Millsop, J. W. & Fazel, N. Oral candidiasis. Clin. Dermatol. 34, 487–494 (2016).
Alrayyes, S. F. et al. Oral candidal carriage and associated risk indicators among adults in Sakaka. Saudi Arabia BMC Oral Health 19, 86 (2019).
Mun, M., Yap, T., Alnuaimi, A. D., Adams, G. G. & McCullough, M. J. Oral candidal carriage in asymptomatic patients. Aust. Dent. J. 6, 190–195 (2016).
Akpan, A. & Morgan, R. Oral candidiasis. Postgrad. Med. J. 78, 455–459 (2002).
Axéll, T., Samaranayake, L. P., Reichart, P. A. & Olsen, I. A proposal for reclassification of oral candidosis. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 84(2), 111–112 (1997).
Samaranayake, L. P. & Yaacob, H. B. Classification of oral candidosis. In Oral Candidosis (eds Samaranayake, L. P. & MacFarlane, T. W.) (Oxford: Wright, 1990).
Scully, C., El-Kabir, M. & Samaranayake, L. P. Candida and oral candidosis: a review. Crit. Rev. Oral Biol. Med. 5(2), 125–157 (1994).
Lewis, M. A. O. & Williams, D. W. Diagnosis and management of oral candidosis. Br. Dent. J. 223, 675–681 (2017).
Davenport, J. C. & Wilton, J. M. A. Incidence of immediate and delayed hypersensitivity to Candida albicans in denture stomatitis. J. Dent. Res. 50(4), 892–896 (1971).
Wang, Y. Looking into Candida albicans infection, host response, and antifungal strategies. Virulence 6, 307–308 (2015).
Chin, V. K., Lee, T. Y., Rusliza, B. & Chong, P. P. Dissecting Candida albicans infection from the perspective of C. albicans virulence and omics approaches on host-pathogen interaction: A review. Int. J. Mol. Sci. 17, 1643 (2016).
Zhang, Y., Chen, R., Zhang, D., Qi, S. & Liu, Y. Metabolite interactions between host and microbiota during health and disease: Which feeds the other?. Biomed. Pharmacother. 160, 114295 (2023).
O’Neill, L. A., Kishton, R. J. & Rathmell, J. Guide to immunometabolism for immunologists. Nat. Rev. Immunol. 16, 553–565 (2016).
Wang, J. H., Byun, J. & Pennathur, S. Analytical approaches to metabolomics and applications to systems biology. Semin. Nephrol. 30, 500–511 (2010).
Guijas, C., Montenegro-Burke, J. R., Warth, B., Spilker, M. E. & Siuzdak, G. Metabolomics activity screening for identifying metabolites that modulate phenotype. Nat. Biotechnol. 36, 316–320 (2018).
Havsed, K. et al. Bacterial composition and metabolomics of dental plaque from adolescents. Front. Cell. Infect. Microbiol. 11, 716493 (2021).
Barnes, V. M. et al. Global metabolomic analysis of human saliva and plasma from healthy and diabetic subjects, with and without periodontal disease. PLoS ONE 9, e10518 (2014).
Ishikawa, S. et al. Identification of salivary metabolomic biomarkers for oral cancer screening. Sci. Rep. 6, 31520 (2016).
Weerasinghe, H. & Traven, A. Immunometabolism in fungal infections: the need to eat to compete. Curr. Opin. Microbiol. 58, 32–40 (2020).
Traven, A. & Naderer, T. Central metabolic interactions of immune cells and microbes: prospects for defeating infections. EMBO Rep. 20, e47995 (2019).
Adachi, T. et al. A preliminary pilot study: Metabolomic analysis of saliva in oral candidiasis. Metabolites 12, 1294 (2022).
Ghannoum, M. A. et al. Metabolomics reveals differential levels of oral metabolites in HIV-infected patients: toward novel diagnostic targets. OMICS A J. Integ. Biol. 17, 5–15 (2013).
Talapko, J. et al. A putative role of Candida albicans in promoting cancer development: a current state of evidence and proposed mechanisms. Microorganisms 11(6), 1476 (2023).
Karajacob, A. S. et al. Candida species and oral mycobiota of patients clinically diagnosed with oral thrush. PLoS ONE 18, e0284043 (2023).
Yang, J., Zhao, X., Lu, X., Lin, X. & Xu, G. A data preprocessing strategy for metabolomics to reduce the mask effect in data analysis. Front. Mol. Biosci. 2, 4 (2015).
Bijlsma, S. et al. Large-scale human metabolomics studies: A strategy for data (pre-) processing and validation. Anal. Chem. 78, 567–574 (2006).
Worley, B. & Powers, R. Multivariate analysis in metabolomics. Curr. Metabolomics 1(1), 92–107 (2013).
Brown, A. J., Brown, G. D., Netea, M. G. & Gow, N. A. Metabolism impacts upon Candida immunogenicity and pathogenicity at multiple levels. Trends Microbiol. 22, 614–622 (2014).
Neumann, A. et al. Lipid alterations in human blood-derived neutrophils lead to formation of neutrophil extracellular traps. Eur. J. Cell Biol. 93, 347–354 (2014).
Assenov, Y., Ramírez, F., Schelhorn, S. E., Lengauer, T. & Albrecht, M. Computing topological parameters of biological networks. Bioinformatics 24, 282–284 (2008).
Duran-Pinedo, A. E. & Frias-Lopez, J. Beyond microbial community composition: functional activities of the oral microbiome in health and disease. Microbes Infect. 17, 505–516 (2015).
Wang, J. et al. Functions of sphingolipids in pathogenesis during host–pathogen interactions. Front. Microbiol. 12, 701041 (2021).
Wu, Y., Liu, Y., Gulbins, E. & Grassmé, H. The anti-infectious role of sphingosine inmicrobial diseases. Cells 10, 1105 (2021).
Veerman, E. C. et al. Phytosphingosine kills Candida albicans by disrupting its cell membrane. Biol. Chem. 391, 65–71 (2010).
Lee, M., Lee, S. Y. & Bae, Y. S. Functional roles of sphingolipids in immunity andtheir implication in disease. Exp. Mol. Med. 55, 1110–1130 (2023).
Tafesse, F. G. et al. Disruption of sphingolipid biosynthesis blocks phagocytosis of Candida albicans. PLoS Pathog. 11, e1005188 (2015).
Singh, A. & Del Poeta, M. Lipid signalling in pathogenic fungi. Cell. Microbiol. 13, 177–185 (2016).
Liu, X. H. et al. Metabolomics analysis identifies sphingolipids as key signaling moieties in appressorium morphogenesis and function in Magnaporthe oryzae. MBio 10, e01467-e1519 (2019).
Chalfant, C. E. & Spiegel, S. Sphingosine 1-phosphate and ceramide 1-phosphate: expanding roles in cell signaling. J. Cell Sci. 118, 4605–4612 (2005).
Urbanek, A. K. et al. The role of ergosterol and sphingolipids in the localization and activity of Candida albicans’ multidrug transporter Cdr1p and plasma membrane ATPase Pma1p. Int. J. Mol. Sci. 23, 9975 (2022).
Wu, G. et al. Proline and hydroxyproline metabolism: implications for animal and human nutrition. Amino Acids 40, 1053–1063 (2011).
Krishnan, N., Dickman, M. B. & Becker, D. F. Proline modulates the intracellular redox environment and protects mammalian cells against oxidative stress. Free Radic. Biol. Med. 44, 671–681 (2008).
Liao, X. H., Majithia, A., Huang, X. & Kimmel, A. R. Growth control via TOR kinase signaling, an intracellular sensor of amino acids and energy availability, with crosstalk potential to proline metabolism. Amino Acids 35, 761–770 (2008).
Grahl, N. et al. Mitochondrial activity and Cyr1 are key regulators of Ras1 activation of C albicans virulence pathways. PLoS Pathog. 11, e1005133 (2015).
Devries, M. C. et al. Leucine, not total protein, content of a supplement is the primary determinant of muscle protein anabolic responses in healthy older women. J. Nutr. 148, 1088–1095 (2018).
Holeček, M. Branched-chain amino acids in health and disease: metabolism, alterations in blood plasma, and as supplements. Nutr. Metab. (Lond.) 15, 33 (2018).
Kingsbury, J. M. & McCusker, J. H. Cytocidal amino acid starvation ofSaccharomyces cerevisiae and Candida albicans acetolactate synthase (ilv2{Delta}) mutants is influenced by the carbon source and rapamycin. Microbiology (Reading, England). 156, 929–939 (2010).
Garbe, E. & Vylkova, S. Role of amino acid metabolism in the virulence of human pathogenic fungi. Curr. Clin. Microbiol. Rep. 6, 108–119 (2019).
Sagar, N. A., Tarafdar, S., Agarwal, S., Tarafdar, A. & Sharma, S. Polyamines: Functions, metabolism, and role in human disease management. Med. Sci. (Basel) 9, 44 (2021).
Zhang, M., Borovikova, L. V., Wang, H., Metz, C. & Tracey, K. J. Spermine inhibition of monocyte activation and inflammation. Mol. Med. 5, 595–605 (1999).
Hesterberg, R. S., Cleveland, J. L. & Epling-Burnette, P. K. Role of polyamines in immune cell functions. Med. Sci. 6, 22 (2018).
Tang, G., Xia, H., Liang, J., Ma, Z. & Liu, W. Spermidine is critical for growth, development, environmental adaptation, and virulence in Fusarium graminearum. Front. Microbiol. 12, 765398 (2021).
Dorighetto Cogo, A. J. et al. Spermine modulates fungal morphogenesis and activates plasmamembrane H+-ATPase during yeast to hyphae transition. Biol. Open 27, bio029660 (2018).
Papackova, Z. & Cahova, M. Fatty acid signaling: the new function of intracellular lipases. Int. J. Mol. Sci. 16, 3831–3855 (2015).
Pan, J., Hu, C. & Yu, J. H. Lipid biosynthesis as an antifungal target. J. Fungi (Basel) 4, 50 (2018).
Sandager, L. et al. Storage lipid synthesis is non-essential in yeast. J. Biol. Chem. 277, 6478–6482 (2002).
Small, D. M. The effects of glyceride structure on absorption and metabolism. Ann. Rev. Nutr. 11, 413–434 (1991).
Gácser, A. et al. Lipase 8 affects thepathogenesis of Candida albicans. Infect. Immun. 75, 4710–4718 (2007).
Bagnat, M., Keränen, S., Shevchenko, A., Shevchenko, A. & Simons, K. Lipid rafts function in biosynthetic delivery of proteins to the cell surface in yeast. Proc. Natl. Acad. Sci. 97, 3254–3259 (2000).
Martin, S. W. & Konopka, J. B. Lipid raft polarization contributes to hyphal growth in Candida albicans. Eukaryot. Cel. 3, 675–684 (2004).
Vance, J. E. Phospholipid synthesis and transport in mammalian cells. Traffic 16, 1–18 (2015).
O’Donnell, V. B., Rossjohn, J. & Wakelam, M. J. Phospholipid signaling in innate immune cells. J. Clin. Invest. 128(7), 2670–2679 (2018).
Hasim, S. et al. Influence of phosphatidylserine and phosphatidylethanolamine on farnesol tolerance in Candida albicans. Yeast 35, 343–351 (2018).
Rella, A., Farnoud, A. M. & Del Poeta, M. Plasma membrane lipids and their role in fungal virulence. Prog. Lipid Res. 61, 63–72 (2016).
Dugail, I., Kayser, B. D. & Lhomme, M. Specific roles of phosphatidylglycerols in hosts and microbes. Biochimie 141, 47–53 (2017).
Nie, J. et al. A novel function of the human CLS1 in phosphatidylglycerol synthesis and remodeling. Biochim. Biophys. Acta. 1801, 438–445 (2010).
Su, X. & Dowhan, W. Regulation of cardiolipin synthase levels in Saccharomyces cerevisiae. Yeast 23, 279–291 (2006).
Han, T. L., Cannon, R. D. & Villas-Boas, S. G. Metabolome analysis during the morphological transition of Candida albicans. Metabolomics 8, 1204–1217 (2012).
De Craene, J. O., Bertazzi, D. L., Bär, S. & Friant, S. Phosphoinositides, major actors in membrane trafficking and lipid signaling pathways. Int. J. Mol. Sci. 18, 634 (2017).
Balla, T. Phosphoinositides: Tiny lipids with giant impact on cell regulation. Physiol. Rev. 93, 1019–1137 (2013).
Konopka, J. B. Plasma membrane phosphatidylinositol 4-phosphate is necessary for virulence of Candida albicans. MBio 13, e0036622 (2022).
Wang, J. et al. The role of phosphatidylserine on the membrane in immunity and blood coagulation. Biomark. Res. 10, 4 (2022).
Hasegawa, J. et al. A role of phosphatidylserine in the function of recycling endosomes. Front. Cell Dev. Biol. 24, 783857 (2021).
Chen, Y. L. et al. Phosphatidylserine synthase and phosphatidylserine decarboxylase are essential for cell wall integrity and virulence in Candida albicans. Mol. Microbiol. 75, 1112–1113 (2010).
Matos, G. S., Fernandes, C. M. & Del Poeta, M. Role of sphingolipids in the host-pathogen interaction. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 1868, 159384 (2023).
Singh, A. & Del Poeta, M. Sphingolipidomics: An important mechanistic tool for studying fungal pathogens. Front. Microbiol. 7, 501 (2016).
Bryan, A. M., Del Poeta, M. & Luberto, C. Sphingolipids as regulators of the phagocytic response to fungal infections. Mediators Inflamm. 2015, 640540 (2015).
Hartel, J. C., Merz, N. & Grösch, S. How sphingolipids affect T cells in the resolution of inflammation. Front Pharmacol. 13, 1002915 (2022).
Ren, W. et al. mTORC1 signaling and IL-17 expression: defining pathways and possible therapeutic targets. Eur. J. Immunol. 46, 291–299 (2016).
Handa, A. K., Fatima, T. & Mattoo, A. K. Polyamines: bio-molecules with diverse functions in plant and human health and disease. Front. Chem. 6, 10 (2018).
Pegg, A. E. Functions of polyamines in mammals. J. Biol. Chem. 291, 14904–14912 (2016).
Ilmarinen, P., Moilanen, E., Erjefält, J. S. & Kankaanranta, H. The polyamine spermine promotes survival and activation of human eosinophils. J. Allergy Clin. Immunol. 136, 482–4.e11 (2015).
Levy, M. et al. Microbiota-modulated metabolites shape the intestinal microenvironment by regulating NLRP6 inflammasome signaling. Cell 163, 1428–1443 (2015).
Haskó, G. et al. Spermine differentially regulates the production of interleukin-12 p40 and interleukin-10 and suppresses the release of the T helper 1 cytokine interferon-gamma. Shock 14, 144–149 (2000).
Vance, J. E. & Steenbergen, R. Metabolism and functions of phosphatidylserine. Prog. Lipid Res. 44, 207–234 (2005).
Maratou, E. et al. Glucose transporter expression on the plasma membrane of resting and activated white blood cells. Eur. J. Clin. Invest. 37, 282–290 (2007).
Hresko, R. C., Kraft, T. E., Quigley, A., Carpenter, E. P. & Hruz, P. W. Mammalian glucose transporter activity is dependent upon anionic and conical phospholipids. J. Biol. Chem. 291, 17271–17282 (2016).
Bergsson, G., Arnfinnsson, J., Steingrímsson, O. & Thormar, H. In vitro killing of Candida albicans by fatty acids and monoglycerides. Antimicrob. Agents Chemother. 45, 3209–3212 (2001).
Shareck, J., Nantel, A. & Belhumeur, P. Conjugated linoleic acid inhibits hyphal growth in Candida albicans by modulating Ras1p cellular levels and downregulating TEC1 expression. Eukaryot. Cell. 10, 565–577 (2011).
Lee, J. H., Kim, Y. G., Khadke, S. K. & Lee, J. Antibiofilm and antifungal activities of medium-chain fatty acids against Candida albicans via mimicking of the quorum-sensing molecule farnesol. Microb. Biotechnol. 14, 1353–1366 (2021).
Altieri, C., Cardillo, D., Bevilacqua, A. & Sinigaglia, M. Inhibition of Aspergillus spp. and Penicillium spp by fatty acids and their monoglycerides. J. Food Prot. 70, 1206–1212 (2007).
Odds, F. C., Brown, A. J. & Gow, N. A. Antifungal agents: Mechanism of action. Trends Microbiol. 11, 272–279 (2003).
Chapela, S., Congost, C., Alonso, M., Burgos, H. & Stella, C. Synergistic effect of dichloroacetate and fluconazole on the growth of Saccharomyces cerevisiae. J. Pharmacol. Clin. Toxicol. 10, 1162 (2022).
Pfaller, M. A., Riley, J. & Gerarden, T. Polyamine depletion and growth inhibition in Candida albicans and Candida tropicalis by alpha-difluoromethylornithine andcyclohexylamine. J. Med. Vet. Mycol. 26, 119–126 (1988).
Rossi, A. et al. Reassessing the use of undecanoic acid as a therapeutic strategy for treating fungal infections. Mycopathologia 186, 327–340 (2021).
Prasad, R., Shah, A. H. & Rawal, M. K. Antifungals: Mechanism of action and drug resistance. Adv. Exp. Med. Biol. 892, 327–349 (2016).
Seleem, D., Chen, E., Benso, B., Pardi, V. & Murata, R. M. In vitro evaluation of antifungal activity of monolaurin against Candida albicans biofilms. PeerJ 4, 1–17 (2016).
Jumina Nurmala, A. et al. Monomyristin and monopalmitin derivatives: Synthesis and evaluation as potential antibacterial and antifungal agents. Molecules 23, 3141 (2018).
Vandeputte, P., Ferrari, S. & Coste, A. T. Antifungal resistance and new strategies to control fungal infections. Int. J. Med. Microbiol. 2012, 1–26 (2012).
James, M. O. et al. Therapeutic applications of dichloroacetate and the role of glutathione transferase zeta-1. Pharmacol. Ther. 170, 166–180 (2017).
Netea, M. G., Joosten, L. A., van der Meer, J. W., Kullberg, B. J. & van de Veerdonk, F. L. Immune defence against Candida fungal infections. Nat. Rev. Immunol. 15, 630–642 (2015).
Domínguez-Andrés, J. et al. Rewiring monocyte glucose metabolism via C-type lectin signaling protects against disseminated candidiasis. PLoS Pathog. 13, e1006632 (2017).
Vikram, A., Jesudhasan, P. R., Pillai, S. D. & Patil, B. S. Isolimonic acid interferes with Escherichia coli O157:H7 biofilm and TTSS in QseBC and QseA dependent fashion. BMC Microbiol. 12, 261 (2012).
Guimarães, A. & Venâncio, A. The potential of fatty acids and their derivatives as antifungal agents: a review. Toxins (Basel) 14(3), 188 (2022).
Badr, M. M., Qinna, N. A., Qadan, F. & Matalka, K. Z. Dichloroacetate modulates cytokines toward T helper 1 function via induction of the interleukin-12-interferon-γ pathway. Oncol. Targ. Ther. 7, 193–201 (2014).
Guenther, G. G. et al. Ceramide starves cells to death by downregulating nutrient transporter proteins. Proc. Natl. Acad. Sci. USA 105, 17402–17407 (2008).
Zheng, W. et al. Ceramides and other bioactive sphingolipid backbones in health and disease: lipidomic analysis, metabolism and roles in membrane structure, dynamics, signaling and autophagy. Biochim. Biophys. Acta 1758, 1864–1884 (2006).
Barnes, V. M. et al. Acceleration of purine degradation by periodontal diseases. J. Dent. Res. 88, 851–855 (2009).
Hynne, H. et al. Saliva metabolomics in dry mouth patients with head and neck cancer or Sjögren’s syndrome. Cells 11(3), 323 (2022).
Kanehisa, M., Furumichi, M., Sato, Y., Ishiguro-Watanabe, M. & Tanabe, M. KEGG: integrating viruses and cellular organisms. Nucleic Acids Res. 49(D1), D545–D551 (2021).
Chen, W. et al. A novel integrated method for large-scale detection, identification, and quantification of widely targeted metabolites: application in the study of rice metabolomics. Mol. Plant. 6(6), 1769–1780 (2013).
Tamai, R. & Kiyoura, Y. Candida infections: the role of saliva in oral health—a narrative review. Microorganisms 13(4), 717 (2025).
de Sá Alves, M. et al. Identification of possible salivary metabolic biomarkers and altered metabolic pathways in South American patients diagnosed with oral squamous cell carcinoma. Metabolites 11, 650 (2021).
Tanemura, Y., Murakami, K., Haneda, M. & Yoshino, M. Polyamine enhances the regeneration of reduced glutathione by the activation of NADP-dependent dehydrogenases in yeast. Biomed. Res. 25, 69–74 (2004).
Mira, N. P. et al. Working Group 3 of the COST Action EuroMicropH. On the potential role of naturally occurring carboxylic organic acids as anti-infective agents: opportunities and challenges. Int. J. Infect. Dis. 140, 119–123 (2024).
Gutierrez, D. et al. Antibiotic-induced gut metabolome and microbiome alterations increase the susceptibility to Candida albicans colonization in the gastrointestinal tract. FEMS Microbiol. Ecol. 96(1), 187 (2020).
Ananieva, E. A., Powell, J. D. & Hutson, S. M. Leucine metabolism in T cell activation: mTOR signaling and beyond. Adv Nutr. 7(4), 798S-805S (2016).
Jump, R. L. et al. Metabolomics analysis identifies intestinal microbiota-derived biomarkers of colonization resistance in clindamycin-treated mice. PLoS ONE 9(7), e101267 (2014).
Heng, X., Jiang, Y. & Chu, W. Influence of fluconazole administration on gut microbiome, intestinal barrier, and immune response in mice. Antimicrob. Agents Chemother. 65(6), e02552-e2620 (2021).
Ogretmen, B. & Hannun, Y. A. Biologically active sphingolipids in cancer pathogenesis and treatment. Nat. Rev. Cancer. 4(8), 604–616 (2004).
Liu, J., Beckman, B. S. & Foroozesh, M. A review of ceramide analogs as potential anticancer agents. Future Med Chem. 5(12), 1405–1421 (2013).
Chen, W. et al. Downregulation of ceramide synthase 1 promotes oral cancer through endoplasmic reticulum stress. Int. J. Oral Sci. 13, 10 (2021).
Beloribi-Djefaflia, S., Vasseur, S. & Guillaumond, F. Lipid metabolic reprogramming in cancer cells. Oncogenesis 5, e189 (2016).
Carballeira, N. M. New advances in fatty acids as antimalarial, antimycobacterial and antifungal agents. Prog. Lipid Res. 47(1), 50–61 (2008).
Gertsch, J. Immunomodulatory lipids in plants: plant fatty acid amides and the human endocannabinoid system. Planta Med. 74(6), 638–650 (2008).
Wei, K. et al. Potential changes in microorganisms and metabolites associated with oral cancer: a preliminary study. BMC Cancer 25(1), 611 (2025).
Chang, J. et al. 12/15 Lipoxygenase regulation of colorectal tumorigenesis is determined by the relative tumor levels of its metabolite 12-HETE and 13-HODE in animal models. Oncotarget 6(5), 2879–2888 (2015).
Park, H. G. et al. A rare eicosanoid precursor analogue, sciadonic acid (5Z,11Z,14Z–20:3), detected in vivo in hormone positive breast cancer tissue. Prostaglandins Leukot Essent Fatty Acids 134, 1–6 (2018).
Murray-Stewart, T. R., Woster, P. M. & Casero, R. A. Jr. Targeting polyamine metabolism for cancer therapy and prevention. Biochem. J. 473(19), 2937–2953 (2016).
Tse, R. T., Wong, C. Y., Chiu, P. K. & Ng, C. F. The potential role of spermine and its acetylated derivative in human malignancies. Int. J. Mol. Sci. 23(3), 1258 (2022).
Liu, X. et al. The significant role of amino acid metabolic reprogramming in cancer. Cell Commun. Signal. 22(1), 380 (2024).
Chen, J., Cui, L., Lu, S. & Xu, S. Amino acid metabolism in tumor biology and therapy. Cell Death Dis. 15(1), 42 (2024).
Akbari, Z., Dijojin, R. T., Zamani, Z., Hosseini, R. H. & Arjmand, M. Aromatic amino acids play a harmonizing role in prostate cancer: a metabolomics-based cross-sectional study. Int. J. Reprod. Biomed. 19(8), 741–750 (2021).
Phang, J. M., Liu, W., Hancock, C. & Christian, K. J. The proline regulatory axis and cancer. Front. Oncol. 2, 60 (2012).
Jia, Y., Chen, X., Guo, H., Zhang, B. & Liu, B. Comprehensive characterization of β-alanine metabolism-related genes in HCC identified a novel prognostic signature related to clinical outcomes. Aging (Albany NY). 16(8), 7073–7100 (2024).
Niu, L. et al. 1-Monopalmitin promotes lung cancer cells apoptosis through PI3K/Akt pathway in vitro. Environ. Toxicol. 38(11), 2621–2631 (2023).
Küpeli Akkol, E., Genç, Y., Karpuz, B., Sobarzo-Sánchez, E. & Capasso, R. Coumarins and coumarin-related compounds in pharmacotherapy of cancer. Cancers (Basel) 12(7), 1959 (2020).
Hunt, S., Thyagarajan, A. & Sahu, R. P. Dichloroacetate and salinomycin as therapeutic agents in cancer. Med. Sci. 13(2), 47 (2025).
Yavuz, M. & Demircan, T. Hydroquinidine demonstrates remarkable antineoplastic effects on non-small cell lung cancer cells. Curr. Mol. Med. 24(9), 1159–1168 (2024).
Yavuz, M., Şahin, B., Baykal, A. T. & Demircan, T. Hydroquinidine displays a significant anticarcinogenic activity in breast and ovarian cancer cells via inhibiting cell-cycle and stimulating apoptosis. Turk. J. Biol. 47(1), 44–60 (2023).
DOSM. DOSM (updated 15 July 2020). https://www.dosm.gov.my/v1/index.php?r=column/cthemeByCat%26cat=155%26bul_id=OVByWjg5YkQ3MWFZRTN5bDJiaEVhZz09%26menu_id=L0pheU43NWJwRWVSZklWdzQ4TlhUUT09. (2020).
BAPEN. Redditch, (2011).
Jager, D. H. J., Bots, C. P., Forouzanfar, T. & Brand, H. S. Clinical oral dryness score: Evaluation of a new screening method for oral dryness. Odontology 106, 439–444 (2018).
Osailan, S. M., Pramanik, R., Shirlaw, P., Proctor, G. B. & Challacombe, S. J. Clinical assessment of oral dryness: development of a scoring system related to salivary flow and mucosal wetness. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 114, 597–603 (2012).
Imabayashi, Y. et al. Molecular analysis of fungal populations in patients with oral candidiasis using next-generation sequencing. Sci. Rep. 6, 1–8 (2016).
Khosravi, Y., Dieye, Y., Loke, M. F., Goh, K. L. & Vadivelu, J. Streptococcus mitis induces conversion of Helicobacter pylori to coccoid cells during co-culture in vitro. PLoS ONE 9, e112214 (2014).
Villas-Bôas, S. G., Højer-Pedersen, J., Akesson, M., Smedsgaard, J. & Nielsen, J. Global metabolite analysis of yeast: Evaluation of sample preparation methods. Yeast (Chichester, England) 22, 1155–1169 (2005).
Wong, E. H. J. et al. Metabolomic analysis of low and high biofilm-forming Helicobacter pylori strains. Sci. Rep. 8, 1409 (2018).
Chong, J. et al. MetaboAnalyst 4.0: towards more transparent and integrative metabolomics analysis. Nucleic Acids Res. 46, W486–W494 (2018).
Quindós, G. et al. Therapeutic tools for oral candidiasis: Current and new antifungal drugs. Med. Oral Patol. Oral Cir. Bucal. 24, e172–e180 (2019).





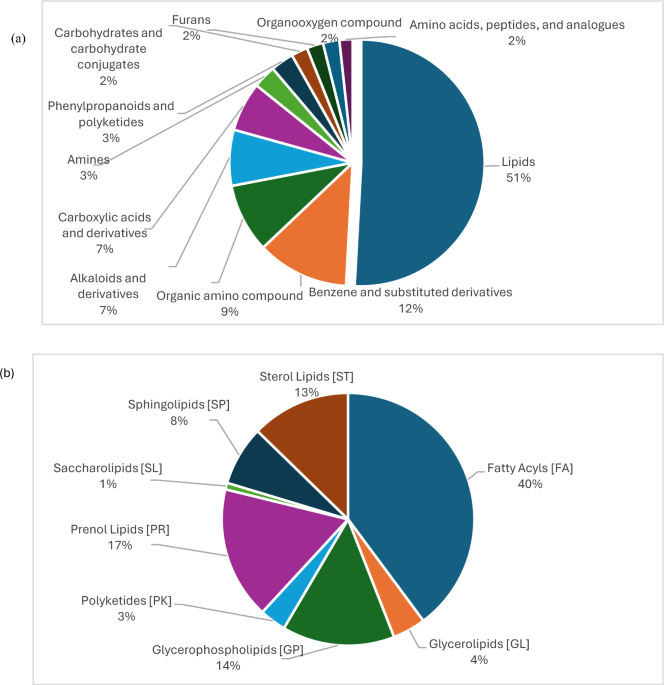












Leave a Reply