TP2-5 synergizes with multiple antibiotics and attenuates MIC escalation during serial passage
We first investigated whether TP2-5 can act as an antibiotic adjuvant against MDR E. coli BAA 2452. Checkerboard assays showed strong synergistic interaction between TP2-5 and meropenem (MEM), colistin (COL), vancomycin (VA) and ciprofloxacin (CIP). In the presence of 1/4× MIC of TP2-5, the MIC of MEM was reduced 4-fold (from 1 µg/mL to 0.25 µg/mL), while the minimum inhibitory concentrations (MICs) of COL and CIP were each reduced 4-fold. VA exhibited a high MIC of 64 µg/mL but also showed an 8-fold MIC reduction in combination with TP2-5 (Fig. 1a–d and Table S2). Because vancomycin is normally excluded by the Gram-negative outer membrane, the observed MIC reduction is interpreted as evidence of envelope barrier permeabilization rather than a clinically intended treatment combination. Synergy was defined as a fractional inhibitory concentration index (FICI) < 0.5 and was observed for TP2-5 in combination with MEM (FICI = 0.31), COL (FICI = 0.38), CIP (FICI = 0.38), and VA (FICI = 0.38) against MDR E. coli in Mueller-Hinton broth (MHB). This synergistic activity was retained when tested in 50% human serum (Fig. 1a–d). To broaden our assessment of TP2-5 as an adjuvant, we determined FIC index values for combinations of TP2-5 with vancomycin, ciprofloxacin, meropenem and colistin across multiple wild-type and MDR pathogens (Fig. 1e–h). All combinations exhibited synergistic activity when used to treat different pathogens, except for the combination of TP2-5 with vancomycin to treat S. epidermidis 32015, P. aeruginosa 19660, and MRSA 50015, and the combination of TP2-5 with ciprofloxacin against P. aeruginosa 19660. In these strains, the interaction with vancomycin or ciprofloxacin was generally additive (0.5 < FICI < 1) rather than synergistic (FICI ≤ 0.5), consistent with strain dependent intrinsic determinants of antibiotic entry and target access.

Synergistic antibacterial activity of TP2-5 combined with a meropenem (MEM), b colistin (COL), c vancomycin (VA), and d ciprofloxacin (CIP) against MDR E. coli BAA 2452 in Mueller-Hinton broth (MHB) and 50% (v/v) human serum using checkerboard broth microdilution assays under static conditions. e–h show fractional inhibitory concentration index (FICI) values for combinations including e TP2-5 plus meropenem, f TP2-5 plus colistin, g TP2-5 plus vancomycin, and h TP2-5 plus ciprofloxacin, tested against diverse Gram-positive and Gram-negative isolates in MHB. Checkerboard assays were performed with twofold serial dilutions of each compound ranging from 0 to 2× MIC. FICI values were calculated from the lowest inhibitory concentration combination in each assay, defined as the lowest inhibitory TP2-5 plus antibiotic concentration pair identified within the checkerboard matrix; therefore, the underlying fractional MICs do not represent a single fixed ratio. Interactions were defined as synergistic (FICI ≤ 0.5), additive (0.5 < FICI ≤ 1), indifferent (1 < FICI ≤ 4), and antagonistic (FICI > 4). Panels i to l show a 21-day serial passage resistance evolution assay with daily MIC determination in MDR E. coli BAA 2452 under static conditions. Individual lineages were passaged under TP2-5 alone at 1× MIC, antibiotic alone, or antibiotic in combination with TP2-5 fixed at 0.25× MIC relative to the day 0 TP2-5 MIC, while antibiotic concentrations were updated daily based on measured MIC. MIC fold change is plotted relative to day 0, with higher fold increase indicating reduced susceptibility. TP2-5 was combined with i meropenem, j colistin, k vancomycin, and l ciprofloxacin. For the VA monotherapy lineage, passaging was terminated at passage 9 when the MIC reached the assay upper limit (4096 μg/mL). Data are shown as mean ± standard deviation of three replicates. Note that the TP2-5 monotherapy lineage was passaged at an inhibitory concentration (1× MIC) and is therefore not directly comparable to the sub-MIC TP2-5 condition used in the combination lineage.
Growth inhibition assays (Fig. S1a–d) revealed that neither TP2-5 nor antibiotics at 1/4× MIC alone could significantly inhibit bacterial growth during a 24-h period. However, their combination at the same concentrations (1/4× MIC) led to marked growth suppression. Bactericidal kinetics assays further showed that the combined treatment achieved complete bacterial clearance within 4–8 h (Fig. S1e–h), whereas either agent alone allowed only partial killing. These results confirm that the combined treatment achieves real synergistic bactericidal activity at sub-MIC levels that are individually non-lethal.
We next evaluated the potential of TP2-5 to enhance antibiotic-mediated inhibition and elimination of MDR E. coli BAA 2452 biofilms using crystal violet staining assays. In the presence of sub-MIC TP2-5, the inhibitory effects of sub-MIC antibiotics were greater than those of antibiotics alone (Fig. S2a–e). Additionally, treating mature biofilms with the combination of sub-MIC TP2-5 and sub-MIC antibiotics caused the biofilms to disperse (Fig. S2f–h).
Finally, we evaluated the effect of TP2-5 on antibiotic MIC escalation during a 21-day serial passage experiment (Fig. 1i–l). When MDR E. coli BAA 2452 was cultured with antibiotics alone, high-level resistance emerged rapidly, with MICs increasing 8-fold for MEM, 16-fold for COL, 128-fold for VA, and 64-fold for CIP. For the vancomycin monotherapy lineage, the experiment was concluded in 9 passages because the MIC reached 4096 μg/mL. At this extreme concentration, further testing was discontinued as the MIC had reached the upper limit of the assay, and the magnitude of resistance was considered biologically definitive. In contrast, when antibiotics were serially passaged in the presence of TP2-5 fixed at 1/4× MIC (day 0), the magnitude of antibiotic MIC escalation was markedly attenuated throughout the 21-day experiment. In parallel, the TP2-5 alone group was conducted at 1× MIC to assess whether bacteria could develop resistance to the peptide under inhibitory conditions, and the minimal change in TP2-5 MIC observed over the same period is consistent with the intrinsic resistance-evasive property of membrane-active AMPs (Fig. 1i–l). As TP2-5 was passaged at 1× MIC in the monotherapy lineage, whereas it was maintained at a fixed sub-MIC concentration in the combination lineage, these conditions are not directly equivalent, and cross-condition comparison should be interpreted with this limitation in mind.
Sub-MIC TP2-5 remodels the bacterial envelope and is associated with inner-membrane hyperpolarization in Gram-negative bacteria
To understand how TP2-5 is associated with antibiotic potentiation and attenuated MIC escalation, we next examined its effects on bacterial envelope structure and membrane potential. 2D-NMR spectroscopy and molecular dynamics simulations revealed that TP2-5 exhibits an amphipathic α-helical conformation (Fig. S3b) with hydrophobic (light brown), hydrophilic (blue) moieties (Fig. S3c), and a positively charged surface. This characteristic is expected to be essential for antimicrobial function, as it facilitates peptide interaction with the bacterial membrane. In support of this mechanism, we also observed TP2-5 interaction with a model bacterial membrane in a 2000-ns in silico study (Fig. S3d). This intramolecular interaction was further supported by enhanced hydrogen bonding and hydrophobic interactions as a function of time (Fig. S3e, f), consistent with preferential interaction with anionic bacterial lipids.
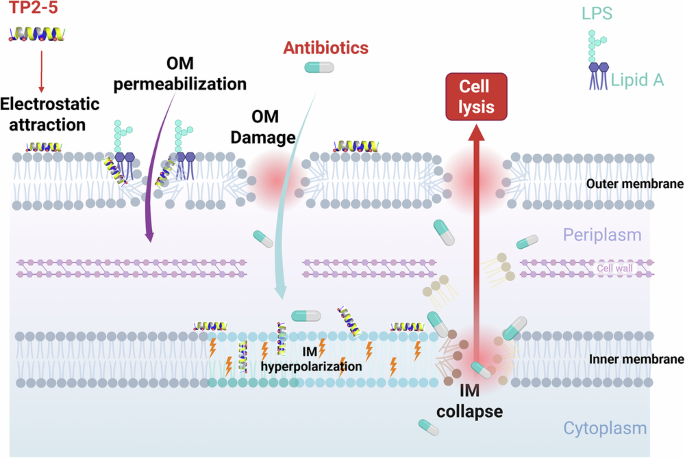
After demonstrating that TP2-5 interacts with bacterial membranes, we examined its effects on MDR E. coli BAA 2452. N-phenyl-1-naphthylamine (NPN) assays revealed that TP2-5 significantly increased outer membrane permeability in a dose-dependent manner (Fig. 2a). We then monitored cytoplasmic membrane potential using the anionic dye DiBAC₄(3). At high bactericidal concentrations (1×, 5× and 10× MIC), TP2-5 induced strong depolarization, indicated by a marked increase in DiBAC₄(3) fluorescence (Fig. 2b) with physical membrane disruption consistent with loss of membrane potential integrity. Notably, at 1/4× MIC, DiBAC₄(3) fluorescence significantly decreased below baseline, consistent with inner-membrane hyperpolarization under sub-lethal peptide exposure (Fig. 2b). At 1/2× MIC, the signal returned close to baseline. These data suggest that, at sublethal doses, TP2-5 does not immediately collapse membrane potential; rather, sub-MIC TP2-5 is associated with a hyperpolarized membrane state, whereas higher doses trigger classical depolarization. The propidium iodide (PI) assay, which detects complete inner membrane disruption, also showed a sharp increase in fluorescence intensity with increasing concentrations of TP2-5, suggesting that TP2-5 disrupts inner membrane integrity (Fig. 2c).

a Outer membrane permeabilization of MDR E. coli according to the N-Phenyl-1-naphthylamine (NPN) assay, b inner membrane potential change according to DiBAC₄(3), and c inner membrane rupture according to the propidium iodide (PI) assay at 0.25× MIC to 10× MIC. A dose-dependent transition was observed from a DiBAC₄(3) signal consistent with inner-membrane hyperpolarization at 0.25× MIC to extensive membrane disruption at 5–10× MIC. Cryo-electron tomography d–l shows corresponding morphological changes in E. coli minicells treated with or without TP2-5. d A tomographic slice of an untreated minicell. e Enlarged image of the blue box in (d) shows outer membrane (OM), peptidoglycan (PG), and inner membrane (IM). f The average distances between OM and PG were measured from 50 minicells, and the distance was determined to be 6.2 ± 1.4 nm. g, h Tomographic slices of minicells treated with TP2-5 at 0.25× and 0.5× MIC. i, j cells were treated with 1× MIC; k, l images of cells treated with 5× and 10× MIC. Scale bar represents 100 nm. Yellow arrow: pore formation. Red arrow: blister. Green arrow: lipid cluster. Violet arrow: shrunken cell. White arrow: bilayer shrunken cell. Pink arrow: low density membrane.
We visualized E. coli treated with TP2-5 using cryo-electron tomography to investigate TP2-5-induced membrane disruption. Without treatment, the outer and inner membranes of E. coli minicells remained intact, and the distance between the outer and inner membranes was fixed (Fig. 2d–f). After treatment with TP2-5, dose-dependent morphological changes were observed. At lower concentrations (1/4× MIC, Figs. 2g and 1/2× MIC, Fig. 2h), membrane thinning with irregular holes on the outer membrane were observed. The minicells also showed blister formations protruding from the outer membrane, likely due to a membrane-thinning effect caused by TP2-5. In addition, small lipid clusters were present in close proximity to the outer and inner membranes, suggesting lipids had been removed from the membrane and collapsed into lipid clusters.
At higher TP2-5 concentrations (1× MIC, Fig. 2i, j), lipid clusters, holes and blisters on the outer membrane were apparent, along with shrunken minicells with double membranes. When the concentration of TP2-5 was increased to 5× or 10× MIC (Fig. 2k, l), some residual minicells were found to be trapped in the lipid clusters. Five hundred tomograms were collected and analyzed to understand these morphological changes (Fig. S4a, b).
The results showed that pore formation, low-density inner membranes and lipid clusters were frequently observed at a low TP2-5 concentration (1/4× MIC, 4 μg/mL). Although monotherapy with sub-MIC TP2-5 was ineffective at eliminating bacteria, the membrane remodeling observed at this dose was temporally associated with the synergistic activity observed in combination treatment. At higher TP2-5 concentrations (1× MIC to 10× MIC), shrunken minicells, lipid clusters with or without residual minicells were observed. Three-dimensional reconstructions of tomograms from TP2-5-treated minicells were generated using the back-projection method (Fig. S5, Supplementary Movies 1 and 2). The resulting images clearly showed membrane disruption at multiple sites on cells after TP2-5 treatment.
TP2-5-induced inner-membrane hyperpolarization is temporally associated with antibiotic sensitization
We performed real-time kinetic assays using sequential addition of 1/4× MIC TP2-5 and 1/4× MIC antibiotics to understand how TP2-5 and antibiotics interact dynamically at the outer and inner membranes (Fig. 3a, b). First, outer membrane permeability was assessed using NPN dye (Fig. 3c–f) with TP2-5 or antibiotics added at t = 20 and t = 40 min. At t = 20 min, TP2-5 alone (red line) induced a slight increase in NPN fluorescence, whereas MEM (Fig. 3c), VA (Fig. 3e) and CIP (Fig. 3f) added alone at t = 20 min (blue curves) failed to alter NPN signal. By contrast, COL alone (blue line) caused a dramatic and immediate increase in fluorescence (Fig. 3d). For the TP2-5 + Antibiotic group, addition of MEM, VA, or CIP at t = 40 min did not cause any further increase in NPN signal, resulting in a similar result to TP2-5 alone. Thus, under these conditions, outer membrane permeabilization is primarily attributable to TP2-5 or colistin, not to MEM, VA, or CIP. For inner-membrane potential, we used DiBAC₄(3) with the same t = 20 and t = 40 sequential design (Fig. 3a). When TP2-5 was added first at t = 20 min, DiBAC₄(3) fluorescence immediately decreased, indicating hyperpolarization (Fig. 3g–j, red curves). Subsequent addition of MEM, COL, VA, or CIP at t = 40 min caused a rapid increase in DiBAC₄(3) fluorescence from hyperpolarization, consistent with depolarization. Conversely, when MEM, COL, VA or CIP were added at t = 20 min, DiBAC₄(3) signal remained essentially unchanged relative to the PBS control (Fig. 3g–j, blue curves). Only after addition of TP2-5 at t = 40 min did fluorescence change, reflecting the intrinsic ability of TP2-5, rather than the antibiotics, to cause membrane potential change. These kinetics suggest that MDR E. coli treated with sub-MIC TP2-5 enters a relatively stable hyperpolarized state that is temporally associated with rapid membrane collapse upon subsequent antibiotic exposure. Whether this hyperpolarized state is mechanistically required for observed synergy or represents a correlated biophysical response remains to be established by functional perturbation experiments.

Schematic of sequential addition experimental design for NPN/DiBAC₄(3) (a) and PI (b) assays. MDR E. coli BAA 2452 were incubated with different probes at 37 °C for 20 min in (a). Three aliquots of cultures were then treated with TP2-5 (1/4× MIC), antibiotics (1/4× MIC) and PBS. Dynamic curves of the permeability of the outer membrane probed with NPN (c–f), membrane potential of the inner membrane probed with DiBAC4(3)(g–j), and permeability of the inner membrane probed with PI (k–n). All experiments were performed as three biologically independent experiments.
We then monitored inner membrane permeability using propidium iodide (PI). In the TP2-5 + antibiotic group (red line), addition of TP2-5 at t = 0 min caused PI fluorescence to rise rapidly and remain elevated, indicating that 1/4× TP2-5 alone is sufficient to create permeable lesions in the inner membrane (Fig. 3k-n, red curves). Subsequent addition of MEM, or VA at t = 20 min did not appreciably increase PI signal, whereas COL and CIP caused a further modest rise. Conversely, in the antibiotic + TP2-5 group (blue line), when antibiotics were added first at t = 0 min, none of the four drugs produced any signal change relative to the PBS control (green line) from 0 to 20 min (Fig. 3k–n). Only upon addition of TP2-5 at t = 20 min did PI fluorescence abruptly increase. Under the sub-MIC (1/4× MIC) used for synergy, only addition of TP2-5 induced PI uptake, whereas antibiotics alone caused little change, indicating that TP2-5 is the primary contributor to inner-membrane permeabilization.
TP2-5 targets LPS and peptidoglycan-associated anionic phospholipids
To identify the molecular targets underlying TP2-5-induced envelope remodeling, we examined different cell wall and membrane components on TP2-5 activity. The addition of exogenous LPS (LPS of E. coli O111:B4) caused a dose-dependent increase in the MIC of TP2-5 against MDR E. coli, indicating that LPS can bind to the peptide (Fig. 4a). Among phospholipids, pre-incubation with phosphatidylglycerol (POPG) markedly increased the MIC of TP2-5, whereas phosphatidylethanolamine (POPE) and phosphatidylcholine (POPC) showed negligible effects (Fig. 4b). These data indicate that TP2-5 preferentially targets anionic moieties enriched in both the outer (LPS) and inner (POPG) membrane.
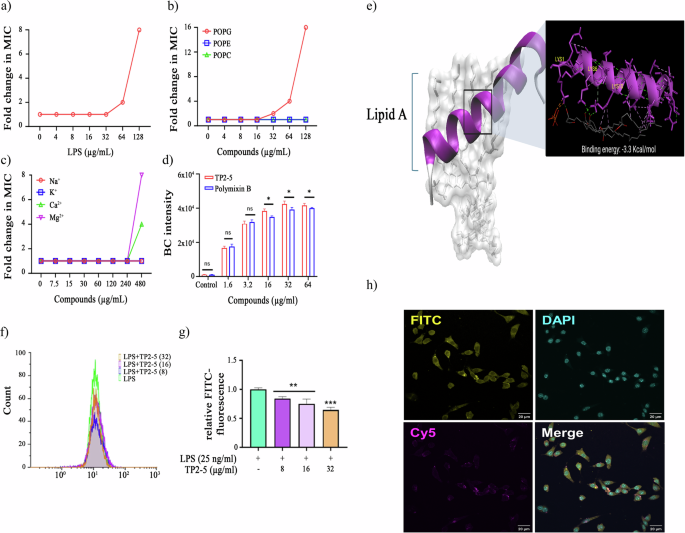
Antimicrobial activity of TP2-5 against MDR E. coli BAA 2452 in the presence of a LPS, b membrane phospholipids (POPG, POPE and POPC), and c physiological ions (Na+, K+, Ca2+, Mg2+). d Competitive displacement of BODIPY-TR-cadaverine (BC) dye attached to lipopolysaccharide (LPS) after incubation with TP2-5 and colistin. e Molecular docking analysis predicted binding of TP2-5 to LPS, with a calculated binding energy of -3.3 kcal/mol. f Flow cytometry analysis showing the effect of TP2-5 on the binding of 100 ng/mL FITC-LPS to RAW 264.7 cells. RAW 264.7 cells were treated with LPS (green), LPS + 8 μg/mL TP2-5 (purple), LPS + 16 μg/mL TP2-5 (pink), and LPS + 32 μg/mL TP2-5 (yellow). g The relative-FITC fluorescence intensity of FITC-LPS for different treatments. h Confocal images showing the binding of TP2-5 with LPS. RAW 264.7 cells were induced by 100 ng/mL FITC-LPS for 30 min, then treated with 16 μg/mL Cy5.5-TP2-5. Data are shown as the mean ± standard deviation of three replicates.
Cation competition assays showed that high concentrations (480 µg/mL) of divalent cations (Mg²⁺, Ca²⁺) suppressed TP2-5 activity, whereas monovalent cations (Na⁺, K⁺) had no significant effect (Fig. 4c). Given that divalent cations bridge lipid A phosphate groups and stabilize the LPS leaflet, these findings are consistent with TP2-5 interfering with this divalent-cation-mediated stabilization through electrostatic competition (Fig. 4c). The LPS interaction was further confirmed by BODIPY-TR-cadaverine displacement assay, in which TP2-5 efficiently displaced the fluorescent probe from LPS, although with a distinct binding profile compared to polymyxin B (Fig. 4d). Molecular docking simulations visualized and predicted TP2-5 along the lipid A–core region of LPS via forming multiple hydrogen bonds and hydrophobic interactions, with a calculated binding energy of -3.3 kcal/mol (Fig. 4e, f).
Finally, to understand the functional implications of TP2-5–LPS binding, we performed a cell-free neutralization assay. FITC-labeled LPS was co-incubated with TP2-5 and analyzed by flow cytometry. TP2-5 caused a concentration-dependent leftward shift of the FITC–LPS fluorescence together with a significant reduction in mean fluorescence intensity, indicating that TP2-5 directly binds and neutralizes LPS, thereby reducing the accessible FITC-LPS signal (Fig. 4f, g). Confocal microscopy further showed co-localization of TP2-5–Cy5.5 with FITC–LPS at the macrophage surface (Fig. 4h), suggesting that TP2-5 associates with LPS within receptor complexes. Collectively, these data support a dual-target model wherein TP2-5 targets both LPS in the outer membrane and peptidoglycan-associated anionic phospholipids in the inner membrane, thereby disrupting both the structural integrity and bioenergetics of the MDR Gram-negative bacteria.
TP2-5 modulates LPS-induced inflammatory responses in macrophages
Beyond its direct effects on the bacterial membrane, we investigated whether TP2-5 modulates LPS-induced inflammatory signaling. Flow cytometry analysis revealed a direct interaction on the surface of LPS-stimulated RAW 264.7 cells, showing that TP2-5 dose-dependently inhibited the binding of FITC-conjugated LPS to macrophages and also interacted with pre-bound LPS on the cell surface (Fig. 5a, b). This surface-level neutralization of LPS translated into a potent suppression of downstream inflammatory signaling. TP2-5 significantly attenuated LPS-induced nitrite accumulation at 8 and 16 μg/mL; however, a slight relative increase was observed at higher concentration (32 and 64 μg/mL), which may reflect sublethal cellular stress at these doses rather than direct cytotoxicity, as these concentrations remain well below the IC₅₀ for RAW 264.7 cells (309 μg/mL) (Fig. 5c). Western blot showed that TP2-5 attenuated LPS-induced phosphorylation of p38 and c-Jun, and suppressed the expression of key inflammatory proteins, including iNOS, COX-2, IL-6, and TNF-α. Notably, the expression of the upstream adaptor protein MyD88 was unaffected, suggesting that TP2-5 acts downstream of this initial signaling event (Fig. 5d-k).
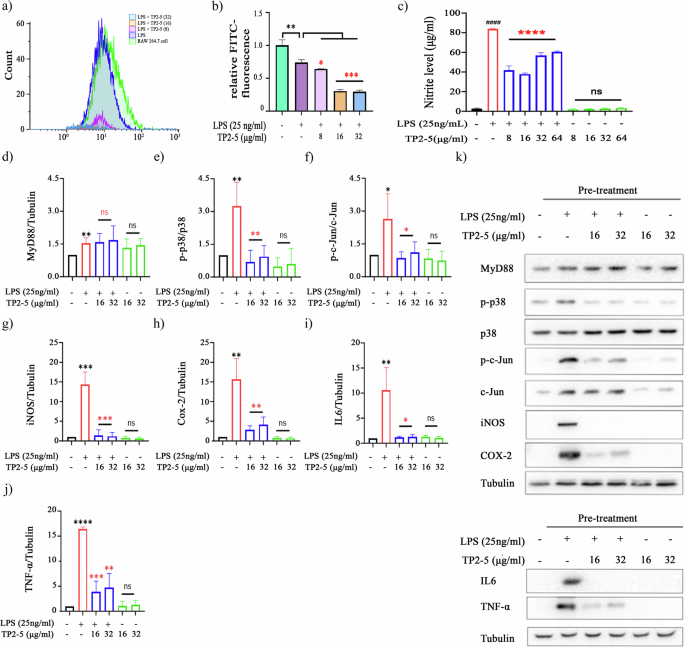
a, b Flow cytometry analysis showing the effects of pre-treated TP2-5 on FITC-LPS on RAW 264.7 cells induced with 25 ng/mL LPS. RAW 264.7 cells without LPS induction (green) or induced by LPS (purple); cells pretreated with 8 μg/mL TP2-5 (pink), 16 μg/mL TP2-5 (yellow), and 32 μg/mL TP2-5 (blue). The cells were then stimulated by LPS. c TP2-5 caused a dose-dependent reduction of LPS-induced nitrite levels at 8 and 16 μg/mL; a slight increase was observed at 32 and 64 μg/mL. d MyD88, e p38, f c-JUN, g iNOS, h COX-2, i IL6, and j TNF-α protein expression levels, quantified from western blot analysis and normalized to α-tubulin, showing relative expression compared to LPS-stimulated control. RAW 264.7 cells were treated with 16 μg/mL TP2-5 followed by LPS (25 ng/mL, 30 min) stimulation, and the k) protein levels were normalized to α-tubulin as determined by western blot analysis.
To assess the global impact of TP2-5 on the inflammatory transcriptome, we performed RNA-seq analysis on LPS-activated macrophages with or without TP2-5 treatment. This revealed a broad suppression of LPS-induced proinflammatory genes, such as Rsad2[27], Mx1 [28], Cmpk2 [20], CXCl10 [29], CXCl11 [30], and Fcgr1 [31]. Interestingly, we also noted the upregulation of Mmp9 [34], a gene with complex roles in LPS-mediated inflammation (Fig. S6a, b). Despite this, the overall transcriptomic profile was clearly anti-inflammatory. Gene Ontology (GO) and pathway enrichment analyses confirmed that TP2-5 significantly downregulated biological processes and molecular pathways related to defense response, leukocyte migration, and cytokine activity (Fig. S6c, d, Fig. S7). Collectively, these findings indicate that TP2-5 not only neutralizes LPS at the membrane but also inhibits the downstream inflammatory transcriptional program, which may contribute to its protective effects in vivo.
Safety and toxicological evaluation of TP2-5 in vitro and in vivo
TP2-5 exhibited limited activity against Gram-positive wild-type and drug-resistant strains (Table 1), with MICs of 128 μg/mL for all tested strains except for Staphylococcus epidermidis. In contrast, TP2-5 demonstrated greater efficacy against Gram-negative bacteria, with MIC values ranging from 4 to 16 μg/mL for most tested strains, and 32 μg/mL for Pseudomonas aeruginosa isolates. To assess the safety and selectivity of TP2-5, the selectivity index (SI; ratio of IC₅₀ to MIC) was calculated for four mammalian cell lines (Fig. 6a–d, Table 1).

Cytotoxicity (IC50) of TP2-5 against a HaCaT, 267 µg/mL; b RAW 264.7, 309 µg/mL; c A549, 194 µg/mL; and d) HepG2, 590 µg/mL. In vivo toxicity of TP2-5 by e–j intravenous or k–r intratracheal administration in C57BL/6 female mice. e Change of body weight in mouse models at the indicated dose and f Percentage of surviving test animals at doses ranging from 0.78 mg/kg to 15 mg/kg. Data are shown as mean ± standard deviation of 6 animals per group. Kidney function was assessed by measuring g blood urea nitrogen (BUN) and h creatinine (CRE), and liver function by measuring i GOT and j GPT following intravenous TP2-5 administration. Intra-tracheal toxicity of TP2-5. Negligible toxicity was observed at doses up to 20 mg/kg (n = 6). k Body weight was unchanged for 5 days. The blood cell counts l–n were similar between untreated healthy controls and treated animals (n = 5). The kidney function (BUN, o) and (CRE, p) and liver function (GOT, q) and (GPT, r) showed no indication of change and were comparable between treated animals and untreated controls. Data are shown as mean ± standard deviation.
Human keratinocytes (HaCaT, Fig. 6a), mouse macrophages (RAW 264.7, Fig. 6b), A549 cells (Fig. 6c) and HepG2 cells (Fig. 6d) were treated with TP2-5. The concentrations causing 50% cytotoxicity (IC50) were as follows for each cell line: HaCaT, 267 µg/mL; RAW 264.7, 309 µg/mL; A549, 194 µg/mL; and HepG2, 590 µg/mL. Based on these data, the SI was calculated. TP2-5 showed highly favorable SI values against Gram-negative MDR strains, ranging from 6.1 to 147.5 (Table 1), while values against Gram-positive strains were lower (1.5–18.4). These results indicate a preferable biocompatibility profile, particularly for treating Gram-negative bacterial infections.
To evaluate the in vivo safety of TP2-5, female C57BL/6 mice received a single intravenous administration of TP2-5 up to 12.5 mg/kg. Over a 7-day observation period, no significant changes in body weight or survival were noted (Fig. 6e, f). Furthermore, analysis of blood samples revealed that biomarkers for hepatic function (GOT, GPT) in TP2-5-treated mice were comparable to those of the saline-treated control group (Fig. 6i, j). Regarding renal function, while a minor statistically significant variation in CRE was observed at low doses in healthy mice (Fig. 6h), all values including BUN (Fig. 6g) remained within the established physiological baseline for this strain. Notably, this transient change was not observed in the subsequent CLP sepsis model (Fig. 7m) and did not correlate with any signs of systemic toxicity, suggesting that the observed CRE levels reflect individual animal differences rather than functional renal impairment.

a Schematic representation of the MDR A. baumannii 2088 lung infection and polymicrobial sepsis caused by the cecal ligation and puncture (CLP) models. Both models were mitigated by TP2-5 treatment. Readouts for the lung infection model included bacterial (MDR A. baumannii 2088) loads in b bronchoalveolar lavage fluid (BALF), c lungs, and d liver following TP2-5 treatment. Data are shown as mean ± standard deviation of 4 animals per group. e Similarly, TP2-5 treatment led to lower nitrite levels in the lung infection model, indicating less severe infection or promotion of healing. f Percent survival of CLP-induced polymicrobial sepsis mice treated with TP2-5 and antibiotics (MEM, meropenem and COL, colistin) alone or in combination (n = 8). TP2-5 treatment alone significantly reduced bacterial loads in g peritoneal fluid, h lungs, i liver, j kidney, and k spleen. Kidney and liver injury markers were improved in TP2-5-treated groups, as reflected by l BUN, m CRE, n GOT, and o GPT. TP2-5 alone or in combination with antibiotics significantly reduced p IL6 and q TNF-α levels. Data are shown as mean ± standard deviation of 5 animals per group.
To determine the in vivo biodistribution of TP2-5, the peptide was conjugated with Cy5.5 and administered intravenously to mice. In vivo fluorescence imaging (IVIS) revealed that the peptide’s signal peaked within 6 h post-administration and subsequently declined over a 72-h period (Fig. S8a). Quantitative analysis of regions of interest (ROIs) showed that TP2-5 accumulated most significantly in the liver, with detectable but lower levels observed in the kidney, heart, lungs, and spleen (Fig. S8). These data support the use of TP2-5 at doses sufficient for adjuvant activity without unacceptable toxicity.
TP2-5 demonstrates in vivo efficacy and antibiotic synergy in a Galleria mellonella infection model
The in vivo efficacy of TP2-5 was evaluated in a Galleria mellonella model of MDR E. coli BAA 2452 infection. In the infection model, larvae challenged with the pathogen alone showed fewer than 20% survival over 5 days (Fig. S9). While sub-MIC concentrations of antibiotics alone provided limited or partial protection, the addition of sub-MIC TP2-5 consistently and significantly improved survival outcomes across all four antibiotic classes tested. Specifically, combining TP2-5 with either colistin or vancomycin resulted in 100% survival of the infected larvae (Fig. S9c, d). Moreover, TP2-5 restored the efficacy of ciprofloxacin, increasing survival from 40% to 80% (Fig. S9d), and markedly enhanced the protective effect of meropenem, boosting survival to 90% in the combination group (1/2× MIC TP2-5 + 1/4× MIC MEM) (Fig. S9b).
Combination of TP2-5 and antibiotics improves outcomes in MDR pneumonia and polymicrobial sepsis
The therapeutic efficacy of TP2-5 was first assessed in a murine model of pneumonia induced by MDR Acinetobacter baumannii 2088. A single 8 mg/kg dose of TP2-5 administered post-infection resulted in a significant, approximately 3-log reduction in the pulmonary bacterial burden compared to the untreated group (Fig. 7c). While bacterial loads in the bronchoalveolar lavage fluid (BALF) and liver showed a non-significant trend towards reduction (Fig. 7b, d), the potent local effect in the lungs was clear. Consistent with the reduction in bacterial load, a trend toward lower nitrite levels, an indicator of inflammation, was also observed (p = 0.08, Fig. 7e).
To evaluate TP2-5 in a more severe systemic infection, we used the cecal ligation and puncture (CLP) model of polymicrobial sepsis. TP2-5 monotherapy (2 mg/kg) achieved 100% survival over the 7-day study, compared to 62.5% for meropenem and 75% for colistin (Fig. 7f). This result suggests that the in vivo efficacy of TP2-5 is multifunctional, likely reflecting contributions from both direct antibacterial activity and potent host-directed immunomodulatory effects, including LPS neutralization and cytokine reduction, rather than antibacterial activity alone. Co-administration of TP2-5 with meropenem also resulted in 100% survival over the same period (Fig. 7f), consistent with synergistic in vivo activity. Besides, co-administration of TP2-5 with colistin resulted in 90% survival (Fig. 7f), which was numerically lower than TP2-5 monotherapy but higher than colistin alone (75%) and did not reach statistical significance compared to the TP2-5 alone group, suggesting that this difference reflects individual animal variability rather than a true antagonistic interaction.
This survival benefit was underpinned by robust bacterial clearance. TP2-5, both alone and in combination with antibiotics, significantly reduced the bacterial load in the peritoneal fluid and systemically cleared bacteria from multiple vital organs, including the lungs, liver, kidneys, and spleen (Fig. 7g–k). This effective infection control led to significant organ protection, as evidenced by the normalization of biomarkers for renal function (BUN, CRE) and hepatic function (GOT, GPT) in TP2-5-treated groups (Fig. 7l–o). Furthermore, TP2-5 treatment profoundly suppressed the systemic levels of key proinflammatory cytokines IL-6 and TNF-α, confirming its potent immunomodulatory activity in vivo (Fig. 7p–q).

















Leave a Reply