading to the destruction of melanocytes in a mouse model.
Nichols, L. A. et al. Deletional self-tolerance to a melanocyte/melanoma antigen derived from tyrosinase is mediated by a radio-resistant cell in peripheral and mesenteric lymph nodes. J. Immunol. 179, 993–1003 (2007).
Truckenbrod, E. N. et al. CD8+ T cell self-tolerance permits responsiveness but limits tissue damage. eLife 10, e65615 (2021).
Rizzuto, G. A. et al. Self-antigen-specific CD8+ T cell precursor frequency determines the quality of the antitumor immune response. J. Exp. Med. 206, 849–866 (2009).
Trager, U. et al. The immune response to melanoma is limited by thymic selection of self-antigens. PLoS ONE 7, e35005 (2012).
Barnes, L. Vitiligo and the Vogt–Koyanagi–Harada syndrome. Dermatol. Clin. 6, 229–239 (1988).
Frisoli, M. L., Essien, K. & Harris, J. E. Vitiligo: mechanisms of pathogenesis and treatment. Annu. Rev. Immunol. 38, 621–648 (2020).
Agarwal, S., Ojha, A. & Gupta, S. Profile of vitiligo in Kumaun region of Uttarakhand, India. Indian J. Dermatol. 59, 209 (2014).
Nishimura, E. K. et al. Dominant role of the niche in melanocyte stem-cell fate determination. Nature 416, 854–860 (2002).
Bae, J. M., Kwon, H. S., Lee, J. H. & Kim, G. M. Repigmentation of poliosis in a patient with segmental vitiligo. J. Am. Acad. Dermatol. 75, e23–e24 (2016).
Tabbara, K. F. Reversal of poliosis and vitiligo following Vogt–Koyanagi–Harada disease. Arch. Ophthalmol. 130, 394–396 (2012).
Overwijk, W. W. et al. Tumor regression and autoimmunity after reversal of a functionally tolerant state of self-reactive CD8+ T cells. J. Exp. Med. 198, 569–580 (2003). This study introduced the gp100-specific TCR transgenic mouse which has been widely used as a model of vitiligo.
Zhang, P., Cote, A. L., de Vries, V. C., Usherwood, E. J. & Turk, M. J. Induction of postsurgical tumor immunity and T-cell memory by a poorly immunogenic tumor. Cancer Res. 67, 6468–6476 (2007).
Eby, J. M. et al. Immune responses in a mouse model of vitiligo with spontaneous epidermal de- and repigmentation. Pigment Cell Melanoma Res. 27, 1075–1085 (2014).
Guevara-Patino, J. A. et al. Optimization of a self antigen for presentation of multiple epitopes in cancer immunity. J. Clin. Invest. 116, 1382–1390 (2006).
Bakker, A. B. et al. Analogues of CTL epitopes with improved MHC class-I binding capacity elicit anti-melanoma CTL recognizing the wild-type epitope. Int. J. Cancer 70, 302–309 (1997).
Ostankovitch, M., Altrich-Vanlith, M., Robila, V. & Engelhard, V. H. N-glycosylation enhances presentation of a MHC class I-restricted epitope from tyrosinase. J. Immunol. 182, 4830–4835 (2009).
Ostankovitch, M., Robila, V. & Engelhard, V. H. Regulated folding of tyrosinase in the endoplasmic reticulum demonstrates that misfolded full-length proteins are efficient substrates for class I processing and presentation. J. Immunol. 174, 2544–2551 (2005).
Byrne, K. T. & Turk, M. J. New perspectives on the role of vitiligo in immune responses to melanoma. Oncotarget 2, 684–694 (2011).
Overwijk, W. W. et al. gp100/pmel17 is a murine tumor rejection antigen: induction of “self”-reactive, tumoricidal T cells using high-affinity, altered peptide ligand. J. Exp. Med. 188, 277–286 (1998).
Weber, L. W. et al. Tumor immunity and autoimmunity induced by immunization with homologous DNA. J. Clin. Invest. 102, 1258–1264 (1998).
Bowne, W. B. et al. Coupling and uncoupling of tumor immunity and autoimmunity. J. Exp. Med. 190, 1717–1722 (1999).
Mehrotra, S. et al. A coreceptor-independent transgenic human TCR mediates anti-tumor and anti-self immunity in mice. J. Immunol. 189, 1627–1638 (2012).
Harris, J. E. et al. A mouse model of vitiligo with focused epidermal depigmentation requires IFN-gamma for autoreactive CD8+ T-cell accumulation in the skin. J. Invest. Dermatol. 132, 1869–1876 (2012). This study established CD8+ T cells and IFNγ as dominant mediators of vitiligo pathogenesis in a mouse model.
Tembhre, M. K. et al. Alteration in regulatory T cells and programmed cell death 1-expressing regulatory T cells in active generalized vitiligo and their clinical correlation. Br. J. Dermatol. 172, 940–950 (2015).
Willemsen, M. et al. Immunophenotypic analysis reveals differences in circulating immune cells in the peripheral blood of patients with segmental and nonsegmental vitiligo. J. Invest. Dermatol. 142, 876–883.e873 (2022).
Elela, M. A., Hegazy, R. A., Fawzy, M. M., Rashed, L. A. & Rasheed, H. Interleukin 17, interleukin 22 and FoxP3 expression in tissue and serum of non-segmental vitiligo: a case- controlled study on eighty-four patients. Eur. J. Dermatol. 23, 350–355 (2013).
Dwivedi, M., Laddha, N. C., Arora, P., Marfatia, Y. S. & Begum, R. Decreased regulatory T-cells and CD4+/CD8+ ratio correlate with disease onset and progression in patients with generalized vitiligo. Pigment Cell Melanoma Res. 26, 586–591 (2013).
Abdallah, M., Lotfi, R., Othman, W. & Galal, R. Assessment of tissue FoxP3+, CD4+ and CD8+ T-cells in active and stable nonsegmental vitiligo. Int. J. Dermatol. 53, 940–946 (2014).
Chen, J. et al. Th1-like Treg in vitiligo: an incompetent regulator in immune tolerance. J. Autoimmun. 131, 102859 (2022).
Lili, Y. et al. Global activation of CD8+ cytotoxic T lymphocytes correlates with an impairment in regulatory T cells in patients with generalized vitiligo. PLoS ONE 7, e37513 (2012).
Klarquist, J. et al. Reduced skin homing by functional treg in vitiligo. Pigment Cell Melanoma Res. 23, 276–286 (2010).
Xu, Z. et al. Anatomically distinct fibroblast subsets determine skin autoimmune patterns. Nature 601, 118–124 (2022). These studies implicate fibroblasts for their role in IFNg response and CD8 T cell recruitment to the skin, and illustrate a model whereby regional fibroblast populations govern patterns of autoimmune skin depigmentation.
Shabaneh, T. B. et al. Oncogenic BRAF(V600E) governs regulatory T-cell recruitment during melanoma tumorigenesis. Cancer Res. 78, 5038–5049 (2018).
Essien, K. I., Katz, E. L., Strassner, J. P. & Harris, J. E. Regulatory T cells require CCR6 for skin migration and local suppression of vitiligo. J. Invest. Dermatol. 142, 3158–3166.e3157 (2022).
Eby, J. M. et al. CCL22 to activate Treg migration and suppress depigmentation in vitiligo. J. Invest. Dermatol. 135, 1574–1580 (2015).
Muranski, P. et al. Tumor-specific Th17-polarized cells eradicate large established melanoma. Blood 112, 362–373 (2008).
Lambe, T. et al. CD4 T cell-dependent autoimmunity against a melanocyte neoantigen induces spontaneous vitiligo and depends upon Fas–Fas ligand interactions. J. Immunol. 177, 3055–3062 (2006).
Le Poole, I. C. et al. A novel, antigen-presenting function of melanocytes and its possible relationship to hypopigmentary disorders. J. Immunol. 151, 7284–7292 (1993).
van den Wijngaard, R. et al. Local immune response in skin of generalized vitiligo patients. Destruction of melanocytes is associated with the prominent presence of CLA+ T cells at the perilesional site. Lab. Invest. 80, 1299–1309 (2000).
Naughton, G. K., Eisinger, M. & Bystryn, J. C. Antibodies to normal human melanocytes in vitiligo. J. Exp. Med. 158, 246–251 (1983).
Kemp, E. H., Gawkrodger, D. J., MacNeil, S., Watson, P. F. & Weetman, A. P. Detection of tyrosinase autoantibodies in patients with vitiligo using 35S-labeled recombinant human tyrosinase in a radioimmunoassay. J. Invest. Dermatol. 109, 69–73 (1997).
Kemp, E. H., Waterman, E. A., Gawkrodger, D. J., Watson, P. F. & Weetman, A. P. Autoantibodies to tyrosinase-related protein-1 detected in the sera of vitiligo patients using a quantitative radiobinding assay. Br. J. Dermatol. 139, 798–805 (1998).
Kemp, E. H., Gawkrodger, D. J., Watson, P. F. & Weetman, A. P. Autoantibodies to human melanocyte-specific protein pmel17 in the sera of vitiligo patients: a sensitive and quantitative radioimmunoassay (RIA). Clin. Exp. Immunol. 114, 333–338 (1998).
Teulings, H. E. et al. The antibody response against MART-1 differs in patients with melanoma-associated leucoderma and vitiligo. Pigment Cell Melanoma Res. 27, 1086–1096 (2014).
Overwijk, W. W. et al. Vaccination with a recombinant vaccinia virus encoding a “self” antigen induces autoimmune vitiligo and tumor cell destruction in mice: requirement for CD4+ T lymphocytes. Proc. Natl Acad. Sci. USA 96, 2982–2987 (1999).
Thomson, T. M., Mattes, M. J., Roux, L., Old, L. J. & Lloyd, K. O. Pigmentation-associated glycoprotein of human melanomas and melanocytes: definition with a mouse monoclonal antibody. J. Invest. Dermatol. 85, 169–174 (1985).
Hara, I., Takechi, Y. & Houghton, A. N. Implicating a role for immune recognition of self in tumor rejection: passive immunization against the brown locus protein. J. Exp. Med. 182, 1609–1614 (1995).
Trcka, J. et al. Redundant and alternative roles for activating Fc receptors and complement in an antibody-dependent model of autoimmune vitiligo. Immunity 16, 861–868 (2002).
Bertolotti, A. et al. Type I interferon signature in the initiation of the immune response in vitiligo. Pigment Cell Melanoma Res. 27, 398–407 (2014).
Boukhedouni, N. et al. Type-1 cytokines regulate MMP-9 production and E-cadherin disruption to promote melanocyte loss in vitiligo. JCI Insight 5, e133772 (2020).
Gregg, R. K., Nichols, L., Chen, Y., Lu, B. & Engelhard, V. H. Mechanisms of spatial and temporal development of autoimmune vitiligo in tyrosinase-specific TCR transgenic mice. J. Immunol. 184, 1909–1917 (2010).
Tang, Q. et al. Rational design of a JAK1-selective siRNA inhibitor for the modulation of autoimmunity in the skin. Nat. Commun. 14, 7099 (2023).
Hsueh, Y. C. et al. A keratinocyte-tethered biologic enables location-precise treatment in mouse vitiligo. J. Invest. Dermatol. 142, 3294–3303 (2022).
Richmond, J. M. et al. CXCR3 depleting antibodies prevent and reverse vitiligo in mice. J. Invest. Dermatol. 137, 982–985 (2017).
Wang, X. X. et al. Increased expression of CXCR3 and its ligands in patients with vitiligo and CXCL10 as a potential clinical marker for vitiligo. Br. J. Dermatol. 174, 1318–1326 (2016).
Richmond, J. M. et al. Keratinocyte-derived chemokines orchestrate T-cell positioning in the epidermis during vitiligo and may serve as biomarkers of disease. J. Invest. Dermatol. 137, 350–358 (2017).
Zhang, X. et al. Characteristics and pathogenesis of koebner phenomenon. Exp. Dermatol. 32, 310–323 (2023).
Zhou, L. et al. Increased circulating Th17 cells and elevated serum levels of TGF-β and IL-21 are correlated with human non-segmental vitiligo development. Pigment Cell Melanoma Res. 28, 324–329 (2015).
Carbone, M. L. et al. Insight into immune profile associated with vitiligo onset and anti-tumoral response in melanoma patients receiving anti-PD-1 immunotherapy. Front. Immunol. 14, 1197630 (2023).
Nelson, M. H. et al. The inducible costimulator augments Tc17 cell responses to self and tumor tissue. J. Immunol. 194, 1737–1747 (2015).
Kim, J. C. & Lee, E. S. Progression of pre-existing vitiligo during secukinumab treatment for psoriasis. Ann. Dermatol. 35, S117–S121 (2023).
Kim, N. H., Torchia, D., Rouhani, P., Roberts, B. & Romanelli, P. Tumor necrosis factor-alpha in vitiligo: direct correlation between tissue levels and clinical parameters. Cutan. Ocul. Toxicol. 30, 225–227 (2011).
Bae, J. M. et al. Increased risk of vitiligo following anti-tumor necrosis factor therapy: a 10-year population-based cohort study. J. Invest. Dermatol. 138, 768–774 (2018).
Webb, K. C. et al. Tumour necrosis factor-α inhibition can stabilize disease in progressive vitiligo. Br. J. Dermatol. 173, 641–650 (2015).
Han, J. et al. Resident and circulating memory T cells persist for years in melanoma patients with durable responses to immunotherapy. Nat. Cancer 2, 300–311 (2021).
Byrne, K. T. et al. Autoimmune melanocyte destruction is required for robust CD8+ memory T cell responses to mouse melanoma. J. Clin. Invest. 121, 1797–1809 (2011).
Richmond, J. M. et al. Resident memory and recirculating memory T cells cooperate to maintain disease in a mouse model of vitiligo. J. Invest. Dermatol. 139, 769–778 (2019).
Byrne, K. T., Zhang, P., Steinberg, S. M. & Turk, M. J. Autoimmune vitiligo does not require the ongoing priming of naive CD8 T cells for disease progression or associated protection against melanoma. J. Immunol. 192, 1433–1439 (2014).
Molodtsov, A. & Turk, M. J. Tissue resident CD8 memory T cell responses in cancer and autoimmunity. Front. Immunol. 9, 2810 (2018).
Malik, B. T. et al. Resident memory T cells in the skin mediate durable immunity to melanoma. Sci. Immunol. 2, eaam6346 (2017).
Molodtsov, A. K. et al. Resident memory CD8+ T cells in regional lymph nodes mediate immunity to metastatic melanoma. Immunity 54, 2117–2132.e2117 (2021).
Vella, J. L. et al. Dendritic cells maintain anti-tumor immunity by positioning CD8 skin-resident memory T cells. Life Sci. Alliance 4, e202101056 (2021).
Kruger, C. & Schallreuter, K. U. A review of the worldwide prevalence of vitiligo in children/adolescents and adults. Int. J. Dermatol. 51, 1206–1212 (2012).
Azzolino, V. et al. Jak inhibitors reverse vitiligo in mice but do not deplete skin resident memory T cells. J. Invest. Dermatol. 141, 182–184.e181 (2021).
Schenkel, J. M., Fraser, K. A., Vezys, V. & Masopust, D. Sensing and alarm function of resident memory CD8+ T cells. Nat. Immunol. 14, 509–513 (2013).
Millington, G. W. & Levell, N. J. Vitiligo: the historical curse of depigmentation. Int. J. Dermatol. 46, 990–995 (2007).
Bae, J. M. et al. Phototherapy for vitiligo: a systematic review and meta-analysis. JAMA Dermatol. 153, 666–674 (2017).
Bohm, M. et al. α-Melanocyte-stimulating hormone protects from ultraviolet radiation-induced apoptosis and DNA damage. J. Biol. Chem. 280, 5795–5802 (2005).
Chakraborty, A., Slominski, A., Ermak, G., Hwang, J. & Pawelek, J. Ultraviolet B and melanocyte-stimulating hormone (MSH) stimulate mRNA production for αMSH receptors and proopiomelanocortin-derived peptides in mouse melanoma cells and transformed keratinocytes. J. Invest. Dermatol. 105, 655–659 (1995).
Lim, H. W. et al. Afamelanotide and narrowband UV-B phototherapy for the treatment of vitiligo: a randomized multicenter trial. JAMA Dermatol. 151, 42–50 (2015).
Regazzetti, C. et al. Transcriptional analysis of vitiligo skin reveals the alteration of wnt pathway: a promising target for repigmenting vitiligo patients. J. Invest. Dermatol. 135, 3105–3114 (2015).
Yamada, T. et al. Wnt/β-catenin and kit signaling sequentially regulate melanocyte stem cell differentiation in UVB-induced epidermal pigmentation. J. Invest. Dermatol. 133, 2753–2762 (2013).
Goldstein, N. B. et al. Narrow band ultraviolet b treatment for human vitiligo is associated with proliferation, migration, and differentiation of melanocyte precursors. J. Invest. Dermatol. 135, 2068–2076 (2015).
Mulekar, S. V. Long-term follow-up study of 142 patients with vitiligo vulgaris treated by autologous, non-cultured melanocyte-keratinocyte cell transplantation. Int. J. Dermatol. 44, 841–845 (2005).
Jafarzadeh, A. et al. A systematic review of case series and clinical trials investigating systemic oral or injectable therapies for the treatment of vitiligo. Skin Res. Technol. 30, e13642 (2024).
Lee, J. H. et al. Treatment outcomes of topical calcineurin inhibitor therapy for patients with vitiligo: a systematic review and meta-analysis. JAMA Dermatol. 155, 929–938 (2019).
Craiglow, B. G. & King, B. A. Tofacitinib citrate for the treatment of vitiligo: a pathogenesis-directed therapy. JAMA Dermatol. 151, 1110–1112 (2015).
Harris, J. E. et al. Rapid skin repigmentation on oral ruxolitinib in a patient with coexistent vitiligo and alopecia areata (AA). J. Am. Acad. Dermatol. 74, 370–371 (2016).
Harris, J. E. et al. Safety and efficacy of ruxolitinib cream for the treatment of vitiligo: a randomised controlled trial secondary analysis at 3 years. Skin Health Dis. 4, e404 (2024).
Rosmarin, D. et al. Two phase 3, randomized, controlled trials of ruxolitinib cream for vitiligo. N. Engl. J. Med. 387, 1445–1455 (2022). Two phase II clinical trials demonstrated the efficacy of a topical JAK1/2 inhibitor for repigmentation of vitiligo lesions, leading to the first FDA approval of a drug for the treatment of vitiligo.
Ezzedine, K. et al. Efficacy and safety of oral ritlecitinib for the treatment of active nonsegmental vitiligo: a randomized phase 2b clinical trial. J. Am. Acad. Dermatol. 88, 395–403 (2023).
Guttman-Yassky, E. et al. Improvements in immune/melanocyte biomarkers with JAK3/TEC family kinase inhibitor ritlecitinib in vitiligo. J. Allergy Clin. Immunol. 153, 161–172.e168 (2024).
Phan, K., Phan, S., Shumack, S. & Gupta, M. Repigmentation in vitiligo using janus kinase (JAK) inhibitors with phototherapy: systematic review and meta-analysis. J. Dermatol. Treat. 33, 173–177 (2022).
Seneschal, J. et al. Combination of baricitinib and phototherapy in adults with active vitiligo: a randomized clinical trial. JAMA Dermatol. 161, 375–382 (2025).
Hu, X., Li, J., Fu, M., Zhao, X. & Wang, W. The JAK/STAT signaling pathway: from bench to clinic. Signal Transduct. Target. Ther. 6, 402 (2021).
Jacquemin, C., Taieb, A., Boniface, K., Seneschal, J. & Fhu, A. Imbalance of peripheral follicular helper T lymphocyte subsets in active vitiligo. Pigment Cell Melanoma Res. 32, 588–592 (2019).
Picone, V. et al. Potential future biologic therapies for the treatment of vitiligo: focus on phase 2 and 3. Expert Rev. Clin. Immunol. 21, 711–721 (2025).
Chatterjee, S. et al. A quantitative increase in regulatory T cells controls development of vitiligo. J. Invest. Dermatol. 134, 1285–1294 (2014).
Chinen, T. et al. An essential role for the IL-2 receptor in Treg cell function. Nat. Immunol. 17, 1322–1333 (2016).
Scheid, J. F. et al. Safety, pharmacokinetics, and pharmacodynamics of MK-6194, an IL-2 mutein designed to selectively activate regulatory T cells: single ascending dose and multiple ascending dose trial data. Immunohorizons 9, vlaf005 (2025).














Leave a Reply