Chien, Y. H., Meyer, C. & Bonneville, M. γδ T cells: first line of defense and beyond. Annu. Rev. Immunol. 32, 121–155 (2014).
Pellicci, D. G., Koay, H. F. & Berzins, S. P. Thymic development of unconventional T cells: how NKT cells, MAIT cells and γδ T cells emerge. Nat. Rev. Immunol. 20, 756–770 (2020).
Lee, S. W., Park, H. J., Van Kaer, L. & Hong, S. Role of CD1d and iNKT cells in regulating intestinal inflammation. Front. Immunol. 14, 1343718 (2024).
Wu, Z., Chen, X., Han, F. & Leeansyah, E. MAIT cell homing in intestinal homeostasis and inflammation. Sci. Adv. 11, eadu4172 (2025).
Beringer, D. X. et al. Disrupting the balance between activating and inhibitory receptors of γδT cells for effective cancer immunotherapy. Nat. Rev. Cancer 25, 590–612 (2025).
Ribot, J. C., Lopes, N. & Silva-Santos, B. γδ T cells in tissue physiology and surveillance. Nat. Rev. Immunol. 21, 221–232 (2021).
Jameson, J. et al. A role for skin gammadelta T cells in wound repair. Science 296, 747–749 (2002).
Nielsen, M. M., Witherden, D. A. & Havran, W. L. γδ T cells in homeostasis and host defence of epithelial barrier tissues. Nat. Rev. Immunol. 17, 733–745 (2017).
Bonneville, M., O’Brien, R. L. & Born, W. K. γδ T cell effector functions: a blend of innate programming and acquired plasticity. Nat. Rev. Immunol. 10, 467–478 (2010).
Hu, Y. et al. γδ T cells: origin and fate, subsets, diseases and immunotherapy. Signal Transduct. Target. Ther. 8, 434 (2023).
Parker, C. M. et al. Evidence for extrathymic changes in the T cell receptor gamma/delta repertoire. J. Exp. Med. 171, 1597–1612 (1990).
Wesch, D., Hinz, T. & Kabelitz, D. Analysis of the TCR Vgamma repertoire in healthy donors and HIV-1-infected individuals. Int. Immunol. 10, 1067–1075 (1998).
Perriman, L. et al. A three-stage developmental pathway for human Vγ9Vδ2 T cells within the postnatal thymus. Sci. Immunol. 8, eabo4365 (2023).
Deusch, K. et al. A major fraction of human intraepithelial lymphocytes simultaneously expresses the γ/δ T cell receptor, the CD8 accessory molecule and preferentially uses the Vδ1 gene segment. Eur. J. Immunol. 121, 1053–1059 (1991).
Kenna, T. et al. Distinct subpopulations of γδ T cells are present in normal and tumor-bearing human liver. Clin. Immunol. 113, 56–63 (2004).
Gray, J. I. et al. Human γδ T cells in diverse tissues exhibit site-specific maturation dynamics across the life span. Sci. Immunol. 9, eadn3954 (2024).
Kierkels, G. J. J. et al. Identification of a tumor-specific allo-HLA-restricted γδTCR. Blood Adv. 3, 2870–2882 (2019).
Harly, C. et al. Key implication of CD277/butyrophilin-3 (BTN3A) in cellular stress sensing by a major human γδ T-cell subset. Blood 120, 2269–2279 (2012).
Herrmann, T. & Karunakaran, M. M. Phosphoantigen recognition by Vγ9Vδ2 T cells. Eur. J. Immunol. 54, e2451068 (2024).
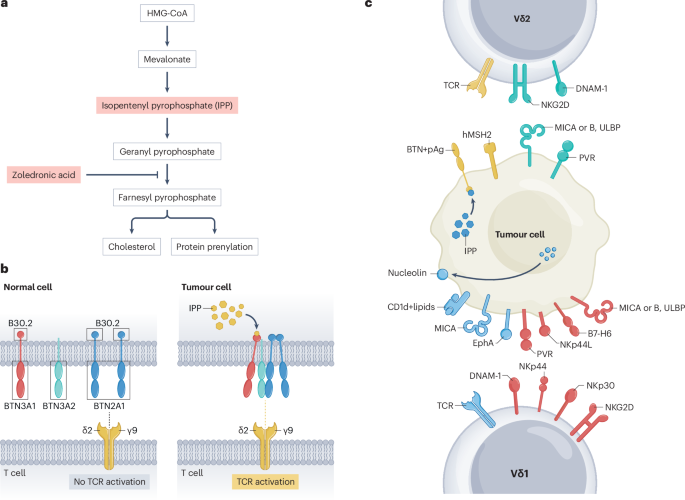
Gruenbacher, G. & Thurnher, M. Mevalonate metabolism in immuno-oncology. Front. Immunol. 8, 1714 (2017).
Roelofs, A. J. et al. Peripheral blood monocytes are responsible for γδ T cell activation induced by zoledronic acid through accumulation of IPP/DMAPP. Br. J. Haematol. 144, 245–250 (2009).
Hintz, M. et al. Identification of (E)-4-hydroxy-3-methyl-but-2-enyl pyrophosphate as a major activator for human gammadelta T cells in Escherichia coli. FEBS Lett. 509, 317–322 (2001).
Sandstrom, A. et al. The intracellular B30.2 domain of butyrophilin 3A1 binds phosphoantigens to mediate activation of human Vγ9Vδ2 T cells. Immunity 40, 490–500 (2014).
Yuan, L. et al. Phosphoantigens glue butyrophilin 3A1 and 2A1 to activate Vγ9Vδ2 T cells. Nature 621, 840–848 (2023).
Gu, S. et al. Phosphoantigen-induced conformational change of butyrophilin 3A1 (BTN3A1) and its implication on Vγ9Vδ2 T cell activation. Proc. Natl Acad. Sci. USA 114, E7311–E7320 (2017).
Fulford, T. S. et al. Vγ9Vδ2 T cells recognize butyrophilin 2A1 and 3A1 heteromers. Nat. Immunol. 25, 1355–1366 (2024).
Karunakaran, M. M. et al. A distinct topology of BTN3A IgV and B30.2 domains controlled by juxtamembrane regions favors optimal human γδ T cell phosphoantigen sensing. Nat. Commun. 14, 7617 (2023).
Rincon-Orozco, B. et al. Activation of Vγ9Vδ2 T cells by NKG2D. J. Immunol. 175, 2144–2151 (2005).
Toutirais, O. et al. DNAX accessory molecule-1 (CD226) promotes human hepatocellular carcinoma cell lysis by Vγ9Vδ2 T cells. Eur. J. Immunol. 39, 1361–1368 (2009).
Silva-Santos, B. & Strid, J. Working in “NK mode”: natural killer group 2 member D and natural cytotoxicity receptors in stress-surveillance by γδ T cells. Front. Immunol. 9, 851 (2018).
Cazzetta, V. et al. NKG2A expression identifies a subset of human Vδ2 T cells exerting the highest antitumor effector functions. Cell Rep. 37, 109871 (2021).
Dokouhaki, P. et al. NKG2D regulates production of soluble TRAIL by ex vivo expanded human γδ T cells. Eur. J. Immunol. 43, 3175–3182 (2013).
Lettau, M. et al. Mechanistic peculiarities of activation-induced mobilization of cytotoxic effector proteins in human T cells. Int. Immunol. 30, 215–228 (2018).
Sandoz, P. A. et al. Modulation of lytic molecules restrain serial killing in γδ T lymphocytes. Nat. Commun. 14, 6035 (2023).
Angelini, D. F. et al. FcγRIII discriminates between 2 subsets of Vγ9Vδ2 effector cells with different responses and activation pathways. Blood 104, 1801–1807 (2004).
Urban, E. M. et al. Control of CD56 expression and tumor cell cytotoxicity in human Vγ2Vδ2 T cells. BMC Immunol. 10, 50 (2009).
Alexander, A. A. et al. Isopentenyl pyrophosphate-activated CD56 + γδ T lymphocytes display potent antitumor activity toward human squamous cell carcinoma. Clin. Cancer Res. 14, 4232–4240 (2008).
Lee, D. et al. Unlocking the potential of allogeneic Vδ2 T cells for ovarian cancer therapy through CD16 biomarker selection and CAR/IL-15 engineering. Nat. Commun. 14, 6942 (2023).
Wesch, D., Glatzel, A. & Kabelitz, D. Differentiation of resting human peripheral blood γδ T cells toward Th1- or Th2-phenotype. Cell. Immunol. 212, 110–117 (2001).
Peters, C., Häsler, R., Wesch, D. & Kabelitz, D. Human Vδ2 T cells are a major source of interleukin-9. Proc. Natl Acad. Sci. USA 113, 12520–12525 (2016).
Ness-Schwickerat, K. J., Jin, C. & Morita, C. T. Cytokine requirements for the differentiation and expansion of IL-17A- and IL-22-producing human Vγ2Vδ2 T cells. J. Immunol. 184, 7268–7280 (2010).
Ribot, J. C. et al. CD27 is a thymic determinant of the balance between interferon-γ- and interleukin 17-producing γδ T cell subsets. Nat. Immunol. 10, 427–436 (2009).
Inácio, D. et al. Signature cytokine-associated transcriptome analysis of effector γδ T cells identifies subset-specific regulators of peripheral activation. Nat. Immunol. 26, 497–510 (2025).
Duan, J., Chung, H., Troy, E. & Kasper, D. L. Microbial colonization drives expansion of IL-1 receptor 1-expressing and IL-17-producing γ/δ T cells. Cell Host Microbe 7, 140–150 (2010).
Ribot, J. C., Ribeiro, S. T., Correia, D. V., Sousa, A. E. & Silva-Santos, B. Human γδ thymocytes are functionally immature and differentiate into cytotoxic type 1 effector T cells upon IL-2/IL-15 signaling. J. Immunol. 192, 2237–2243 (2014).
Schilbach, K. et al. Suppressive activity of Vδ2+ γδ T cells on αβ T cells is licensed by TCR signaling and correlates with signal strength. Cancer Immunol. Immunother. 69, 593–610 (2020).
Barjon, C. et al. IL-21 promotes the development of a CD73-positive Vγ9Vδ2 T cell regulatory population. Oncoimmunology 7, e1379642 (2017).
Xia, C., Yin, S., To, K. K. W. & Fu, L. CD39/CD73/A2AR pathway and cancer immunotherapy. Mol. Cancer 22, 44 (2023).
Mao, Y. et al. A new effect of IL-4 on human γδ T cells: promoting regulatory Vδ1 T cells via IL-10 production and inhibiting function of Vδ2 T cells. Cell. Mol. Immunol. 13, 217–228 (2016).
Chabab, G., Barjon, C., Bonnefoy, N. & Lafont, V. Pro-tumor γδ T cells in human cancer: polarization, mechanisms of action, and implications for therapy. Front. Immunol. 11, 2186 (2020).
Harmon, C. et al. γδ T cell dichotomy with opposing cytotoxic and wound healing functions in human solid tumors. Nat. Cancer 4, 1122–1137 (2023).
Kouakanou, L. et al. Vitamin C supports conversion of human γδ T cells into FOXP3-expressing regulatory cells by epigenetic regulation. Sci. Rep. 10, 6550 (2020).
de Visser, K. E. & Joyce, J. A. The evolving tumor microenvironment: from cancer initiation to metastatic outgrowth. Cancer Cell 41, 374–403 (2023).
Girardi, M. et al. Regulation of cutaneous malignancy by γδ T cells. Science 294, 605–609 (2001).
Kabelitz, D., Serrano, R., Kouakanou, L., Peters, C. & Kalyan, S. Cancer immunotherapy with γδ T cells: many paths ahead of us. Cell. Mol. Immunol. 17, 925–939 (2020).
Sebestyen, Z., Prinz, I., Déchanet-Merville, J., Silva-Santos, B. & Kuball, J. Translating gammadelta (γδ) T cells and their receptors into cancer cell therapies. Nat. Rev. Drug Discov. 19, 169–184 (2020).
Mensurado, S., Blanco-Domínguez, R. & Silva-Santos, B. The emerging roles of γδ T cells in cancer immunotherapy. Nat. Rev. Clin. Oncol. 20, 178–191 (2023).
Hayday, A., Dechanet-Merville, J., Rossjohn, J. & Silva-Santos, B. Cancer immunotherapy by γδ T cells. Science 386, eabq7248 (2024).
Arias-Badia, M., Chang, R. & Fong, L. γδ T cells as critical anti-tumor immune effectors. Nat. Cancer 5, 1145–1157 (2024).
Lv, J. et al. γδT cells, a key subset of T cell for cancer immunotherapy. Front. Immunol. 16, 1562188 (2025).
Gentles, A. J. et al. The prognostic landscape of genes and infiltrating immune cells across human cancers. Nat. Med. 21, 938–945 (2015).
Wu, Y. et al. An innate-like Vδ1+ γδ T cell compartment in the human breast is associated with remission in triple-negative breast cancer. Sci. Transl. Med. 11, eaax9364 (2019).
Lu, H. et al. High abundance of intratumoral γδ T cells favors a better prognosis in head and neck squamous cell carcinoma: a bioinformatic analysis. Front. Immunol. 11, 573920 (2020).
Wu, Y. et al. A local human Vδ1 T cell population is associated with survival in nonsmall-cell lung cancer. Nat. Cancer 3, 696–709 (2022).
Wang, J. et al. Tumor-infiltrating γδT cells predict prognosis and adjuvant chemotherapeutic benefit in patients with gastric cancer. Oncoimmunology 6, e1353858 (2017).
Gherardin, N. A. et al. γδ T cells in Merkel cell carcinomas have a proinflammatory profile prognostic of patient survival. Cancer Immunol. Res. 9, 612–623 (2021).
Nguyen, S. et al. Vδ2 T cells are associated with favorable clinical outcomes in patients with bladder cancer and their tumor reactivity can be boosted by BCG and zoledronate treatments. J. Immunother. Cancer 10, e004880 (2022).
Ma, C. et al. Tumor-infiltrating γδ T lymphocytes predict clinical outcome in human breast cancer. J. Immunol. 189, 5029–5036 (2012).
Wu, P. et al. γδT17 cells promote the accumulation and expansion of myeloid-derived suppressor cells in human colorectal cancer. Immunity 40, 785–800 (2014).
Patil, R. S. et al. IL17 producing γδT cells induce angiogenesis and are associated with poor survival in gallbladder cancer patients. Int. J. Cancer 139, 869–881 (2016).
Wu, Z. et al. Unsynchronized butyrophilin molecules dictate cancer cell evasion of Vγ9Vδ2 T-cell killing. Cell. Mol. Immunol. 21, 362–373 (2024).
Mamedov, M. R. et al. CRISPR screens decode cancer cell pathways that trigger γδ T cell detection. Nature 621, 188–195 (2023).
Gober, H. J. et al. Human T cell receptor gammadelta cells recognize endogenous mevalonate metabolites in tumor cells. J. Exp. Med. 197, 163–168 (2003).
Mattarollo, S. R., Kenna, T., Nieda, M. & Nicol, A. J. Chemotherapy and zoledronate sensitize solid tumour cells to Vγ9Vδ2 T cell cytotoxicity. Cancer Immunol. Immunother. 56, 1285–1297 (2007).
Wrobel, P. et al. Lysis of a broad range of epithelial tumour cells by human γδ T cells: involvement of NKG2D ligands and T-cell receptor- versus NKG2D-dependent recognition. Scand. J. Immunol. 66, 320–328 (2007).
Mensurado, S. et al. CD155/PVR determines acute myeloid leukemia targeting by Delta One T cells. Blood 143, 1488–1495 (2024).
Mikulak, J. et al. NKp46-expressing human gut-resident intraepithelial Vδ1 T cell subpopulation exhibits high antitumor activity against colorectal cancer. JCI Insight 4, e125884 (2019).
Almeida, A. R. et al. Delta One T cells for immunotherapy of chronic lymphocytic leukemia: clinical-grade expansion/differentiation and preclinical proof of concept. Clin. Cancer Res. 22, 5795–5804 (2016).
Blanco-Domínguez, R. et al. Dual modulation of cytotoxic and checkpoint receptors tunes the efficacy of adoptive Delta One T cell therapy against colorectal cancer. Nat. Cancer 6, 1056–1972 (2025).
Lockhart, A., Mucida, D. & Bilate, A. M. Intraepithelial lymphocytes of the Intestine. Annu. Rev. Immunol. 42, 289–316 (2024).
Horn, V. & Sonnenberg, G. F. Group 3 innate lymphoid cells in intestinal health and disease. Nat. Rev. Gastroenterol. Hepatol. 21, 428–443 (2024).
Di Marco Barros, R. et al. Epithelia use butyrophilin-like molecules to shape organ-specific γδ T cell compartments. Cell 167, 203–218 (2016).
Ullrich, R., Schieferdecker, H. L., Ziegler, K., Riecken, E. O. & Zeitz, M. gamma delta T cells in the human intestine express surface markers of activation and are preferentially located in the epithelium. Cell. Immunol. 128, 619-627 (1990)
Han, J. et al. TGF-β controls development of TCRγδ+CD8αα+ intestinal intraepithelial lymphocytes. Cell Discov. 9, 52 (2023).
Suzuki, T., Hayman, L., Kilbey, A., Edwards, J. & Coffelt, S. B. Gut γδ T cells as guardians, disruptors, and instigators of cancer. Immunol. Rev. 298, 198–217 (2020).
Dalton, J. E. et al. Intraepithelial γδ+ lymphocytes maintain the integrity of intestinal epithelial tight junctions in response to infection. Gastroenterology 131, 818–829 (2006).
Ismail, A. S. et al. γδ intraepithelial lymphocytes are essential mediators of host-microbial homeostasis at the intestinal mucosal surface. Proc. Natl Acad. Sci. USA 108, 8743–8748 (2011).
Rampoldi, F. & Prinz, I. Three layers of intestinal γδ T cells talk different languages with the microbiota. Front. Immunol. 13, 849954 (2022).
Malinarich, F. H. et al. Constant TCR triggering suggests that the TCR expressed on intestinal intraepithelial γδ T cells is functional in vivo. Eur. J. Immunol. 40, 3378–3388 (2010).
Meehan, T. F. et al. Protection against colitis by CD100-dependent modulation of intraepithelial γδ T lymphocyte function. Mucosal Immunol. 7, 134–142 (2014).
Chen, Y., Chou, K., Fuchs, E., Havran, W. L. & Boismenu, R. Protection of the intestinal mucosa by intraepithelial γδ T cells. Proc. Natl Acad. Sci. USA 99, 14338–14343 (2002).
Watanabe, M. et al. Interleukin 7 is produced by human intestinal epithelial cells and regulates the proliferation of intestinal mucosal lymphocytes. J. Clin. Invest. 95, 2945–2953 (1995).
Yu, Q. et al. MyD88-dependent signaling for IL-15 production plays an important role in maintenance of CD8αα TCRαβ and TCRγδ intestinal intraepithelial lymphocytes. J. Immunol. 176, 6180–6185 (2006).
Li, Y. et al. Exogenous stimuli maintain intraepithelial lymphocytes via aryl hydrocarbon receptor activation. Cell 147, 629–640 (2011).
Sullivan, Z. A. et al. γδ T cells regulate the intestinal response to nutrient sensing. Science 371, eaba8310 (2021).
Wang, X. et al. Interleukin-22 alleviates metabolic disorders and restores mucosal immunity in diabetes. Nature 514, 237–241 (2014).
Lindemans, C. A. et al. Interleukin-22 promotes intestinal-stem-cell-mediated epithelial regeneration. Nature 528, 560–564 (2015).
Xie, B. et al. Gut-derived memory γδ T17 cells exacerbate sepsis-induced acute lung injury in mice. Nat. Commun. 15, 6737 (2024).
Zhu, X. et al. Dectin-1 signaling on colonic γδ T cells promotes psychosocial stress responses. Nat. Immunol. 24, 625–636 (2023).
Li, G. Q. et al. The intestinal γδ T cells: functions in the gut and in the distant organs. Front. Immunol. 14, 1206299 (2023).
McCarthy, N. E. et al. Proinflammatory Vδ2+ T cells populate the human intestinal mucosa and enhance IFN-γ production by colonic αβ T cells. J. Immunol. 191, 2752–2763 (2013).
Kyes, S., Carew, E., Carding, S. R., Janeway, C. A. Jr. & Hayday, A. Diversity in T-cell receptor gamma gene usage in intestinal epithelium. Proc. Natl Acad. Sci. USA 86, 5527–5531 (1989).
Bas, A. et al. Butyrophilin-like 1 encodes an enterocyte protein that selectively regulates functional interactions with T lymphocytes. Proc. Natl Acad. Sci. USA 108, 4376–4381 (2011).
Melandri, D. et al. The γδTCR combines innate immunity with adaptive immunity by utilizing spatially distinct regions for agonist selection and antigen responsiveness. Nat. Immunol. 19, 1352–1365 (2018).
Jandke, A. et al. Butyrophilin-like proteins display combinatorial diversity in selecting and maintaining signature intraepithelial γδ T cell compartments. Nat. Commun. 11, 3769 (2020).
Panea, C. et al. Butyrophilin-like 2 regulates site-specific adaptations of intestinal γδ intraepithelial lymphocytes. Commun. Biol. 4, 913 (2021).
Willcox, C. R. et al. Butyrophilin-like 3 directly binds a human Vγ4+ T cell receptor using a modality distinct from clonally-restricted antigen. Immunity 51, 813–825 (2019).
McKenzie, D. R. et al. Normality sensing licenses local T cells for innate-like tissue surveillance. Nat. Immunol. 23, 411–422 (2022).
Dart, R. J. et al. Conserved γδ T cell selection by BTNL proteins limits progression of human inflammatory bowel disease. Science 381, eadh0301 (2023).
Rice, M. T. et al. Recognition of the antigen-presenting molecule MR1 by a Vδ3+ γδ T cell receptor. Proc. Natl Acad. Sci. USA 118, e2110288118 (2021).
Wegrecki, M. et al. Atypical sideways recognition of CD1a by autoreactive γδ T cell receptors. Nat. Commun. 13, 3872 (2022).
Reijneveld, J. F. et al. Human γδ T cells recognize CD1b by two distinct mechanisms. Proc. Natl Acad. Sci. USA 117, 22944–22952 (2020).
Morgan, E. et al. Global burden of colorectal cancer in 2020 and 2040: incidence and mortality estimates from GLOBOCAN. Gut 72, 338–344 (2023).
Galon, J. et al. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313, 1960–1964 (2006).
Overman, M. J. et al. Durable clinical benefit with nivolumab plus ipilimumab in DNA mismatch repair-deficient/microsatellite instability-high metastatic colorectal cancer. J. Clin. Oncol. 36, 773–779 (2018).
Germano, G. et al. Inactivation of DNA repair triggers neoantigen generation and impairs tumour growth. Nature 552, 116–120 (2017).
Heregger, R. et al. Unraveling resistance to immunotherapy in MSI-high colorectal cancer. Cancers 15, 5090 (2023).
Corvaisier, M. et al. Vγ9Vδ2 T cell response to colon carcinoma cells. J. Immunol. 175, 5481–5488 (2005).
Maeurer, M. J. et al. Human intestinal Vdelta1 + lymphocytes recognize tumor cells of epithelial origin. J. Exp. Med. 183, 1681–1696 (1996).
Halary, F. et al. Shared reactivity of Vδ2neg γδ T cells against cytomegalovirus-infected cells and tumor intestinal epithelial cells. J. Exp. Med. 201, 1567–1578 (2005).
de Vries, N. L. et al. High-dimensional cytometric analysis of colorectal cancer reveals novel mediators of antitumour immunity. Gut 69, 691–703 (2020).
Meraviglia, S. et al. Distinctive features of tumor-infiltrating γδ T lymphocytes in human colorectal cancer. Oncoimmunology 6, e1347742 (2017).
Rong, L. et al. Analysis of tumor-infiltrating gamma delta T cells in rectal cancer. World J. Gastroenterol. 22, 3573–3580 (2016).
Rodin, W. et al. γδ T cells in human colon adenocarcinomas comprise mainly Vδ1, Vδ2, and Vδ3 cells with distinct phenotype and function. Cancer Immunol. Immunother. 73, 174 (2024).
Chabab, G. et al. Diversity of tumor-infiltrating, γδ T-cell abundance in solid cancers. Cells 9, 1537 (2020).
Yu, L. et al. Tumor-infiltrating gamma delta T-cells reveal exhausted subsets with remarkable heterogeneity in colorectal cancer. Int. J. Cancer 153, 1684–1697 (2023).
Karjalainen, H. et al. Characteristics and significance of γδ T cells in colorectal cancer. Oncoimmunology 14, 2532231 (2025).
Yakou, M. H. et al. TCF-1 limits intraepithelial lymphocyte antitumor immunity in colorectal carcinoma. Sci. Immunol. 8, eadf2163 (2023).
Stary, V. et al. Dysfunctional tumor-infiltrating Vδ1 + T lymphocytes in microsatellite-stable colorectal cancer. Nat. Commun. 15, 6949 (2024).
Li, X. et al. Tim-3 suppresses the killing effect of Vγ9Vδ2 T cells on colon cancer cells by reducing perforin and granzyme B expression. Exp. Cell Res. 386, 111719 (2010).
Deaglio, S. et al. Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression. J. Exp. Med. 204, 1257–1265 (2007).
Hu, G. et al. Tumor-infiltrating CD39+γδ Tregs are novel immunosuppressive T cells in human colorectal cancer. Oncoimmunology 6, e1277305 (2017).
Shida, D. et al. Prognostic value of primary tumor sidedness for unresectable stage IV colorectal cancer: a retrospective study. Ann. Surg. Oncol. 26, 1358–1365 (2019).
Zhan, Y. et al. PLA2G4A promotes right-sided colorectal cancer progression by inducing CD39 + γδ Treg polarization. JCI Insight 6, e148028 (2021).
Suzuki, T. et al. β-Catenin drives butyrophilin-like molecule loss and γδ T-cell exclusion in colon cancer. Cancer Immunol. Res. 11, 1137–1155 (2023).
Zhao, X., Shan, Q. & Xue, H. H. TCF1 in T cell immunity: a broadened frontier. Nat. Rev. Immunol. 22, 147–157 (2022).
Wang, K. et al. Interleukin-17 receptor a signaling in transformed enterocytes promotes early colorectal tumorigenesis. Immunity 41, 1052–1063 (2014).
Coffelt, S. B. et al. IL-17-producing γδ T cells and neutrophils conspire to promote breast cancer metastasis. Nature 522, 345–348 (2015).
Ma, S. et al. IL-17A produced by γδ T cells promotes tumor growth in hepatocellular carcinoma. Cancer Res. 74, 1969–1982 (2014).
Wu, S. et al. A human colonic commensal promotes colon tumorigenesis via activation of T helper type 17 T cell responses. Nat. Med. 15, 1016–1022 (2009).
Housseau, F. et al. Redundant innate and adaptive sources of IL17 production drive colon tumorigenesis. Cancer Res. 76, 2115–2124 (2016).
Jin, C. et al. Commensal microbiota promote lung cancer development via γδ T cells. Cell 176, 998–1013 (2019).
Begagic, E., Vranic, S. & Sominanda, A. The role of interleukin 17 in cancer: a systematic review. Carcinogenesis 46, bgae079 (2025).
Reis, B. S. et al. TCR-Vγδ usage distinguishes protumor from antitumor intestinal γδ T cell subsets. Science 377, 276–284 (2022).
Amicarella, F. et al. Dual role of tumour-infiltrating T helper 17 cells in human colorectal cancer. Gut 66, 692–704 (2017).
Ran, R., Trapecar, M. & Brubaker, D. K. Systematic analysis of human colorectal cancer scRNA-seq revealed limited pro-tumoral IL-17 production potential in gamma delta T cells. Neoplasia 58, 101072 (2024).
Yang, A. Y., Wistuba-Hamprecht, K., Greten, T. F. & Ruf, B. Innate-like T cells in liver disease. Trends Immunol. 45, 535–548 (2024).
Gao, B., Jeong, W. I. & Tian, Z. Liver: an organ with predominant innate immunity. Hepatology 47, 729–736 (2008).
Jenne, C. N. & Kubes, P. Immune surveillance by the liver. Nat. Immunol. 14, 996–1006 (2013).
Hu, Y. et al. Single-cell analysis reveals the origins and intrahepatic development of liver-resident IFN-γ-producing γδ T cells. Cell. Mol. Immunol. 18, 954–968 (2021).
Norris, S. et al. Resident human hepatic lymphocytes are phenotypically different from circulating lymphocytes. J. Hepatol. 28, 84–90 (1998).
Hunter, S. et al. Human liver infiltrating γδ T cells are composed of clonally expanded circulating and tissue-resident populations. J. Hepatol. 69, 654–665 (2018).
Mangan, B. A. et al. Cutting edge: CD1d restriction and Th1/Th2/Th17 cytokine secretion by human Vδ3 T cells. J. Immunol. 191, 30–34 (2013).
Hou, W. & Wu, X. Diverse functions of γδ T cells in the progression of hepatitis B virus and hepatitis C virus infection. Front. Immunol. 11, 619872 (2021).
Ibidapo-Obe, O. & Bruns, T. Tissue-resident and innate-like T cells in patients with advanced chronic liver disease. JHEP Rep. 5, 100812 (2023).
Agrati, C. et al. Vδ1 T lymphocytes expressing a Th1 phenotype are the major γδ T cell subset infiltrating the liver of HCV-infected persons. Mol. Med. 7, 11–19 (2001).
Wu, X. et al. Decreased Vδ2 γδ T cells associated with liver damage by regulation of Th17 response in patients with chronic hepatitis B. J. Infect. Dis. 208, 1294–1304 (2013).
Chen, S. et al. Reciprocal alterations in circulating and hepatic gamma-delta T cells in patients with primary biliary cholangitis. Hepatol. Int. 16, 195–206 (2022).
Llovet, J. M. et al. Hepatocellular carcinoma. Nat. Rev. Dis. Primers 2, 16018 (2016).
Sangro, B., Sarobe, P., Hervás-Stubbs, S. & Melero, I. Advances in immunotherapy for hepatocellular carcinoma. Nat. Rev. Gastroenterol. Hepatol. 18, 525–543 (2021).
Di Blasi, D. et al. Unique T-cell populations define immune-inflamed hepatocellular carcinoma. Cell. Mol. Gastroenterol. Hepatol. 9, 195–218 (2020).
Cai, X. Y. et al. Low counts of γδ T cells in peritumoral liver tissue are related to more frequent recurrence in patients with hepatocellular carcinoma after curative resection. Asian Pac. J. Cancer Prev. 15, 775–780 (2014).
Zhao, N. et al. Intratumoral γδ T-cell infiltrates, chemokine (C-C motif) ligand 4/chemokine (C-C motif) ligand 5 protein expression and survival in patients with hepatocellular carcinoma. Hepatology 73, 1045–1060 (2021).
Zakeri, N. et al. Characterisation and induction of tissue-resident gamma delta T-cells to target hepatocellular carcinoma. Nat. Commun. 13, 1372 (2022).
Jiang, H. et al. γδ T cells in hepatocellular carcinoma patients present cytotoxic activity but are reduced in potency due to IL-2 and IL-21 pathways. Int. Immunopharmacol. 70, 167–173 (2019).
Yi, Y. et al. The functional impairment of HCC-infiltrating γδ T cells, partially mediated by regulatory T cells in a TGFβ- and IL-10-dependent manner. J. Hepatol. 58, 977–983 (2013).
Xia, J., Wang, C. & Li, B. Hepatocellular carcinoma cells induce γδ T cells through metabolic reprogramming into tumor-progressive subpopulation. Front. Oncol. 14, 1451650 (2024).
Hu, Y. et al. Apoptosis, pyroptosis, and ferroptosis conspiringly induce immunosuppressive hepatocellular carcinoma microenvironment and γδ T-cell imbalance. Front. Immunol. 13, 845974 (2022).
He, W. et al. Hepatocellular carcinoma-infiltrating γδ T cells are functionally defected and allogenic Vδ2+ γδ T cell can be a promising complement. Clin. Transl. Med. 12, e800 (2022).
You, H. et al. CD69+ Vδ1γδ T cells are anti-tumor subpopulations in hepatocellular carcinoma. Mol. Immunol. 172, 76–84 (2024).
Bruni, E. et al. Intrahepatic CD69+Vδ1 T cells re-circulate in the blood of patients with metastatic colorectal cancer and limit tumor progression. J. Immunother. Cancer 10, e004579 (2022).
Bruni, E. et al. Chemotherapy accelerates immune-senescence and functional impairments of Vδ2pos T cells in elderly patients affected by liver metastatic colorectal cancer. J. Immunother. Cancer 7, 347 (2019).
Handgretinger, R. & Schilbach, K. The potential role of γδ T cells after allogeneic HCT for leukemia. Blood 131, 1063–1072 (2018).
Xu, Y. et al. Allogeneic Vγ9Vδ2 T-cell immunotherapy exhibits promising clinical safety and prolongs the survival of patients with late-stage lung or liver cancer. Cell. Mol. Immunol. 18, 427–439 (2021).
Saura-Esteller, J. et al. Gamma delta T-cell based cancer immunotherapy: past-present-future. Front. Immunol. 13, 915837 (2022).
Wu, D. et al. Ex vivo expanded human circulating Vδ1 γδT cells exhibit favorable therapeutic potential for colon cancer. Oncoimmunology 4, e992749 (2015).
Devaud, C. et al. Anti-metastatic potential of human Vδ1+ γδ T cells in an orthotopic mouse xenograft model of colon carcinoma. Cancer Immunol. Immunother. 62, 1199–1210 (2013).
Van Acker, H. H. et al. Interleukin-15 enhances the proliferation, stimulatory phenotype, and antitumor effector functions of human gamma delta T cells. J. Hematol. Oncol. 9, 101 (2016).
Aehnlich, P., Carnaz Simões, A. M., Skadborg, S. K., Holmen Olofsson, G. & Thor Straten, P. Expansion with IL-15 increases cytotoxicity of Vγ9Vδ2 T cells and is associated with higher levels of cytotoxic molecules and T-bet. Front. Immunol. 11, 1868 (2020).
Fowler, D. et al. Payload-delivering engineered γδ T cells display enhanced cytotoxicity, persistence, and efficacy in preclinical models of osteosarcoma. Sci. Transl. Med. 16, eadg9814 (2024).
Nattress, C. B. et al. Phenoscaping reveals multimodal γδ T cell cytotoxicity as a strategy to overcome cancer cell-mediated immunomodulation. Cancer Res. 85, 4415–4432 (2025).
Kouakanou, L. et al. Vitamin C promotes the proliferation and effector functions of human γδ T cells. Cell. Mol. Immunol. 17, 462–473 (2020).
Uslu, U. & June, C. H. Beyond the blood: expanding CAR T cell therapy to solid tumors. Nat. Biotechnol. 43, 506–515 (2025).
Thomas, P., Paris, P. & Pecqueur, C. Arming Vδ2 T cells with chimeric antigen receptors to combat cancer. Clin. Cancer Res. 20, 3105–3116 (2024).
Zhai, X. et al. MUC1-Tn-targeting chimeric antigen receptor-modified Vγ9Vδ2 T cells with enhanced antigen-specific anti-tumor activity. Am. J. Cancer Res. 11, 79–91 (2021).
Makkouk, A. et al. Off-the-shelf Vδ1 gamma delta T cells engineered with glypican-3 (GPC-3)-specific chimeric antigen receptor (CAR) and soluble IL-15 display robust antitumor efficacy against hepatocellular carcinoma. J. Immunother. Cancer 9, e003441 (2021).
Magee, M. S. et al. Human GUCY2C-targeted chimeric antigen receptor (CAR)-expressing T cells eliminate colorectal cancer metastases. Cancer Immunol. Res. 6, 509–516 (2018).
Li, M. et al. CD318 is a target of chimeric antigen receptor T cells for the treatment of colorectal cancer. Clin. Exp. Med. 23, 2409–2419 (2023).
de Bruin, R. C. G. et al. A bispecific nanobody approach to leverage the potent and widely applicable tumor cytolytic capacity of Vγ9Vδ2-T cells. Oncoimmunology 7, e1375641 (2017).
van Diest, E. et al. Gamma delta TCR anti-CD3 bispecific molecules (GABs) as novel immunotherapeutic compounds. J. Immunother. Cancer 9, e003850 (2021).
Marcu-Malina, V. et al. Redirecting αβ T cells against cancer cells by transfer of a broadly tumor-reactive γδT-cell receptor. Blood 118, 50–59 (2011).
Straetemans, T. et al. GMP-grade manufacturing of T cells engineered to express a defined γδTCR. Front. Immunol. 9, 1062 (2018).
Meringa, A. D. et al. Strategies to improve γδTCRs engineered T-cell therapies for the treatment of solid malignancies. Front. Immunol. 14, 1159337 (2023).
De Gassart, A. et al. Development of ICT01, a first-in-class, anti-BTN3A antibody for activating Vγ9Vδ2 T cell-mediated antitumor immune response. Sci. Transl. Med. 13, eabj0835 (2021).
Champiat, S. et al. EVICTION study: ICT01, an anti-butyrophilin 3A monoclonal antibody activating γ9δ2 T cells in combination with pembrolizumab in checkpoint inhibitor refractory melanoma. J. Clin. Oncol. 42, 9534 (2024).
Maiti, A. et al. ICT01, an investigational γ9δ2 T cell activator, added to azacitidine-venetoclax achieves frequent and early complete remissions in adults with AML unfit for intensive induction chemotherapy: interim results from the ongoing open-label, randomized phase 1 study eviction. Blood 144, 2876 (2024).
Payne, K. K. et al. BTN3A1 governs antitumor responses by coordinating αβ and γδ T cells. Science 369, 942–949 (2020).
Todaro, M. et al. Efficient killing of human colon cancer stem cells by γδ T lymphocytes. J. Immunol. 182, 7287–7196 (2009).
Zocchi, M. R. et al. Zoledronate can induce colorectal cancer microenvironment expressing BTN3A1 to stimulate effector γδ T cells with antitumor activity. Oncoimmunology 6, e1278099 (2017).
Reid, I. R. et al. Zoledronate. Bone 137, 115390 (2020).
Di Mascolo, D. et al. Nanoformulated zoledronic acid boosts the Vδ2 T cell immunotherapeutic potential in colorectal cancer. Cancers 12, 104 (2019).
Benelli, R. et al. Targeting of colorectal cancer organoids with zoledronic acid conjugated to the anti-EGFR antibody cetuximab. J. Immunother. Cancer 10, e005660 (2022).
Tyler, C. J. et al. Antigen-presenting human γδ T cells promote intestinal CD4+ T cell expression of IL-22 and mucosal release of calprotectin. J. Immunol. 198, 3417–3425 (2017).
Holmen Olofsson, G. et al. Vγ9Vδ2 T cells concurrently kill cancer cells and cross-present tumor antigens. Front. Immunol. 12, 645131 (2021).
Rancan, C. et al. Exhausted intratumoral Vδ2− γδ T cells in human kidney cancer retain effector function. Nat. Immunol. 24, 612–624 (2023).
Davies, D. et al. PD-1 defines a distinct, functional, tissue-adapted state in Vδ1+ T cells with implications for cancer immunotherapy. Nat. Cancer 5, 420–432 (2024).
de Vries, N. L. et al. γδ T cells are effectors of immunotherapy in cancers with HLA class I defects. Nature 613, 743–750 (2023).
Lu, H. et al. B7-H3 inhibits the IFN-γ-dependent cytotoxicity of Vγ9Vδ2 T cells against colon cancer cells. Oncoimmunology 9, 1748991 (2020).
André, P. et al. Anti-NKG2A mAb is a checkpoint inhibitor that promotes anti-tumor immunity by unleashing both T and NK cells. Cell 175, 1731–1743 (2018).
Mantovani, A., Allavena, P., Marchesi, F. & Garlanda, C. Macrophages as tools and targets in cancer therapy. Nat. Rev. Drug Discov. 21, 799–820 (2022).
Wesch, D., Kabelitz, D. & Oberg, H. H. Tumor resistance mechanisms and their consequences on γδ T cell activation. Immunol. Rev. 298, 84–98 (2020).
Wang, Q., Yu, M. & Zhang, S. The characteristics of the tumor immune microenvironment in colorectal cancer with different MSI status and current therapeutic strategies. Front. Immunol. 15, 1440830 (2025).
Shen, K. Y., Zhu, Y., Xie, S. Z. & Qin, L. X. Immunosuppressive tumor microenvironment and immunotherapy of hepatocellular carcinoma: current status and prospectives. J. Hematol. Oncol. 17, 25 (2024).
Jing, Y. et al. Remodelling hypoxic TNBC microenvironment restores antitumor efficacy of Vγ9Vδ2 T cell therapy. Br. J. Cancer 133, 365–380 (2025).
Hayati, M. J. et al. Gamma delta T cells in the tumour microenvironment: a double-edged sword. Immunology 177, 44–58 (2026).
Lanng, K. R. B., Lauridsen, E. L. & Jakobsen, M. R. The balance of STING signaling orchestrates immunity in cancer. Nat. Immunol. 25, 1144–1157 (2024).
Richter, F., Paget, C. & Apetoh, L. STING-driven activation of T cells: relevance for the adoptive cell therapy of cancer. Cell Stress 7, 95–104 (2023).
Serrano, R. et al. Stimulatory and inhibitory activity of STING ligands on tumor-reactive human gamma/delta T cells. Oncoimmunology 11, 2030021 (2022).
Luo, J. et al. γδ T cell-mediated tumor immunity is tightly regulated by STING and TGF-β signaling pathways. Adv. Sci. 12, e2404432 (2025).
Scirgolea, C. et al. NaCl enhances CD8+ T cell effector functions in cancer immunotherapy. Nat. Immunol. 25, 1845–1857 (2024).
Soll, D. et al. Sodium chloride in the tumor microenvironment enhances T cell metabolic fitness and cytotoxicity. Nat. Immunol. 25, 1830–1844 (2024).
Parthasarathy, A., Li, T. & Edelblum, K. L. Crosstalk between the microbiota and intestinal γδ T cell compartments in health and IBD. Gut Microbes 18, 2604908 (2026).
Dai, Y., Chen, H., Mo, C., Cui, L. & He, W. Ectopically expressed human tumor biomarker MutS homologue 2 is a novel endogenous ligand that is recognized by human γδ T cells to induce innate anti-tumor/virus immunity. J. Biol. Chem. 287, 16812–16819 (2012).
Luoma, A. M. et al. Crystal structure of Vδ1 T cell receptor in complex with CD1d-sulfatide shows MHC-like recognition of a self-lipid by human γδ T cells. Immunity 39, 1032–1042 (2013).
Uldrich, A. P. et al. CD1d-lipid antigen recognition by the γδ TCR. Nat. Immunol. 14, 1137–1145 (2013).
Xu, B. et al. Crystal structure of a γδ T-cell receptor specific for the human MHC class I homolog MICA. Proc. Natl Acad. Sci. USA 108, 2414–2419 (2011).
You, H. et al. γδ T-cell autoresponses to ectopic membrane proteins: a new type of pattern recognition. Cell. Mol. Immunol. 22, 356–370 (2025).
Harly, C. et al. Human γδ T cell sensing of AMPK-dependent metabolic tumor reprogramming through TCR recognition of EphA2. Sci. Immunol. 6, eaba9010 (2021).
Marlin, R. et al. Sensing of cell stress by human γδ TCR-dependent recognition of annexin A2. Proc. Natl Acad. Sci. USA 114, 3163–3168 (2017).
Willcox, C. R. et al. Cytomegalovirus and tumor stress surveillance by binding of a human γδ T cell antigen receptor to endothelial protein C receptor. Nat. Immunol. 13, 872–879 (2012).














Leave a Reply