Miller KD, Nogueira L, Mariotto AB, Rowland JH, Yabroff KR, Alfano CM, et al. Cancer treatment and survivorship statistics, 2019. CA Cancer J Clin. 2019;69:363–85.
Luo N. Advances in Targeted Immunotherapy in Cancers. Int J Mol Sci. 2023;24:17475.
D’Angelo SP, Araujo DM, Abdul Razak AR, Agulnik M, Attia S, Blay JY, et al. Afamitresgene autoleucel for advanced synovial sarcoma and myxoid round cell liposarcoma (SPEARHEAD-1): an international, open-label, phase 2 trial. Lancet. 2024;403:1460–71.
Kantoff PW, Higano CS, Shore ND, Berger ER, Small EJ, Penson DF, et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med. 2010;363:411–22.
Liu S, Galat V, Galat Y, Lee YKA, Wainwright D, Wu J. NK cell-based cancer immunotherapy: from basic biology to clinical development. J Hematol Oncol. 2021;14:7.
Neelapu SS, Locke FL, Bartlett NL, Lekakis LJ, Miklos DB, Jacobson CA, et al. Axicabtagene Ciloleucel CAR T-Cell Therapy in Refractory Large B-Cell Lymphoma. N Engl J Med. 2017;377:2531–44.
Sarnaik AA, Hamid O, Khushalani NI, Lewis KD, Medina T, Kluger HM, et al. Lifileucel, a Tumor-Infiltrating Lymphocyte Therapy, in Metastatic Melanoma. J Clin Oncol. 2021;39:2656–66.
Sabado RL, Balan S, Bhardwaj N. Dendritic cell-based immunotherapy. Cell Res. 2017;27:74–95.
Lin X, Sun Y, Dong X, Liu Z, Sugimura R, Xie G. IPSC-derived CAR-NK cells for cancer immunotherapy. Biomed Pharmacother. 2023;165:115123.
Sterner RC, Sterner RM. CAR-T cell therapy: current limitations and potential strategies. Blood Cancer J. 2021;11:69.
Zhang Y, Liu Z, Wei W, Li Y. TCR engineered T cells for solid tumor immunotherapy. Exp Hematol Oncol. 2022;11:38.
Hu W, Bian Y, Ji H. TIL Therapy in Lung Cancer: Current Progress and Perspectives. Adv Sci. 2024;11:e2409356.
Levi ST, Copeland AR, Nah S, Crystal JS, Ivey GD, Lalani A, et al. Neoantigen Identification and Response to Adoptive Cell Transfer in Anti-PD-1 Naïve and Experienced Patients with Metastatic Melanoma. Clin Cancer Res. 2022;28:3042–52.
Matsueda S, Chen L, Li H, Yao H, Yu F. Recent clinical researches and technological development in TIL therapy. Cancer Immunol Immunother. 2024;73:232.
Rosenberg SA. Lymphocytes as a living drug for cancer. Science. 2024;385:25–6.
Stevanović S, Draper LM, Langhan MM, Campbell TE, Kwong ML, Wunderlich JR, et al. Complete regression of metastatic cervical cancer after treatment with human papillomavirus-targeted tumor-infiltrating T cells. J Clin Oncol. 2015;33:1543–50.
Califf RM, Zarin DA, Kramer JM, Sherman RE, Aberle LH, Tasneem A. Characteristics of clinical trials registered in ClinicalTrials.gov, 2007-2010. JAMA. 2012;307:1838–47.
DeAngelis CD, Drazen JM, Frizelle FA, Haug C, Hoey J, Horton R, et al. Clinical trial registration: a statement from the International Committee of Medical Journal Editors. Jama. 2004;292:1363–4.
Aizaz M, Khan AS, Khan M, Musazade E, Yang G. Advancements in tumor-infiltrating lymphocytes: Historical insights, contemporary milestones, and future directions in oncology therapy. Crit Rev Oncol Hematol. 2024;202:104471.
Julve M, Lythgoe MP, Larkin J, Furness AJS. Lifileucel: the first cellular therapy approved for solid tumours. Trends Cancer. 2024;10:475–7.
Klobuch S, Seijkens TTP, Schumacher TN, Haanen J. Tumour-infiltrating lymphocyte therapy for patients with advanced-stage melanoma. Nat Rev Clin Oncol. 2024;21:173–84.
Plaugher DR, Childress AR, Gosser CM, Esoe DP, Naughton KJ, Hao Z, et al. Therapeutic potential of tumor-infiltrating lymphocytes in non-small cell lung cancer. Cancer Lett. 2024;605:217281.
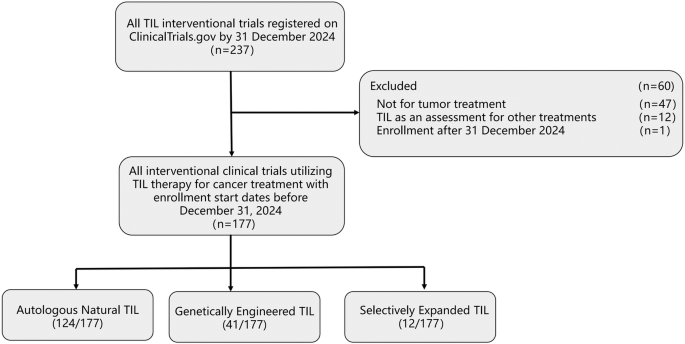
Zhang Y, Fu H, Zhao Q. Current status and perspectives of clinical trials for tumor-infiltrating lymphocyte therapy. Clin Transl Oncol. 2025;27:466–72.
Dudley ME, Gross CA, Somerville RP, Hong Y, Schaub NP, Rosati SF, et al. Randomized selection design trial evaluating CD8⁺-enriched versus unselected tumor-infiltrating lymphocytes for adoptive cell therapy for patients with melanoma. J Clin Oncol. 2013;31:2152–9.
Borch TH, Andersen R, Ellebaek E, Met Ö, Donia M, Svane IM. Future role for adoptive T-cell therapy in checkpoint inhibitor-resistant metastatic melanoma. J Immunother Cancer. 2020;8:e000668.
Lowery FJ, Goff SL, Gasmi B, Parkhurst MR, Ratnam NM, Halas HK, et al. Neoantigen-specific tumor-infiltrating lymphocytes in gastrointestinal cancers: a phase 2 trial. Nat Med. 2025;31:1994–2003.
Chandran SS, Somerville RPT, Yang JC, Sherry RM, Klebanoff CA, Goff SL, et al. Treatment of metastatic uveal melanoma with adoptive transfer of tumour-infiltrating lymphocytes: a single-centre, two-stage, single-arm, phase 2 study. Lancet Oncol. 2017;18:792–802.
Nguyen LT, Saibil SD, Sotov V, Le MX, Khoja L, Ghazarian D, et al. Phase II clinical trial of adoptive cell therapy for patients with metastatic melanoma with autologous tumor-infiltrating lymphocytes and low-dose interleukin-2. Cancer Immunol Immunother. 2019;68:773–85.
Rohaan MW, Borch TH, van den Berg JH, Met Ö, Kessels R, Geukes Foppen MH, et al. Tumor-infiltrating lymphocyte therapy or ipilimumab in advanced melanoma. N Engl J Med. 2022;387:2113–25.
Liang YJ, Chen QY, Xu JX, Liu XF, Xia JC, Liu LT, et al. A phase II randomised controlled trial of adjuvant tumour-infiltrating lymphocytes for pretreatment Epstein-Barr virus DNA-selected high-risk nasopharyngeal carcinoma patients. Eur J Cancer. 2023;191:112965.
Ferris RL, Leidner RS, Chung CH, Jimeno A, Lee SM, Sukari A, et al. Efficacy and safety of one-time autologous tumor-infiltrating lymphocyte cell therapy in patients with recurrent and/or metastatic head and neck squamous cell carcinoma. J Immunother Cancer. 2025;13:e011633.
Creelan BC, Wang C, Teer JK, Toloza EM, Yao J, Kim S, et al. Tumor-infiltrating lymphocyte treatment for anti-PD-1-resistant metastatic lung cancer: a phase 1 trial. Nat Med. 2021;27:1410–8.
Kverneland AH, Chamberlain CA, Borch TH, Nielsen M, Mørk SK, Kjeldsen JW, et al. Adoptive cell therapy with tumor-infiltrating lymphocytes supported by checkpoint inhibition across multiple solid cancer types. J Immunother Cancer. 2021;9:e003499.
Amaria R, Knisely A, Vining D, Kopetz S, Overman MJ, Javle M, et al. Efficacy and safety of autologous tumor-infiltrating lymphocytes in recurrent or refractory ovarian cancer, colorectal cancer, and pancreatic ductal adenocarcinoma. J Immunother Cancer. 2024;12:e006822.
Verdegaal EME, Verpoorte ALC, van der Kooij MK, de Bruin L, Visser M, van der Minne CE, et al. Effective TIL therapy for patients with checkpoint-resistant melanoma without lymphodepleting regimens requires IFNα. Clin Cancer Res. 2025;31:2628–38.
Verdegaal EME, Santegoets SJ, Welters MJP, de Bruin L, Visser M, van der Minne CE, et al. Timed adoptive T cell transfer during chemotherapy in patients with recurrent platinum-sensitive epithelial ovarian cancer. J Immunother Cancer. 2023;11:e007697.
König D, Kasenda B, Sandholzer M, Chirindel A, Zingg A, Ritschard R, et al. Adoptive cell therapy with tumor-infiltrating lymphocytes in combination with nivolumab in patients with advanced melanoma. Immunooncol Technol. 2024;24:100728.
Lou E, Choudhry MS, Starr TK, Folsom TD, Bell J, Rathmann B, et al. Targeting the intracellular immune checkpoint CISH with CRISPR-Cas9-edited T cells in patients with metastatic colorectal cancer: a first-in-human, single-centre, phase 1 trial. Lancet Oncol. 2025;26:559–70.
Huang H, Nie CP, Liu XF, Song B, Yue JH, Xu JX, et al. Phase I study of adjuvant immunotherapy with autologous tumor-infiltrating lymphocytes in locally advanced cervical cancer. J Clin Invest. 2022;132:e157726.
Guo J, Wang C, Luo N, Wu Y, Huang W, Zhu J, et al. IL-2-free tumor-infiltrating lymphocyte therapy with PD-1 blockade demonstrates potent efficacy in advanced gynecologic cancer. BMC Med. 2024;22:207.
Li F, Wang Y, Yan J, Wu H, Du X, Feng W, et al. Autologous tumor-infiltrating lymphocyte mono-therapy can rapidly shrink tumor in Asian patient with stage III/IV cervical cancer: two cases report. Int J Womens Health. 2024;16:31–9.
Kazemi MH, Sadri M, Najafi A, Rahimi A, Baghernejadan Z, Khorramdelazad H, et al. Tumor-infiltrating lymphocytes for treatment of solid tumors: It takes two to tango?. Front Immunol. 2022;13:1018962.
Hong H, He Y, Li Y, Shen Y, Qu Y. Clinical trial landscape for TIL therapy: emerging insights and future directions in oncology. J Transl Med. 2024;22:1008.
Xu C, Zhang S, Zhang Y, Tang SQ, Fang XL, Zhu GL, et al. Evolving landscape and academic attitudes toward the controversies of global immuno-oncology trials. Int J Cancer. 2021;149:108–18.
Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229–63.
Sun Y, Shen Y, Liu Q, Zhang H, Jia L, Chai Y, et al. Global trends in melanoma burden: A comprehensive analysis from the Global Burden of Disease Study, 1990-2021. J Am Acad Dermatol. 2025;92:100–7.
Salem ML, Cole DJ. Dendritic cell recovery post-lymphodepletion: a potential mechanism for anti-cancer adoptive T cell therapy and vaccination. Cancer Immunol Immunother. 2010;59:341–53.
Sarnaik AA, Hwu P, Mulé JJ, Pilon-Thomas S. Tumor-infiltrating lymphocytes: A new hope. Cancer Cell. 2024;42:1315–8.
Goff SL, Dudley ME, Citrin DE, Somerville RP, Wunderlich JR, Danforth DN, et al. Randomized, Prospective Evaluation Comparing Intensity of Lymphodepletion Before Adoptive Transfer of Tumor-Infiltrating Lymphocytes for Patients With Metastatic Melanoma. J Clin Oncol. 2016;34:2389–97.
Heemskerk B, Liu K, Dudley ME, Johnson LA, Kaiser A, Downey S, et al. Adoptive cell therapy for patients with melanoma, using tumor-infiltrating lymphocytes genetically engineered to secrete interleukin-2. Hum Gene Ther. 2008;19:496–510.
Fernandez-Poma SM, Salas-Benito D, Lozano T, Casares N, Riezu-Boj JI, Mancheño U, et al. Expansion of Tumor-Infiltrating CD8(+) T cells Expressing PD-1 Improves the Efficacy of Adoptive T-cell Therapy. Cancer Res. 2017;77:3672–84.
Gros A, Robbins PF, Yao X, Li YF, Turcotte S, Tran E, et al. PD-1 identifies the patient-specific CD8⁺ tumor-reactive repertoire infiltrating human tumors. J Clin Invest. 2014;124:2246–59.
Ye Q, Song DG, Poussin M, Yamamoto T, Best A, Li C, et al. CD137 accurately identifies and enriches for naturally occurring tumor-reactive T cells in tumor. Clin Cancer Res. 2014;20:44–55.
Chatani PD, Lowery FJ, Parikh NB, Hitscherich KJ, Yossef R, Hill V, et al. Cell surface marker-based capture of neoantigen-reactive CD8(+) T-cell receptors from metastatic tumor digests. J Immunother Cancer. 2023;11:e006264.
Darragh LB, Oweida AJ, Karam SD. Overcoming Resistance to Combination Radiation-Immunotherapy: A Focus on Contributing Pathways Within the Tumor Microenvironment. Front Immunol. 2018;9:3154.
Keam SP, Halse H, Nguyen T, Wang M, Van Kooten Losio N, Mitchell C, et al. High dose-rate brachytherapy of localized prostate cancer converts tumors from cold to hot. J Immunother Cancer. 2020;8:e000792.
Yoneda K, Kuwata T, Kanayama M, Mori M, Kawanami T, Yatera K, et al. Alteration in tumoural PD-L1 expression and stromal CD8-positive tumour-infiltrating lymphocytes after concurrent chemo-radiotherapy for non-small cell lung cancer. Br J Cancer. 2019;121:490–6.
Chen S, Li J, Dong A, Liu Z, Zhu M, Jin M, et al. Nab-paclitaxel and gemcitabine plus camrelizumab and radiotherapy versus nab-paclitaxel and gemcitabine alone for locally advanced pancreatic adenocarcinoma: a prospective cohort study. J Hematol Oncol. 2023;16:26.
Chu X, Niu L, Xiao G, Peng H, Deng F, Liu Z, et al. The Long-Term and Short-Term Efficacy of Immunotherapy in Non-Small Cell Lung Cancer Patients With Brain Metastases: A Systematic Review and Meta-Analysis. Front Immunol. 2022;13:875488.















Leave a Reply