Smolen, J. S., Aletaha, D. & McInnes, I. B. Rheumatoid arthritis. Lancet 388, 2023–2038 (2016).
Rech, J. et al. Abatacept inhibits inflammation and onset of rheumatoid arthritis in individuals at high risk (ARIAA): a randomised, international, multicentre, double-blind, placebo-controlled trial. Lancet 403, 850–859 (2024).
Aletaha, D. & Smolen, J. S. Diagnosis and management of rheumatoid arthritis: a review. Jama 320, 1360–1372 (2018).
Lamba, A. & Taneja, V. Gut microbiota as a sensor of autoimmune response and treatment for rheumatoid arthritis. Immunol. Rev. 325, 90–106 (2024).
Zaiss, M. M., Joyce Wu, H. J., Mauro, D., Schett, G. & Ciccia, F. The gut-joint axis in rheumatoid arthritis. Nat. Rev. Rheumatol. 17, 224–237 (2021).
Yang, W. & Cong, Y. Gut microbiota-derived metabolites in the regulation of host immune responses and immune-related inflammatory diseases. Cell Mol. Immunol. 18, 866–877 (2021).
Qi, X. Y. et al. Gut microbiota in rheumatoid arthritis: mechanistic insights, clinical biomarkers, and translational perspectives. Autoimmun. Rev. 24, 103912 (2025).
Cao, Z. et al. The gut virome: a new microbiome component in health and disease. EBioMedicine 81, 104113 (2022).
Mangalea, M. R. et al. Individuals at risk for rheumatoid arthritis harbor differential intestinal bacteriophage communities with distinct metabolic potential. Cell Host Microbe 29, 726–739.e725 (2021).
Tomofuji, Y. et al. Whole gut virome analysis of 476 Japanese revealed a link between phage and autoimmune disease. Ann. Rheum. Dis. 81, 278–288 (2022).
Hu, F. et al. Rheumatoid arthritis patients harbour aberrant enteric bacteriophages with autoimmunity-provoking potential: a paired sibling study. Ann. Rheum. Dis. 83, 1677–1690 (2024).
Yu, Y., Wang, W. & Zhang, F. The next generation fecal Microbiota transplantation: to transplant bacteria or virome. Adv. Sci. 10, e2301097 (2023).
Lam, S. et al. Roles of the gut virome and mycobiome in faecal microbiota transplantation. Lancet Gastroenterol. Hepatol. 7, 472–484 (2022).
Sinha, A. et al. Transplantation of bacteriophages from ulcerative colitis patients shifts the gut bacteriome and exacerbates the severity of DSS colitis. Microbiome 10, 105 (2022).
Majzoub, M. E. et al. The phageome of patients with ulcerative colitis treated with donor fecal microbiota reveals markers associated with disease remission. Nat. Commun. 15, 8979 (2024).
Mao, X. et al. Transfer of modified gut viromes improves symptoms associated with metabolic syndrome in obese male mice. Nat. Commun. 15, 4704 (2024).
Rasmussen, T. S. et al. Faecal virome transplantation decreases symptoms of type 2 diabetes and obesity in a murine model. Gut 69, 2122–2130 (2020).
Zhao, X. et al. Commensal viruses promote intestinal stem cell regeneration following radiation damage by inhibiting hyperactivation of RIG-I and notch signals. Adv. Sci. 12, e05204 (2025).
Rasmussen, T. S. et al. Overcoming donor variability and risks associated with fecal microbiota transplants through bacteriophage-mediated treatments. Microbiome 12, 119 (2024).
Ji, S. et al. Engrafting gut bacteriophages have potential to modulate microbial metabolism in fecal microbiota transplantation. Microbiome 13, 149 (2025).
Christensen, A. D., Haase, C., Cook, A. D. & Hamilton, J. A. K/BxN Serum-transfer arthritis as a model for human inflammatory arthritis. Front. Immunol. 7, 213 (2016).
Li, J. & Kuhn, K. A. Microbial threads in the tapestry of rheumatoid arthritis. J. Clin. Investig. 135, e195374 (2025).
Saint-Jean, M., Claisse, O., Marrec, C. L. & Samot, J. Structural and genetic diversity of lysis modules in bacteriophages infecting the genus streptococcus. Genes 16, https://doi.org/10.3390/genes16070842 (2025).
Schoch, C. L. et al. NCBI Taxonomy: a comprehensive update on curation, resources, and tools. Database 2020, https://doi.org/10.1093/database/baaa062 (2020).
Fan, L. et al. A. muciniphila suppresses colorectal tumorigenesis by inducing TLR2/NLRP3-mediated M1-Like TAMs. Cancer Immunol. Res. 9, 1111–1124 (2021).
Jiang, L. et al. A high-fiber diet synergizes with Prevotella copri and exacerbates rheumatoid arthritis. Cell Mol. Immunol. 19, 1414–1424 (2022).
Liu, H. et al. Live Akkermansia muciniphila boosts dendritic cell retinoic acid synthesis to modulate IL-22 activity and mitigate colitis in mice. Microbiome 12, 275 (2024).
Hu, J. et al. Tripterygium hypoglaucum extract ameliorates adjuvant-induced arthritis in mice through the gut microbiota. Chin. J. Nat. Med. 21, 730–744 (2023).
Luo, Y. et al. Alteration of gut microbiota in individuals at high-risk for rheumatoid arthritis associated with disturbed metabolome and the initiation of arthritis through the triggering of mucosal immunity imbalance. Arthritis Rheumatol. 75, 1736–1748 (2023).
Seo, S. K. & Kwon, B. Immune regulation through tryptophan metabolism. Exp. Mol. Med. 55, 1371–1379 (2023).
Roager, H. M. & Licht, T. R. Microbial tryptophan catabolites in health and disease. Nat. Commun. 9, 3294 (2018).
Pei, T. et al. The relationship between tryptophan metabolism and gut microbiota: Interaction mechanism and potential effects in infection treatment. Microbiol. Res. 298, 128211 (2025).
Xiang, T. et al. Aberrant tryptophan metabolism manipulates osteochondral homeostasis. Research 8, 0728 (2025).
Wortelboer, K. et al. Phage-microbe dynamics after sterile faecal filtrate transplantation in individuals with metabolic syndrome: a double-blind, randomised, placebo-controlled clinical trial assessing efficacy and safety. Nat. Commun. 14, 5600 (2023).
Borin, J. M. et al. Fecal virome transplantation is sufficient to alter fecal microbiota and drive lean and obese body phenotypes in mice. Gut Microbes 15, 2236750 (2023).
Brunse, A. et al. Fecal filtrate transplantation protects against necrotizing enterocolitis. ISME J. 16, 686–694 (2022).





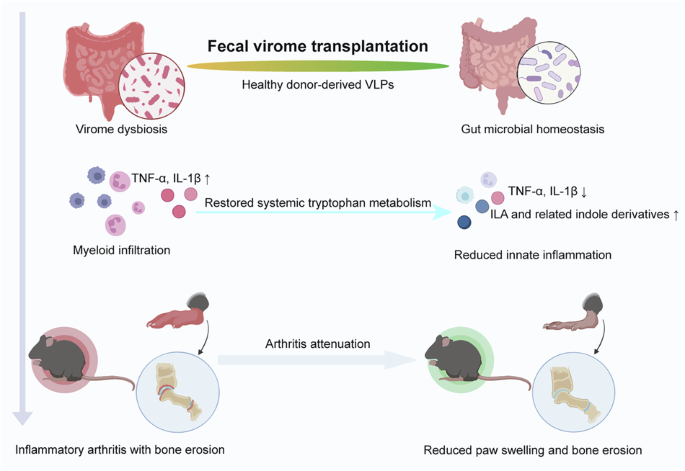









Leave a Reply