Nicklas, T. A. et al. Nutrient intake, introduction of baby cereals and other complementary foods in the diets of infants and toddlers from birth to 23 months of age. AIMS Public Health 7, 123 (2020).
China Nutrition and Health Food Association. China Infant And Toddler Complementary Food Industry Development Report (2020) CNHFA (Beijing, 2022).
Moroishi, Y. et al. Infant infections, respiratory symptoms, and allergy in relation to timing of rice cereal introduction in a United States cohort. Sci. Rep. 12, 4450 (2022).
Anania, C. et al. Hydrolyzed rice formula: an appropriate choice for the treatment of cow’s milk allergy. J. Clin. Med. 11, 4823 (2022).
Pantazi, A. C. et al. Relationship between gut microbiota and allergies in children: a literature review. Nutrients 15, 2529 (2023).
Anvari, S., Miller, J., Yeh, C. Y. & Davis, C. M. IgE-mediated food allergy. Clin. Rev. Allergy Immunol. 57, 244–260 (2019).
Zhang, S., Sicherer, S., Berin, M. C. & Agyemang, A. Pathophysiology of non-IgE-mediated food allergy. ImmunoTargets Ther. 10, 431–446 (2021).
Ferreira, F., Hawranek, T., Gruber, P., Wopfner, N. & Mari, A. Allergic cross-reactivity: from gene to the clinic. Allergy 59, 243–267 (2004).
Hu, W. et al. Identification and validation of key amino acids in IgE linear epitopes of β-lactoglobulin: comparison of recognition patterns of Chinese bovine milk-allergic sera with different symptoms. J. Agric Food Chem. 73, 5537–5547 (2025).
Florsheim, E. B. et al. Immune sensing of food allergens promotes avoidance behaviour. Nature 620, 643–650 (2023).
Lin, Y. J., Zimmermann, J. & Schülke, S. Novel adjuvants in allergen-specific immunotherapy: where do we stand? Front. Immunol. 15, 1348305 (2024).
Ong, G. H., Lian, B. S. X., Kawasaki, T. & Kawai, T. Exploration of pattern recognition receptor agonists as candidate adjuvants. Front. Cell Infect. Microbiol. 11, 745016 (2021).
Batista Duharte, A., Téllez Martínez, D., Fuentes, D. L. P. & Carlos, I. Z. Molecular adjuvants that modulate regulatory T cell function in vaccination: a critical appraisal. Pharm. Res. 129, 237–250 (2018).
Pifferi, C., Fuentes, R. & Fernández Tejada, A. Natural and synthetic carbohydrate-based vaccine adjuvants and their mechanisms of action. Nat. Rev. Chem. 5, 197–216 (2021).
Ou, B., Yang, Y., Lv, H., Lin, X. & Zhang, M. Current progress and challenges in the study of adjuvants for oral vaccines. BioDrugs 37, 143–180 (2023).
Mathias, F. A. S. et al. The use of an adjuvant system improves innate and adaptive immune response when associated with a Leishmania (viannia) braziliensis antigen in a vaccine candidate against L. (Leishmania) infantum infection. Vaccines 11, 395 (2023).
Sun, N. et al. Allergic reactions were compared between BN and Wistar rats after oral exposure to ovalbumin. J. Immunotoxicol. 10, 67–74 (2013).
Yang, X. et al. Antiallergic activity of Lactobacillus plantarum against peanut allergy in a balb/c mouse model. Food Agric. Immunol. 30, 762–773 (2019).
Santoro, D. & Marsella, R. Animal models of allergic diseases. Vet. Sci. 1, 192–212 (2014).
Burton, O. T. et al. A humanized mouse model of anaphylactic peanut allergy. J. Allergy Clin. Immunol. 139, 314–322.e319 (2017).
Ahuja, V. et al. Evaluation of biotechnology-derived novel proteins for the risk of food-allergic potential: advances in the development of animal models and future challenges. Arch. Toxicol. 84, 909–917 (2010).
Van Gramberg, J. L., De Veer, M. J., O’Hehir, R. E., Meeusen, E. N. T. & Bischof, R. J. Use of animal models to investigate major allergens associated with food allergy. J. Allergy 2013, 1–10 (2013).
Chen, C. Y. et al. Induction of interleukin-9-producing mucosal mast cells promotes susceptibility to IgE-mediated experimental food allergy. Immunity 43, 788–802 (2015).
Smith, N. A. & Nagamoto Combs, K. Induction of hypersensitivity with purified beta-lactoglobulin as a mouse model of cow’s milk allergy. Methods Mol. Biol. 2223, 67–78 (2021).
Kotzé Hörstmann, L. et al. Characterization and comparison of the divergent metabolic consequences of high-sugar and high-fat diets in male Wistar rats. Front. Physiol. 13, 904366 (2022).
Thakur, V. R., Khuman, V., Beladiya, J. V., Chaudagar, K. K. & Mehta, A. A. An experimental model of asthma in rats using ovalbumin and lipopolysaccharide allergens. Heliyon 5, e02864 (2019).
Xie, A. et al. Influence of diet on the effect of the probiotic lactobacillus paracasei in rats suffering from allergic asthma. Front. Microbiol. 12, 737622 (2021).
Abril Gil, M., Garcia Just, A., Pérez Cano, F. J., Franch, À. & Castell, M. Development and characterization of an effective food allergy model in brown norway rats. PLoS ONE 10, e0125314 (2015).
Reyes Pavón, D. et al. Protective effect of glycomacropeptide on food allergy with gastrointestinal manifestations in a rat model through down-regulation of type 2 immune response. Nutrients 12, 2942 (2020).
Keddar, K. et al. Probiotic bacteria from human milk can alleviate oral bovine casein sensitization in juvenile Wistar rats. Microorganisms 11, 1030 (2023).
Mazhar, A. et al. Development of tolerogenic casein encapsulated quercetin and curcumin nanoparticles to mitigate cow milk allergic responses. Int. J. Biol. Macromol. 314, 144396 (2025).
Noviski, M. et al. IgM and IgD B cell receptors differentially respond to endogenous antigens and control B cell fate. Elife 7, e35074 (2018).
Jin, Y. et al. Mechanisms of wheat allergenicity in mice: comparison of adjuvant-free vs. Alum-adjuvant models. Int. J. Mol. Sci. 21, 3205 (2020).
Aguilar Pimentel, J. A. et al. Specific CD8 T cells in IgE-mediated allergy correlate with allergen dose and allergic phenotype. Am. J. Respir. Crit. Care Med 181, 7–16 (2010).
Plum, T. et al. Mast cells link immune sensing to antigen-avoidance behaviour. Nature 620, 634–642 (2023).
Duan, C. et al. Comparison of allergenicity among cow, goat, and horse milks using a murine model of atopy. Food Funct. 12, 5417–5428 (2021).
Bao, C. et al. A mast cell–thermoregulatory neuron circuit axis regulates hypothermia in anaphylaxis. Sci. Immunol. 8, eadc9417 (2023).
Ma, Y. et al. Olive oil ameliorates allergic response in murine ovalbumin-induced food allergy by promoting intestinal mucosal immunity. Food Sci. Hum. Wellness 12, 801–808 (2023).
Wongsaengchan, C., McCafferty, D. J., Evans, N. P., McKeegan, D. E. F. & Nager, R. G. Body surface temperature of rats reveals both magnitude and sex differences in the acute stress response. Physiol. Behav. 264, 114138 (2023).
Berin, M. C. & Sampson, H. A. Food allergy: an enigmatic epidemic. Trends Immunol. 34, 390–397 (2013).
Mantis, N. J., Rol, N. & Corthésy, B. Secretory IgA’s complex roles in immunity and mucosal homeostasis in the gut. Mucosal Immunol. 4, 603–611 (2011).
Di Lorenzo, F. et al. Pairing bacteroides vulgatus LPS structure with its immunomodulatory effects on human cellular models. ACS Cent. Sci. 6, 1602–1616 (2020).
Li, Y. et al. Sesamin is an effective spleen tyrosine kinase inhibitor against IgE-mediated food allergy in computational, cell-based and animal studies. Food Sci. Hum. Wellness 14, 9250081 (2025).
Stierschneider, A. & Wiesner, C. Shedding light on the molecular and regulatory mechanisms of TLR4 signaling in endothelial cells under physiological and inflamed conditions. Front. Immunol. 14, 1264889 (2023).
Zhang, Q., Yu, X., Tian, L., Cong, Y. & Li, L. Therapeutic effects of epigallocatechin and epigallocatechin gallate on the allergic reaction of s1-casein sensitized mice. Food Sci. Hum. Wellness 12, 882–888 (2023).
Li, L., Zhang, X. H., Liu, G. R., Liu, C. & Dong, Y. M. Isoquercitrin suppresses the expression of histamine and pro-inflammatory cytokines by inhibiting the activation of MAP kinases and NF-κB in human KU812 cells. Chin. J. Nat. Med. 14, 407–412 (2016).
Odenwald, M. A. & Turner, J. R. The intestinal epithelial barrier: a therapeutic target? Nat. Rev. Gastroenterol. Hepatol. 14, 9–21 (2017).
Shamji, M. H. et al. The role of allergen-specific IgE, IgG and IgA in allergic disease. Allergy 76, 3627–3641 (2021).
Seikrit, C. & Pabst, O. The immune landscape of IgA induction in the gut. Semin. Immunopathol. 43, 627–637 (2021).
Liu, J. et al. Role of the IgM fc receptor in immunity and tolerance. Front. Immunol. 10, 529 (2019).
Corthésy, B. Multi-faceted functions of secretory IgA at mucosal surfaces. Front. Immunol. 4, 185 (2013).
Smolinska, S., Jutel, M., Crameri, R. & O’Mahony, L. Histamine and gut mucosal immune regulation. Allergy 69, 273–281 (2014).
Ricklin, D., Reis, E. S., Mastellos, D. C., Gros, P. & Lambris, J. D. Complement component C3 – the “swiss army knife” of innate immunity and host defense. Immunol. Rev. 274, 33–58 (2016).
Wang, H. & Liu, M. Complement C4, infections, and autoimmune diseases. Front. Immunol. 12, 694928 (2021).
Kihl, P. et al. Oral LPS dosing induces local immunological changes in the pancreatic lymph nodes in mice. J. Diabetes Res. 2019, 1–9 (2019).
Mizobuchi, H. Oral route lipopolysaccharide as a potential dementia preventive agent inducing neuroprotective microglia. Front Immunol. 14, 1110583 (2023).
Inagawa, H., Kohchi, C. & Soma, G. I. Oral administration of lipopolysaccharides for the prevention of various diseases: benefit and usefulness. Anticancer Res. 31, 2431–2436 (2011).
Lasselin, J. et al. Comparison of bacterial lipopolysaccharide-induced sickness behavior in rodents and humans: relevance for symptoms of anxiety and depression. Neurosci. Biobehav Rev. 115, 15–24 (2020).
Harvei, S. et al. Chronic oral LPS administration does not increase inflammation or induce metabolic dysregulation in mice fed a western-style diet. Front. Nutr. 11, 1376493 (2024).
Mota, C. M. D. & Madden, C. J. Neural control of the spleen as an effector of immune responses to inflammation: mechanisms and treatments. Am. J. Physiol. Regul. Integr. Comp. Physiol. 323, R375–R384 (2022).
Kamath, S. D. et al. Cross-reactive epitopes and their role in food allergy. J. Allergy Clin. Immunol. 151, 1178–1190 (2023).
Sathya, P. & Fenton, T. R. Cow’s milk protein allergy in infants and children. Paediatr. Child Health 29, 382–388 (2024).
Bani, C. et al. Unexpected cow’s milk proteins in a “vegan” easter egg as a cause of anaphylaxis. Foods 14, 1737 (2025).
Cardona, V. et al. World Allergy Organization Anaphylaxis Guidance 2020. World Allergy Organ J. 13, 100472 (2020).
Mishra, A., Gaur, S. N., Singh, B. P. & Arora, N. In silico assessment of the potential allergenicity of transgenes used for the development of GM food crops. Food Chem. Toxicol. 50, 1334–1339 (2012).





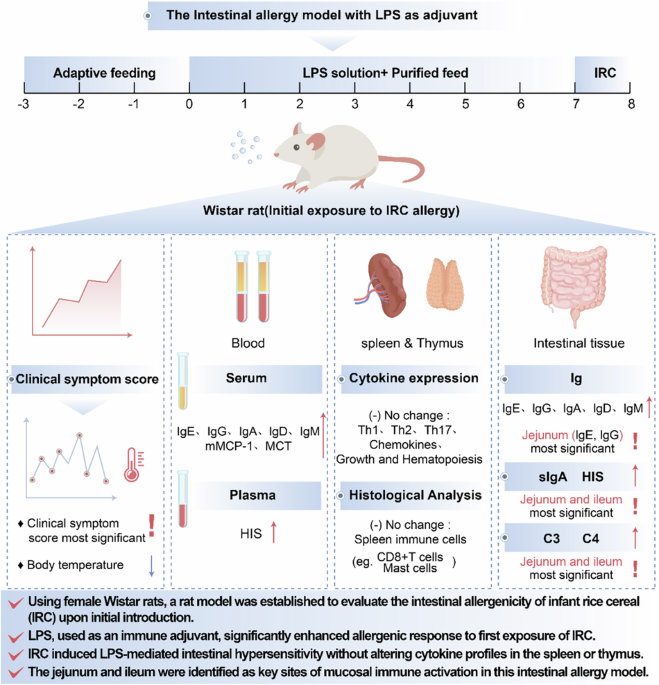






Leave a Reply