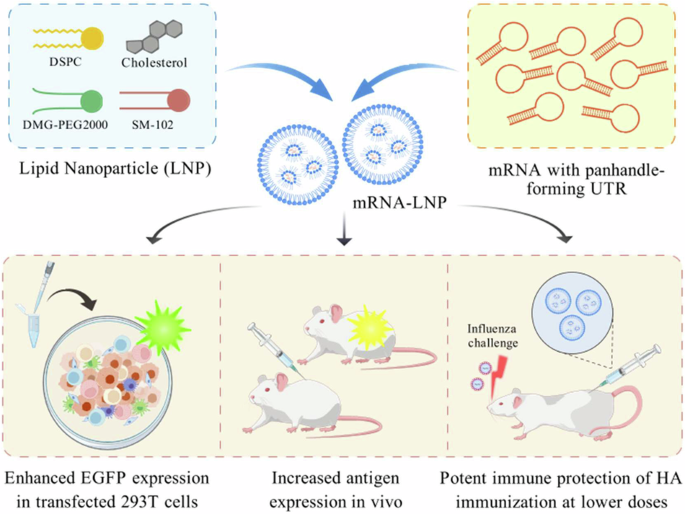
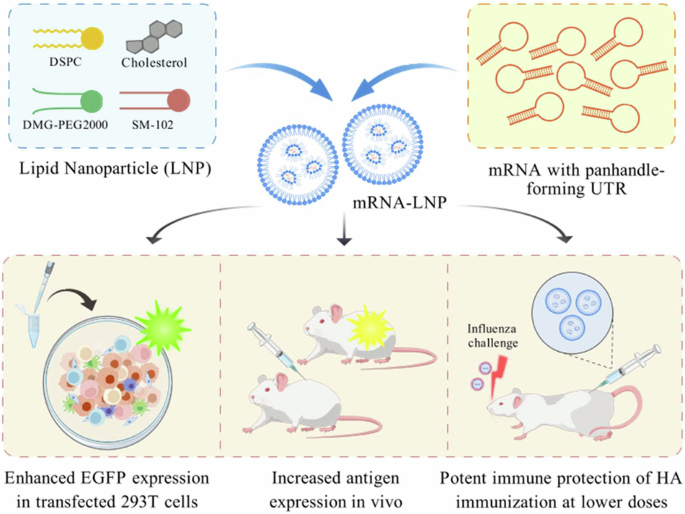
Influenza viruses pose a persistent threat to global public health, causing widespread respiratory illness and significant morbidity. Vaccination remains the most effective strategy to reduce the burden of both seasonal and pandemic influenza. mRNA vaccines represent a promising alternative to conventional vaccine platforms due to their rapid development, flexibility, and high efficacy. Nonetheless, optimizing non-coding regulatory elements such as untranslated regions (UTRs) remains crucial for enhancing mRNA vaccine performance. In this study, we designed a novel UTR derived from the influenza A virus M segment, engineered to form optimal panhandle structures through selective base-pair enhancing mutations (M1 + 2), aiming to improve mRNA translation efficiency and immunogenicity. Using both reporter and HA antigen-encoding mRNAs, we demonstrated that the M1 + 2 UTR significantly enhanced protein expression in vitro and in vivo compared to unmodified UTRs and the canonical α-globin UTR control. In a murine model, low-dose (0.1 μg) vaccination with HA mRNA-lipid nanoparticles (LNPs) incorporating the M1 + 2 UTR elicited robust innate, humoral, and T cell-mediated immune responses, and conferred complete protection against lethal challenge with seasonal influenza strains, including H1N1, H3N2, and IBV. Our findings underscore the potential of rational UTR design in developing more efficacious and dose-sparing mRNA vaccines.












Leave a Reply