Ross, R. Atherosclerosis–an inflammatory disease. N. Engl. J. Med. 340, 115–126 (1999). Established the paradigm of atherosclerosis as a chronic inflammatory disease, providing the conceptual foundation for anti-inflammatory therapy in ASCVD.
Ridker, P. M. et al. Inflammation and cholesterol as predictors of cardiovascular events among patients receiving statin therapy: a collaborative analysis of three randomised trials. Lancet 401, 1293–1301 (2023). Demonstrated that residual inflammation and LDL-C each predict cardiovascular risk despite statin therapy, motivating inflammation-targeted prevention.
Ridker, P. M. et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N. Engl. J. Med. 377, 1119–1131 (2017). CANTOS showed that targeting IL-1β with canakinumab reduced recurrent cardiovascular events, validating inflammation as a causal therapeutic target in ASCVD.
Ridker, P. M. et al. Low-dose methotrexate for the prevention of atherosclerotic events. N. Engl. J. Med. 380, 752–762 (2019).
Tardif, J. -C. et al. Efficacy and safety of low-dose colchicine after myocardial infarction. N. Engl. J. Med. 381, 2497–2505 (2019). COLCOT demonstrated that low-dose colchicine after myocardial infarction reduces ischemic cardiovascular events, supporting colchicine as effective secondary prevention.
Nidorf, S. M. et al. Colchicine in patients with chronic coronary disease. N. Engl. J. Med. 383, 1838–1847 (2020). LoDoCo2 showed that 0.5 mg per day colchicine lowers major cardiovascular events in chronic coronary disease, establishing robust long-term efficacy.
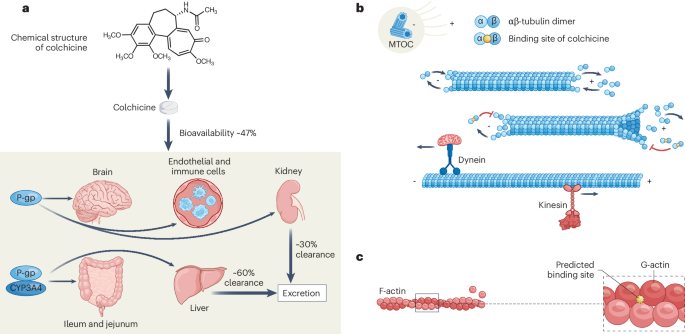
Ferron, G. M., Rochdi, M., Jusko, W. J. & Scherrmann, J. M. Oral absorption characteristics and pharmacokinetics of colchicine in healthy volunteers after single and multiple doses. J. Clin. Pharmacol. 36, 874–883 (1996).
Hunter, A. L. & Klaassen, C. D. Biliary excretion of colchicine. J. Pharmacol. Exp. Ther. 192, 605–617 (1975).
Rochdi, M., Sabouraud, A., Baud, F. J., Bismuth, C. & Scherrmann, J. M. Toxicokinetics of colchicine in humans: analysis of tissue, plasma and urine data in ten cases. Hum. Exp. Toxicol. 11, 510–516 (1992).
Gómez-Lumbreras, A. et al. Drugs that interact with colchicine via inhibition of cytochrome P450 3A4 and P-glycoprotein: a signal detection analysis using a database of spontaneously reported adverse events (FAERS). Ann. Pharmacother. 57, 1137–1146 (2023).
Alfehaid, L. S. et al. Drug-drug interactions and the clinical tolerability of colchicine among patients with COVID-19: a secondary analysis of the COLCORONA randomized clinical trial. JAMA Netw. Open 7, e2431309 (2024).
Wallace, S. L. & Ertel, N. H. Plasma levels of colchicine after oral administration of a single dose. Metabolism 22, 749–753 (1973).
Chappey, O. N. et al. Colchicine disposition in human leukocytes after single and multiple oral administration. Clin. Pharmacol. Ther. 54, 360–367 (1993).
Ben-Chetrit, E. & Levy, M. Does the lack of the P-glycoprotein efflux pump in neutrophils explain the efficacy of colchicine in familial Mediterranean fever and other inflammatory diseases?. Med. Hypotheses 51, 377–380 (1998).
Declèves, X., Niel, E., Debray, M. & Scherrmann, J. -M. Is P-glycoprotein (ABCB1) a phase 0 or a phase 3 colchicine transporter depending on colchicine exposure conditions?. Toxicol. Appl. Pharmacol. 217, 153–160 (2006).
Klimecki, W. T., Futscher, B. W., Grogan, T. M. & Dalton, W. S. P-glycoprotein expression and function in circulating blood cells from normal volunteers. Blood 83, 2451–2458 (1994).
Lidar, M. Colchicine nonresponsiveness in familial Mediterranean fever: clinical, genetic, pharmacokinetic, and socioeconomic characterization. Semin. Arthritis Rheum. 33, 273–282 (2004).
Holy, T. E. & Leibler, S. Dynamic instability of microtubules as an efficient way to search in space. Proc. Natl Acad. Sci. USA 91, 5682–5685 (1994).
Manka, S. W. & Moores, C. A. Microtubule structure by cryo-EM: snapshots of dynamic instability. Essays Biochem 62, 737–751 (2018).
Vorobjev, I. A., Rodionov, V. I., Maly, I. V. & Borisy, G. G. Contribution of plus and minus end pathways to microtubule turnover. J. Cell Sci. 112, 2277–2289 (1999).
Schnapp, B. J. & Reese, T. S. Dynein is the motor for retrograde axonal transport of organelles. Proc. Natl Acad. Sci. USA 86, 1548–1552 (1989).
De Rossi, M. C., Levi, V. & Bruno, L. Retraction of rod-like mitochondria during microtubule-dependent transport. Biosci. Rep. 38, BSR20180208 (2018).
Ravelli, R. B. G. et al. Insight into tubulin regulation from a complex with colchicine and a stathmin-like domain. Nature 428, 198–202 (2004). Provided structural insight into the tubulin–colchicine complex, establishing tubulin as the canonical molecular target underlying microtubule modulation.
Rocha De Abreu, I., Barkdull, A. & Craddock, T. J. A. A computational analysis of colchicine structural analogs as potential microtubule destabilizing agents. FASEB J. 36, fasebj.2022.36.S1.R2187 (2022).
Taylor, E. W. The mechanism of colchicine inhibition of mitosis. I. kinetics of inhibition and the binding of H3-colchicine. J. Cell Biol. 25, 145–160 (1965).
Basu, R., Bose, A., Thomas, D. & Das Sarma, J. Microtubule-assisted altered trafficking of astrocytic gap junction protein connexin 43 is associated with depletion of connexin 47 during mouse hepatitis virus infection. J. Biol. Chem. 292, 14747–14763 (2017).
Garland, D. & Teller, D. C. A reexamination of the reaction between colchicine and tubulin. Ann. N. Y. Acad. Sci. 253, 232–238 (1975).
Rochdi, M., Sabouraud, A., Girre, C., Venet, R. & Scherrmann, J. M. Pharmacokinetics and absolute bioavailability of colchicine after i. v. and oral administration in healthy human volunteers and elderly subjects. Eur. J. Clin. Pharmacol. 46, 351–354 (1994).
Paschke, S. et al. Technical advance: inhibition of neutrophil chemotaxis by colchicine is modulated through viscoelastic properties of subcellular compartments. J. Leukocyte Biol. 94, 1091–1096 (2013).
Hamm-Alvarez, S. F. & Sheetz, M. P. Microtubule-dependent vesicle transport: modulation of channel and transporter activity in liver and kidney. Physiol. Rev. 78, 1109–1129 (1998).
Barlan, K. & Gelfand, V. I. Microtubule-based transport and the distribution, tethering, and organization of organelles. Cold Spring Harb. Perspect. Biol. 9, a025817 (2017).
Moujaber, O. & Stochaj, U. The cytoskeleton as regulator of cell signaling pathways. Trends Biochem. Sci 45, 96–107 (2020).
Ong, M. S. et al. Cytoskeletal proteins in cancer and intracellular stress: a therapeutic perspective. Cancers 12, 238 (2020).
Muñoz-Lasso, D. C., Romá-Mateo, C., Pallardó, F. V. & Gonzalez-Cabo, P. Much more than a scaffold: cytoskeletal proteins in neurological disorders. Cells 9, 358 (2020).
Brivio, V., Faivre-Sarrailh, C., Peles, E., Sherman, D. L. & Brophy, P. J. Assembly of CNS nodes of ranvier in myelinated nerves is promoted by the axon cytoskeleton. Curr. Biol. 27, 1068–1073 (2017).
Weng, J. -H. et al. Colchicine acts selectively in the liver to induce hepatokines that inhibit myeloid cell activation. Nat. Metab. 3, 513–522 (2021).
Sheedy, F. J. et al. CD36 coordinates NLRP3 inflammasome activation by facilitating intracellular nucleation of soluble ligands into particulate ligands in sterile inflammation. Nat. Immunol. 14, 812–820 (2013). Identified CD36-driven particulate ligand formation as a key step enabling NLRP3 inflammasome activation, connecting lipid handling to sterile inflammation.
Duewell, P. et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature 464, 1357–1361 (2010). Demonstrated that cholesterol crystals activate NLRP3 and that NLRP3 inflammasomes are required for atherogenesis, providing a mechanistic rationale for inflammasome-targeted therapy.
Marques-da-Silva, C., Chaves, M., Castro, N., Coutinho-Silva, R. & Guimaraes, M. Colchicine inhibits cationic dye uptake induced by ATP in P2X2 and P2X7 receptor-expressing cells: implications for its therapeutic action. Br. J. Pharmacol. 163, 912–926 (2011).
Li, X. et al. MARK4 regulates NLRP3 positioning and inflammasome activation through a microtubule-dependent mechanism. Nat. Commun. 8, 15986 (2017). Demonstrated that MARK4 controls NLRP3 positioning and activation through a microtubule-dependent mechanism, mechanistically linking microtubules to inflammasome signaling.
Misawa, T. et al. Microtubule-driven spatial arrangement of mitochondria promotes activation of the NLRP3 inflammasome. Nat. Immunol. 14, 454–460 (2013). Showed that microtubule-driven mitochondrial positioning promotes NLRP3 inflammasome activation, explaining how cytoskeletal dynamics regulate innate immune activation.
Martinon, F., Pétrilli, V., Mayor, A., Tardivel, A. & Tschopp, J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440, 237–241 (2006).
Park, Y. H., Wood, G., Kastner, D. L. & Chae, J. J. Pyrin inflammasome activation and RhoA signaling in the autoinflammatory diseases FMF and HIDS. Nat. Immunol. 17, 914–921 (2016).
Magupalli, V. G. et al. HDAC6 mediates an aggresome-like mechanism for NLRP3 and pyrin inflammasome activation. Science 369, eaas8995 (2020).
Bouabdallaoui, N. et al. Time-to-treatment initiation of colchicine and cardiovascular outcomes after myocardial infarction in the Colchicine Cardiovascular Outcomes Trial (COLCOT). Eur. Heart J. 41, 4092–4099 (2020).
Zhou, H. et al. Colchicine prevents oxidative stress-induced endothelial cell senescence via blocking NF-κB and MAPKs: implications in vascular diseases. J. Inflamm. 20, 41 (2023).
Chia, E. W., Grainger, R. & Harper, J. L. Colchicine suppresses neutrophil superoxide production in a murine model of gouty arthritis: a rationale for use of low-dose colchicine. Br. J. Pharmacol. 153, 1288–1295 (2008).
Rai, A., Kapoor, S., Singh, S., Chatterji, B. P. & Panda, D. Transcription factor NF-κB associates with microtubules and stimulates apoptosis in response to suppression of microtubule dynamics in MCF-7 cells. Biochem. Pharmacol. 93, 277–289 (2015).
Modriansky, M. Anti-/pro-oxidant effects of phenolic compounds in cells: are colchicine metabolites chain-breaking antioxidants?. Toxicology 177, 105–117 (2002).
Kadoma, Y., Ishihara, M., Yokoe, I. & Fujisawa, S. Kinetic radical-scavenging activity of colchicine and tropolone. In Vivo 21, 481–486 (2007).
Aldogan, E. H. et al. Colchicine modulates the actin cytoskeleton by direct binding to the monomer and facilitating polymerization. FASEB J. 39, e71054 (2025).
Bueno, O. F. & Leidenheimer, N. J. Colchicine inhibits GABAA receptors independently of microtubule depolymerization. Neuropharmacology 37, 383–390 (1998).
Zhou, X. et al. Identification of glycine receptor α3 as a colchicine-binding protein. Front. Pharmacol. 9, 1238 (2018).
Hu, Y. -J., Liu, Y., Zhao, R. -M. & Qu, S. -S. Interaction of colchicine with human serum albumin investigated by spectroscopic methods. Int. J. Biol. Macromol. 37, 122–126 (2005).
Mughal, J., Katkoori, V. R., Nidorf, S. M., Manu, M. & Abela, G. S. The formation of cholesterol crystals and embolization during myocardial infarction. Am. Heart J. Plus 51, 100509 (2025).
Rajamäki, K. et al. Cholesterol crystals activate the NLRP3 inflammasome in human macrophages: a novel link between cholesterol metabolism and inflammation. PLoS ONE 5, e11765 (2010).
Luo, Y. et al. Modeling of mechanical stress exerted by cholesterol crystallization on atherosclerotic plaques. PLoS ONE 11, e0155117 (2016).
Abela, G. S. et al. Cholesterol crystals induce mechanical trauma, inflammation, and neo-vascularization in solid cancers as in atherosclerosis. Am. Heart J. Plus 35, 100317 (2023).
Abideen, Z. U., Pathak, D. R., Sabanci, R., Manu, M. & Abela, G. S. The effect of colchicine on cholesterol crystal formation, expansion and morphology: a potential mechanism in atherosclerosis. Front. Cardiovasc. Med. 11, 1345521 (2024).
Abela, G. S. et al. Effect of statins on cholesterol crystallization and atherosclerotic plaque stabilization. Am. J. Cardiol. 107, 1710–1717 (2011).
Fry, L. et al. Effect of aspirin on cholesterol crystallization: a potential mechanism for plaque stabilization. Am. Heart J. Plus 13, 100083 (2022).
Kataoka, Y. et al. Plaque microstructures during metformin therapy in type 2 diabetic subjects with coronary artery disease: optical coherence tomography analysis. Cardiovasc. Diagn. Ther. 12, 77–87 (2022).
Opstal, T. S. J. et al. Colchicine attenuates inflammation beyond the inflammasome in chronic coronary artery disease: a LoDoCo2 proteomic substudy. Circulation 142, 1996–1998 (2020). LoDoCo2 proteomic substudy showed broad suppression of inflammatory pathways beyond hsCRP, supporting multi-pathway immunomodulation by colchicine.
Demidowich, A. P. et al. Colchicine’s effects on metabolic and inflammatory molecules in adults with obesity and metabolic syndrome: results from a pilot randomized controlled trial. Int. J. Obes. 44, 1793–1799 (2020).
Rao, S. R. et al. Proteomic effects of colchicine on human macrophages loaded with oxidised low-density lipoprotein cholesterol. Heart Lung Circ. 34, 980–983 (2025).
Warnatsch, A., Ioannou, M., Wang, Q. & Papayannopoulos, V. Inflammation. Neutrophil extracellular traps license macrophages for cytokine production in atherosclerosis. Science 349, 316–320 (2015).
Dorweiler, B. et al. Subendothelial infiltration of neutrophil granulocytes and liberation of matrix-destabilizing enzymes in an experimental model of human neo-intima. Thromb. Haemost. 99, 373–381 (2008).
Pezzato, E. et al. Proteinase-3 directly activates MMP-2 and degrades gelatin and Matrigel; differential inhibition by (-)epigallocatechin-3-gallate. J. Leukoc. Biol. 74, 88–94 (2003).
Döring, Y., Soehnlein, O. & Weber, C. Neutrophil extracellular traps in atherosclerosis and atherothrombosis. Circ. Res. 120, 736–743 (2017).
Cao, J. et al. DNA-sensing inflammasomes cause recurrent atherosclerotic stroke. Nature 633, 433–441 (2024).
Cronstein, B. N. et al. Colchicine alters the quantitative and qualitative display of selectins on endothelial cells and neutrophils. J. Clin. Invest. 96, 994–1002 (1995).
Vaidya, K. et al. Colchicine inhibits neutrophil extracellular trap formation in patients with acute coronary syndrome after percutaneous coronary intervention. J. Am. Heart Assoc. 10, e018993 (2021). Demonstrated in patients with ACS that colchicine inhibits NET formation after PCI, supporting a human mechanistic link to reduced thromboinflammation.
Roberge, C. J. et al. Crystal-induced neutrophil activation. IV. Specific inhibition of tyrosine phosphorylation by colchicine. J. Clin. Invest. 92, 1722–1729 (1993).
Meyer-Lindemann, U. et al. Colchicine impacts leukocyte trafficking in atherosclerosis and reduces vascular inflammation. Front. Immunol. 13, 898690 (2022).
Moore, K. J., Sheedy, F. J. & Fisher, E. A. Macrophages in atherosclerosis: a dynamic balance. Nat. Rev. Immunol. 13, 709–721 (2013).
Wang, J. et al. LRG1 promotes atherosclerosis by inducing macrophage M1-like polarization. Proc. Natl Acad. Sci. USA 121, e2405845121 (2024).
Tucker, B. et al. Colchicine as a novel therapy for suppressing chemokine production in patients with an acute coronary syndrome: a pilot study. Clin. Ther. 41, 2172–2181 (2019).
Schwarz, N. et al. Colchicine exerts anti-atherosclerotic and ‑plaque-stabilizing effects targeting foam cell formation. FASEB J. 37, e22846 (2023).
Panda, D., Daijo, J. E., Jordan, M. A. & Wilson, L. Kinetic stabilization of microtubule dynamics at steady state in vitro by substoichiometric concentrations of tubulin-colchicine complex. Biochemistry 34, 9921–9929 (1995).
Wang, Y., Viollet, B., Terkeltaub, R. & Liu-Bryan, R. AMP-activated protein kinase suppresses urate crystal-induced inflammation and transduces colchicine effects in macrophages. Ann. Rheum. Dis. 75, 286–294 (2016).
Gimbrone, M. A. & García-Cardeña, G. Endothelial cell dysfunction and the pathobiology of atherosclerosis. Circ. Res. 118, 620–636 (2016).
Duplàa, C. et al. Monocyte/macrophage recruitment and expression of endothelial adhesion proteins in human atherosclerotic lesions. Atherosclerosis 121, 253–266 (1996).
Fan, X. et al. Non-canonical NF-κB contributes to endothelial pyroptosis and atherogenesis dependent on IRF-1. Transl. Res. 255, 1–13 (2023).
Fujiyoshi, K. et al. Incidence, factors, and clinical significance of cholesterol crystals in coronary plaque: an optical coherence tomography study. Atherosclerosis 283, 79–84 (2019).
Yang, M. et al. Colchicine alleviates cholesterol crystal-induced endothelial cell pyroptosis through activating AMPK/SIRT1 pathway. Oxid. Med. Cell. Longev. 2020, 9173530 (2020).
Li, Z., Davis, G. S., Mohr, C., Nain, M. & Gemsa, D. Inhibition of LPS-induced tumor necrosis factor-alpha production by colchicine and other microtubule disrupting drugs. Immunobiology 195, 624–639 (1996).
Tang, J. et al. Colchicine delivered by a novel nanoparticle platform alleviates atherosclerosis by targeted inhibition of NF-κB/NLRP3 pathways in inflammatory endothelial cells. J. Nanobiotechnology 21, 460 (2023).
Skålén, K. et al. Subendothelial retention of atherogenic lipoproteins in early atherosclerosis. Nature 417, 750–754 (2002).
Papakonstantinou, E., Karakiulakis, G., Roth, M. & Block, L. H. Platelet-derived growth factor stimulates the secretion of hyaluronic acid by proliferating human vascular smooth muscle cells. Proc. Natl Acad. Sci. USA 92, 9881–9885 (1995).
Robbins, C. S. et al. Local proliferation dominates lesional macrophage accumulation in atherosclerosis. Nat. Med. 19, 1166–1172 (2013).
Basatemur, G. L., Jørgensen, H. F., Clarke, M. C. H., Bennett, M. R. & Mallat, Z. Vascular smooth muscle cells in atherosclerosis. Nat. Rev. Cardiol. 16, 727–744 (2019).
Nakagawa, K. & Nakashima, Y. Pathologic intimal thickening in human atherosclerosis is formed by extracellular accumulation of plasma-derived lipids and dispersion of intimal smooth muscle cells. Atherosclerosis 274, 235–242 (2018).
Elishaev, M. et al. Multiplex imaging for cell phenotyping of early human atherosclerosis. J. Am. Heart Assoc. 13, e034990 (2024).
Clarke, M. C. H., Talib, S., Figg, N. L. & Bennett, M. R. Vascular smooth muscle cell apoptosis induces interleukin-1-directed inflammation: effects of hyperlipidemia-mediated inhibition of phagocytosis. Circ. Res. 106, 363–372 (2010).
Zhang, B. et al. Colchicine reduces neointima formation and VSMC phenotype transition by modulating SRF-MYOCD activation and autophagy. Acta Pharmacol. Sin. 46, 951–963 (2025).
Zhao, Y. et al. Colchicine protects against the development of experimental abdominal aortic aneurysm. Clin. Sci. 137, 1533–1545 (2023).
Chen, M. et al. Colchicine blocks abdominal aortic aneurysm development by maintaining vascular smooth muscle cell homeostasis. Int. J. Biol. Sci. 20, 2092–2110 (2024).
Jiang, H. et al. Colchicine inhibits smooth muscle cell phenotypic switch and aortic dissection in mice-brief report. Arterioscler. Thromb. Vasc. Biol. 45, 979–984 (2025).
Nording, H., Baron, L. & Langer, H. F. Platelets as therapeutic targets to prevent atherosclerosis. Atherosclerosis 307, 97–108 (2020).
Bombeli, T., Schwartz, B. R. & Harlan, J. M. Adhesion of activated platelets to endothelial cells: evidence for a GPIIbIIIa-dependent bridging mechanism and novel roles for endothelial intercellular adhesion molecule 1 (ICAM-1), αvβ3 Integrin, and GPIbα. J. Exp. Med. 187, 329–339 (1998).
Giannini, S. et al. Interaction with damaged vessel wall in vivo in humans induces platelets to express CD40L resulting in endothelial activation with no effect of aspirin intake. Am. J. Physiol Heart. Circ. Physiol. 300, H2072–H2079 (2011).
Baaten, C. C. F. M. J., Nagy, M., Bergmeier, W., Spronk, H. M. H. & Van Der Meijden, P. E. J. Platelet biology and function: plaque erosion vs. rupture. Eur. Heart J. 45, 18–31 (2024).
Cirillo, P. et al. Effects of colchicine on platelet aggregation in patients on dual antiplatelet therapy with aspirin and clopidogrel. J. Thromb. Thrombolysis 50, 468–472 (2020).
Sneddon, J. M. Effect of mitosis inhibitors on blood platelet microtubules and aggregation. J. Physiol. 214, 145–158 (1971).
White, J. G. Effects of colchicine and vinca alkaloids on human platelets. 3. Influence on primary internal contraction and secondary aggregation. Am. J. Pathol. 54, 467–478 (1969).
Rayes, J., Watson, S. P. & Nieswandt, B. Functional significance of the platelet immune receptors GPVI and CLEC-2. J. Clin. Invest. 129, 12–23 (2019).
Menche, D., Israel, A. & Karpatkin, S. Platelets and microtubules. Effect of colchicine and D2O on platelet aggregation and release induced by calcium ionophore A23187. J. Clin. Invest. 66, 284–291 (1980).
Shah, B. et al. Effect of colchicine on platelet-platelet and platelet-leukocyte interactions: a pilot study in healthy subjects. Inflammation 39, 182–189 (2016).
Cimmino, G. et al. Colchicine reduces platelet aggregation by modulating cytoskeleton rearrangement via inhibition of cofilin and LIM domain kinase 1. Vascul. Pharmacol. 111, 62–70 (2018).
Yu, M. et al. Different anti-inflammatory drugs on high-sensitivity C-reactive protein in patients after percutaneous coronary intervention: a pilot randomized clinical trial. J. Cardiovasc. Pharmacol. 83, 234–242 (2024).
Nidorf, M. & Thompson, P. L. Effect of colchicine (0.5 mg twice daily) on high-sensitivity c-reactive protein independent of aspirin and atorvastatin in patients with stable coronary artery disease. Am. J. Cardiol. 99, 805–807 (2007).
Kajikawa, M. et al. Effect of short-term colchicine treatment on endothelial function in patients with coronary artery disease. Int. J. Cardiol. 281, 35–39 (2019).
Hennessy, T. et al. The Low Dose Colchicine after Myocardial Infarction (LoDoCo-MI) study: a pilot randomized placebo controlled trial of colchicine following acute myocardial infarction. Am. Heart J. 215, 62–69 (2019).
Raju, N. C. et al. Effect of colchicine compared with placebo on high sensitivity C-reactive protein in patients with acute coronary syndrome or acute stroke: a pilot randomized controlled trial. J. Thromb. Thrombolysis 33, 88–94 (2012).
Akodad, M. et al. COLIN trial: value of colchicine in the treatment of patients with acute myocardial infarction and inflammatory response. Arch. Cardiovasc. Dis. 110, 395–402 (2017).
Gholoobi, A. et al. Colchicine effectively attenuates inflammatory biomarker high-sensitivity C-reactive protein (hs-CRP) in patients with non-ST-segment elevation myocardial infarction: a randomised, double-blind, placebo-controlled clinical trial. Inflammopharmacology 29, 1379–1387 (2021).
Martínez, G. J. et al. Colchicine acutely suppresses local cardiac production of inflammatory cytokines in patients with an acute coronary syndrome. J. Am. Heart Assoc. 4, e002128 (2015).
Zuriaga, M. A. et al. Colchicine prevents accelerated atherosclerosis in TET2-mutant clonal haematopoiesis. Eur. Heart J. 45, 4601–4615 (2024).
Mohammadnia, N. et al. Colchicine and longitudinal dynamics of clonal hematopoiesis: an exploratory substudy of the LoDoCo2 trial. J. Am. Coll. Cardiol. https://doi.org/10.1016/j.jacc.2025.08.025 (2025).
Tardif, J. -C. et al. Reduction of clonal hematopoiesis mutation burden in coronary patients treated with low-dose colchicine. Preprint at medRxiv https://doi.org/10.1101/2024.10.17.24315679 (2024).
Vaidya, K. et al. Colchicine therapy and plaque stabilization in patients with acute coronary syndrome: a CT coronary angiography study. JACC Cardiovasc. Imaging 11, 305–316 (2018).
Verghese, D. et al. Effect of colchicine on progression of known coronary atherosclerosis in patients with STable CoROnary artery disease CoMpared to placebo (EKSTROM) trial-rationale and design. Am. Heart J. 277, 20–26 (2024).
Iatan, I., Guan, M., Humphries, K. H., Yeoh, E. & Mancini, G. B. J. Atherosclerotic coronary plaque regression and risk of adverse cardiovascular events: a systematic review and updated meta-regression analysis. JAMA Cardiol. 8, 937–945 (2023).
Yu, M. et al. Effect of colchicine on coronary plaque stability in acute coronary syndrome as assessed by optical coherence tomography: the COLOCT randomized clinical trial. Circulation 150, 981–993 (2024). COLOCT trial showed colchicine can increase FCT in ACS lipid-rich plaques, consistent with plaque-stabilizing effects.
Psaltis, P. J. et al. Optical coherence tomography assessment of the impact of colchicine on non-culprit coronary plaque composition after myocardial infarction. Cardiovasc. Res. 121, 468–478 (2025).
Fiolet, A. T. L. et al. Effect of low-dose colchicine on pericoronary inflammation and coronary plaque composition in chronic coronary disease: a subanalysis of the LoDoCo2 trial. Heart https://doi.org/10.1136/heartjnl-2024-325527 (2025).
Nidorf, S. M., Eikelboom, J. W., Budgeon, C. A. & Thompson, P. L. Low-dose colchicine for secondary prevention of cardiovascular disease. J. Am. Coll. Cardiol. 61, 404–410 (2013).
Visseren, F. L. J. et al. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice. Eur. Heart J. 42, 3227–3337 (2021).
Virani, S. S. et al. 2023 AHA/ACC/ACCP/ASPC/NLA/PCNA Guideline for the Management of Patients With Chronic Coronary Disease: a report of the American Heart Association/American College of Cardiology Joint Committee on Clinical Practice Guidelines. Circulation 148, e9–e119 (2023).
Vrints, C. et al. 2024 ESC Guidelines for the management of chronic coronary syndromes. Eur. Heart J 45, 3415–3537 (2024).
Mensah, G. A., Arnold, N., Prabhu, S. D., Ridker, P. M. & Welty, F. K. Inflammation and cardiovascular disease: 2025 ACC Scientific Statement: a report of the American College of Cardiology. J. Am. Coll. Cardiol. https://doi.org/10.1016/j.jacc.2025.08.047 (2025).
Dubé, M. -P. et al. Pharmacogenomics of the efficacy and safety of colchicine in COLCOT. Circ. Genom. Precis. Med. 14, e003183 (2021).
Tong, D. C. et al. Colchicine in patients with acute coronary syndrome: two-year follow-up of the Australian COPS randomized clinical trial. Circulation 144, 1584–1586 (2021).
Jolly, S. S. et al. Colchicine in acute myocardial infarction. N. Engl. J. Med. 392, 633–642 (2025).
Jolly, S. S. et al. Routine spironolactone in acute myocardial infarction. N. Engl. J. Med. 392, 643–652 (2025).
Solomon, M. D. et al. The COVID-19 pandemic and the incidence of acute myocardial infarction. N. Engl. J. Med. 383, 691–693 (2020).
Bhatt, A. S. et al. Fewer hospitalizations for acute cardiovascular conditions during the COVID-19 pandemic. J. Am. Coll. Cardiol. 76, 280–288 (2020).
Mehta, S. R. et al. Complete revascularization with multivessel PCI for myocardial infarction. N. Engl. J. Med. 381, 1411–1421 (2019).
Pitt, B. et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized aldactone evaluation study investigators. N. Engl. J. Med. 341, 709–717 (1999).
Rivero-Santana, B. et al. Early anti-inflammatory therapy in acute myocardial infarction: a network meta-analysis of timing-dependent effects in 23 randomized trials and 28,220 patients. Atherosclerosis 408, 120443 (2025).
Byrne, R. A. et al. 2023 ESC Guidelines for the management of acute coronary syndromes: developed by the task force on the management of acute coronary syndromes of the European Society of Cardiology (ESC). Eur. Heart J. 44, 3720–3826 (2023).
Rao, S. V. et al. 2025 ACC/AHA/ACEP/NAEMSP/SCAI Guideline for the Management of Patients With Acute Coronary Syndromes: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 85, 2135–2237 (2025).
Fiolet, A. T. L. et al. Efficacy and safety of low-dose colchicine in patients with coronary disease: a systematic review and meta-analysis of randomized trials. Eur. Heart J. 42, 2765–2775 (2021).
Tsivgoulis, G. et al. The role of colchicine in the prevention of cerebrovascular ischemia. Curr. Pharm. Des. 24, 668–674 (2018).
Kelly, P. et al. Long-term colchicine for the prevention of vascular recurrent events in non-cardioembolic stroke (CONVINCE): a randomised controlled trial. Lancet 404, 125–133 (2024).
Li, J. et al. Colchicine in patients with acute ischaemic stroke or transient ischaemic attack (CHANCE-3): multicentre, double blind, randomised, placebo controlled trial. BMJ 385, e079061 (2024).
Li, J. et al. Colchicine for preventing stroke in patients with and without intracranial atherosclerotic stenosis: a prespecified analysis of a randomized clinical trial. eClinicalMedicine 84, 103226 (2025).
Jaiswal, V. et al. Abstract 16: short and long term efficacy of colchicine for prevention of stroke and major adverse cardiovascular events: a meta-analysis with CHANCE 3, and CONVINCE randomized controlled trials. Stroke 56, A16 (2025).
Samuel, M. et al. Long-term trials of colchicine for secondary prevention of vascular events: a meta-analysis. Eur. Heart J. 46, 2552–2563 (2025).
d’Entremont, M. -A. et al. Colchicine for secondary prevention of vascular events: a meta-analysis of trials. Eur. Heart J. 46, 2564–2575 (2025).
Lin, D. S. -H., Huang, K. -C., Lin, T. -T., Lee, J. -K. & Lin, L. -Y. Effects of colchicine on major adverse limb and cardiovascular events in patients with peripheral artery disease. Mayo Clin. Proc. 99, 1374–1387 (2024).
Tramujas, L. et al. Association of colchicine use with cardiovascular and limb events in peripheral artery disease: Insights from a retrospective cohort study. Atherosclerosis 398, 118563 (2024).
Bao, R. -H. et al. Balancing the benefits and risks of colchicine use among patients with atherosclerotic cardiovascular disease: an umbrella review of meta-analyses of randomised controlled trials. eClinicalMedicine 84, 103277 (2025).
Yu, M. et al. Residual inflammatory risk in outcomes of Chinese patients after percutaneous coronary intervention. JACC Asia 4, 636–638 (2024).
Lee, S. -Y. et al. P2Y12 inhibitor monotherapy combined with colchicine following PCI in ACS patients: the MACT pilot study. JACC Cardiovasc. Interv. 16, 1845–1855 (2023).
Martí-Carvajal, A. J. et al. Colchicine for the primary prevention of cardiovascular events. Cochrane Database Syst. Rev. 2025, CD015003 (2025).
Tong, D. C. et al. Colchicine in patients with acute coronary syndrome: the Australian COPS randomized clinical trial. Circulation 142, 1890–1900 (2020).
Goldfinger, S. E. Colchicine for familial Mediterranean fever. N. Engl. J. Med. 287, 1302–1302 (1972).
French FMF Consortium. A candidate gene for familial Mediterranean fever. Nat. Genet. 17, 25–31 (1997).
Ozen, S. et al. EULAR/PReS endorsed recommendations for the management of familial Mediterranean fever (FMF): 2024 update. Ann. Rheum. Dis. 84, 899–909 (2025).
De La Serna, A. R., Soldevila, J. G., Claramunt, V. M. & De Luna, A. B. Colchicine for recurrent pericarditis. Lancet 330, 1517 (1987).
Adler, Y. et al. 2015 ESC Guidelines for the diagnosis and management of pericardial diseases: the Task Force for the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology (ESC). Endorsed by: The European Association for Cardio-Thoracic Surgery (EACTS). Eur. Heart J. 36, 2921–2964 (2015).
Richette, P. et al. 2016 updated EULAR evidence-based recommendations for the management of gout. Ann. Rheum. Dis. 76, 29–42 (2017).
Zhang, J. et al. Enhanced trained immunity in peripheral monocytes in unstable angina with elevated high-sensitivity C-reactive protein. JACC Basic Transl. Sci. 10, 101300 (2025).
Jaiswal, S. et al. Clonal hematopoiesis and risk of atherosclerotic cardiovascular disease. N. Engl. J. Med. 377, 111–121 (2017). Established clonal hematopoiesis (CHIP) as a risk factor for ASCVD, linking somatic mutations to heightened inflammatory risk and adverse outcomes.
Von Scheidt, M. et al. Clonal haematopoiesis of indeterminate potential and mortality in coronary artery disease. Eur. Heart J. https://doi.org/10.1093/eurheartj/ehaf602 (2025).
Motoyama, S. et al. Computed tomographic angiography characteristics of atherosclerotic plaques subsequently resulting in acute coronary syndrome. J. Am. Coll. Cardiol. 54, 49–57 (2009).
Araki, M. et al. Optical coherence tomography in coronary atherosclerosis assessment and intervention. Nat. Rev. Cardiol. 19, 684–703 (2022).
Prati, F. et al. Relationship between coronary plaque morphology of the left anterior descending artery and 12 months clinical outcome: the CLIMA study. Eur. Heart J. 41, 383–391 (2020).
Räber, L. et al. Effect of alirocumab added to high-intensity statin therapy on coronary atherosclerosis in patients with acute myocardial infarction: the PACMAN-AMI randomized clinical trial. JAMA 327, 1771–1781 (2022).
Antonopoulos, A. S. et al. Detecting human coronary inflammation by imaging perivascular fat. Sci. Transl. Med. 9, eaal2658 (2017).
Oikonomou, E. K. et al. Non-invasive detection of coronary inflammation using computed tomography and prediction of residual cardiovascular risk (the CRISP CT study): a post-hoc analysis of prospective outcome data. Lancet 392, 929–939 (2018).














Leave a Reply