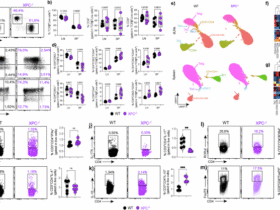
In this study we demonstrate that uveal tissue contains TRM cells. These findings, along with the recent description of T cells in the human cornea5,12, challenge the conventional view that normal uveal tissue is devoid of lymphocytes. Our data suggest that uveal TRM cells likely originate from activated T cells that infiltrate during inflammatory episodes. Supporting this model, we found that clonally expanded T cells in aqueous humour showed enrichment for tissue retention genes while depleting egress markers (Fig. 3J), and in our EAU model, TRM cells accumulated during active inflammation and persisted locally as disease resolved (Fig. 5). This is consistent with developmental pathways where circulating effector T cells encounter antigen in peripheral tissue and, upon receiving appropriate signals (including TGF-β, which induces CD103), adopt a resident memory phenotype8,9.
An important clinical implication from our findings is that TRM cells remaining in post-uveitic eyes may serve as a persistent cellular reservoir capable of triggering disease flares. This concept is analogous to mechanisms described in other tissues: in psoriatic skin, TRM cells persist in resolved lesions and are reactivated to cause new flares21,22; in vitiligo, autoreactive TRM cells are implicated in episodes of recurrent depigmentation21,22. Similarly, we propose that persistent post-uveitic TRM cells may mediate relapses.
Our study adds to accumulating evidence that structures in the eye experience different levels of ‘immune-privilege’23,24,25. To protect vision, the retina must maintain a high degree of immunotolerance whereas the uveal tract, cornea and sclera may tolerate and possibly benefit from the presence of some types of lymphocytes. Greater permeability of the blood aqueous than the blood retinal barrier, and ‘leakiness’ of choroidal blood vessels, are likely to facilitate these differences26. This concept is supported by Shechter et al., who propose that immune-privileged sites are equipped with either restrictive endothelial barriers or immunoregulatory epithelial gates that permit selective leukocyte entry and immune modulation27.
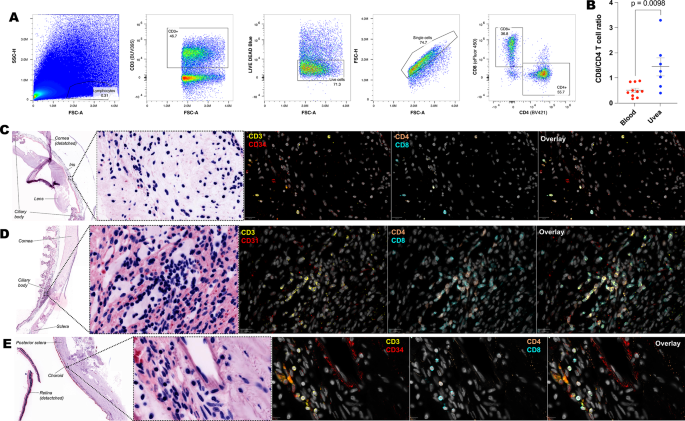
This is also representative of other immune-privileged sites. For example, TRM cells accumulate in the choroid plexus in the brain which has a less restrictive blood barrier than parenchymal tissue28. Likewise, in the testes, normal seminiferous epithelium (the site of spermatogenesis) is considered to be totally absent of lymphocytes, whereas TRM cells are present in other testicular sites29. A breakdown in these barriers such as during local infection, trauma or severe inflammation would permit lymphocyte infiltration and facilitate formation of long-lived TRM cells. This is evident in the eyes with T cell infiltrate in the retina of a human eye with severe chronic uveitis, but not in the retina of an eye without uveitis (Supplementary Fig. 3). Similarly, there is an accumulation of TRM cells in the retina in mice during chronic experimental autoimmune uveoretinitis30. While the retina remains devoid of T cells in the absence of inflammation, the uveal tract harbours TRM populations even under homeostatic conditions. This suggests that the uveal tract, as a border tissue with fenestrated capillaries and greater immune surveillance, operates under distinct immunoregulatory constraints compared to the neural retina. TRM cell persistence in post-uveitic uveal tissue may therefore reflect a shift rather than loss of the local immune equilibrium that favours memory responses and increases susceptibility to recurrent inflammation.
An important question is whether TRM cells contribute similarly to disease flares in anterior uveitis and in posterior uveoretinitis. Our data suggest TRM cells populate both anterior uveal tissue and, in chronic inflammation, the retina (Supplementary Fig. 3). In our EAU model where posterior uveitis predominates, TRM cells persisted in the anterior uvea. (Fig. 5). These findings suggest that TRM mediated mechanisms may operate across anatomical compartments, though differences in barrier permeability and local immune regulation between anterior and posterior tissues may influence disease dynamics.
Targeting TRM cells may offer new approaches for preventing uveitis. TRM cells at other sites are implicated in the pathogenesis of autoimmune and autoinflammatory diseases and may contribute to disease relapse. Recently, synovial CD8 TRM cells were demonstrated to initiate relapse in murine models of inflammatory arthritis and depleting joint specific TRM cells during remission attenuated disease recurrence31. In humans, TRM targeting therapy has shown promise in psoriasis and other skin conditions that are characterised by recurrent lesions in same locations32. TRM cell-depleting therapy potentially has value in other relapsing T-cell mediated, uveitides, such as spondyloarthropathy-associated anterior uveitis and Bechet’s uveitis.
TRM cell depletion may likewise prevent certain instances of immune-checkpoint inhibitor (ICI) uveitis. Most patients on ICI therapy experience an immune-related adverse event (irAE) that is reminiscent of autoimmune disease33. Notably, there is a higher incidence of irAEs in people with pre-existing autoimmune or autoinflammatory conditions where they can manifest as flare at previously inflamed sites33,34,35. ICI-uveitis occurs in about 1% of patients on ICIs for unrelated malignancy36. ICIs block T cell inhibitory proteins, including CTLA-4 and PD-1 which are highly expressed on TRM cells37. ICI-induced TRM activation mechanistically underpins some irAEs. For example, CD8+ TRM cells are key effector cells in ICI-colitis38,39 and are implicated in ICI-dermatitis40. Although the mechanisms underlying ICI-associated uveitis likely involve multiple pathways such as, activation of autoreactive peripheral T cells, suppression of Tregs, and cross-reactivity of tumour-targeting activated T cells41, the local reactivation of uveal TRM cells may also contribute, particularly in anterior uveal tissues where the blood-aqueous barrier is more permeable than the blood-retinal barrier and may permit antibody access. Although the non-pigmented ciliary epithelium normally restricts antibody entry, barrier integrity can be compromised for instance, by trauma, surgery, or inflammation, potentially permitting therapeutic antibody access to uveal tissues. In such contexts, local modulation of resident immune cells, including TRM cells, could contribute to the pathogenesis of ICI-associated uveitis.
The presence of clonally expanded TRM cells within aqueous humour in active uveitis informs our understanding of TCR-antigen interactions that promote uveitis. Our observations align with reports in other settings. For example, TRM cells accumulate in both synovial fluid17 and joint tissue31 in arthritis. Likewise, during chronic neuroinflammatory and neurodegenerative diseases TRM cells accumulate in cerebral spinal fluid11 and central nervous system tissue42. Given multiple expanded T cell clonotypes within all aqueous humour donors, it is possible that following a trigger that facilitates T cell influx, inflammation-associated autoantigens contribute to the local activation of infiltrating T cells. Following activation these cells upregulate TRM genes which facilitates their migration into tissue. There, they remain long-lived and are positioned to promote subsequent episodes of uveitis following an inflammatory trigger (e.g., microtrauma).
Our data indicates that in addition to TRM cells, other T cell subtypes reside in the uveal tissue. Both flow cytometry and immunofluorescence revealed, numerous CD3+ CD103– cells within ocular tissue indicating that additional T cells subtypes reside in the uveal tract. Of particular interest are the TCR-Vd1+ IL-23R+ population identified by flow cytometry. This T cell population resides in entheses and are linked to the development of spondyloarthropathy13, and possibly spondyloarthropathy-associated uveitis6,43. There are several structures within the eye which might be considered analogous to an enthesis, such as the scleral spur or the iris root, that might harbour this T cell population. Unfortunately, our attempts to confirm the presence of these cells by immunofluorescence imaging and spatially locate them to ocular entheses were thwarted by the scarcity of these cells in non-inflamed human tissue combined with challenges in staining for IL-23R and γδ T cells. We detected MAIT cells in non-inflamed control tissue as well in uveitic aqueous humour where they were highly expanded in 3 donors, each with anterior uveitis. Further work is needed to confirm if these cells have a role in reducing inflammation, as described in experimental autoimmune uveitis44.
Although our study predominantly characterised CD8+ TRM cells, we also identified CD4+ populations with T RM markers (CD103+, CD69+) in uveal tissue (Fig. 2C, cluster C2). CD4+ T cells are well-established drivers of uveitis pathogenesis, particularly in Th17-mediated disease, and the presence of CD4+ TRM cells suggests they may contribute to both immune surveillance and disease relapse.
While our study provides phenotypic and transcriptional characterisation of TRM cells in human uveal tissue, we did not assess antigen specificity. Future studies employing tetramer-based approaches or functional assays with candidate antigens (e.g., cytomegalovirus, herpes simplex virus, or ocular autoantigens) would be valuable to determine whether TRM populations in inflamed eyes are enriched for pathogen-specific or autoreactive clones. Such investigations could provide critical insights into the triggers of uveitic flares and inform targeted therapeutic strategies.
Although we did not test the protective capacity of uveal TRM cells in this study, their phenotypic similarity to corneal TRM cells, which have demonstrated protective functions against viral reinfection and environmental insults5,12, suggests the possibility of an analogous, dual role in uveal tissue where TRM cells play a protective role in viral causes of uveitis, providing rapid local recall responses that limit viral replication and tissue damage.
The availability of healthy ocular tissues presents a challenge for human eye research. Our study may be limited in that some of our control tissue may not truly represent normal eyes. In particular, non-uveitic tissue used for immunofluorescence was obtained from enucleated eyes containing posterior ocular melanoma, or from post-mortem donors (Supplementary Table 3). While the uveal tract and surrounding structures in these donors appear histologically normal, they may not fully reflect healthy tissue.
We observed substantial variability among samples from patients with clinically similar uveitis subtypes. This heterogeneity likely reflects several factors: (1) clinical diagnostic categories may encompass immunologically distinct disease subtypes with different underlying pathogenic mechanisms; (2) aqueous humour sampling captures only cells present in the fluid compartment at a single timepoint, providing a limited window into the tissue-resident TRM populations. The heterogeneity and small size of our patient cohort preclude comment about clinical subtypes. Larger prospective studies with well-defined clinical phenotypes, standardised collection processes, and where possible, longitudinal follow-up will be important in determining cellular subtypes in uveitis.
Until recently, it was thought that T cells were only recruited to tissues following infection or damage and then underwent egress or apoptosis following resolution of inflammation8. It is now evident that long-lived resident T cells populate most tissues throughout the body and have important physiological functions. We suggest a contributory role for TRM cells in the pathophysiology of immune-mediated inflammatory diseases including in the eye. Future studies are warranted to clarify their involvement in uveitis and if targeting them therapeutically improve control of some uveitides and their associated systemic diseases.













Leave a Reply