Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229–63.
Sterner RC, Sterner RM. CAR-T cell therapy: Current limitations and potential strategies. Blood Cancer J. 2021;11:69.
Bagley SJ, O’Rourke DM. Clinical investigation of CAR T cells for solid tumors: Lessons learned and future directions. Pharm Ther. 2020;205:107419.
Patel U, Abernathy J, Savani BN, Oluwole O, Sengsayadeth S, Dholaria B. CAR T cell therapy in solid tumors: A review of current clinical trials. EJHaem. 2022;3:24–31.
Muhammad N, Mao Q, Xia H. CAR T-cells for cancer therapy. Biotechnol Genet Eng Rev. 2017;33:190–226.
Zheng J. CAR T-cell therapy in solid tumors: Current review and future perspectives. HSET. 2023;54:517–27.
Umut Ö, Gottschlich A, Endres S, Kobold S. CAR T cell therapy in solid tumors: A short review. Memo. 2021;14:143–9.
Karati D, Sarkar S, Kundu M, Majumder P, Biswas A, Mondal T, et al. CAR-T cell therapy: Illuminating the path to solid tumour recovery. Curr Cell Sci. 2025;1:E27726215340105.
Zeng Y. Progress and prospects of CAR-T therapy for the treatment of solid tumors. Highlights Sci, Eng Technol. 2023;74:1323–33.
Uslu U, June CH. Beyond the blood: Expanding CAR T cell therapy to solid tumors. Nat Biotechnol. 2025;43:506–15.
Lee Y-H, Kim CH. Evolution of chimeric antigen receptor (CAR) T cell therapy: Current status and future perspectives. Arch Pharm Res. 2019;42:607–16.
Messaoudi D, Perez F, Gouveia Z. [the new generations of CAR-T cells]. Med Sci (Paris). 2024;40:848–57.
Zhang W. CAR T cell therapy: Evolutions and emergent improvements. TNS. 2023;3:726–36.
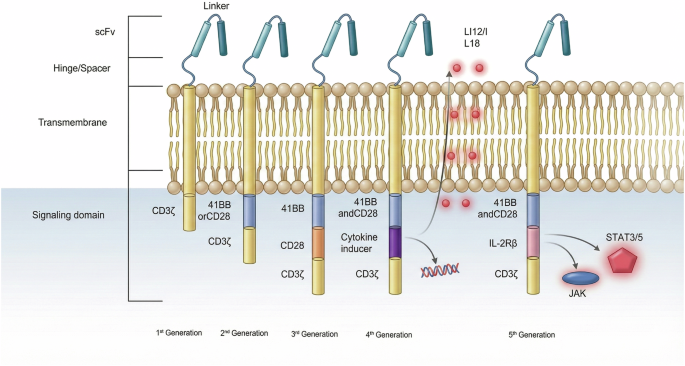
Gallego-Valle J, Pérez-Fernández VA, Rosales-Magallares J, Gil-Manso S, Castellá M, Gonzalez-Navarro EA, et al. High specificity of engineered T cells with third generation CAR (CD28-4-1BB-CD3-ζ) based on biotin-bound monomeric streptavidin for potential tumor immunotherapy. Front Immunol. 2024;15:1448752.
Thomas S, Abken H. CAR T cell therapy becomes CHIC: ‘cytokine help intensified CAR’ T cells. Front Immunol. 2022;13:1090959.
Zheng Z, Li S, Liu M, Chen C, Zhang L, Zhou D. Fine-tuning through generations: Advances in structure and production of CAR-T therapy. Cancers (Basel). 2023;15:3476.
Roybal KT, Rupp LJ, Morsut L, Walker WJ, McNally KA, Park JS, et al. Precision tumor recognition by T cells with combinatorial antigen-sensing circuits. Cell. 2016;164:770–9.
Fedorov VD, Themeli M, Sadelain M. PD-1- and CTLA-4-based inhibitory chimeric antigen receptors (iCARs) divert off-target immunotherapy responses. Sci Transl Med. 2013;5:215ra172.
Kloss CC, Condomines M, Cartellieri M, Bachmann M, Sadelain M. Combinatorial antigen recognition with balanced signaling promotes selective tumor eradication by engineered T cells. Nat Biotechnol. 2013;31:71–75.
Guedan S, Calderon H, Posey AD, Maus MV. Engineering and design of chimeric antigen receptors. Mol Ther Methods Clin Dev. 2019;12:145–56.
Safarzadeh Kozani P, Naseri A, Mirarefin SMJ, Salem F, Nikbakht M, Evazi Bakhshi S, et al. Nanobody-based CAR-T cells for cancer immunotherapy. Biomark Res. 2022;10:24.
Jovčevska I, Muyldermans S. The therapeutic potential of nanobodies. BioDrugs. 2020;34:11–26.
Wagner DL, Fritsche E, Pulsipher MA, Ahmed N, Hamieh M, Hegde M, et al. Immunogenicity of CAR T cells in cancer therapy. Nat Rev Clin Oncol. 2021;18:379–93.
Brudno JN, Lam N, Vanasse D, Shen Y-W, Rose JJ, Rossi J, et al. Safety and feasibility of anti-CD19 CAR T cells with fully human binding domains in patients with B-cell lymphoma. Nat Med. 2020;26:270–80.
Lu X. Structural and research advances in CAR-T cell therapy. TNS. 2023;3:200–5.
Wesson W, Ayoobkhan FS, Mahmoudjafari Z, Mushtaq MU, Abdallah A-OA, Sun W, et al. Characterizing clinical trials for CAR T targeting solid tumors from 2018 to 2023: A systematic review. JCO. 2024;42:e15080.
Zhu S. CAR-T in cancer therapeutics and updates. J Natl Cancer Cent. 2024;4:189–94.
Tony LT, Stabile A, Schauer MP, Hudecek M, Weber J. CAR-T cell therapy for solid tumors. Transfus Med Hemother. 2025;52:96–108.
Martínez-Gamboa DA, Hans R, Moreno-Cortes E, Figueroa-Aguirre J, Garcia-Robledo JE, Vargas-Cely F, et al. CAR T-cell therapy landscape in pediatric, adolescent and young adult oncology – a comprehensive analysis of clinical trials. Crit Rev Oncol Hematol. 2025;209:104648.
Zhu D, Kim WJ, Lee H, Bao X, Kim P. Engineering CAR-T therapeutics for enhanced solid tumor targeting. Adv Mater. 2025;37:e2414882.
Albelda SM. CAR T cell therapy for patients with solid tumours: Key lessons to learn and unlearn. Nat Rev Clin Oncol. 2024;21:47–66.
Misawa K, Bhat H, Adusumilli PS, Hou Z. Combinational CAR T-cell therapy for solid tumors: Requisites, rationales, and trials. Pharm Ther. 2025;266:108763.
Minn I, Rowe SP, Pomper MG. Enhancing CAR T-cell therapy through cellular imaging and radiotherapy. Lancet Oncol. 2019;20:e443–e451.
D’Aloia MM, Zizzari IG, Sacchetti B, Pierelli L, Alimandi M. CAR-T cells: The long and winding road to solid tumors. Cell Death Dis. 2018;9:282.
Beatty GL, O’Hara M. Chimeric antigen receptor-modified T cells for the treatment of solid tumors: Defining the challenges and next steps. Pharm Ther. 2016;166:30–9.
Morgan RA, Yang JC, Kitano M, Dudley ME, Laurencot CM, Rosenberg SA. Case report of a serious adverse event following the administration of T cells transduced with a chimeric antigen receptor recognizing ERBB2. Mol Ther. 2010;18:843–51.
Parkhurst MR, Yang JC, Langan RC, Dudley ME, Nathan D-AN, Feldman SA, et al. T cells targeting carcinoembryonic antigen can mediate regression of metastatic colorectal cancer but induce severe transient colitis. Mol Ther. 2011;19:620–6.
Ghilardi G, Fraietta JA, Gerson JN, Van Deerlin VM, Morrissette JJD, Caponetti GC, et al. T cell lymphoma and secondary primary malignancy risk after commercial CAR T cell therapy. Nat Med. 2024;30:984–9.
Bushman FD. Retroviral integration and human gene therapy. J Clin Invest. 2007;117:2083–6.
Ben AA, Niculescu M-V, Migliorini D. [CAR-T cells in solid tumours: Present and future]. Rev Med Suisse. 2021;17:985–93.
Sirini C, De Rossi L, Moresco MA, Casucci M. CAR T cells in solid tumors and metastasis: Paving the way forward. Cancer Metastasis Rev. 2024;43:1279–96.
Ramakrishna S, Barsan V, Mackall C. Prospects and challenges for use of CAR T cell therapies in solid tumors. Expert Opin Biol Ther. 2020;20:503–16.
Guzman G, Reed MR, Bielamowicz K, Koss B, Rodriguez A. CAR-T therapies in solid tumors: Opportunities and challenges. Curr Oncol Rep. 2023;25:479–89.
Khan SH, Choi Y, Veena M, Lee JK, Shin DS. Advances in CAR T cell therapy: Antigen selection, modifications, and current trials for solid tumors. Front Immunol. 2024;15:1489827.
Lyu M. CAR-T cell therapy in solid tumor. Highlights Sci, Eng Technol. 2023;74:1421–5.
Rojas-Quintero J, Díaz MP, Palmar J, Galan-Freyle NJ, Morillo V, Escalona D, et al. Car T cells in solid tumors: Overcoming obstacles. Int J Mol Sci. 2024;25:4170.
Fucà G, Reppel L, Landoni E, Savoldo B, Dotti G. Enhancing chimeric antigen receptor T-cell efficacy in solid tumors. Clin Cancer Res. 2020;26:2444–51.
Alcantara M, Du Rusquec P, Romano E. Current clinical evidence and potential solutions to increase benefit of CAR T-cell therapy for patients with solid tumors. Oncoimmunology. 2020;9:1777064.
Martinez M, Moon EK. CAR T cells for solid tumors: New strategies for finding, infiltrating, and surviving in the tumor microenvironment. Front Immunol. 2019;10:128.
Ghahri-Saremi N, Akbari B, Soltantoyeh T, Hadjati J, Ghassemi S, Mirzaei HR. Genetic modification of cytokine signaling to enhance efficacy of CAR T cell therapy in solid tumors. Front Immunol. 2021;12:738456.
Fu R, Li H, Li R, McGrath K, Dotti G, Gu Z. Delivery techniques for enhancing CAR T cell therapy against solid tumors. Adv Funct Mater. 2021;31:2009489.
Akbari P, Katsarou A, Daghighian R, van Mil LWHG, Huijbers EJM, Griffioen AW, et al. Directing CAR T cells towards the tumor vasculature for the treatment of solid tumors. Biochim Biophys Acta Rev Cancer. 2022;1877:188701.
Das S, Valton J, Duchateau P, Poirot L. Stromal depletion by TALEN-edited universal hypoimmunogenic FAP-CAR T cells enables infiltration and anti-tumor cytotoxicity of tumor antigen-targeted CAR-T immunotherapy. Front Immunol. 2023;14:1172681.
Safarzadeh Kozani P, Safarzadeh Kozani P, Rahbarizadeh F. Addressing the obstacles of CAR T cell migration in solid tumors: Wishing a heavy traffic. Crit Rev Biotechnol. 2022;42:1079–98.
Kankeu Fonkoua LA, Sirpilla O, Sakemura R, Siegler EL, Kenderian SS. CAR T cell therapy and the tumor microenvironment: Current challenges and opportunities. Mol Ther Oncolytics. 2022;25:69–77.
Wang Y, Drum DL, Sun R, Zhang Y, Chen F, Sun F, et al. Stressed target cancer cells drive nongenetic reprogramming of CAR T cells and solid tumor microenvironment. Nat Commun. 2023;14:5727.
Quach HT, Skovgard MS, Villena-Vargas J, Bellis RY, Chintala NK, Amador-Molina A, et al. Tumor-targeted nonablative radiation promotes solid tumor CAR T-cell therapy efficacy. Cancer Immunol Res. 2023;11:1314–31.
Song D. Epigenetic strategies to optimize CAR-T therapy. HSET. 2024;102:253–9.
Tian Y, Li Y, Shao Y, Zhang Y. Gene modification strategies for next-generation CAR T cells against solid cancers. J Hematol Oncol. 2020;13:54.
Pietrobon V, Todd LA, Goswami A, Stefanson O, Yang Z, Marincola F. Improving CAR T-cell persistence. Int J Mol Sci. 2021;22:10828.
Srinivasan S, Armitage J, Nilsson J, Waithman J. Transcriptional rewiring in CD8+ T cells: Implications for CAR-T cell therapy against solid tumours. Front Immunol. 2024;15:1412731.
O’Rourke DM, Nasrallah MP, Desai A, Melenhorst JJ, Mansfield K, Morrissette JJD, et al. A single dose of peripherally infused EGFRvIII-directed CAR T cells mediates antigen loss and induces adaptive resistance in patients with recurrent glioblastoma. Sci Transl Med. 2017;9:eaaa0984.
Labanieh L, Mackall CL. CAR immune cells: Design principles, resistance and the next generation. Nature. 2023;614:635–48.
Liu S, Deng B, Yin Z, Lin Y, An L, Liu D, et al. Combination of CD19 and CD22 CAR-T cell therapy in relapsed B-cell acute lymphoblastic leukemia after allogeneic transplantation. Am J Hematol. 2021;96:671–9.
Chen J, Jiang H Current challenges and strategies for chimeric antigen receptor-T-cell therapy for solid tumors. CRI 2021; 41. https://doi.org/10.1615/CritRevImmunol.2020036178.
Liu S, Zhang Y. Challenges and interventions of chimeric antigen receptor-T cell therapy in solid tumors. Chin J Cancer Res. 2023;35:239–44.
Kyte JA. Strategies for improving the efficacy of CAR T cells in solid cancers. Cancers (Basel). 2022;14:571.
Bagley SJ, Logun M, Fraietta JA, Wang X, Desai AS, Bagley LJ, et al. Intrathecal bivalent CAR T cells targeting EGFR and IL13Rα2 in recurrent glioblastoma: Phase 1 trial interim results. Nat Med. 2024;30:1320–9.
Choi BD, Gerstner ER, Frigault MJ, Leick MB, Mount CW, Balaj L, et al. Intraventricular CARv3-TEAM-E T cells in recurrent glioblastoma. N Engl J Med. 2024;390:1290–8.
Choi BD, Yu X, Castano AP, Bouffard AA, Schmidts A, Larson RC, et al. CAR-T cells secreting BiTEs circumvent antigen escape without detectable toxicity. Nat Biotechnol. 2019;37:1049–58.
Cho JH, Collins JJ, Wong WW. Universal chimeric antigen receptors for multiplexed and logical control of T cell responses. Cell. 2018;173:1426–1438.e11.
Wu C-Y, Roybal KT, Puchner EM, Onuffer J, Lim WA. Remote control of therapeutic T cells through a small molecule-gated chimeric receptor. Science. 2015;350:aab4077.
Rodgers DT, Mazagova M, Hampton EN, Cao Y, Ramadoss NS, Hardy IR, et al. Switch-mediated activation and retargeting of CAR-T cells for B-cell malignancies. Proc Natl Acad Sci USA. 2016;113:E459–468.
Ma JSY, Kim JY, Kazane SA, Choi S-H, Yun HY, Kim MS, et al. Versatile strategy for controlling the specificity and activity of engineered T cells. Proc Natl Acad Sci USA. 2016;113:E450–458.
Tamada K, Geng D, Sakoda Y, Bansal N, Srivastava R, Li Z, et al. Redirecting gene-modified T cells toward various cancer types using tagged antibodies. Clin Cancer Res. 2012;18:6436–45.
Sridhar P, Petrocca F. Regional delivery of chimeric antigen receptor (CAR) T-cells for cancer therapy. Cancers (Basel). 2017;9:92.
Adusumilli PS, Cherkassky L, Villena-Vargas J, Colovos C, Servais E, Plotkin J, et al. Regional delivery of mesothelin-targeted CAR T cell therapy generates potent and long-lasting CD4-dependent tumor immunity. Sci Transl Med. 2014;6:261ra151.
Chai LF, Hardaway JC, Heatherton KR, O’Connell KP, LaPorte JP, Guha P, et al. Antigen receptor T cells (CAR-T) effectively control tumor growth in a colorectal liver metastasis model. J Surg Res. 2022;272:37–50.
Cherkassky L, Hou Z, Amador-Molina A, Adusumilli PS. Regional CAR T cell therapy: An ignition key for systemic immunity in solid tumors. Cancer Cell. 2022;40:569–74.
Sagnella SM, White AL, Yeo D, Saxena P, van Zandwijk N, Rasko JEJ. Locoregional delivery of CAR-T cells in the clinic. Pharm Res. 2022;182:106329.
Whilding LM, Halim L, Draper B, Parente-Pereira AC, Zabinski T, Davies DM, et al. CAR T-cells targeting the integrin αvβ6 and co-expressing the chemokine receptor CXCR2 demonstrate enhanced homing and efficacy against several solid malignancies. Cancers (Basel). 2019;11:674.
Hosking M, Shirinbak S, Grant J, Pan Y, Gentile A, Mehta A et al. 120 chemokine receptor engineering enhances trafficking and homing of primary and iPSC-derived CAR-T cells to solid tumors. J Immunother Cancer 2021;9. https://doi.org/10.1136/jitc-2021-SITC2021.120.
Cadilha BL, Dorman K, Huynh D, Lorenzini T, Vänttinen M, Benmebarek M-R et al. P06.03 C-C chemokine receptor 8 tumor-directed recruitment enables CAR T cells to reject solid tumors. J Immunother Cancer 2020;8. https://doi.org/10.1136/jitc-2020-ITOC7.82.
Wan W, Han W, Chen J, Li Y, Zhao L, Gao M, et al. Chemokine-based injectable navigation system for enhancing CAR-T cell therapy against solid tumors. Adv Funct Mater. 2024;34:2401161.
Zou Q, Liao K, Li G, Huang X, Zheng Y, Yang G, et al. Photo-metallo-immunotherapy: Fabricating chromium-based nanocomposites to enhance CAR-T cell infiltration and cytotoxicity against solid tumors. Adv Mater. 2025;37:e2407425.
Katz SC, Burga RA, McCormack E, Wang LJ, Mooring W, Point GR, et al. Phase I hepatic immunotherapy for metastases study of intra-arterial chimeric antigen receptor-modified T-cell therapy for CEA+ liver metastases. Clin Cancer Res. 2015;21:3149–59.
Brown CE, Alizadeh D, Starr R, Weng L, Wagner JR, Naranjo A, et al. Regression of glioblastoma after chimeric antigen receptor T-cell therapy. N Engl J Med. 2016;375:2561–9.
Mead PA, Safdieh JE, Nizza P, Tuma S, Sepkowitz KA. Ommaya reservoir infections: A 16-year retrospective analysis. J Infect. 2014;68:225–30.
Ali A, Bosse RC, Doonan BP, Narayan P, Jester GA, Delaune JD, et al. Ommaya reservoir related complications: A single center experience and review of current literature. Int J Clin Oncol Cancer Res. 2019;4:10–24.
Nagarsheth N, Wicha MS, Zou W. Chemokines in the cancer microenvironment and their relevance in cancer immunotherapy. Nat Rev Immunol. 2017;17:559–72.
Rafiq S, Hackett CS, Brentjens RJ. Engineering strategies to overcome the current roadblocks in CAR T cell therapy. Nat Rev Clin Oncol. 2020;17:147–67.
Satapathy BP, Sheoran P, Yadav R, Chettri D, Sonowal D, Dash CP, et al. The synergistic immunotherapeutic impact of engineered CAR-T cells with PD-1 blockade in lymphomas and solid tumors: A systematic review. Front Immunol. 2024;15:1389971.
Pérez-Moreno MA, Ciudad-Gutiérrez P, Jaramillo-Ruiz D, Reguera-Ortega JL, Abdel-Kader Martín L, Flores-Moreno S. Combined or sequential treatment with immune checkpoint inhibitors and car-T cell therapies for the management of haematological malignancies: A systematic review. Int J Mol Sci. 2023;24:14780.
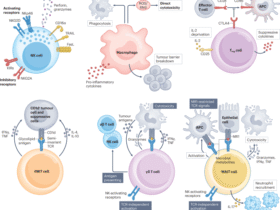
Xia X, Yang Z, Lu Q, Liu Z, Wang L, Du J, et al. Reshaping the tumor immune microenvironment to improve CAR-T cell-based cancer immunotherapy. Mol Cancer. 2024;23:175.
Liu G, Rui W, Zhao X, Lin X. Enhancing CAR-T cell efficacy in solid tumors by targeting the tumor microenvironment. Cell Mol Immunol. 2021;18:1085–95.
Rafiq S, Yeku OO, Jackson HJ, Purdon TJ, van Leeuwen DG, Drakes DJ, et al. Targeted delivery of a PD-1-blocking scFv by CAR-T cells enhances anti-tumor efficacy in vivo. Nat Biotechnol. 2018;36:847–56.
Liu H, Lei W, Zhang C, Yang C, Wei J, Guo Q, et al. CD19-specific CAR T cells that express a PD-1/CD28 chimeric switch-receptor are effective in patients with PD-L1-positive B-cell lymphoma. Clin Cancer Res. 2021;27:473–84.
Rupp LJ, Schumann K, Roybal KT, Gate RE, Ye CJ, Lim WA, et al. CRISPR/Cas9-mediated PD-1 disruption enhances anti-tumor efficacy of human chimeric antigen receptor T cells. Sci Rep. 2017;7:737.
Liang Y, Liu H, Lu Z, Lei W, Zhang C, Li P, et al. CD19 CAR-T expressing PD-1/CD28 chimeric switch receptor as a salvage therapy for DLBCL patients treated with different CD19-directed CAR T-cell therapies. J Hematol Oncol. 2021;14:26.
Kwon N, Chen YY. Overcoming solid-tumor barriers: Armored CAR-T cell therapy. Trends Cancer. 2025;11:1019–29.
Lynn RC, Weber EW, Sotillo E, Gennert D, Xu P, Good Z, et al. c-jun overexpression in CAR T cells induces exhaustion resistance. Nature. 2019;576:293–300.
Kagoya Y, Tanaka S, Guo T, Anczurowski M, Wang C-H, Saso K, et al. A novel chimeric antigen receptor containing a JAK-STAT signaling domain mediates superior antitumor effects. Nat Med. 2018;24:352–9.
Sukumar M, Liu J, Mehta GU, Patel SJ, Roychoudhuri R, Crompton JG, et al. Mitochondrial membrane potential identifies cells with enhanced stemness for cellular therapy. Cell Metab. 2016;23:63–76.
Frlic T, Pavlin M. Metabolic reprogramming of CAR T cells: A new frontier in cancer immunotherapy. Front Immunol. 2025;16:1688995.
Kawalekar OU, O’Connor RS, Fraietta JA, Guo L, McGettigan SE, Posey AD, et al. Distinct signaling of coreceptors regulates specific metabolism pathways and impacts memory development in CAR T cells. Immunity. 2016;44:380–90.
Yeku OO, Purdon TJ, Koneru M, Spriggs D, Brentjens RJ. Armored CAR T cells enhance antitumor efficacy and overcome the tumor microenvironment. Sci Rep. 2017;7:10541.
Stadtmauer EA, Fraietta JA, Davis MM, Cohen AD, Weber KL, Lancaster E, et al. CRISPR-engineered T cells in patients with refractory cancer. Science. 2020;367:eaba7365.
Lasek W, Zagożdżon R, Jakobisiak M. Interleukin 12: Still a promising candidate for tumor immunotherapy?. Cancer Immunol Immunother. 2014;63:419–35.
Vormittag P, Gunn R, Ghorashian S, Veraitch FS. A guide to manufacturing CAR T cell therapies. Curr Opin Biotechnol. 2018;53:164–81.
Levine BL, Miskin J, Wonnacott K, Keir C. Global manufacturing of CAR T cell therapy. Mol Ther Methods Clin Dev. 2017;4:92–101.
Grada Z, Hegde M, Byrd T, Shaffer DR, Ghazi A, Brawley VS, et al. TanCAR: A novel bispecific chimeric antigen receptor for cancer immunotherapy. Mol Ther Nucleic Acids. 2013;2:e105.
Zah E, Lin M-Y, Silva-Benedict A, Jensen MC, Chen YY. T cells expressing CD19/CD20 bispecific chimeric antigen receptors prevent antigen escape by malignant B cells. Cancer Immunol Res. 2016;4:498–508.
Roybal KT, Williams JZ, Morsut L, Rupp LJ, Kolinko I, Choe JH, et al. Engineering T cells with customized therapeutic response programs using synthetic notch receptors. Cell. 2016;167:419–432.e16.
Simeone DM, Hecht JR, Smith C, Fakih M, Welling T, Garon E et al. 636 BASECAMP-1: A master prescreening study to identify patients with high-risk or metastatic solid tumors with HLA loss of heterozygosity (LOH) in preparation for tmod CAR T-cell therapy trials. J Immunother Cancer. 2023;11. https://doi.org/10.1136/jitc-2023-SITC2023.0636.
Larson S First results of IMPT-314, an autologous bispecific CD19/CD20 chimeric antigen receptor (CAR) in enriched naive and central memory T cells, for the treatment of large B cell lymphoma (LBCL). ASH, 2024 https://ash.confex.com/ash/2024/webprogram/Paper200697.html (accessed 8 Feb2026).
Bangayan NJ, Wang L, Burton Sojo G, Noguchi M, Cheng D, Ta L, et al. Dual-inhibitory domain iCARs improve the efficiency of the AND-NOT gate CAR T strategy. Proc Natl Acad Sci USA. 2023;120:e2312374120.
Bassan D, Weinberger L, Yi J, Kim T, Weist MR, Adams GB, et al. HER2 and HLA-a*02 dual CAR-T cells utilize LOH in a NOT logic gate to address on-target off-tumor toxicity. J Immunother Cancer. 2023;11:e007426.
Oh J, Kirsh C, Hsin J-P, Radecki KC, Zampieri A, Manry D, et al. NOT gated T cells that selectively target EGFR and other widely expressed tumor antigens. iScience. 2024;27:109913.
Di Stasi A, Tey S-K, Dotti G, Fujita Y, Kennedy-Nasser A, Martinez C, et al. Inducible apoptosis as a safety switch for adoptive cell therapy. N Engl J Med. 2011;365:1673–83.
Straathof KC, Pulè MA, Yotnda P, Dotti G, Vanin EF, Brenner MK, et al. An inducible caspase 9 safety switch for T-cell therapy. Blood. 2005;105:4247–54.
Del Bufalo F, De Angelis B, Caruana I, Del Baldo G, De Ioris MA, Serra A, et al. GD2-CART01 for relapsed or refractory high-risk neuroblastoma. N Engl J Med. 2023;388:1284–95.
Foster MC, Savoldo B, Lau W, Rubinos C, Grover N, Armistead P, et al. Utility of a safety switch to abrogate CD19.CAR T-cell-associated neurotoxicity. Blood. 2021;137:3306–9.
2025. NCCN Clinical Practice Guidelines in Oncology: Management of CAR T-Cell and Lymphocyte Engager-Related Toxicities. Version 2.2026
Rejeski K, Subklewe M, Aljurf M, Bachy E, Balduzzi A, Barba P, et al. Immune effector cell-associated hematotoxicity: EHA/EBMT consensus grading and best practice recommendations. Blood. 2023;142:865–77.
Botta GP, Chao J, Ma H, Hahn M, Sierra G, Jia J, et al. Metastatic gastric cancer target lesion complete response with Claudin18.2-CAR T cells. J Immunother Cancer. 2024;12:e007927.
Zhong G, Zhang X, Guo Z, Gao Y, Zhao B, Liu X, et al. Complete remission of advanced pancreatic cancer induced by claudin18.2-targeted CAR-T cell therapy: A case report. Front Immunol. 2024;15:1325860.
Shen SH, Woroniecka K, Barbour AB, Fecci PE, Sanchez-Perez L, Sampson JH. CAR T cells and checkpoint inhibition for the treatment of glioblastoma. Expert Opin Biol Ther. 2020;20:579–91.
Brown CE, Rodriguez A, Palmer J, Ostberg JR, Naranjo A, Wagner JR, et al. Off-the-shelf, steroid-resistant, IL13Rα2-specific CAR T cells for treatment of glioblastoma. Neuro Oncol. 2022;24:1318–30.
Ohta K, Sakoda Y, Adachi K, Shinozaki T, Nakajima M, Yasuda H, et al. Therapeutic efficacy of IL7/CCL19-expressing CAR-T cells in intractable solid tumor models of glioblastoma and pancreatic cancer. Cancer Res Commun. 2024;4:2514–24.
Anwar MY, Williams GR, Paluri RK. CAR T cell therapy in pancreaticobiliary cancers: A focused review of clinical data. J Gastrointest Cancer. 2021;52:1–10.
Lin Y-J, Mashouf LA, Lim M. CAR T cell therapy in primary brain tumors: Current investigations and the future. Front Immunol. 2022;13:817296.
Titov A, Valiullina A, Zmievskaya E, Zaikova E, Petukhov A, Miftakhova R, et al. Advancing CAR T-cell therapy for solid tumors: Lessons learned from lymphoma treatment. Cancers (Basel). 2020;12:125.
Siddiqui RS, Sardar M. A systematic review of the role of chimeric antigen receptor T (CAR-T) cell therapy in the treatment of solid tumors. Cureus. 2021;13:e14494.
Martínez Bedoya D, Dutoit V, Migliorini D. Allogeneic CAR T cells: An alternative to overcome challenges of CAR T cell therapy in glioblastoma. Front Immunol. 2021;12:640082.
Huang J, Zheng M, Zhang Z, Tang X, Chen Y, Peng A, et al. Interleukin-7-loaded oncolytic adenovirus improves CAR-T cell therapy for glioblastoma. Cancer Immunol Immunother. 2021;70:2453–65.
Klampatsa A, Dimou V, Albelda SM. Mesothelin-targeted CAR-T cell therapy for solid tumors. Expert Opin Biol Ther. 2021;21:473–86.
Zhang DY, Wang X, Sun Y, Cui Q, Nelson E, Zhang Z, et al. Temporal multi-modal single-cell analyses reveal dynamic interactions of CAR. T cells glioblastoma Target antigen-Negat Neoplast cells. 2024. https://doi.org/10.1101/2024.10.03.616537.













Leave a Reply