Siddiqui ZA, Walker A, Pirwani MM, Tahiri M, Syed I. Allergic rhinitis: diagnosis and management. Br J Hosp Med (Lond). 2022;83(2):1–9.
Meng Y, Wang C, Zhang L. Recent developments and highlights in allergic rhinitis. Allergy. 2019;74(12):2320–8.
Wise SK, Damask C, Roland LT, Ebert C, Levy JM, Lin S, et al. International consensus statement on allergy and rhinology: Allergic rhinitis – 2023. Int Forum Allergy Rhinol. 2023;13(4):293–859.
Brożek JL, Bousquet J, Agache I, Agarwal A, Bachert C, Bosnic-Anticevich S, et al. Allergic Rhinitis and its Impact on Asthma (ARIA) guidelines-2016 revision. J Allergy Clin Immunol. 2017;140(4):950–8.
Bousquet J, Anto JM, Bachert C, Baiardini I, Bosnic-Anticevich S, Walter Canonica G, et al. Allergic rhinitis. Nat Rev Dis Primers. 2020;6(1):95.
Wang XD, Zheng M, Lou HF, Wang CS, Zhang Y, Bo MY, et al. An increased prevalence of self-reported allergic rhinitis in major Chinese cities from 2005 to 2011. Allergy. 2016;71(8):1170–80.
Scadding GK, Kariyawasam HH, Scadding G, Mirakian R, Buckley RJ, Dixon T, et al. BSACI guideline for the diagnosis and management of allergic and non-allergic rhinitis (Revised Edition 2017; First edition 2007). Clin Exp Allergy. 2017;47(7):856–89.
Tosca MA, Trincianti C, Naso M, Nosratian V, Ciprandi G. Treatment of Allergic Rhinitis in Clinical Practice. Curr Pediatr Rev. 2024;20(3):271–7.
Saulyte J, Regueira C, Montes-Martínez A, Khudyakov P, Takkouche B. Active or passive exposure to tobacco smoking and allergic rhinitis, allergic dermatitis, and food allergy in adults and children: a systematic review and meta-analysis. PLoS Med. 2014;11(3):e1001611.
Greiner AN, Hellings PW, Rotiroti G, Scadding GK. Allergic rhinitis. Lancet. 2011;378(9809):2112–22.
Okubo K, Kurono Y, Ichimura K, Enomoto T, Okamoto Y, Kawauchi H, et al. Japanese guidelines for allergic rhinitis 2020. Allergol Int. 2020;69(3):331–45.
Chinese guidelines for diagnosis and treatment of allergic rhinitis. Zhonghua Er Bi Yan Hou Tou Jing Wai Ke Za Zhi, 2016. 51(1): p. 6–24.
He Y, Chen Y, Xu S, Luo Y, Qin F, Hu W. Pathogenesis and Key Cells in Allergic Rhinitis. Int Arch Allergy Immunol. 2025;186:418–29.
Qin Y, Gao C, Luo J. Metabolism Characteristics of Th17 and Regulatory T Cells in Autoimmune Diseases. Front Immunol. 2022;13:828191.
Siddhuraj P, Nordström FU, Jogdand P, Elangovan G, Mori M, Bjermer L, et al. Histology-based blood leukocyte profiling reveals parallel Th2 and Th17 signatures in seasonal allergic rhinitis. Acta Otolaryngol. 2022;142(9-12):696–704.
Xu T, Yang Y. Clinical value of serum IL-27 in allergic rhinitis patients and its relationship with Treg associated cytokine levels. Hum Immunol. 2023;84(2):130–5.
Lee GR. The Balance of Th17 versus Treg Cells in Autoimmunity. Int J Mol Sci. 2018;19(3):730.
Huang X, Chen Y, Zhang F, Yang Q, Zhang G. Peripheral Th17/Treg cell-mediated immunity imbalance in allergic rhinitis patients. Braz J Otorhinolaryngol. 2014;80(2):152–5.
Dong C. Cytokine Regulation and Function in T Cells. Annu Rev Immunol. 2021;39:51–76.
Kimura A, Kishimoto T. IL-6: regulator of Treg/Th17 balance. Eur J Immunol. 2010;40(7):1830–5.
Long D, Chen Y, Wu H, Zhao M, Lu Q. Clinical significance and immunobiology of IL-21 in autoimmunity. J Autoimmun. 2019;99:1–14.
Cerboni S, Gehrmann U, Preite S, Mitra S. Cytokine-regulated Th17 plasticity in human health and diseases. Immunology. 2021;163(1):3–18.
Harris F, Berdugo YA, Tree T. IL-2-based approaches to Treg enhancement. Clin Exp Immunol. 2023;211(2):149–63.
Peter J, Sabu V, Aswathy IS, Krishnan S, Lal Preethi SS, Simon M, et al. Dietary amaranths modulate the immune response via balancing Th1/Th2 and Th17/Treg response in collagen-induced arthritis. Mol Cell Biochem. 2020;472(1-2):57–66.
Wang G, Su Z, Li H, Xiao L, Li C, Lian G. The role of metabolism in Th17 cell differentiation and autoimmune diseases. Int Immunopharmacol. 2022;103:108450.
Xiong H, Wei L, Peng B. IL-17 stimulates the production of the inflammatory chemokines IL-6 and IL-8 in human dental pulp fibroblasts. Int Endod J. 2015;48(6):505–11.
Ivanov II, McKenzie BS, Zhou L, Tadokoro CE, Lepelley A, Lafaille JJ, et al. The orphan nuclear receptor RORgammat directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell. 2006;126(6):1121–33.
Liu S, Cao Y, Cui K, Ren G, Zhao T, Wang X, et al. Regulation of T helper cell differentiation by the interplay between histone modification and chromatin interaction. Immunity. 2024;57(5):987–1004.e5.
Lee S, Kim J, Min H, Seong RH. RORγt-driven T(H)17 Cell Differentiation Requires Epigenetic Control by the Swi/Snf Chromatin Remodeling Complex. iScience. 2020;23(5):101106.
Zhang F, Meng G, Strober W. Interactions among the transcription factors Runx1, RORgammat and Foxp3 regulate the differentiation of interleukin 17-producing T cells. Nat Immunol. 2008;9(11):1297–306.
Pham D, Silberger DJ, Nguyen KN, Gao M, Weaver CT, Hatton RD. Batf stabilizes Th17 cell development via impaired Stat5 recruitment of Ets1-Runx1 complexes. Embo J. 2023;42(8):e109803.
Mudter J, Yu J, Zufferey C, Brüstle A, Wirtz S, Weigmann B, et al. IRF4 regulates IL-17A promoter activity and controls RORγt-dependent Th17 colitis in vivo. Inflamm Bowel Dis. 2011;17(6):1343–58.
Camporeale A, Poli V. IL-6, IL-17 and STAT3: a holy trinity in auto-immunity? Front Biosci (Landmark Ed), 2012. 17(6):2306–26.
Bettelli E, Carrier Y, Gao W, Korn T, Strom TB, Oukka M, et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature. 2006;441(7090):235–8.
Mesas-Fernández A, Bodner E, Hilke FJ, Meier K, Ghoreschi K, Solimani F. Interleukin-21 in autoimmune and inflammatory skin diseases. Eur J Immunol. 2023;53(4):e2250075.
Sharma A, Upadhyay DK, Gupta GD, Narang RK, Rai VK. IL-23/Th17 Axis: A Potential Therapeutic Target of Psoriasis. Curr Drug Res Rev. 2022;14(1):24–36.
Yang XP, Ghoreschi K, Steward-Tharp SM, Rodriguez-Canales J, Zhu J, Grainger JR, et al. Opposing regulation of the locus encoding IL-17 through direct, reciprocal actions of STAT3 and STAT5. Nat Immunol. 2011;12(3):247–54.
Yoon JH, Sudo K, Kuroda M, Kato M, Lee IK, Han JS, et al. Phosphorylation status determines the opposing functions of Smad2/Smad3 as STAT3 cofactors in TH17 differentiation. Nat Commun. 2015;6:7600.
He C, Li Y, Gan L, Lin Y, Zhang B, Ma L, et al. Notch signaling regulates Th17 cells differentiation through PI3K/AKT/mTORC1 pathway and involves in the thyroid injury of autoimmune thyroiditis. J Endocrinol Invest. 2024;47(8):1971–86.
Zhang RY, Zhang XS, Lu C, Wang ZR, Shi Y, Wang YG, et al. TLR4-MyD88-NF-κB signaling imbalances Th17 and Treg cells in thymoma with myasthenia gravis. Eur Rev Med Pharmacol Sci. 2023;27(21):10342–64.
Dang EV, Barbi J, Yang HY, Jinasena D, Yu H, Zheng Y, et al. Control of T(H)17/T(reg) balance by hypoxia-inducible factor 1. Cell. 2011;146(5):772–84.
Geng J, Yu S, Zhao H, Sun X, Li X, Wang P, et al. The transcriptional coactivator TAZ regulates reciprocal differentiation of T(H)17 cells and T(reg) cells. Nat Immunol. 2017;18(7):800–12.
Sugiura A, Andrejeva G, Voss K, Heintzman DR, Xu X, Madden MZ, et al. MTHFD2 is a metabolic checkpoint controlling effector and regulatory T cell fate and function. Immunity. 2022;55(1):65–81.e9.
Yao R, Ma YL, Liang W, Li HH, Ma ZJ, Yu X, et al. MicroRNA-155 modulates Treg and Th17 cells differentiation and Th17 cell function by targeting SOCS1. PLoS One. 2012;7(10):e46082.
Wang M, Jin XE, Xue CM, Liao QB, Pu YF, Liu GQ, et al. MiR-155-5p facilitates acute lung injury progression via Th17 cells through the ETS-1/STAT3 pathway. Gene. 2025;963:149605.
Hou J, Hu X, Chen B, Chen X, Zhao L, Chen Z, et al. miR-155 targets Est-1 and induces ulcerative colitis via the IL-23/17/6-mediated Th17 pathway. Pathol Res Pract. 2017;213(10):1289–95.
Escobar TM, Kanellopoulou C, Kugler DG, Kilaru G, Nguyen CK, Nagarajan V, et al. miR-155 activates cytokine gene expression in Th17 cells by regulating the DNA-binding protein Jarid2 to relieve polycomb-mediated repression. Immunity. 2014;40(6):865–79.
Tian K, Xu W, Chen M, Deng F. miR-155 promotes Th17 differentiation by targeting FOXP3 to aggravate inflammation in MRSA pneumonia. Cytokine. 2024;180:156662.
Zhang G, Wu S, Xia G. MiR-326 sponges TET2 triggering imbalance of Th17/Treg differentiation to exacerbate pyroptosis of hepatocytes in concanavalin A-induced autoimmune hepatitis. Ann Hepatol. 2024;29(2):101183.
Liu Y, Cui X, Wang S, Liu J, Zhao N, Huang M, et al. Elevated MicroRNA-326 Levels Regulate the IL-23/IL-23R/Th17 Cell Axis in Hashimoto’s Thyroiditis by Targeting a Disintegrin and Metalloprotease 17. Thyroid. 2020;30(9):1327–37.
Zhao N, Zou H, Qin J, Fan C, Liu Y, Wang S, et al. MicroRNA-326 contributes to autoimmune thyroiditis by targeting the Ets-1 protein. Endocrine. 2018;59(1):120–9.
Liu C, Zou Q. miR-21 Regulates Immune Balance Mediated by Th17/Treg in Peripheral Blood of Septic Rats during the Early Phase through Apoptosis Pathway. Biochem Res Int. 2022;2022:9948229.
He S, Sun S, Lu J, Chen L, Mei X, Li L, et al. The effects of the miR-21/SMAD7/TGF-β pathway on Th17 cell differentiation in COPD. Sci Rep. 2021;11(1):6338.
Murugaiyan G, da Cunha AP, Ajay AK, Joller N, Garo LP, Kumaradevan S, et al. MicroRNA-21 promotes Th17 differentiation and mediates experimental autoimmune encephalomyelitis. J Clin Invest. 2015;125(3):1069–80.
Ichiyama K, Gonzalez-Martin A, Kim BS, Jin HY, Jin W, Xu W, et al. The MicroRNA-183-96-182 Cluster Promotes T Helper 17 Cell Pathogenicity by Negatively Regulating Transcription Factor Foxo1 Expression. Immunity. 2016;44(6):1284–98.
Singer M, Elsayed AM, Husseiny MI. Regulatory T-cells: The Face-off of the Immune Balance. Front Biosci (Landmark Ed.). 2024;29(11):377
Kanamori M, Nakatsukasa H, Okada M, Lu Q, Yoshimura A. Induced Regulatory T Cells: Their Development, Stability, and Applications. Trends Immunol. 2016;37(11):803–11.
Lifshitz GV, Zhdanov DD, Lokhonina AV, Eliseeva DD, Lyssuck EY, Zavalishin IA, et al. Ex vivo expanded regulatory T cells CD4(+)CD25(+)FoxP3(+)CD127(Low) develop strong immunosuppressive activity in patients with remitting-relapsing multiple sclerosis. Autoimmunity. 2016;49(6):388–96.
Henríquez C, Morán G, Carrasco C, Sarmiento J, Barría M, Folch H, et al. Modulatory role of regulatory T cells in a murine model of severe equine asthma. BMC Vet Res. 2017;13(1):117.
Xu L, Kitani A, Stuelten C, McGrady G, Fuss I, Strober W. Positive and negative transcriptional regulation of the Foxp3 gene is mediated by access and binding of the Smad3 protein to enhancer I. Immunity. 2010;33(3):313–25.
Iizuka-Koga M, Nakatsukasa H, Ito M, Akanuma T, Lu Q, Yoshimura A. Induction and maintenance of regulatory T cells by transcription factors and epigenetic modifications. J Autoimmun. 2017;83:113–21.
Xiao Y, Li B, Zhou Z, Hancock WW, Zhang H, Greene MI. Histone acetyltransferase mediated regulation of FOXP3 acetylation and Treg function. Curr Opin Immunol. 2010;22(5):583–91.
Zhang H, Xiao Y, Zhu Z, Li B, Greene MI. Immune regulation by histone deacetylases: a focus on the alteration of FOXP3 activity. Immunol Cell Biol. 2012;90(1):95–100.
Ouyang W, Beckett O, Ma Q, Paik JH, DePinho RA, Li MO. Foxo proteins cooperatively control the differentiation of Foxp3+ regulatory T cells. Nat Immunol. 2010;11(7):618–27.
Abdullah L, Hills LB, Winter EB, Huang YH. Diverse Roles of Akt in T cells. Immunometabolism. 2021;3(1):e210007.
Wu C, Chen Z, Xiao S, Thalhamer T, Madi A, Han T, et al. SGK1 Governs the Reciprocal Development of Th17 and Regulatory T Cells. Cell Rep. 2018;22(3):653–65.
Gibson SA, Yang W, Yan Z, Qin H, Benveniste EN. CK2 Controls Th17 and Regulatory T Cell Differentiation Through Inhibition of FoxO1. J Immunol. 2018;201(2):383–92.
Jhun J, Woo JS, Lee SH, Jeong JH, Jung K, Hur W. et al. GRIM19 Impedes Obesity by Regulating Inflammatory White Fat Browning and Promoting Th17/Treg Balance. Cells. 2021;10(1):162.
Murawski MR, Litherland SA, Clare-Salzler MJ, Davoodi-Semiromi A. Upregulation of Foxp3 expression in mouse and human Treg is IL-2/STAT5 dependent: implications for the NOD STAT5B mutation in diabetes pathogenesis. Ann N Y Acad Sci. 2006;1079:198–204.
Thangavelu G, Andrejeva G, Bolivar-Wagers S, Jin S, Zaiken MC, Loschi M, et al. Retinoic acid signaling acts as a rheostat to balance Treg function. Cell Mol Immunol. 2022;19(7):820–33.
Xiao S, Yan Y, Shao M, Zhou X, Niu Z, Wu Y, et al. Kuijieling decoction regulates the Treg/Th17 cell balance in ulcerative colitis through the RA/RARα signaling pathway. J Ethnopharmacol. 2024;318(Pt A):116909.
Yu J, Mei J, Zuo D, Zhang M, Yu S, Li F, et al. Inflammatory factor-mediated miR-155/SOCS1 signaling axis leads to Treg impairment in systemic lupus erythematosus. Int Immunopharmacol. 2024;141:113013.
Zeng Q, Liu W, Luo R, Lu G. MicroRNA-181a and microRNA-155 are involved in the regulation of the differentiation and function of regulatory T cells in allergic rhinitis children. Pediatr Allergy Immunol. 2019;30(4):434–42.
Lu J, Wang W, Li P, Wang X, Gao C, Zhang B, et al. MiR-146a regulates regulatory T cells to suppress heart transplant rejection in mice. Cell Death Discov. 2021;7(1):165.
Zhang Y, Yang Y, Guo J, Cui L, Yang L, Li Y, et al. miR-146a enhances regulatory T-cell differentiation and function in allergic rhinitis by targeting STAT5b. Allergy. 2022;77(2):550–8.
Zhou Q, Haupt S, Kreuzer JT, Hammitzsch A, Proft F, Neumann C, et al. Decreased expression of miR-146a and miR-155 contributes to an abnormal Treg phenotype in patients with rheumatoid arthritis. Ann Rheum Dis. 2015;74(6):1265–74.
Jeker LT, Zhou X, Gershberg K, de Kouchkovsky D, Morar MM, Stadthagen G, et al. MicroRNA 10a marks regulatory T cells. PLoS One. 2012;7(5):e36684.
Takahashi H, Kanno T, Nakayamada S, Hirahara K, Sciumè G, Muljo SA, et al. TGF-β and retinoic acid induce the microRNA miR-10a, which targets Bcl-6 and constrains the plasticity of helper T cells. Nat Immunol. 2012;13(6):587–95.
Dong J, Huth WJ, Marcel N, Zhang Z, Lin LL, Lu LF. miR-15/16 clusters restrict effector Treg cell differentiation and function. J Exp Med. 2023;220:e20230321.
Rouas R, Fayyad-Kazan H, El Zein N, Lewalle P, Rothé F, Simion A, et al. Human natural Treg microRNA signature: role of microRNA-31 and microRNA-21 in FOXP3 expression. Eur J Immunol. 2009;39(6):1608–18.
Zhang L, Ke F, Liu Z, Bai J, Liu J, Yan S, et al. MicroRNA-31 negatively regulates peripherally derived regulatory T-cell generation by repressing retinoic acid-inducible protein 3. Nat Commun. 2015;6:7639.
Lu Y, Gao J, Zhang S, Gu J, Lu H, Xia Y, et al. miR-142-3p regulates autophagy by targeting ATG16L1 in thymic-derived regulatory T cell (tTreg). Cell Death Dis. 2018;9(3):290.
Gao J, Gu J, Pan X, Gan X, Ju Z, Zhang S, et al. Blockade of miR-142-3p promotes anti-apoptotic and suppressive function by inducing KDM6A-mediated H3K27me3 demethylation in induced regulatory T cells. Cell Death Dis. 2019;10(5):332.
Lu Y, Li Y, Liu Q, Tian N, Du P, Zhu F, et al. MondoA-Thioredoxin-Interacting Protein Axis Maintains Regulatory T-Cell Identity and Function in Colorectal Cancer Microenvironment. Gastroenterology. 2021;161(2):575–591.e16.
Hinshaw DC, Benavides GA, Metge BJ, Swain CA, Kammerud SC, Alsheikh HA, et al. Hedgehog Signaling Regulates Treg to Th17 Conversion Through Metabolic Rewiring in Breast Cancer. Cancer Immunol Res. 2023;11(5):687–702.
Yang YH, Istomine R, Alvarez F, Al-Aubodah TA, Shi XQ, Takano T, et al. Salt Sensing by Serum/Glucocorticoid-Regulated Kinase 1 Promotes Th17-like Inflammatory Adaptation of Foxp3(+) Regulatory T Cells. Cell Rep. 2020;30(5):1515–1529.e4.
Cai J, Wang F, Shi SL. Expression of serum miR-135a in patients with allergic rhinitis and its relationship with Treg/Th17 balance. Kaohsiung J Med Sci. 2025;41(1):e12918.
Ueda S, Miura K, Kawasaki H, Ogata S, Yamasaki N, Miura S. et al. Th17-Dependent Nasal Hyperresponsiveness Is Mitigated by Steroid Treatment. Biomolecules. 2022;12(5):674.
Amin K, Issa SM, Ali KM, Aziz MI, Hama Amieen HM, Bystrom J, et al. Evidence for eosinophil and IL-17 mediated inflammation in allergic rhinitis. Clin Mol Allergy. 2020;18:6.
Gu ZW, Wang YX, Cao ZW. Neutralization of interleukin-17 suppresses allergic rhinitis symptoms by downregulating Th2 and Th17 responses and upregulating the Treg response. Oncotarget. 2017;8(14):22361–9.
Navarro-Compán V, Puig L, Vidal S, Ramírez J, Llamas-Velasco M, Fernández-Carballido C, et al. The paradigm of IL-23-independent production of IL-17F and IL-17A and their role in chronic inflammatory diseases. Front Immunol. 2023;14:1191782.
Chung SH, Ye XQ, Iwakura Y. Interleukin-17 family members in health and disease. Int Immunol. 2021;33(12):723–9.
Gao S, Yu L, Zhang J, Li X, Zhou J, Zeng P, et al. Expression and clinical significance of VCAM-1, IL-6, and IL-17A in patients with allergic rhinitis. Ann Palliat Med. 2021;10(4):4516–22.
Sun H, Liu B, Ma W, Ji D, Guan B. Changing Characteristics of Treg/Th17 Cells in the Nasal Mucosa of a Mouse Model of Allergic Rhinitis and the Effect of Intervention with Anti-IL-17 Antibody. Discov Med. 2024;36(185):1260–7.
Guo C, Chen G, Ge R. IL-23, rather than IL-17, is crucial for the development of ovalbumin-induced allergic rhinitis. Mol Immunol. 2015;67(2 Pt B):436–43.
Hirano T. IL-6 in inflammation, autoimmunity and cancer. Int Immunol. 2021;33(3):127–48.
Song XY, Sun Q, Wei SZ, Wang HR, Wang Y, Zhang WB, et al. IL-6 mediates olfactory dysfunction in a mouse model of allergic rhinitis. Brain Res. 2024;1833:148885.
Cui Q, Li J, Wang J. The Assessment of TNF-α Gene Polymorphism Association with the Risk of Allergic Rhinitis in the Chinese Han Population. Int J Gen Med. 2021;14:5183–92.
Iwasaki M, Saito K, Takemura M, Sekikawa K, Fujii H, Yamada Y, et al. TNF-alpha contributes to the development of allergic rhinitis in mice. J Allergy Clin Immunol. 2003;112(1):134–40.
Göschl L, Scheinecker C, Bonelli M. Treg cells in autoimmunity: from identification to Treg-based therapies. Semin Immunopathol. 2019;41(3):301–14.
Nasiri R, Hirbod-Mobarakeh A, Movahedi M, Farhadi E, Ansaripour B, Amirzargar AA, et al. Gene polymorphisms of interleukin-10 and transforming growth factor beta in allergic rhinitis. Allergol Immunopathol (Madr). 2016;44(2):125–30.
Kang MG, Han SW, Kang HR, Hong SJ, Kim DH, Choi JH. Probiotic NVP-1703 Alleviates Allergic Rhinitis by Inducing IL-10 Expression: A Four-week Clinical Trial. Nutrients. 2020;12(5):1427.
Qu Y, Yamada T, Aoi N, Morikura I, Fuchiwaki T, Hotta Y, et al. Sublingual Immunotherapy Attenuates Nasal Symptoms Upon Allergen Exposure in Murine Allergic Rhinitis Model via an Induction of IL-10 producing T cells in Submandibular Lymph Node. Ann Otol Rhinol Laryngol. 2019;128:26s–35s.
Ouyang Y, Miyata M, Hatsushika K, Ohnuma Y, Katoh R, Ogawa H, et al. TGF-beta signaling may play a role in the development of goblet cell hyperplasia in a mouse model of allergic rhinitis. Allergol Int. 2010;59(3):313–9.
Pavón-Romero GF, Ramírez-Jiménez MI, Ram¡rez-Jim‚nez F, Melgoza-Ruiz E, Serrano-Pérez NH, Teran LM. Allergen Immunotherapy: Current and Future Trends. Cells. 2022;11(2):212.
Meng XT, Shi YY, Zhang H, Zhou HY. The Role of Th17 Cells and IL-17 in Th2 Immune Responses of Allergic Conjunctivitis. J Ophthalmol. 2020;2020:6917185.
Oboki K, Ohno T, Saito H, Nakae S. Th17 and allergy. Allergol Int. 2008;57(2):121–34.
Kinyanjui MW, Shan J, Nakada EM, Qureshi ST, Fixman ED. Dose-dependent effects of IL-17 on IL-13-induced airway inflammatory responses and airway hyperresponsiveness. J Immunol. 2013;190(8):3859–68.
Wakashin H, Hirose K, Maezawa Y, Kagami S, Suto A, Watanabe N, et al. IL-23 and Th17 cells enhance Th2-cell-mediated eosinophilic airway inflammation in mice. Am J Respir Crit Care Med. 2008;178(10):1023–32.
El-Behi M, Ciric B, Dai H, Yan Y, Cullimore M, Safavi F, et al. The encephalitogenicity of T(H)17 cells is dependent on IL-1- and IL-23-induced production of the cytokine GM-CSF. Nat Immunol. 2011;12(6):568–75.
Griseri T, Arnold IC, Pearson C, Krausgruber T, Schiering C, Franchini F, et al. Granulocyte Macrophage Colony-Stimulating Factor-Activated Eosinophils Promote Interleukin-23 Driven Chronic Colitis. Immunity. 2015;43(1):187–99.
Mitsdoerffer M, Lee Y, Jäger A, Kim HJ, Korn T, Kolls JK, et al. Proinflammatory T helper type 17 cells are effective B-cell helpers. Proc Natl Acad Sci USA. 2010;107(32):14292–7.
Walker LS, Sansom DM. Confusing signals: recent progress in CTLA-4 biology. Trends Immunol. 2015;36(2):63–70.
Solomon I, Amann M, Goubier A, Arce Vargas F, Zervas D, Qing C, et al. CD25-T(reg)-depleting antibodies preserving IL-2 signaling on effector T cells enhance effector activation and antitumor immunity. Nat Cancer. 2020;1(12):1153–66.
Martín-Orozco E, Norte-Muñoz M, Martínez-García J. Regulatory T Cells in Allergy and Asthma. Front Pediatr. 2017;5:117.
Bagherinia E, Falahi S, Mortazavi SH, Salari F, Rezaiemanesh A, Karaji AG. Co-treatment with Fexofenadine and Budesonide Increases FoxP3 Gene Expression in Patients with Allergic Rhinitis. Am J Rhinol Allergy. 2023;37(6):623–9.
Chen H, Lou H, Wang Y, Cao F, Zhang L, Wang C. Comparison of the efficacy and mechanisms of intranasal budesonide, montelukast, and their combination in treatment of patients with seasonal allergic rhinitis. Int Forum Allergy Rhinol. 2018;8(11):1242–52.
Kim DH, Kim BY, Shin JH, Kim SW, Kim SW. Intranasal azelastine and mometasone exhibit a synergistic effect on a murine model of allergic rhinitis. Am J Otolaryngol. 2017;38(2):198–203.
Ruan H, Long M, Li J, Zhang D, Feng N, Zhang Y. Sustained-Release Hydrogen-Powered Bilateral Microneedles Integrating CD-MOFs for In Situ Treating Allergic Rhinitis. Adv Healthc Mater. 2024;13(22):e2400637.
Vocca L, Di Sano C, Uasuf CG, Sala A, Riccobono L, Gangemi S, et al. IL-33/ST2 axis controls Th2/IL-31 and Th17 immune response in allergic airway diseases. Immunobiology. 2015;220(8):954–63.
Qiao YL, Jiao WE, Xu S, Kong YG, Deng YQ, Yang R, et al. Allergen immunotherapy enhances the immunosuppressive effects of Treg cells to alleviate allergic rhinitis by decreasing PU-1+ Treg cell numbers. Int Immunopharmacol. 2022;112:109187.
Yang J, Lei S. Efficacy and safety of sublingual versus subcutaneous immunotherapy in children with allergic rhinitis: a systematic review and meta-analysis. Front Immunol. 2023;14:1274241.
Tian GX, Peng KP, Liu MH, Tian DF, Xie HQ, Wang LW, et al. CD38(+) B cells affect immunotherapy for allergic rhinitis. J Allergy Clin Immunol. 2022;149(5):1691–1701.e9.
Huang J, Zhang W, Xiang R, Tan L, Liu P, Tao Z, et al. The early-phase transcriptome and the clinical efficacy analysis in three modes of subcutaneous immunotherapy for allergic rhinitis. World Allergy Organ J. 2023;16(8):100811.
Starchenka S, Oluwayi K, Heath M, Armfield O, Shamji M, Layhadi J, et al. Peripheral blood mononuclear cell transcriptome profile in a clinical trial with subcutaneous, grass pollen allergoid immunotherapy. Clin Exp Allergy. 2024;54(2):130–42.
Wang J, Qiu L, Chen Y, Chen M. Sublingual immunotherapy increases Treg/Th17 ratio in allergic rhinitis. Open Med (Wars). 2021;16(1):826–32.
Suzuki S, Sakurai D, Sakurai T, Yonekura S, Iinuma T, Okuma Y, et al. Sublingual administration of liposomes enclosing alpha-galactosylceramide as an effective adjuvant of allergen immunotherapy in a murine model of allergic rhinitis. Allergol Int. 2019;68(3):352–62.
Pan Y, Zhang X, Geng H, Yu Y, Liu J, Li M, et al. Increased Nasal Blimp1 + Treg Cells After Sublingual Immunotherapy Reflect the Efficacy of Treatment in Allergic Rhinitis. Adv Ther. 2024;41(4):1698–710.
Cook LM, Longfellow GA, Kessel JC, Thorp BD, Kimple AJ, Klatt-Cromwell CN. et al. Direct Comparison of Quality of Life in Patients with Allergic Rhinitis Undergoing Sublingual Versus Subcutaneous Immunotherapy. J Clin Med. 2024;13(21):6397.
Gu T, Zhang W, Tan L, Xiang R, Liu P, Huang J, et al. Intratonsillar Immunotherapy: A Convenient and Effective Alternative to Subcutaneous Immunotherapy for Allergic Rhinitis. Research (Wash D C). 2025;8:0573.
Heldner A, Heath MD, Schnautz B, Kotz S, Chaker A, Kramer MF, et al. Ex Vivo Immunomodulatory Effects of Lactobacillus-, Lacticaseibacillus-, and Bifidobacterium-Containing Synbiotics on Human Peripheral Blood Mononuclear Cells and Monocyte-Derived Dendritic Cells in the Context of Grass Pollen Allergy. Probiotics Antimicrob Proteins. 2023;15(4):868–79.
Kim WG, Kang GD, Kim HI, Han MJ, Kim DH. Bifidobacterium longum IM55 and Lactobacillus plantarum IM76 alleviate allergic rhinitis in mice by restoring Th2/Treg imbalance and gut microbiota disturbance. Benef Microbes. 2019;10(1):55–67.
Cavalcanti RFP, Gadelha F, Paiva Ferreira LKD, Paiva Ferreira LAM, Chaves Júnior JV, de Araújo Batista RS, et al. Limosilactobacillus fermentum modulates the gut-airway axis by improving the immune response through FOXP3 activation on combined allergic rhinitis and asthma syndrome (CARAS). Immunobiology. 2023;228(5):152721.
Ren J, Zhao Y, Huang S, Lv D, Yang F, Lou L, et al. Immunomodulatory effect of Bifidobacterium breve on experimental allergic rhinitis in BALB/c mice. Exp Ther Med. 2018;16:3996–4004.
Ren JJ, Yu Z, Yang FL, Lv D, Hung S, Zhang J, et al. Effects of Bifidobacterium Breve Feeding Strategy and Delivery Modes on Experimental Allergic Rhinitis Mice. PLoS One. 2015;10(10):e0140018.
Shin JH, Kim DH, Kim BY, Kim SW, Hwang SH, Lee J, et al. Anti-Interleukin-9 Antibody Increases the Effect of Allergen-Specific Immunotherapy in Murine Allergic Rhinitis. Allergy Asthma Immunol Res. 2017;9(3):237–46.
Gu ZW, Wang YX, Cao ZW. Neutralization of interleukin-9 ameliorates symptoms of allergic rhinitis by reducing Th2, Th9, and Th17 responses and increasing the Treg response in a murine model. Oncotarget. 2017;8(9):14314–24.
Xu B, Liu X, Gao S. IL2-inducible T-cell kinase inhibitor ibrutinib reduces symptoms and Th2 differentiation in mouse allergic-rhinitis model. Drug Dev Res. 2022;83(2):544–51.
Ouyang H, Cheng J, Du J, Gan H, Zheng L. Interleukin-27 Suppresses T Helper-17 Inflammation in Allergic Rhinitis. Iran J Immunol. 2020;17(4):275–82.
Xie F, Hu Q, Cai Q, Yao R, Ouyang S. IL-35 Inhibited Th17 Response in Children with Allergic Rhinitis. ORL J Otorhinolaryngol Relat Spec. 2020;82(1):47–52.
Yokota M, Suzuki M, Nakamura Y, Ozaki S, Murakami S. Cytokine modulation by IL-35 in mice with allergic rhinitis. Am J Rhinol Allergy. 2015;29(4):251–6.
Wang J, Shen Y, Li C, Liu C, Wang ZH, Li YS, et al. IL-37 attenuates allergic process via STAT6/STAT3 pathways in murine allergic rhinitis. Int Immunopharmacol. 2019;69:27–33.
Kim DH, Kim SW, Kim SW, Kang JM. Interleukin-37 Relieves Allergic Inflammation in a House Dust Mite Allergic Rhinitis Murine Model. Iran J Allergy Asthma Immunol. 2017;16(5):404–17.
Zou B, Zhuang RX, Sun XY, Liang J. Analysis of the expression changes of IL-17+ γδ T cells and Treg cells in bone marrow mesenchymal stem cells targeted therapy for allergic rhinitis. Eur Rev Med Pharmacol Sci. 2021;25(7):2858–65.
Peng YQ, Wu ZC, Xu ZB, Fang SB, Chen DH, Zhang HY, et al. Mesenchymal stromal cells-derived small extracellular vesicles modulate DC function to suppress Th2 responses via IL-10 in patients with allergic rhinitis. Eur J Immunol. 2022;52(7):1129–40.
Wang Z, Shahzad KA, Li X, Cai B, Xu M, Li J, et al. Immunomodulatory effect of mesenchymal stem cells-derived extracellular vesicles to modulate the regulatory T cells and Th1/Th2 imbalance in peripheral blood mononuclear cells of patients with allergic rhinitis. Scand J Immunol. 2024;100(6):e13416.
Fan XL, Zeng QX, Li X, Li CL, Xu ZB, Deng XQ, et al. Induced pluripotent stem cell-derived mesenchymal stem cells activate quiescent T cells and elevate regulatory T cell response via NF-κB in allergic rhinitis patients. Stem Cell Res Ther. 2018;9(1):170.
Hou Y, Li FF, Kong JW, Wang Y, Pan Y, Ding H. et al. Adipose Tissue-Derived Mesenchymal Stem Cells Regulate Th17/Treg in a Rat Model of Allergic Rhinitis by Activating IL-2/JAK3/STAT5 Signaling Pathway. Int Arch Allergy Immunol. 2025;186(9):811–23.
Li, W, Wang Y, Cheng F, Qi X, An Y, Zhao C. [Research advances of mesenchymal stem cell in allergic rhinitis]. Lin Chuang Er Bi Yan Hou Tou Jing Wai Ke Za Zhi, 2024. 38(5): 442–7;452.
Liu J, Wang M, Tian X, Wu S, Peng H, Zhu Y, et al. New insights into allergic rhinitis treatment: MSC nanovesicles targeting dendritic cells. J Nanobiotechnology. 2024;22(1):575.
Yang Y, Li S, Xu H. BPIFA1 alleviates allergic rhinitis by regulating the NF-κB signaling pathway and Treg/Th17 balance. Int J Rheum Dis. 2024;27(10):e15372.
Xiang R, Xu Y, Zhang W, Kong YG, Tan L, Chen SM, et al. Semaphorin 3A inhibits allergic inflammation by regulating immune responses in a mouse model of allergic rhinitis. Int Forum Allergy Rhinol. 2019;9(5):528–37.
Liu H, Xia J, Chen Y, Ai J, Wang T, Tan G. Immunosuppressive Regulation of Dendritic Cells and T Cells in Allergic Rhinitis by Semaphorin 3A. Am J Rhinol Allergy. 2021;35(6):846–53.
Yang HC, Won EJ, Kim MJ, Sung CM, Rhee JH, Nam KI. Intralymphatic Administration of Metagonimus yokogawai-Extracted Protein Attenuates Experimental Murine Allergic Rhinitis Model. Int Arch Allergy Immunol. 2021;182(5):381–7.
Van Nguyen T, Piao CH, Fan YJ, Shin DU, Kim SY, Song HJ, et al. Anti-allergic rhinitis activity of α-lipoic acid via balancing Th17/Treg expression and enhancing Nrf2/HO-1 pathway signaling. Sci Rep. 2020;10(1):12528.
Nakano T, Hsu LW, Lai CY, Takaoka Y, Inomata M, Kitano S, et al. Therapeutic potential of α-lipoic acid derivative, sodium zinc histidine dithiooctanamide, in a mouse model of allergic rhinitis. Int Forum Allergy Rhinol. 2017;7(11):1095–103.
Qu S, Qin T, Li M, Zhang S, Ye L, Wei J, et al. The effects of resiquimod in an ovalbumin-induced allergic rhinitis model. Int Immunopharmacol. 2018;59:233–42.
Son HL, Park HR, Park YJ, Kim SW. Effect of Retinoic Acid in a Mouse Model of Allergic Rhinitis. Allergy Asthma Immunol Res. 2015;7(6):590–8.
Xu T, Xia S, Zhang X, Yuan Y. Abnormal purine metabolism in nasal epithelial cells affects allergic rhinitis by regulating Th17/Treg cells. Am J Physiol Cell Physiol. 2025;328(4):C1193–205.
Xu F, Yu S, Qin M, Mao Y, Jin L, Che N, et al. Hydrogen-Rich Saline Ameliorates Allergic Rhinitis by Reversing the Imbalance of Th1/Th2 and Up-Regulation of CD4+CD25+Foxp3+Regulatory T Cells, Interleukin-10, and Membrane-Bound Transforming Growth Factor-β in Guinea Pigs. Inflammation. 2018;41(1):81–92.
Fan Y, Yang C, Zhou J, Cheng X, Dong Y, Wang Q, et al. Regulatory effect of glutathione on treg/Th17 cell balance in allergic rhinitis patients through inhibiting intracellular autophagy. Immunopharmacol Immunotoxicol. 2021;43(1):58–67.
Xiao C, Feng L, Yang W. Inhibition of dendritic cell autophagy alleviates the progression of allergic rhinitis by inhibiting Th1/Th2/Th17 immune imbalance and inflammation. Histol Histopathol. 2025;40(2):237–47.
Cho JS, Kang JH, Han IH, Um JY, Park IH, Lee SH, et al. Antiallergic Effects of Trichostatin A in a Murine Model of Allergic Rhinitis. Clin Exp Otorhinolaryngol. 2015;8(3):243–9.
Dong L, Tang Y, Wen S, He Y, Li F, Deng Y, et al. Fecal Microbiota Transplantation Alleviates Allergic Rhinitis via CD4(+) T Cell Modulation Through Gut Microbiota Restoration. Inflammation. 2024;47(4):1278–97.
Cheng J, Zhang M, Zheng Y, Wang J, Wang Q. Integrative analysis of network pharmacology and proteomics to identify key targets of Tuomin-Zhiti-Decoction for allergic rhinitis. J Ethnopharmacol. 2022;296:115448.
Li P, Tsang MS, Kan LL, Hou T, Hon SS, Chan BC. et al. The Immuno-Modulatory Activities of Pentaherbs Formula on Ovalbumin-Induced Allergic Rhinitis Mice via the Activation of Th1 and Treg Cells and Inhibition of Th2 and Th17 Cells. Molecules. 2021;27(1):239.
Liang X, Liu CS, Wei XH, Xia T, Chen FL, Tang QF, et al. Mahuang Fuzi Xixin Decoction Ameliorates Allergic Rhinitis in Rats by Regulating the Gut Microbiota and Th17/Treg Balance. J Immunol Res. 2020;2020:6841078.
Fu Y, Kong Y, Li J, Wang Y, Li M, Wang Y, et al. Mesenchymal stem cells combined with traditional Chinese medicine (qi-fang-bi-min-tang) alleviates rodent allergic rhinitis. J Cell Biochem. 2020;121(2):1541–51.
Shi J, Liu Y, Yan S, Yan D. Yiqi Wenyang Fang ameliorates allergic rhinitis through inhibiting inflammatory response and promoting the expression of Foxp3. Int J Immunopathol Pharmacol. 2016;29(4):696–706.
Yu ZN, Fan YJ, Nguyen TV, Piao CH, Lee BH, Lee SY, et al. Undaria pinnatifida ameliorates nasal inflammation by inhibiting eosinophil and mast cell activation and modulating the NF-κB/MAPKs signaling pathway. Immun Inflamm Dis. 2024;12(3):e1215.
Piaoa CH, Zou SC, Bui TT, Song CH, Chai OH. Saikosaponin D inhibits nasal inflammation by regulating the transcription factors T-box protein expressed in T cells/GATA-3 and retinoic acid-related orphan nuclear receptor γt in a murine model of allergic rhinitis. Heliyon. 2023;9(6):e17319.
Piao CH, Song CH, Lee EJ, Chai OH. Saikosaponin A ameliorates nasal inflammation by suppressing IL-6/ROR-γt/STAT3/IL-17/NF-κB pathway in OVA-induced allergic rhinitis. Chem Biol Interact. 2020;315:108874.
Jiang Y, Nguyen TV, Jin J, Yu ZN, Song CH, Chai OH. Bergapten ameliorates combined allergic rhinitis and asthma syndrome after PM2.5 exposure by balancing Treg/Th17 expression and suppressing STAT3 and MAPK activation in a mouse model. Biomed Pharmacother. 2023;164:114959.
Ma Q, Li G, He J, Wang J, Ye B. Hydroxysafflor yellow A attenuates allergic response of ovalbumin induced allergic rhinitis via Nrf2/HO-1 and inflammatory signaling pathways. Environ Toxicol. 2023;38(7):1520–34.
Yang Y, Wang L, Wang S, Wang Y, Du Y, Fan Y. Luteolin restored Treg/Th17 balance to ameliorate allergic rhinitis in a mouse model. Immunopharmacol Immunotoxicol. 2023;45(4):461–8.
Li J, Lin X, Liu X, Ma Z, Li Y. Baicalin regulates Treg/Th17 cell imbalance by inhibiting autophagy in allergic rhinitis. Mol Immunol. 2020;125:162–71.
Ke X, Chen Z, Wang X, Kang H, Hong S. Quercetin improves the imbalance of Th1/Th2 cells and Treg/Th17 cells to attenuate allergic rhinitis. Autoimmunity. 2023;56(1):2189133.
Ji Q, Ren S, Li L. Bufotalin ameliorates ovalbumin-induced allergic rhinitis by restoring the Tregs. Microb Pathog. 2023;174:105918.
Huang Y, Yang G, Fei J, Wu Y, Yan J. Bufotalin ameliorates experimental Sjögren’s syndrome development by inhibiting Th17 generation. Naunyn Schmiedebergs Arch Pharmacol. 2020;393(10):1977–85.
He X, Liu L, Luo X, Zhu J, Yang H, Wang J, et al. Astragalus Polysaccharide Relieves Inflammatory Responses in Guinea Pigs with Allergic Rhinitis via Ameliorating NF-kB-Mediated Treg/Th17 Imbalance. Am J Rhinol Allergy. 2022;36(5):638–48.
Liu L, Deng YQ, Jiao WE, Qiao YL, Tao ZZ, Wang Y, et al. Maggot extracts promote regulatory T cell differentiation by upregulating Foxp3 in allergic rhinitis. Am J Transl Res. 2021;13(12):13540–54.
Zheng X, Sun C, Yu R, Chu X, Xu J, Liu C, et al. CD13-specific ligand facilitates Xanthatin nanomedicine targeting dendritic cells for therapy of refractory allergic rhinitis. Int J Pharm. 2020;577:119034.
Xu S, Kong YG, Jiao WE, Yang R, Qiao YL, Xu Y, et al. Tangeretin promotes regulatory T cell differentiation by inhibiting Notch1/Jagged1 signaling in allergic rhinitis. Int Immunopharmacol. 2019;72:402–12.
Bui TT, Piao CH, Hyeon E, Fan Y, Van Nguyen T, Jung SY, et al. The protective role of Piper nigrum fruit extract in an ovalbumin-induced allergic rhinitis by targeting of NFκBp65 and STAT3 signalings. Biomed Pharmacother. 2019;109:1915–23.
Kim BY, Park HR, Jeong HG, Kim SW. Berberine reduce allergic inflammation in a house dust mite allergic rhinitis mouse model. Rhinology. 2015;53(4):353–8.
Duan FP, Li YS, Hu TY, Pan XQ, Ma F, Feng Y, et al. Dendrobium nobile protects against ovalbumin-induced allergic rhinitis by regulating intestinal flora and suppressing lung inflammation. Chin J Nat Med. 2022;20(6):443–57.
Li J, Wang B, Luo Y, Bian Y, Wang R. Effect of artemisinin and neurectomy of pterygoid canal in ovalbumin-induced allergic rhinitis mouse model. Allergy Asthma Clin Immunol. 2018;14:22.
Tian MH, Zhang YN, Sun WF, Liu H, Tang Y. Acupuncture at “Die E acupoint” alleviates inflammatory reaction via inhibiting TLR4/MyD88/NF-κB signaling in rats with allergic rhinitis. Zhen Ci Yan Jiu. 2024;49(5):456–62.
Tian M, Sun W, Mao Y, Zhang Y, Liu H, Tang Y. Mechanistic study of acupuncture on the pterygopalatine ganglion to improve allergic rhinitis: analysis of multi-target effects based on bioinformatics/network topology strategie. Brief Bioinform. 2024;25(4):bbae287.
Wang Y, Hou XR, Li LH, Zhang Y, Yang H, Liang X, et al. [Acupoint injection improves allergic rhinitis by balancing Th17/Treg in allergic rhinitis rats]. Zhen Ci Yan Jiu. 2019;44(4):276–81.
Ma W, Zhai CT, Shang HM, Xu XS, Zheng T, Tian YF. [Clinical efficacy and safety evaluation of acupuncture combined with moxibustion in treatment of allergic rhinitis]. Zhen Ci Yan Jiu. 2022;47(4):336–42.
Hao Y, Yang Y, Zhao H, Chen Y, Zuo T, Zhang Y, et al. Multi-omics in Allergic Rhinitis: Mechanism Dissection and Precision Medicine. Clin Rev Allergy Immunol. 2025;68(1):19.
Ogulur I, Pat Y, Ardicli O, Barletta E, Cevhertas L, Fernandez-Santamaria R, et al. Advances and highlights in biomarkers of allergic diseases. Allergy. 2021;76(12):3659–86.
Mishra T, Sasanka KK, Sudha Ty S, Kumar D, Ps S, Bano G, et al. Emerging Novel Biomarkers in Allergic Rhinitis: A Narrative Review. Cureus. 2025;17:e84705.
Jevnikar Z, Östling J, Ax E, Calvén J, Thörn K, Israelsson E, et al. Epithelial IL-6 trans-signaling defines a new asthma phenotype with increased airway inflammation. J Allergy Clin Immunol. 2019;143(2):577–90.
Kim B, Lee YE, Yeon JW, Go GY, Byun J, Lee K, et al. A novel therapeutic modality using CRISPR-engineered dendritic cells to treat allergies. Biomaterials. 2021;273:120798.
Skuljec J, Chmielewski M, Happle C, Habener A, Busse M, Abken H, et al. Chimeric Antigen Receptor-Redirected Regulatory T Cells Suppress Experimental Allergic Airway Inflammation, a Model of Asthma. Front Immunol. 2017;8:1125.
Ye YM, Zhao YX, Xiang LR, Zou CY, Xiao H, Lu H, et al. The Immunomodulatory mechanism and research progress of mesenchymal stem cells in the treatment of allergic rhinitis. Stem Cell Res Ther. 2025;16(1):188.





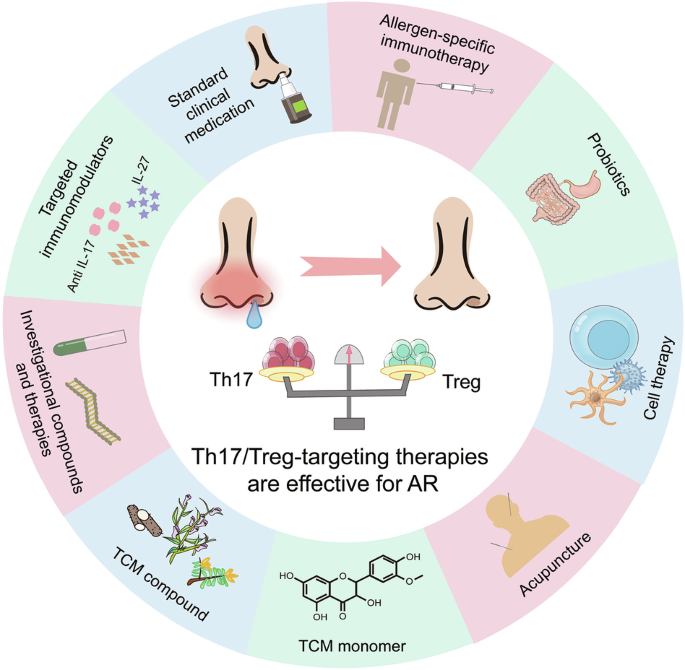











Leave a Reply