Lopez-Otin, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. Hallmarks of aging: an expanding universe. Cell 186, 243–278 (2023).
Kousa, A. I. et al. Age-related epithelial defects limit thymic function and regeneration. Nat. Immunol. 25, 1593–1606 (2024).
de Magalhães, J. P. Cellular senescence in normal physiology. Science 384, 1300–1301 (2024).
Phan, H. V. et al. Host-microbe multiomic profiling reveals age-dependent immune dysregulation associated with COVID-19 immunopathology. Sci. Transl. Med. 16, eadj5154 (2024).
Dallan, B. et al. Age differentially impacts adaptive immune responses induced by adenoviral versus mRNA vaccines against COVID-19. Nat. Aging 4, 1121–1136 (2024).
Jiang, G., Zou, Y., Zhao, D. & Yu, J. Optimising vaccine immunogenicity in ageing populations: key strategies. Lancet Infect. Dis. 25, e23–e33 (2025).
Zhivaki, D. et al. Correction of age-associated defects in dendritic cells enables CD4(+) T cells to eradicate tumors. Cell 187, 3888–3903 e3818 (2024).
Wu, R., Sun, F., Zhang, W., Ren, J. & Liu, G. H. Targeting aging and age-related diseases with vaccines. Nat. Aging 4, 464–482 (2024).
Palacios-Pedrero, M. A. et al. Signs of immunosenescence correlate with poor outcome of mRNA COVID-19 vaccination in older adults. Nat. Aging 2, 896–905 (2022).
Cobey, S. Vaccination against rapidly evolving pathogens and the entanglements of memory. Nat. Immunol. 25, 2015–2023 (2024).
Inoue, T. & Kurosaki, T. Memory B cells. Nat. Rev. Immunol. 24, 5–17 (2024).
van Dorst, M. et al. Immunological factors linked to geographical variation in vaccine responses. Nat. Rev. Immunol. 24, 250–263 (2024).
Jo, N. et al. Impaired CD4(+) T cell response in older adults is associated with reduced immunogenicity and reactogenicity of mRNA COVID-19 vaccination. Nat. Aging 3, 82–92 (2023).
Silva-Cayetano, A. et al. Spatial dysregulation of T follicular helper cells impairs vaccine responses in aging. Nat. Immunol. 24, 1124–1137 (2023).
Xiao, C. et al. Insufficient epitope-specific T cell clones are responsible for impaired cellular immunity to inactivated SARS-CoV-2 vaccine in older adults. Nat. Aging 3, 418–435 (2023).
Bartleson, J. M. et al. SARS-CoV-2, COVID-19 and the ageing immune system. Nat. Aging 1, 769–782 (2021).
Binnewies, M. et al. Unleashing type-2 dendritic cells to drive protective antitumor CD4(+) T cell immunity. Cell 177, 556–571 e516 (2019).
Yin, X., Chen, S. & Eisenbarth, S. C. Dendritic cell regulation of T helper cells. Annu. Rev. Immunol. 39, 759–790 (2021).
Aw, D., Silva, A. B. & Palmer, D. B. Immunosenescence: emerging challenges for an ageing population. Immunology 120, 435–446 (2007).
Chen, J., Deng, J. C. & Goldstein, D. R. How aging impacts vaccine efficacy: known molecular and cellular mechanisms and future directions. Trends Mol. Med. 28, 1100–1111 (2022).
Carrasco, E. et al. The role of T cells in age-related diseases. Nat. Rev. Immunol. 22, 97–111 (2022).
Amon, L. et al. Guidelines for DC preparation and flow cytometry analysis of mouse lymphohematopoietic tissues. Eur. J. Immunol. 53, e2249893 (2023).
Ginhoux, F., Guilliams, M. & Merad, M. Expanding dendritic cell nomenclature in the single-cell era. Nat. Rev. Immunol. 22, 67–68 (2022).
Lopez, L. et al. Dendritic cell-targeted therapy expands CD8 T cell responses to bona-fide neoantigens in lung tumors. Nat. Commun. 15, 2280 (2024).
Jin, Y., Li, P. & Wang, F. β-glucans as potential immunoadjuvants: a review on the adjuvanticity, structure-activity relationship and receptor recognition properties. Vaccine 36, 5235–5244 (2018).
Xu, J. et al. Yeast-derived nanoparticles remodel the immunosuppressive microenvironment in tumor and tumor-draining lymph nodes to suppress tumor growth. Nat. Commun. 13, 110 (2022).
Powell, N., Walker, M. M. & Talley, N. J. The mucosal immune system: master regulator of bidirectional gut-brain communications. Nat. Rev. Gastroenterol. Hepatol. 14, 143–159 (2017).
Agirman, G., Yu, K. B. & Hsiao, E. Y. Signaling inflammation across the gut-brain axis. Science 374, 1087–1092 (2021).
Chougnet, C. A. et al. Loss of phagocytic and antigen cross-presenting capacity in aging dendritic cells is associated with mitochondrial dysfunction. J. Immunol. 195, 2624–2632 (2015).
Grolleau-Julius, A., Harning, E. K., Abernathy, L. M. & Yung, R. L. Impaired dendritic cell function in aging leads to defective antitumor immunity. Cancer Res. 68, 6341–6349 (2008).
Granot, T. et al. Dendritic cells display subset and tissue-specific maturation dynamics over human life. Immunity 46, 504–515 (2017).
Förster, R., Davalos-Misslitz, A. C. & Rot, A. CCR7 and its ligands: balancing immunity and tolerance. Nat. Rev. Immunol. 8, 362–371 (2008).
Eckert, N., Permanyer, M., Yu, K., Werth, K. & Forster, R. Chemokines and other mediators in the development and functional organization of lymph nodes. Immunol. Rev. 289, 62–83 (2019).
Song, J. H. et al. CCR7-CCL19/CCL21-regulated dendritic cells are responsible for effectiveness of sublingual vaccination. J. Immunol. 182, 6851–6860 (2009).
Choi, Y. et al. Immune checkpoint blockade induces gut microbiota translocation that augments extraintestinal antitumor immunity. Sci. Immunol. 8, eabo2003 (2023).
Krebs, C. F. et al. Autoimmune renal disease is exacerbated by S1P-receptor-1-dependent intestinal Th17 cell migration to the kidney. Immunity 45, 1078–1092 (2016).
Tatsumi, N., El-Fenej, J., Davila-Pagan, A. & Kumamoto, Y. CD301b(+) dendritic cell-derived IL-2 dictates CD4(+) T helper cell differentiation. Nat. Commun. 16, 2002 (2025).
Goronzy, J. J. & Weyand, C. M. Understanding immunosenescence to improve responses to vaccines. Nat. Immunol. 14, 428–436 (2013).
Gustafson, C. E., Kim, C., Weyand, C. M. & Goronzy, J. J. Influence of immune aging on vaccine responses. J. Allergy Clin. Immunol. 145, 1309–1321 (2020).
Zhang, X., Yang, B., Ni, Q. & Chen, X. Materials engineering strategies for cancer vaccine adjuvant development. Chem. Soc. Rev. 52, 2886–2910 (2023).
Shi, S. et al. Vaccine adjuvants: understanding the structure and mechanism of adjuvanticity. Vaccine 37, 3167–3178 (2019).
Pereira, B., Xu, X. N. & Akbar, A. N. Targeting inflammation and immunosenescence to improve vaccine responses in the elderly. Front. Immunol. 11, 583019 (2020).
Sanmarco, L. M. et al. Lactate limits CNS autoimmunity by stabilizing HIF-1α in dendritic cells. Nature 620, 881–889 (2023).
Agrawal, A. et al. Altered innate immune functioning of dendritic cells in elderly humans: a role of phosphoinositide 3-kinase-signaling pathway. J. Immunol. 178, 6912–6922 (2007).
Agrawal, A. & Gupta, S. Impact of aging on dendritic cell functions in humans. Ageing Res. Rev. 10, 336–345 (2011).
Shaw, A. C., Goldstein, D. R. & Montgomery, R. R. Age-dependent dysregulation of innate immunity. Nat. Rev. Immunol. 13, 875–887 (2013).
Wong, C. & Goldstein, D. R. Impact of aging on antigen presentation cell function of dendritic cells. Curr. Opin. Immunol. 25, 535–541 (2013).
Salminen, A. Immunosuppressive network promotes immunosenescence associated with aging and chronic inflammatory conditions. J. Mol. Med. 99, 1553–1569 (2021).
Reitsema, R. D., Kumawat, A. K., Hesselink, B. C., van Baarle, D. & van Sleen, Y. Effects of ageing and frailty on circulating monocyte and dendritic cell subsets. NPJ Aging 10, 17 (2024).
Othman, R. A., Moghadasian, M. H. & Jones, P. J. Cholesterol-lowering effects of oat β-glucan. Nutr. Rev. 69, 299–309 (2011).
Xu, Y. P. et al. Aging affects epidermal Langerhans cell development and function and alters their miRNA gene expression profile. Aging 4, 742–754 (2012).
Grewe, M. Chronological ageing and photoageing of dendritic cells. Clin. Exp. Dermatol. 26, 608–612 (2001).
Tang, H. et al. Rescuing dendritic cell interstitial motility sustains antitumour immunity. Nature 645, 244–253 (2025).
Lobel, G. P. et al. Glutamine is critical for the maintenance of type 1 conventional dendritic cells in normal tissue and the tumor microenvironment. Proc. Natl. Acad. Sci. USA 121, e2412157121 (2024).





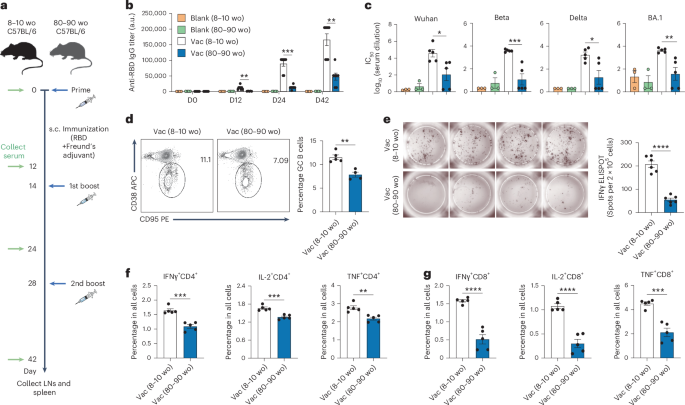









Leave a Reply