Xiong, W., Guo, C., Gozlan, R. E. & Liu, J. Tilapia introduction in China: Economic boom in aquaculture versus ecological threats to ecosystems. Rev. Aquacult. 15, 179–197. https://doi.org/10.1111/raq.12710 (2023).
El-Sayed, A.-F.M. & Fitzsimmons, K. From Africa to the world—The journey of Nile tilapia. Rev. Aquacult. 15, 6–21. https://doi.org/10.1111/raq.12738 (2023).
Hendam, B. M. et al. Effects of water additive probiotic, Pediococcus acidilactici on growth performance, feed utilization, hematology, gene expression and disease resistance against Aspergillus flavus of Nile tilapia (Oreochromis niloticus). Anim. Feed Sci. Technol. 303, 115696. https://doi.org/10.1016/j.anifeedsci.2023.115696 (2023).
Gule, T. T. & Geremew, A. Dietary strategies for better utilization of aquafeeds in tilapia farming. Aquacult. Nutr. 2022, 9463307. https://doi.org/10.1155/2022/9463307 (2022).
Musa, S. et al. The influence of various feeding and pond fertilization strategies on Nile Tilapia (Oreochromis niloticus L.) production and the selectivity for natural versus supplementary diet in semi-intensive aquaculture systems. Aquaculture Fish Fish 5, e70031. https://doi.org/10.1002/aff2.70031 (2025).
Barad, R. R., Verma, D. K., Yusufzai, S. I., Shrivastava, V. & Ram, A. R. Herbal feed additives: Natural boost for aquatic health and growth. In Sustainable Feed Ingredients and Additives for Aquaculture Farming: Perspectives from Africa And Asia (eds Gabriel, N. N. et al.) 405–431 (Springer Nature Singapore, 2024).
Chaklader, M. R. et al. Advantageous roles of top-sprayed exogenous enzymes on growth, liver and gut functions, and growth-related genes in Nile tilapia fed a high plant protein diet. Aquacult. Int. 33, 412. https://doi.org/10.1007/s10499-025-02092-8 (2025).
Vijayaram, S. et al. Synergetic response on herbal and probiotic applications: A review. Fish Physiol. Biochem. 50, 1315–1329. https://doi.org/10.1007/s10695-024-01318-5 (2024).
Rasidi, R. et al. in Sustainable Feed Ingredients and Additives for Aquaculture Farming: Perspectives from Africa and Asia (eds Ndakalimwe Naftal Gabriel, Kenneth Prudence Abasubong, Victoria Ndinelago Erasmus, & Manoj Tukaram Kamble) 481–500 (Springer Nature Singapore, 2024).
Hoseinifar, S. H. et al. Reducing the use of antibiotics in European aquaculture with vaccines, functional feed additives and optimization of the gut microbiota. J. Mar. Sci. Eng. 12, 204. https://doi.org/10.3390/jmse12020204 (2024).
Ng, W.-K. & Koh, C.-B. The utilization and mode of action of organic acids in the feeds of cultured aquatic animals. Rev. Aquacult. 9, 342–368. https://doi.org/10.1111/raq.12141 (2017).
Morales-Covarrubias, M. S. et al. Efficacy of BACTI-NIL® AQUA in experimental infection with Vibrio parahaemolyticus in juvenile of Litopenaeus vannamei. Rev Cient Facul Cienc Vet 32, 1–9, https://doi.org/10.52973/rcfcv-e32148 (2022).
El-Sayed, A.-F.M. et al. Dietary organic acid blend modulates hemato-immunological parameters, digestive and reproductive performances in red tilapia (Oreochromis niloticus × O. mossambicus) broodstock. Fish Physiol. Biochem. 51, 41. https://doi.org/10.1007/s10695-025-01459-1 (2025).
Taylor, T. M. & Doores, S. X. in Antimicrobials in Food 133–190 (CRC Press, 2020).
Lückstädt, C. The use of acidifiers in fish nutrition. CABI Reviews, 8 pp.-, https://doi.org/10.1079/PAVSNNR20083044 (2008).
Dibner, J. J. & Buttin, P. Use of organic acids as a model to study the impact of gut microflora on nutrition and metabolism. J. Appl. Poult. Res. 11, 453–463. https://doi.org/10.1093/japr/11.4.453 (2002).
da Silva, V. G. et al. Effect of an organic acid blend in Nile tilapia growth performance, immunity, gut microbiota, and resistance to challenge against francisellosis. Res. Vet. Sci. 159, 214–224. https://doi.org/10.1016/j.rvsc.2023.04.019 (2023).
Zoheiri, F. et al. Dietary fulvic acid increased growth, stress tolerance and disease resistance against Vibrio harveyi in Asian seabass (Lates calcarifer) juvenile. Aquac. Rep. 32, 101738. https://doi.org/10.1016/j.aqrep.2023.101738 (2023).
Silva, B. C. et al. Improved digestion and initial performance of whiteleg shrimp using organic salt supplements. Aquac. Nutr. 22, 997–1005. https://doi.org/10.1111/anu.12315 (2016).
Castillo, S., Rosales, M., Pohlenz, C. & Gatlin, D. M. Effects of organic acids on growth performance and digestive enzyme activities of juvenile red drum Sciaenops ocellatus. Aquaculture 433, 6–12. https://doi.org/10.1016/j.aquaculture.2014.05.038 (2014).
Ebrahimi, M. et al. Comparing the effects of different dietary organic acids on the growth, intestinal short-chain fatty acids, and liver histopathology of red hybrid tilapia (Oreochromis sp.) and potential use of these as preservatives. Fish Physiol. Biochem. 43, 1195–1207. https://doi.org/10.1007/s10695-017-0365-0 (2017).
Mehrgan, M. S. et al. Synergistic effects of sodium butyrate and sodium propionate on the growth performance, blood biochemistry, immunity, and immune-related gene expression of goldfish (Carassius auratus). Aquac. Int. 30, 3179–3193. https://doi.org/10.1007/s10499-022-00954-z (2022).
Ringø, E. Effects of dietary lactate and propionate on growth and digesta in Arctic charr, Salvelinus alpinus (L.). Aquaculture 96, 321–333. https://doi.org/10.1016/0044-8486(91)90161-Y (1991).
Safari, R., Hoseinifar, S. H., Nejadmoghadam, S. & Khalili, M. Non-specific immune parameters, immune, antioxidant and growth-related genes expression of common carp (Cyprinus carpio L.) fed sodium propionate. Aquaculture Res. 48, 4470–4478. https://doi.org/10.1111/are.13272 (2017).
Hoseinifar, S. H., Safari, R. & Dadar, M. Dietary sodium propionate affects mucosal immune parameters, growth and appetite related genes expression: Insights from zebrafish model. Gen. Comp. Endocrinol. 243, 78–83. https://doi.org/10.1016/j.ygcen.2016.11.008 (2017).
Hoseinifar, S. H., Zoheiri, F. & Caipang, C. M. Dietary sodium propionate improved performance, mucosal and humoral immune responses in Caspian white fish (Rutilus frisii kutum) fry. Fish Shellfish Immunol. 55, 523–528. https://doi.org/10.1016/j.fsi.2016.06.027 (2016).
Morken, T. et al. Sodium diformate and extrusion temperature affect nutrient digestibility and physical quality of diets with fish meal and barley protein concentrate for rainbow trout (Oncorhynchus mykiss). Aquaculture 317, 138–145. https://doi.org/10.1016/j.aquaculture.2011.04.020 (2011).
Gao, Y., Storebakken, T., Shearer, K. D., Penn, M. & Øverland, M. Supplementation of fishmeal and plant protein-based diets for rainbow trout with a mixture of sodium formate and butyrate. Aquaculture 311, 233–240. https://doi.org/10.1016/j.aquaculture.2010.11.048 (2011).
Romano, N. et al. Dietary sodium citrate improved oxidative stability in red hybrid tilapia (Oreochromis sp.) but reduced growth, health status, intestinal short chain fatty acids and induced liver damage. Aquaculture 458, 170–176. https://doi.org/10.1016/j.aquaculture.2016.03.014 (2016).
Fabay, R. V., Serrano, A. E. Jr., Alejos, M. S. & Fabay, J. V. Effects of dietary acidification and acid source on fish growth and feed efficiency (Review). World Acad. Sci. J. 4, 21. https://doi.org/10.3892/wasj.2022.156 (2022).
Zhao, T. et al. Effects of dietary citric acid on growth performance, mineral status, body and muscle composition, muscle growth and mTOR signaling in yellow catfish Pelteobagrus fulvidraco fed with low-manganese diets. Aquaculture 582, 740569. https://doi.org/10.1016/j.aquaculture.2024.740569 (2024).
Yousefi, M. et al. Effects of dietary malic acid supplementation on growth performance, antioxidant and immunological parameters, and intestinal gene expressions in rainbow trout, Oncorhynchus mykiss. Aquaculture 563, 738864. https://doi.org/10.1016/j.aquaculture.2022.738864 (2023).
Yong, A. S. K., Abang Zamhari, D. NJb., Shapawi, R., Zhuo, L.-C. & Lin, Y.-H. Physiological changes of giant grouper (Epinephelus lanceolatus) fed with high plant protein with and without supplementation of organic acid. Aquac. Rep. 18, 100499. https://doi.org/10.1016/j.aqrep.2020.100499 (2020).
Reda, R. M., El-Murr, A., Abd Elhakim, Y. & El-Shahat, W. Aeromonas veronii detection in Egyptian fish farms with summer tilapia mortality outbreaks and the role of formic acid in limiting its spread. Aquac. Res. 53, 940–956. https://doi.org/10.1111/are.15635 (2022).
Macusi, E. D. et al. Protein fishmeal replacement in aquaculture: A systematic review and implications on growth and adoption viability. Sustainability 15, 12500. https://doi.org/10.3390/su151612500 (2023).
Aydın, B. et al. Evaluation of non-conventional plant-based feed ingredients (NPFIs) for a sustainable aquafeed industry. Rev. Aquac. 18, e70127. https://doi.org/10.1111/raq.70127 (2026).
Pervin, M. A., Jahan, H., Akter, R., Omri, A. & Hossain, Z. Appraisal of different levels of soybean meal in diets on growth, digestive enzyme activity, antioxidation, and gut histology of tilapia (Oreochromis niloticus). Fish. Physiol. Biochem. 46, 1397–1407. https://doi.org/10.1007/s10695-020-00798-5 (2020).
Zakaria, M. K. et al. Fermented soybean meal (FSBM) in African catfish (Clarias gariepinus) diets: Effects on growth performance, fish gut microbiota analysis, blood haematology, and liver morphology. Life 12, 1851. https://doi.org/10.3390/life12111851 (2022).
Zhang, J. et al. The role of sodium butyrate in modulating growth, intestinal health, and antimicrobial efficacy in turbot (Scophthalmus maximus L.) fed high soy diets. Sci. Rep. 14, 32033. https://doi.org/10.1038/s41598-024-83704-w (2024).
Chen, W., Chang, K., Chen, J., Zhao, X. & Gao, S. Dietary sodium butyrate supplementation attenuates intestinal inflammatory response and improves gut microbiota composition in largemouth bass (Micropterus salmoides) fed with a high soybean meal diet. Fish. Physiol. Biochem. 47, 1805–1819. https://doi.org/10.1007/s10695-021-01004-w (2021).
Naveed, M., Mateen, A. & Abdullah, S. Assessment of partially replacement of fishmeal with Lactobacillus plantarum fermented soybean meal, probiotics in the diet of Oreochromis niloticus on the growth profile, enzymatic indices and immunity. Pak. J. Agric. Sci. 61, 391–401. https://doi.org/10.21162/PAKJAS/24.137 (2024).
Li, Y. et al. Replacing soybean meal with fermented rapeseed meal in diets: Potential effects on growth performance, antioxidant capacity, and liver and intestinal health of juvenile tilapia (Oreochromis niloticus). Fish. Physiol. Biochem. 50, 1683–1699. https://doi.org/10.1007/s10695-024-01363-0 (2024).
Ding, Q. et al. The effects of sodium propionate on intestinal barrier function of genetically improved farmed tilapia in a high-lipid formulation. Aquaculture 579, 740187. https://doi.org/10.1016/j.aquaculture.2023.740187 (2024).
Abd El-Naby, A. S., Khattaby, A.E.-R.A., Samir, F., Awad, S. M. M. & Abdel-Tawwab, M. Stimulatory effect of dietary butyrate on growth, immune response, and resistance of Nile tilapia, Oreochromis niloticus against Aeromonas hydrophila infection. Anim. Feed. Sci. Technol. 254, 114212. https://doi.org/10.1016/j.anifeedsci.2019.114212 (2019).
NRC. Nutrient requirements of fish and shrimp. (National academies press, 2011).
Bezerra, R. S. et al. Alkaline proteinase from intestine of Nile tilapia (Oreochromis niloticus). Process. Biochem. 40, 1829–1834. https://doi.org/10.1016/j.procbio.2004.06.066 (2005).
Borlongan, I. G. Studies on the digestive lipases of milkfish. Chanos chanos. Aquaculture 89, 315–325. https://doi.org/10.1016/0044-8486(90)90135-A (1990).
Caraway, W. T. A stable starch substrate for the determination of amylase in serum and other body fluids. Am. J. Clin. Pathol. 32, 97–99. https://doi.org/10.1093/ajcp/32.1_ts.97 (1959).
Bancroft, J. D. & Gamble, M. 40–138 (Elsevier health sciences, 2008).
Gewaily, M. S. et al. Dietary synbiotics can help relieve the impacts of Deltamethrin toxicity of Nile Tilapia reared at low temperatures. Animals 11, 1790. https://doi.org/10.3390/ani11061790 (2021).
Benzie, I. F. F. & Strain, J. J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 239, 70–76. https://doi.org/10.1006/abio.1996.0292 (1996).
Ohkawa, H., Ohishi, N. & Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 95, 351–358. https://doi.org/10.1016/0003-2697(79)90738-3 (1979).
Habig, W. H., Pabst, M. J. & Jakoby, W. B. Glutathione S-transferases: The first enzymatic step in mercapturic acid formation. J. Biol. Chem. 249, 7130–7139. https://doi.org/10.1016/S0021-9258(19)42083-8 (1974).
Marklund, S. & Marklund, G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 47, 469–474 (1974).
Aebi, H. in Methods in Enzymology Vol. 105 121–126 (Academic Press, 1984).
Paglia, D. E. & Valentine, W. N. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 70, 158–169 (1967).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25, 402–408. https://doi.org/10.1006/meth.2001.1262 (2001).
Deng, Z. & Kim, S. W. Opportunities and challenges of soy proteins with different processing applications. Antioxidants 13, 569. https://doi.org/10.3390/antiox13050569 (2024).
Di, D. et al. Exploring the dual role of anti-nutritional factors in soybeans: A comprehensive analysis of health risks and benefits. Crit. Rev. Food Sci. Nutr. https://doi.org/10.1080/10408398.2024.2430757 (2024).
Zhang, Q., Zhang, S., Wu, S., Madsen, M. H. & Shi, S. Supplementing the early diet of broilers with soy protein concentrate can improve intestinal development and enhance short-chain fatty acid-producing microbes and short-chain fatty acids, especially butyric acid. J. Anim. Sci. Biotechnol. 13, 97. https://doi.org/10.1186/s40104-022-00749-5 (2022).
Sun, H. et al. The effects of sodium propionate supplementation in the diet with high soybean meal on growth performance, intestinal health, and immune resistance to bacterial infection in turbot (Scophthalmus maximus L.). Aquac. Nutr. 2022, 8952755. https://doi.org/10.1155/2022/8952755 (2022).
Abdel-Latif, H. M. R., Abdel-Tawwab, M., Dawood, M. A. O., Menanteau-Ledouble, S. & El-Matbouli, M. Benefits of dietary butyric acid, sodium butyrate, and their protected forms in aquafeeds: A review. Rev. Fish. Sci. Aquacult. 28, 421–448. https://doi.org/10.1080/23308249.2020.1758899 (2020).
Yu, D. H., Gong, S. Y., Yuan, Y. C. & Lin, Y. C. Effects of replacing fish meal with soybean meal on growth, body composition and digestive enzyme activities of juvenile Chinese sucker, Myxocyprinus asiaticus. Aquacult. Nutr. 19, 84–90. https://doi.org/10.1111/j.1365-2095.2012.00945.x (2013).
Liang, H. et al. Excessive replacement of fish meal by soy protein concentrate resulted in inhibition of growth, nutrient metabolism, antioxidant capacity, immune capacity, and intestinal development in juvenile largemouth bass (Micropterus salmoides). Antioxidants 13, 809. https://doi.org/10.3390/antiox13070809 (2024).
Li, Z. et al. An evaluation of soybean protein concentrate as a replacement for fish meal with methionine supplementation in diets for hybrid sturgeon (Acipenser baerii ♀ × A. schrenckii ♂). Animals 15, 787. https://doi.org/10.3390/ani15060787 (2025).
Krogdahl, Å., Penn, M., Thorsen, J., Refstie, S. & Bakke, A. M. Important antinutrients in plant feedstuffs for aquaculture: An update on recent findings regarding responses in salmonids. Aquacult. Res. 41, 333–344. https://doi.org/10.1111/j.1365-2109.2009.02426.x (2010).
Krogdahl, Å. et al. Soya saponins induce enteritis in Atlantic salmon (Salmo salar L.). J. Agric. Food Chem. 63, 3887–3902. https://doi.org/10.1021/jf506242t (2015).
Romano, N. et al. Effects of bioprocessed soybean meal and nucleotide supplementation on growth, physiology and histomorphology in largemouth bass, Micropterus salmoides, juveniles. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 260, 111038. https://doi.org/10.1016/j.cbpa.2021.111038 (2021).
Gu, J., Bakke, A. M., Valen, E. C., Lein, I. & Krogdahl, Å. Bt-maize (MON810) and non-GM soybean meal in diets for Atlantic salmon (Salmo salar L.) juveniles – Impact on survival, growth performance, development, digestive function, and transcriptional expression of intestinal immune and stress responses. PLoS ONE 9, e99932. https://doi.org/10.1371/journal.pone.0099932 (2014).
Ke, L., Qin, Y., Song, T., Wang, K. & Ye, J. Dietary sodium butyrate administration alleviates high soybean meal-induced growth retardation and enteritis of orange-spotted groupers (Epinephelus coioides). Front. Mar. Sci. https://doi.org/10.3389/fmars.2022.1029397 (2022).
Zhou, C. et al. Effect of dietary sodium butyrate on growth performance, enzyme activities and intestinal proliferation-related gene expression of juvenile golden pompano Trachinotus ovatus. Aquacult. Nutr. 25, 1261–1271. https://doi.org/10.1111/anu.12940 (2019).
Yousefi, M. et al. Dietary propionate administration improves growth performance, hepatic lipid deposition, and intestinal activity of digestive enzymes, inflammation, bacterial population, and antioxidant capacity in rainbow trout, Oncorhynchus mykiss. Aquaculture 578, 740099. https://doi.org/10.1016/j.aquaculture.2023.740099 (2024).
Ahmadifar, E. et al. Effect of a diet enriched with sodium propionate on growth performance, antioxidant property, innate-adaptive immune response, and growth-related genes expression in critically endangered beluga sturgeon (Huso huso). Fish Shellfish Immunol. 125, 101–108. https://doi.org/10.1016/j.fsi.2022.04.031 (2022).
d. Sá, L. S. et al. The zootechnical performance, health state modulation, morphology, and intestinal microbiome of Nile tilapia juveniles fed with a functional blend of immunostimulants associated with a diet high in soybean meal. Fishes 9, 212. https://doi.org/10.3390/fishes9060212 (2024).
Jiao, F. et al. A comparison of digestive strategies for fishes with different feeding habits: Digestive enzyme activities, intestinal morphology, and gut microbiota. Ecol. Evol. 13, e10499. https://doi.org/10.1002/ece3.10499 (2023).
Gu, M. et al. Effects of dietary β-conglycinin and glycinin on digestive enzymes activities, intestinal histology and immune responses of juvenile turbot Scophthalmus maximus. Aquac. Res. https://doi.org/10.1111/are.12528 (2016).
Jiang, J. et al. Lysine and methionine supplementation ameliorates high inclusion of soybean meal inducing intestinal oxidative injury and digestive and antioxidant capacity decrease of yellow catfish. Fish Physiol. Biochem. 44, 319–328. https://doi.org/10.1007/s10695-017-0437-1 (2018).
Wang, C.-Y. & Li, Z.-B. Growth performance, digestive enzyme activity and immune response of Japanese sea bass, Lateolabrax japonicus fed with fructooligosaccharide. Aquacult. Nutr. 26, 296–305. https://doi.org/10.1111/anu.12990 (2020).
Qin, H. et al. Growth performance, digestive capacity and intestinal health of juvenile spotted seabass (Lateolabrax maculatus) fed dietary laminarin supplement. Front. Mar. Sci. https://doi.org/10.3389/fmars.2023.1242175 (2023).
Zhao, H. et al. Effects of dietary sodium butyrate on growth, digestive enzymes, body composition and nutrient retention-related gene expression of juvenile yellow catfish (Pelteobagrus fulvidraco). Anim. Nutr. 7, 539–547. https://doi.org/10.1016/j.aninu.2020.12.007 (2021).
Martin-Gallausiaux, C., Marinelli, L., Blottière, H. M., Larraufie, P. & Lapaque, N. SCFA: Mechanisms and functional importance in the gut. Proc. Nutr. Soc. 80, 37–49. https://doi.org/10.1017/S0029665120006916 (2021).
Tran, N. T. et al. Progress and perspectives of short-chain fatty acids in aquaculture. Rev. Aquacult. 12, 283–298. https://doi.org/10.1111/raq.12317 (2020).
De Marco, G., Cappello, T. & Maisano, M. Histomorphological changes in fish gut in response to prebiotics and probiotics treatment to improve their health status: A review. Animals 13, 2860. https://doi.org/10.3390/ani13182860 (2023).
Abdel-Aziz, M. F. A. et al. Unchanged water stress induces growth retardation, histopathological alterations, and antioxidant-immune disruptions in Oreochromis niloticus: The promising role of dietary organic acids. Aquacult. Int. 32, 6031–6052. https://doi.org/10.1007/s10499-024-01454-y (2024).
Bakke, A. M. Pathophysiological and immunological characteristics of soybean meal-induced enteropathy in salmon: contribution of recent molecular investigations. Avances en Nutrición Acuicola (2011).
Zhang, W. et al. Mechanisms by which fermented soybean meal and soybean meal induced enteritis in marine fish juvenile Pearl gentian grouper. Front. Physiol. https://doi.org/10.3389/fphys.2021.646853 (2021).
Gu, M., Bai, N., Zhang, Y. & Krogdahl, Å. Soybean meal induces enteritis in turbot Scophthalmus maximus at high supplementation levels. Aquaculture 464, 286–295. https://doi.org/10.1016/j.aquaculture.2016.06.035 (2016).
Rimoldi, S. et al. Butyrate and taurine exert a mitigating effect on the inflamed distal intestine of European sea bass fed with a high percentage of soybean meal. Fish. Aquat. Sci. 19, 40. https://doi.org/10.1186/s41240-016-0041-9 (2016).
Gustafsson, J. K. & Johansson, M. E. V. The role of goblet cells and mucus in intestinal homeostasis. Nat. Rev. Gastroenterol. Hepatol. 19, 785–803. https://doi.org/10.1038/s41575-022-00675-x (2022).
Ferrer, M., Buey, B., Grasa, L., Mesonero, J. E. & Latorre, E. Protective role of short-chain fatty acids on intestinal oxidative stress induced by TNF-α. Cell Stress Chaperones 29, 769–776. https://doi.org/10.1016/j.cstres.2024.11.002 (2024).
Fan, Z. et al. Suitable cottonseed protein concentrate supplementation in common carp (Cyprinus carpio) serves as an effective strategy for fish meal sparing based on improvement in intestinal antioxidant capacity, barrier and microbiota composition. Antioxidants 13, 436. https://doi.org/10.3390/antiox13040436 (2024).
Zheng, Q., Wen, X., Han, C., Li, H. & Xie, X. Effect of replacing soybean meal with cottonseed meal on growth, hematology, antioxidant enzymes activity and expression for juvenile grass carp, Ctenopharyngodon idellus. Fish Physiol. Biochem. 38, 1059–1069. https://doi.org/10.1007/s10695-011-9590-0 (2012).
Abdel-Latif, H. M. R. et al. Effects of sodium butyrate nanoparticles on the hemato-immunological indices, hepatic antioxidant capacity, and gene expression responses in Oreochromis niloticus. Fish Shellfish Immunol. 119, 516–523. https://doi.org/10.1016/j.fsi.2021.10.039 (2021).
Alhoshy, M. et al. Nutrigenomics in crustaceans: Current status and future prospects. Fish Shellfish Immunol. 129, 1–12. https://doi.org/10.1016/j.fsi.2022.08.056 (2022).
Rajabiesterabadi, H., Ghelichi, A., Jorjani, S., Hoseini, S. M. & Akrami, R. Dietary olive (Olea europaea) leaf extract suppresses oxidative stress and modulates intestinal expression of antioxidant- and tight junction-related genes in common carp (Cyprinus carpio). Aquaculture 520, 734676. https://doi.org/10.1016/j.aquaculture.2019.734676 (2020).
Al-Hawary, I. I. et al. Evaluating local strains of soybean and corn cultivars in the diets of Nile tilapia (Oreochromis niloticus): Growth and insulin-like growth factor 1, intestinal health, and inflammation features. Ann. Anim. Sci. 22, 1301–1309. https://doi.org/10.2478/aoas-2022-0031 (2022).
Song, Z., Li, P., Wang, J., Sun, Y. & Wang, C. Dietary inclusion of hydrolyzed soybean and cottonseed meals influence digestion, metabolic enzymes, and growth-related hormones and growth of juvenile turbot (Scophthalmus maximus). Aquac. Int. 26, 1017–1033. https://doi.org/10.1007/s10499-018-0265-z (2018).
Shabana, S. M., Elbaz, S., Abass, M. E. & Abu Zahra, N. I. Effects of dietary sodium butyrate on the biological indices, gene expression, and resistance of Oreochromis niloticus to multidrug-resistant Pseudomonas aeruginosa infection. Assiut Vet. Med. J. 70, 335–360. https://doi.org/10.21608/avmj.2024.301270.1291 (2024).
Ullah, S., Feng, F., Zhao, M., Zhang, J. & Shao, Q. Comparative effects of dietary supplementations with microencapsulated sodium butyrate, glycerol monolaurate and tributyrin on growth, immunity, and gut health in Black Sea bream. Animals 15, 810 (2025).
Yin, B. et al. Dietary supplementation of β-conglycinin, with or without sodium butyrate on the growth, immune response and intestinal health of hybrid grouper. Sci. Rep. 11, 17298. https://doi.org/10.1038/s41598-021-96693-x (2021).
Moustafa, E. M. et al. Enhancing growth, immunity and disease resistance in Nile tilapia (Oreochromis niloticus) through dietary sodium butyrate supplementation. Egypt. J. Vet. Sci. https://doi.org/10.21608/ejvs.2025.368505.2701 (2025).
Mirghaed, A. T., Yarahmadi, P., Soltani, M., Paknejad, H. & Hoseini, S. M. Dietary sodium butyrate (Butirex® C4) supplementation modulates intestinal transcriptomic responses and augments disease resistance of rainbow trout (Oncorhynchus mykiss). Fish Shellfish Immunol. 92, 621–628. https://doi.org/10.1016/j.fsi.2019.06.046 (2019).
Sheikhzadeh, N., Ahmadifar, E., Dawood, M. A. O. & Soltani, M. Dietary sodium propionate enhanced the growth performance, immune-related genes expression, and resistance against Ichthyophthirius multifiliis in goldfish (Carassius auratus). Aquaculture 540, 736720. https://doi.org/10.1016/j.aquaculture.2021.736720 (2021).
Fontinha, F., Martins, N., Campos, G., Peres, H. & Oliva-Teles, A. The effects of short-chain fatty acids in gut immune and oxidative responses of European sea bass (Dicentrarchus labrax): An ex vivo approach. Animals 14, 1360. https://doi.org/10.3390/ani14091360 (2024).
Meng, X. et al. Safflower promotes the immune functions of the common carp (Cyprinus carpio L.) by producing the short-chain fatty acids and regulating the intestinal microflora. Aquac. Nutr. 2022, 7068088. https://doi.org/10.1155/2022/7068088 (2022).





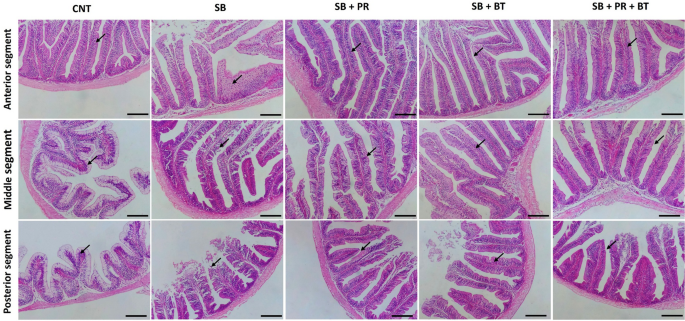










Leave a Reply