Quillin, S. J. & Seifert, H. S. Neisseria gonorrhoeae host adaptation and pathogenesis. Nat. Rev. Microbiol. 16, 226–240 (2018).
WHO. Gonorrhoea (Neisseria gonorrhoeae infection) (WHO, https://www.who.int/news-room/fact-sheets/detail/gonorrhoea-(neisseria-gonorrhoeae-infection) 2024).
Day, M. J. et al. Stably high azithromycin resistance and decreasing ceftriaxone susceptibility in Neisseria gonorrhoeae in 25 European countries, 2016. BMC Infect. Dis. 18, 609 (2018).
Russell, M. W., Jerse, A. E. & Gray-Owen, S. D. Progress toward a gonococcal vaccine: the way forward. Front. Immunol. 10, 2417 (2019).
Petousis-Harris, H. et al. Effectiveness of a group B outer membrane vesicle meningococcal vaccine against gonorrhoea in New Zealand: a retrospective case-control study. Lancet 390, 1603–1610 (2017).
Bruxvoort, K. J. et al. Prevention of Neisseria gonorrhoeae with meningococcal B vaccine: a matched cohort study in Southern California. Clin. Infect. Dis. 76, e1341–e1349 (2023).
Abara, W. E. et al. Effectiveness of a serogroup B outer membrane vesicle meningococcal vaccine against gonorrhoea: a retrospective observational study. Lancet Infect. Dis. 22, 1021–1029 (2022).
Wang, B. et al. 4CMenB sustained vaccine effectiveness against invasive meningococcal B disease and gonorrhoea at three years post programme implementation. J. Infect. 87, 95–102 (2023).
Abara, W. E. et al. Effectiveness of a serogroup B meningococcal vaccine against gonorrhea: a retrospective study. Vaccine 42, 126312 (2024).
UK_Health_Security_Agency. Gonorrhoea: the green book chapter. https://www.gov.uk/government/publications/gonorrhoea-the-green-book-chapter (UK Health Security Agency, 2025).
Sparling, P. F. Genetic transformation of Neisseria gonorrhoeae to streptomycin resistance. J. Bacteriol. 92, 1364–1371 (1966).
Unemo, M. & Shafer, W. M. Antimicrobial resistance in Neisseria gonorrhoeae in the 21st century: past, evolution, and future. Clin. Microbiol. Rev. 27, 587–613 (2014).
Hagblom, P., Segal, E., Billyard, E. & So, M. Intragenic recombination leads to pilus antigenic variation in Neisseria gonorrhoeae. Nature 315, 156–158 (1985).
Murphy, G. L., Connell, T. D., Barritt, D. S., Koomey, M. & Cannon, J. G. Phase variation of gonococcal protein II: regulation of gene expression by slipped-strand mispairing of a repetitive DNA sequence. Cell 56, 539–547 (1989).
Islam, E. A. et al. Specific binding to differentially expressed human carcinoembryonic antigen-related cell adhesion molecules determines the outcome of Neisseria gonorrhoeae infections along the female reproductive tract. Infect. Immun. 86, e00092-18 (2018).
Alcott, A. M. et al. Variable expression of Opa proteins by Neisseria gonorrhoeae influences bacterial association and phagocytic killing by human neutrophils. J. Bacteriol. 204, e0003522 (2022).
Boulton, I. C. & Gray-Owen, S. D. Neisserial binding to CEACAM1 arrests the activation and proliferation of CD4+ T lymphocytes. Nat. Immunol. 3, 229–236 (2002).
Yang, Q. L. & Gotschlich, E. C. Variation of gonococcal lipooligosaccharide structure is due to alterations in poly-G tracts in lgt genes encoding glycosyl transferases. J. Exp. Med. 183, 323–327 (1996).
Banerjee, A. et al. Identification of the gene (lgtG) encoding the lipooligosaccharide beta chain synthesizing glucosyl transferase from Neisseria gonorrhoeae. Proc. Natl. Acad. Sci. USA 95, 10872–10877 (1998).
Bennett, J. S. et al. Species status of Neisseria gonorrhoeae: evolutionary and epidemiological inferences from multilocus sequence typing. BMC Biol. 5, 35 (2007).
Demczuk, W. et al. Neisseria gonorrhoeae sequence typing for antimicrobial resistance, a novel antimicrobial resistance multilocus typing scheme for tracking global dissemination of N. gonorrhoeae strains. J. Clin. Microbiol. 55, 1454–1468 (2017).
Harrison, O. B. et al. Neisseria gonorrhoeae population genomics: use of the gonococcal core genome to improve surveillance of antimicrobial resistance. J. Infect. Dis. 222, 1816–1825 (2020).
Unitt, A. et al. Neisseria gonorrhoeae LIN codes provides a robust, multi-resolution lineage nomenclature. eLife 14:RP107758 (2025).
Jolley, K. A., Bray, J. E. & Maiden, M. C. J. Open-access bacterial population genomics: BIGSdb software, the PubMLST.org website and their applications. Wellcome Open Res. 3, 124 (2018).
Deo, P. et al. Outer membrane vesicles from Neisseria gonorrhoeae target PorB to mitochondria and induce apoptosis. PLoS Pathog. 14, e1006945 (2018).
Nickel, J. C. Management of urinary tract infections: historical perspective and current strategies: part 1-before antibiotics. J. Urol. 173, 21–26 (2005).
Lewis, L. A. & Ram, S. Complement interactions with the pathogenic Neisseriae: clinical features, deficiency states, and evasion mechanisms. FEBS Lett. 594, 2670–2694 (2020).
Edwards, J. L. et al. A co-operative interaction between Neisseria gonorrhoeae and complement receptor 3 mediates infection of primary cervical epithelial cells. Cell Microbiol. 4, 571–584 (2002).
Ram, S. et al. Binding of C4b-binding protein to porin: a molecular mechanism of serum resistance of Neisseria gonorrhoeae. J. Exp. Med. 193, 281–295 (2001).
Ram, S. et al. Binding of complement factor H to loop 5 of porin protein 1A: a molecular mechanism of serum resistance of nonsialylated Neisseria gonorrhoeae. J. Exp. Med. 188, 671–680 (1998).
Rice, P. A., Vayo, H. E., Tam, M. R. & Blake, M. S. Immunoglobulin G antibodies directed against protein III block killing of serum-resistant Neisseria gonorrhoeae by immune serum. J. Exp. Med. 164, 1735–1748 (1986).
Mandrell, R. E., Griffiss, J. M. & Macher, B. A. Lipooligosaccharides (LOS) of Neisseria gonorrhoeae and Neisseria meningitidis have components that are immunochemically similar to precursors of human blood group antigens. Carbohydrate sequence specificity of the mouse monoclonal antibodies that recognize crossreacting antigens on LOS and human erythrocytes. J. Exp. Med. 168, 107–126 (1988).
Zhu, W. et al. Properly folded and functional PorB from Neisseria gonorrhoeae inhibits dendritic cell stimulation of CD4+ T cell proliferation. J. Biol. Chem. 293, 11218–11229 (2018).
Lee, H. S., Ostrowski, M. A. & Gray-Owen, S. D. CEACAM1 dynamics during Neisseria gonorrhoeae suppression of CD4+ T lymphocyte activation. J. Immunol. 180, 6827–6835 (2008).
Leung, S. et al. The cytokine milieu in the interplay of pathogenic Th1/Th17 cells and regulatory T cells in autoimmune disease. Cell Mol. Immunol. 7, 182–189 (2010).
Ramsey, K. H. et al. Inflammatory cytokines produced in response to experimental human gonorrhea. J. Infect. Dis. 172, 186–191 (1995).
Hedges, S. R., Sibley, D. A., Mayo, M. S., Hook, E. W. 3rd & Russell, M. W. Cytokine and antibody responses in women infected with Neisseria gonorrhoeae: effects of concomitant infections. J. Infect. Dis. 178, 742–751 (1998).
Masson, L. et al. Relationship between female genital tract infections, mucosal interleukin-17 production and local T helper type 17 cells. Immunology 146, 557–567 (2015).
Gagliardi, M. C. et al. Circulating levels of interleukin-17A and interleukin-23 are increased in patients with gonococcal infection. FEMS Immunol. Med. Microbiol. 61, 129–132 (2011).
Feinen, B., Jerse, A. E., Gaffen, S. L. & Russell, M. W. Critical role of Th17 responses in a murine model of Neisseria gonorrhoeae genital infection. Mucosal Immunol. 3, 312–321 (2010).
Liu, Y., Islam, E. A., Jarvis, G. A., Gray-Owen, S. D. & Russell, M. W. Neisseria gonorrhoeae selectively suppresses the development of Th1 and Th2 cells, and enhances Th17 cell responses, through TGF-beta-dependent mechanisms. Mucosal Immunol. 5, 320–331 (2012).
Liu, Y., Egilmez, N. K. & Russell, M. W. Enhancement of adaptive immunity to Neisseria gonorrhoeae by local intravaginal administration of microencapsulated interleukin 12. J. Infect. Dis. 208, 1821–1829 (2013).
Liu, Y., Liu, W. & Russell, M. W. Suppression of host adaptive immune responses by Neisseria gonorrhoeae: role of interleukin 10 and type 1 regulatory T cells. Mucosal Immunol. 7, 165–176 (2014).
Oster, P. et al. MeNZB: a safe and highly immunogenic tailor-made vaccine against the New Zealand Neisseria meningitidis serogroup B disease epidemic strain. Vaccine 23, 2191–2196 (2005).
Schwechheimer, C. & Kuehn, M. J. Outer-membrane vesicles from Gram-negative bacteria: biogenesis and functions. Nat. Rev. Microbiol. 13, 605–619 (2015).
GlaxoSmithKline. BEXSERO (Meningococcal Group B Vaccine) injectable suspension, for intramuscular use: prescribing information. https://www.fda.gov/media/90996/download (GSK Vaccines, 2024).
Tzeng, Y. L., Sannigrahi, S. & Stephens, D. S. NHBA antibodies elicited by 4CMenB vaccination are key for serum bactericidal activity against Neisseria gonorrhoeae. NPJ Vaccines 9, 223 (2024).
Semchenko, E. A., Tan, A., Borrow, R. & Seib, K. L. The serogroup B meningococcal vaccine Bexsero elicits antibodies to Neisseria gonorrhoeae. Clin. Infect. Dis. 69, 1101–1111 (2019).
Gray, M. C. et al. Evaluating vaccine-elicited antibody activities against Neisseria gonorrhoeae: cross-protective responses elicited by the 4CMenB meningococcal vaccine. Infect. Immun. 91, e0030923 (2023).
Leduc, I. et al. The serogroup B meningococcal outer membrane vesicle-based vaccine 4CMenB induces cross-species protection against Neisseria gonorrhoeae. PLoS Pathog. 16, e1008602 (2020).
Zeppa, J. J. et al. Meningococcal vaccine 4CMenB elicits a robust cellular immune response that targets but is not consistently protective against Neisseria gonorrhoeae during murine vaginal infection. mSphere 10, e0094024 (2025).
Zhu, W. et al. Protection against N. gonorrhoeae induced by OMV-based meningococcal vaccines are associated with cross-species directed humoral and cellular immune responses. Front. Immunol. 16, 1539795 (2025).
Jerse, A. E. et al. Estradiol-treated female mice as surrogate hosts for Neisseria gonorrhoeae genital tract infections. Front. Microbiol. 2, 107 (2011).
Stejskal, L. et al. Profiling IgG and IgA antibody responses during vaccination and infection in a high-risk gonorrhoea population. Nat. Commun. 15, 6712 (2024).
Ramirez-Bencomo, F. et al. Identification of immunogenic outer membrane vesicle vaccine antigen components using a meningococcal protein microarray. Vaccine 53, 126953 (2025).
Tzeng, Y. L., Sannigrahi, S., Borrow, R. & Stephens, D. S. Neisseria gonorrhoeae lipooligosaccharide glycan epitopes recognized by bactericidal IgG antibodies elicited by the meningococcal group B-directed vaccine, MenB-4C. Front. Immunol. 15, 1350344 (2024).
Troisi, M. et al. Human monoclonal antibodies targeting subdominant meningococcal antigens confer cross-protection against gonococcus. Sci. Transl. Med. 17, eadv0969 (2025).
Li, Z. et al. Transfer of IgG in the female genital tract by MHC class I-related neonatal Fc receptor (FcRn) confers protective immunity to vaginal infection. Proc. Natl. Acad. Sci. USA 108, 4388–4393 (2011).
Chen, K., Magri, G., Grasset, E. K. & Cerutti, A. Rethinking mucosal antibody responses: IgM, IgG and IgD join IgA. Nat. Rev. Immunol. 20, 427–441 (2020).
Stover, E. L. et al. Development and validation of multiplex assays for mouse and human IgG and IgA to Neisseria gonorrhoeae antigens. J. Infect. Dis. 230, 852–856 (2024).
Zariri, A. et al. Meningococcal outer membrane vesicle composition-dependent activation of the innate immune response. Infect. Immun. 84, 3024–3033 (2016).
van de Waterbeemd, B. et al. Quantitative proteomics reveals distinct differences in the protein content of outer membrane vesicle vaccines. J. Proteome Res. 12, 1898–1908 (2013).
Matthias, K. A. et al. Deletion of major porins from meningococcal outer membrane vesicle vaccines enhances reactivity against heterologous serogroup B Neisseria meningitidis strains. Vaccine 38, 2396–2405 (2020).
Matthias, K. A. et al. Meningococcal detoxified outer membrane vesicle vaccines enhance gonococcal clearance in a murine infection model. J. Infect. Dis. 225, 650–660 (2022).
Harrison, O. B. et al. Investigating genetic, antigenic, and structural diversity in the Neisseria gonorrhoeae outer membrane protein, PorB: implications for vaccine design. mBio 16, e0130925 (2025).
Matthias, K. A. et al. Evaluation of immunization route in induction of vaccine-mediated anti-gonococcal immune responses in a murine model of ascending infection. J. Infect. Dis. 232, e765–e777 (2025).
Beernink, P. T. et al. A meningococcal native outer membrane vesicle vaccine with attenuated endotoxin and overexpressed factor H binding protein elicits gonococcal bactericidal antibodies. J. Infect. Dis. 219, 1130–1137 (2019).
Plante, M. et al. Intranasal immunization with gonococcal outer membrane preparations reduces the duration of vaginal colonization of mice by Neisseria gonorrhoeae. J. Infect. Dis. 182, 848–855 (2000).
Zhu, W. et al. Comparison of immune responses to gonococcal PorB delivered as outer membrane vesicles, recombinant protein, or Venezuelan equine encephalitis virus replicon particles. Infect. Immun. 73, 7558–7568 (2005).
Kaparakis, M. et al. Bacterial membrane vesicles deliver peptidoglycan to NOD1 in epithelial cells. Cell Microbiol. 12, 372–385 (2010).
Youssef, A. R. et al. Opa+ and Opa– isolates of Neisseria meningitidis and Neisseria gonorrhoeae induce sustained proliferative responses in human CD4+ T cells. Infect. Immun. 77, 5170–5180 (2009).
Zhu, W. et al. Vaccines for gonorrhea: can we rise to the challenge? Front. Microbiol. 2, 124 (2011).
Mosaheb, M. & Wetzler, L. M. Meningococcal PorB induces a robust and diverse antigen specific T cell response as a vaccine adjuvant. Vaccine 36, 7689–7699 (2018).
Maharjan, S. et al. Dissection of the function of the RmpM periplasmic protein from Neisseria meningitidis. Microbiology 162, 364–375 (2016).
Liu, Y. et al. Experimental vaccine induces Th1-driven immune responses and resistance to Neisseria gonorrhoeae infection in a murine model. Mucosal Immunol. 10, 1594–1608 (2017).
Liu, Y. et al. Microencapsulated IL-12 drives genital tract immune responses to intranasal gonococcal outer membrane vesicle vaccine and induces resistance to vaginal infection with diverse strains of Neisseria gonorrhoeae. mSphere 8, e0038822 (2023).
Jones, R. A. et al. Tackling immunosuppression by Neisseria gonorrhoeae to facilitate vaccine design. PLoS Pathog. 20, e1012688 (2024).
Dhital, S. et al. Characterization of outer membrane vesicles released by clinical isolates of Neisseria gonorrhoeae. Proteomics 24, e2300087 (2024).
Sardinas, G., Reddin, K., Pajon, R. & Gorringe, A. Outer membrane vesicles of Neisseria lactamica as a potential mucosal adjuvant. Vaccine 24, 206–214 (2006).
Piliou, S., Farman, T. A., Marini, A., Manoharan, S. & Mastroeni, P. Commensal Neisseria cinerea outer membrane vesicles as a platform for the delivery of meningococcal and gonococcal antigens to the immune system. Vaccine 41, 7671–7681 (2023).
Kesty, N. C. & Kuehn, M. J. Incorporation of heterologous outer membrane and periplasmic proteins into Escherichia coli outer membrane vesicles. J. Biol. Chem. 279, 2069–2076 (2004).
Molina, J. M. et al. Doxycycline prophylaxis and meningococcal group B vaccine to prevent bacterial sexually transmitted infections in France (ANRS 174 DOXYVAC): a multicentre, open-label, randomised trial with a 2 x 2 factorial design. Lancet Infect. Dis. 24, 1093–1104 (2024).
Williams, E. et al. Neisseria gonorrhoeae vaccines: a contemporary overview. Clin. Microbiol. Rev. 37, e0009423 (2024).
Johnson, B. GSK’s gonorrhea vaccine receives fast-track designation to expedite clinical trials. Nat. Med. 29, 2146–2147 (2023).
Spinsanti, M. et al. A novel GMMA-based gonococcal vaccine demonstrates functional immune responses in mice. NPJ Vaccines 10, 146 (2025).
Taylor, N. P. GSK axes vaccine from $2.1B deal, switching to preclinical successor over ‘increased competition’. https://www.fiercebiotech.com/biotech/gsk-axes-vaccine-21b-deal-switching-preclinical-successor-over-increased-competition (Fierce Biotech, 2024).
Mulks, M. H. & Knapp, J. S. Immunoglobulin A1 protease types of Neisseria gonorrhoeae and their relationship to auxotype and serovar. Infect. Immun. 55, 931–936 (1987).
Drake, S. L. & Koomey, M. The product of the pilQ gene is essential for the biogenesis of type IV pili in Neisseria gonorrhoeae. Mol. Microbiol. 18, 975–986 (1995).
Bettoni, S. et al. C4BP-IgM protein as a therapeutic approach to treat Neisseria gonorrhoeae infections. JCI Insight 4, e131886 (2019).
Shafer, W. M., Joiner, K., Guymon, L. F., Cohen, M. S. & Sparling, P. F. Serum sensitivity of Neisseria gonorrhoeae: the role of lipopolysaccharide. J. Infect. Dis. 149, 175–183 (1984).
Fegan, J. E. et al. Rational selection of TbpB variants yields a bivalent vaccine with broad coverage against Neisseria gonorrhoeae. NPJ Vaccines 10, 10 (2025).
Shaughnessy, J. et al. An optimized factor H-Fc fusion protein against multidrug-resistant Neisseria gonorrhoeae. Front. Immunol. 13, 975676 (2022).
Awanye, A. M. et al. Immunogenicity profiling of protein antigens from capsular group B Neisseria meningitidis. Sci. Rep. 9, 6843 (2019).





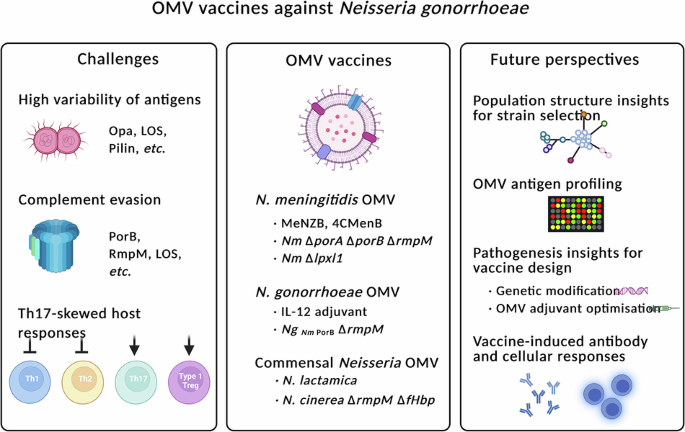










Leave a Reply