Bhatt, S. et al. The global distribution and burden of dengue. Nature 496, 504–507 (2013).
Paz-Bailey, G., Adams, L. E., Deen, J., Anderson, K. B. & Katzelnick, L. C. Dengue. Lancet 403, 667–682 (2024).
Katzelnick, L. C. et al. Antibody-dependent enhancement of severe dengue disease in humans. Science 358, 929–932 (2017).
Katzelnick, L. C. et al. Zika virus infection enhances future risk of severe dengue disease. Science 369, 1123–1128 (2020).
Zambrana, J. V. et al. Primary exposure to Zika virus is linked with increased risk of symptomatic dengue virus infection with serotypes 2, 3, and 4, but not 1. Sci. Transl. Med. 16, 2199 (2024).
Estofolete, C. F. et al. Influence of previous Zika virus infection on acute dengue episode. PLoS Negl. Trop. Dis. 17, e0011710 (2023).
Yamanaka, A. et al. Antibody-dependent enhancement representing in vitro infective progeny virus titer correlates with the viremia level in dengue patients. Sci. Rep. 11, 12354 (2021).
Waggoner, J. J. et al. Antibody-dependent enhancement of severe disease is mediated by serum viral load in pediatric dengue virus infections. J. Infect. Dis. 221, 1846–1854 (2020).
Bournazos, S. et al. Antibody fucosylation predicts disease severity in secondary dengue infection. Science 372, 1102–1105 (2021).
Yamin, R. et al. Human FcγRIIIa activation on splenic macrophages drives dengue pathogenesis in mice. Nat. Microbiol 8, 1468–1479 (2023).
Gordon, A. et al. The Nicaraguan Pediatric Dengue Cohort Study: incidence of inapparent and symptomatic dengue virus infections, 2004–2010. PLoS Negl. Trop. Dis. 7, e2462 (2013).
Bos, S. et al. Serotype-specific epidemiological patterns of inapparent versus symptomatic primary dengue virus infections: a 17-year cohort study in Nicaragua. Lancet Infect. Dis. 25, 346–356 (2025).
Perciani, C. T., Peixoto, P. S., Dias, W. O., Kubrusly, F. S. & Tanizaki, M. M. Improved method to calculate the antibody avidity index. J. Clin. Lab. Anal. 21, 201–206 (2007).
Shackelford, P. G., Granoff, D. M., Madassery, J. V., Scott, M. G. & Nahm, M. H. Clinical and immunologic characteristics of healthy children with subnormal serum concentrations of IgG2. Pediatr. Res 27, 16–21 (1990).
Plebani, A. et al. Serum IgG subclass concentrations in healthy subjects at different age: age normal percentile charts. Eur. J. Pediatr. 149, 164–167 (1989).
Steffen, U. et al. IgA subclasses have different effector functions associated with distinct glycosylation profiles. Nat. Commun. 11, 120 (2020).
Mayer-Hain, S. et al. Systemic activation of neutrophils by immune complexes is critical to IgA vasculitis. J. Immun. 209, 1048–1058 (2022).
Pillebout, E. IgA vasculitis and IgA nephropathy: same disease? J. Clin. Med. 10, 2310 (2021).
Aroca-Crevillén, A., Vicanolo, T., Ovadia, S. & Hidalgo, A. Neutrophils in physiology and pathology. Annu Rev. Pathol. 19, 227–259 (2024).
Dias, A. G. et al. Antibody Fc characteristics and effector functions correlate with protection from symptomatic dengue virus type 3 infection. Sci. Transl. Med. 14, 3151 (2022).
Strich, J. R. et al. Fostamatinib inhibits neutrophils extracellular traps induced by COVID-19 patient plasma: a potential therapeutic. J. Infect. Dis. 223, 981–984 (2021).
Narvaez, F. et al. Dengue severity by serotype and immune status in 19 years of pediatric clinical studies in Nicaragua. PLoS Negl. Trop. Dis. 19, e0012811 (2025).
Colbert, J. A. et al. Ultrasound measurement of gallbladder wall thickening as a diagnostic test and prognostic indicator for severe dengue in pediatric patients. Pediatr. Infect. Dis. J. 26, 850–852 (2007).
Ferreira, B. D. C. & Correia, D. Ultrasound assessment of hepatobiliary and splenic changes in patients with dengue and warning signs during the acute and recovery phases. J. Ultrasound Med 38, 2015–2024 (2019).
Srikiatkhachorn, A. et al. Natural history of plasma leakage in dengue hemorrhagic fever. Pediatr. Infect. Dis. J. 26, 283–290 (2007).
Ruchusatsawat, K. et al. Long-term circulation of Zika virus in Thailand: an observational study. Lancet Infect. Dis. 19, 439–446 (2019).
Monteiro, R. C. & Suzuki, Y. Are there animal models of IgA nephropathy? Semin. Immunopathol. 43, 639–648 (2021).
Seminario, G. et al. The Latin American Society for Immunodeficiencies Registry. J. Clin. Immunol. 45, 28 (2024).
Wang, Z. et al. Enhanced SARS-CoV-2 neutralization by dimeric IgA. Sci. Transl. Med. 13, 1555 (2021).
Wegman, A. D. et al. DENV-specific IgA contributes protective and non-pathologic function during antibody-dependent enhancement of DENV infection. PLoS Pathog. 19, e1011616 (2023).
Upadhaya, B. K. et al. Transient IgA nephropathy with acute kidney injury in a patient with dengue fever. Saudi J. Kidney Dis. Transpl. 21, 521–5 (2010).
Bachal, R. et al. Higher levels of dengue-virus-specific IgG and IgA during pre-defervescence associated with primary dengue hemorrhagic fever. Arch. Virol. 160, 2435–2443 (2015).
Chia, P. Y., Teo, A. & Yeo, T. W. Association of neutrophil mediators with dengue disease severity and cardiac impairment in adults. J. Infect. Dis. 226, 1974–1984 (2022).
Loke, P. et al. Gene expression patterns of dengue virus-infected children from Nicaragua reveal a distinct signature of increased metabolism. PLoS Negl. Trop. Dis. 4, e710 (2010).
Recker, M. et al. Markers of prolonged hospitalisation in severe dengue. PLoS Negl. Trop. Dis. 18, e0011922 (2024).
Robinson, M. et al. A 20-gene set predictive of progression to severe dengue. Cell Rep. 26, 1104–1111 (2019).
Liu, Y. E. et al. An 8-gene machine learning model improves clinical prediction of severe dengue progression. Genome Med. 14, 33 (2022).
Duggal, S. et al. Dengue virus infection in mice induces bone marrow myeloid cell differentiation and generates Ly6Glow immature neutrophils with modulated functions. J. Leukoc. Biol. 115, 130–148 (2024).
Stacey, H. D. et al. IgA potentiates NETosis in response to viral infection. Proc. Natl Acad. Sci. USA 118, e2101497118 (2021).
Gimpel, A.-K. et al. IgA complexes induce neutrophil extracellular trap formation more potently than IgG complexes. Front. Immunol. 12, 761816 (2021).
Allen, K. C. et al. SARS-CoV-2 immune complex–mediated neutrophil activation. Open Forum Infect. Dis. 12, ofaf199 (2025).
Knackstedt, S. L. et al. Neutrophil extracellular traps drive inflammatory pathogenesis in malaria. Sci. Immunol. 4, 336 (2019).
Passelli, K. et al. The c-MET receptor tyrosine kinase contributes to neutrophil-driven pathology in cutaneous leishmaniasis. PLoS Pathog. 18, e1010247 (2022).
Biering, S. B. et al. Structural basis for antibody inhibition of flavivirus NS1-triggered endothelial dysfunction. Science 371, 194–200 (2021).
Modhiran, N. et al. A broadly protective antibody that targets the flavivirus NS1 protein. Science 371, 190–194 (2021).
Németh, T., Sperandio, M. & Mócsai, A. Neutrophils as emerging therapeutic targets. Nat. Rev. Drug Discov. 19, 253–275 (2020).
Pan American Health Organization. Dengue and Dengue Hemorrhagic Fever in the Americas: Guidelines for Prevention and Control Report No. 548 (PAHO, 1997).
Gordon, A. et al. The Nicaraguan pediatric influenza cohort study: design, methods, use of technology, and compliance. BMC Infect. Dis. 15, 504 (2015).
Kuan, G. et al. The Nicaraguan Pediatric Dengue Cohort Study: study design, methods, use of information technology, and extension to other infectious diseases. Am. J. Epidemiol. 170, 120–129 (2009).
Narvaez, F. et al. Evaluation of the traditional and revised WHO classifications of dengue disease severity. PLoS Negl. Trop. Dis. 5, e1397 (2011).
Balmaseda, A. et al. Comparison of four serological methods and two reverse transcription-PCR assays for diagnosis and surveillance of Zika virus infection. J. Clin. Microbiol. 56, e01785-17 (2018).
Waggoner, J. J. et al. Single-reaction, multiplex, real-time RT-PCR for the detection, quantitation, and serotyping of dengue viruses. PLoS Negl. Trop. Dis. 7, e2116 (2013).
Waggoner, J. J. et al. Single-reaction multiplex reverse transcription PCR for detection of Zika, chikungunya, and dengue viruses. Emerg. Infect. Dis. 22, 1295–1297 (2016).
Gordon, A. et al. Prior dengue virus infection and risk of Zika: a pediatric cohort in Nicaragua. PLoS Med. 16, e1002726 (2019).
Cohen, J. Statistical Power Analysis for the Behavioral Sciences (Lawrence Erlbaum Associates, 2013).
Dias, A. G. et al. Anti-dengue virus antibodies that elicit complement-mediated lysis of Zika virion correlate with protection from severe dengue disease. Cell Rep. 44, 115613 (2025).
Kudlacek, S. T. et al. Designed, highly expressing, thermostable dengue virus 2 envelope protein dimers elicit quaternary epitope antibodies. Sci. Adv. 7, 4084–4099 (2021).
Cardona-Ospina, J. A. Antibody profiling and regularization model for pre-secondary DENV2 sera post-primary ZIKV infection—Pediatric Dengue Cohort Study. Zenodo https://doi.org/10.5281/zenodo.14837883 (2025).
Cardona-Ospina, J. A. Avidity index analysis and regularization model for pre-DENV2 sera post-primary ZIKV infection. Zenodo https://doi.org/10.5281/zenodo.14837889 (2025).
Cardona-Ospina, J. A. Post-Zika anti-NS1 serum IgA drives increased neutrophil functions in severe dengue disease. Zenodo https://doi.org/10.5281/zenodo.14837915 (2025).





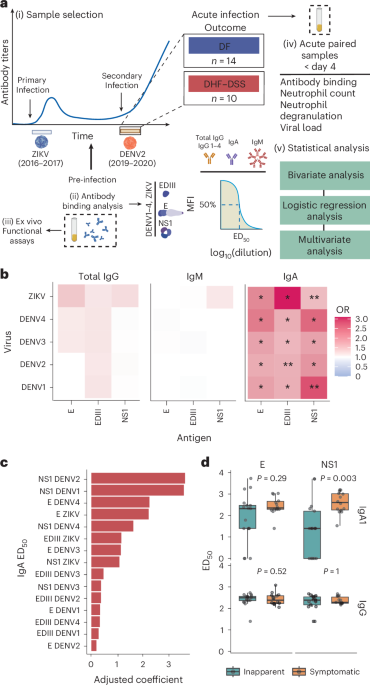






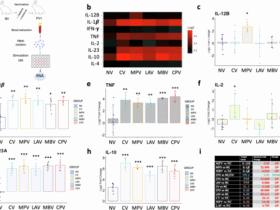

Leave a Reply