Wang, X. et al. The traditional Chinese medicine and non-small cell lung cancer: from a gut microbiome perspective. Front Cell Infect. Microbiol 13, 1151557 (2023).
Bernstein, J. A., Bernstein, J. S., Makol, R. & Ward, S. Allergic Rhinitis: A Review. JAMA 331, 866–877 (2024).
Patel, K. B., Mims, J. W. & Clinger, J. D. The Burden of Asthma and Allergic Rhinitis: Epidemiology and Health Care Costs. Otolaryngologic Clin. North Am. 57, 179–189 (2024).
Leth-Moller, K. B., Skaaby, T. & Linneberg, A. Allergic rhinitis and allergic sensitisation are still increasing among Danish adults. Allergy 75, 660–668 (2020).
Bousquet, J. et al. Allergic rhinitis. Nat. Rev. Dis. Prim. 6, 95 (2020).
Wasilewska, E. et al. Dietary Habits in Children with Respiratory Allergies: A Single-Center Polish Pilot Study. Nutrients 12, 1521 (2020).
Kim, C. W. et al. Gut dysbiosis from high-salt diet promotes glioma via propionate-mediated TGF-beta activation. J. Exp. Med. 222, e20241135 (2025).
Olde Engberink, R. H. G. Salt Intake: Reduce or Substitute? J. Am. Coll. Cardiol. 83, 723–725 (2024).
Musiol, S. et al. The impact of high-salt diet on asthma in humans and mice: Effect on specific T-cell signatures and microbiome. Allergy 79, 1844–1857 (2024).
Bailey, M. A. Salt-sensitive hypertension and the immune system. Exp. Physiol. 105, 767–768 (2020).
Kirabo, A., Masenga, S. K. & Kleyman, T. R. Epithelial Na+ Channels, Immune Cells, and Salt. Annu. Rev. Physiol. 87, 381–395 (2025).
Zheng, T. et al. The process of hypertension induced by high-salt diet: Association with interactions between intestinal mucosal microbiota, and chronic low-grade inflammation, end-organ damage. Front Microbiol 14, 1123843 (2023).
Ertuglu, L. A., Mutchler, A. P., Yu, J. & Kirabo, A. Inflammation and oxidative stress in salt sensitive hypertension; The role of the NLRP3 inflammasome. Front Physiol. 13, 1096296 (2022).
Ruggeri Barbaro, N. et al. Sodium activates human monocytes via the NADPH oxidase and isolevuglandin formation. Cardiovasc Res 117, 1358–1371 (2021).
Matthias, J. et al. Salt generates antiinflammatory Th17 cells but amplifies pathogenicity in proinflammatory cytokine microenvironments. J. Clin. Invest 130, 4587–4600 (2020).
Corte-Real, B. F. et al. Sodium perturbs mitochondrial respiration and induces dysfunctional Tregs. Cell Metab. 35, 299–315 e298 (2023).
Li, Y. et al. Lysine methylation promotes NFAT5 activation and determines temozolomide efficacy in glioblastoma. Nat. Commun. 14, 4062 (2023).
Jobin, K. et al. A high-salt diet compromises antibacterial neutrophil responses through hormonal perturbation. Sci. Transl. Med. 12, eaay3850 (2020).
Ye, B. J. et al. TonEBP in dendritic cells mediates pro-inflammatory maturation and Th1/Th17 responses. Cell Death Dis. 11, 421 (2020).
Zeng, X. et al. Nasal mucosal fibroblasts produce IL-4 to induce Th2 response. Innate Immun. 30, 55–65 (2024).
Fan, L. et al. B. adolescentis ameliorates chronic colitis by regulating Treg/Th2 response and gut microbiota remodeling. Gut Microbes 13, 1–17 (2021).
Yuan, X. et al. Comparative Study of the Effects of Dietary-Free and -Bound Nepsilon-Carboxymethyllysine on Gut Microbiota and Intestinal Barrier. J. Agric Food Chem. 72, 5014–5025 (2024).
Demissie, K., Ernst, P., Gray Donald, K. & Joseph, L. Usual dietary salt intake and asthma in children: a case-control study. Thorax 51, 59–63 (1996).
Knox, A. J. Salt and asthma. BMJ 307, 1159–1160 (1993).
Mo, Q. et al. GSK2334470 attenuates high salt-exacerbated rheumatoid arthritis progression by restoring Th17/Treg homeostasis. iScience 27, 109798 (2024).
Lee, E. et al. Activated pathogenic Th17 lymphocytes induce hypertension following high-fructose intake in Dahl salt-sensitive but not Dahl salt-resistant rats. Dis. Model Mech. 13, dmm044107 (2020).
Yamaguchi, T. et al. Effect of gut microbial composition and diversity on major inhaled allergen sensitization and onset of allergic rhinitis. Allergol. Int 72, 135–142 (2023).
Chen, Z. et al. Fecal and serum metabolomic signatures and gut microbiota characteristics of allergic rhinitis mice model. Front Cell Infect. Microbiol 13, 1150043 (2023).
Houtman, T. A., Eckermann, H. A., Smidt, H. & de Weerth, C. Gut microbiota and BMI throughout childhood: the role of firmicutes, bacteroidetes, and short-chain fatty acid producers. Sci. Rep. 12, 3140 (2022).
Chen, Y. et al. Boeravinone B alleviates gut dysbiosis during myocardial infarction-induced cardiotoxicity in rats. J. Cell Mol. Med 25, 6403–6416 (2021).
Fu, W. et al. Pediococcus acidilactici Strain Alleviates Gluten-Induced Food Allergy and Regulates Gut Microbiota in Mice. Front Cell Infect. Microbiol 12, 845142 (2022).
Miranda, P. M. et al. High salt diet exacerbates colitis in mice by decreasing Lactobacillus levels and butyrate production. Microbiome 6, 57 (2018).
Liu, X. et al. Fucoidan Ameliorated Dextran Sulfate Sodium-Induced Ulcerative Colitis by Modulating Gut Microbiota and Bile Acid Metabolism. J. Agric Food Chem. 70, 14864–14876 (2022).
Li, M. et al. Combined Ganoderma lucidum polysaccharide and ciprofloxacin therapy alleviates Salmonella enterica infection, protects the intestinal barrier, and regulates gut microbiota. Food Funct. 14, 6896–6913 (2023).
Liu, Y. et al. The role of MUC2 mucin in intestinal homeostasis and the impact of dietary components on MUC2 expression. Int J. Biol. Macromol. 164, 884–891 (2020).
Liu, Y. et al. Orchestration of MUC2 – The key regulatory target of gut barrier and homeostasis: A review. Int J. Biol. Macromol. 236, 123862 (2023).
Poritz, L. S., Harris, L. R., Kelly, A. A. 3rd & Koltun, W. A. Increase in the tight junction protein claudin-1 in intestinal inflammation. Dig. Dis. Sci. 56, 2802–2809 (2011).
Yang, S. et al. Akebia saponin D ameliorates metabolic syndrome (MetS) via remodeling gut microbiota and attenuating intestinal barrier injury. Biomed. Pharmacother. 138, 111441 (2021).
Boland, B. et al. 716 Immunodeficiency and Autoimmune Enterocolopathy Linked to NFAT5 Deficiency. Gastroenterology 146, S-125 (2014).
Kleinewietfeld, M. et al. Sodium chloride drives autoimmune disease by the induction of pathogenic TH17 cells. Nature 496, 518–522 (2013).
Aramburu, J. & Lopez-Rodriguez, C. Regulation of Inflammatory Functions of Macrophages and T Lymphocytes by NFAT5. Front Immunol. 10, 535 (2019).
Lee, N., Kim, D. & Kim, W. U. Role of NFAT5 in the Immune System and Pathogenesis of Autoimmune Diseases. Front Immunol. 10, 270 (2019).
Alberdi, M. et al. Context-dependent regulation of Th17-associated genes and IFNgamma expression by the transcription factor NFAT5. Immunol. Cell Biol. 95, 56–67 (2017).
Lu, R. Q. et al. SGK1, a Critical Regulator of Immune Modulation and Fibrosis and a Potential Therapeutic Target in Chronic Graft-Versus-Host Disease. Front Immunol. 13, 822303 (2022).
Matthias, J. et al. Sodium chloride is an ionic checkpoint for human T(H)2 cells and shapes the atopic skin microenvironment. Sci. Transl. Med. 11, eaau0683 (2019).
Delgoffe, G. M. et al. The kinase mTOR regulates the differentiation of helper T cells through the selective activation of signaling by mTORC1 and mTORC2. Nat. Immunol. 12, 295–303 (2011).
Steelant, B. et al. Histamine and T helper cytokine-driven epithelial barrier dysfunction in allergic rhinitis. J. Allergy Clin. Immunol. 141, 951–963 e958 (2018).
Xiao, Z. X. et al. High salt diet accelerates the progression of murine lupus through dendritic cells via the p38 MAPK and STAT1 signaling pathways. Signal Transduct. Target Ther. 5, 34 (2020).
Bai, X. et al. Water-extracted Lonicera japonica polysaccharide attenuates allergic rhinitis by regulating NLRP3-IL-17 signaling axis. Carbohydr. Polym. 297, 120053 (2022).





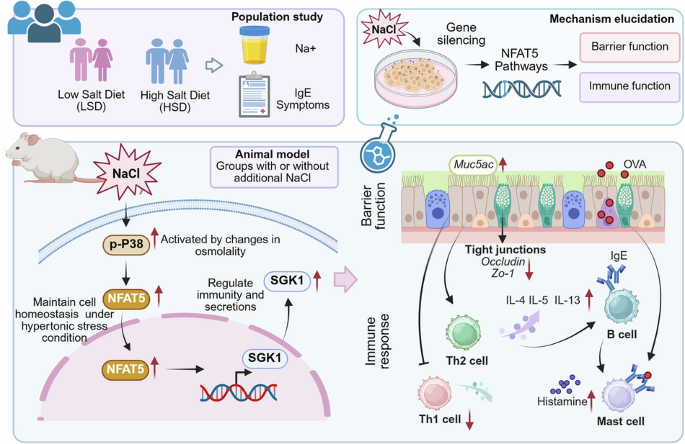
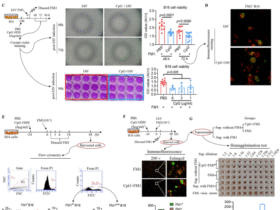









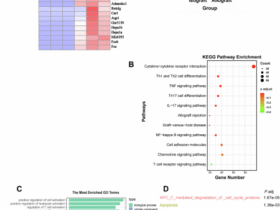

Leave a Reply