Robert, C. A decade of immune-checkpoint inhibitors in cancer therapy. Nat. Commun. 11, 3801 (2020).
Sharma, P., Hu-Lieskovan, S., Wargo, J. A. & Ribas, A. Primary, adaptive, and acquired resistance to cancer immunotherapy. Cell 168, 707–723 (2017).
Giraldo, N. A. et al. The clinical role of the TME in solid cancer. Br. J. Cancer 120, 45–53 (2019).
Binnewies, M. et al. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat. Med. 24, 541–550 (2018).
Chen, D. S. & Mellman, I. Elements of cancer immunity and the cancer-immune set point. Nature 541, 321–330 (2017).
Jiang, Y., Li, Y. & Zhu, B. T-cell exhaustion in the tumor microenvironment. Cell Death Dis. 6, e1792 (2015).
Quatromoni, J. G. & Eruslanov, E. Tumor-associated macrophages: function, phenotype, and link to prognosis in human lung cancer. Am. J. Transl. Res. 4, 376–389 (2012).
Hinshaw, D. C. & Shevde, L. A. The tumor microenvironment innately modulates cancer progression. Cancer Res. 79, 4557–4566 (2019).
Quail, D. F. & Joyce, J. A. Microenvironmental regulation of tumor progression and metastasis. Nat. Med. 19, 1423–1437 (2013).
Zhang, L. et al. Oncolytic viruses improve cancer immunotherapy by reprogramming solid tumor microenvironment. Med. Oncol. https://doi.org/10.1007/s12032-023-02233-0 (2023).
Grzybowski, M. M. et al. Metabolomic reprogramming of the tumor microenvironment by dual arginase inhibitor OATD-02 boosts anticancer immunity. Sci. Rep. 15, 18741 (2025).
Platten, M., Wick, W. & Van den Eynde, B. J. Tryptophan catabolism in cancer: beyond IDO and tryptophan depletion. Cancer Res. 72, 5435–5440 (2012).
Hayes, C., Donohoe, C. L., Davern, M. & Donlon, N. E. The oncogenic and clinical implications of lactate induced immunosuppression in the tumour microenvironment. Cancer Lett. 500, 75–86 (2021).
Mathewson, N. D. et al. Inhibitory CD161 receptor identified in glioma-infiltrating T cells by single-cell analysis. Cell 184, 1281–1298 (2021).
Ribas, A. et al. Oncolytic virotherapy promotes intratumoral T cell infiltration and improves anti-PD-1 immunotherapy. Cell 170, 1109–1119 (2017).
Chiocca, E. A. et al. Regulatable interleukin-12 gene therapy in patients with recurrent high-grade glioma: results of a phase 1 trial. Sci. Transl. Med. https://doi.org/10.1126/scitranslmed.aaw5680 (2019).
Friedman, G. K. et al. Oncolytic HSV-1 G207 immunovirotherapy for pediatric high-grade gliomas. N. Engl. J. Med. 384, 1613–1622 (2021).
Lang, F. F. et al. Phase I study of DNX-2401 (Delta-24-RGD) oncolytic adenovirus: replication and immunotherapeutic effects in recurrent malignant glioma. J. Clin. Oncol. 36, 1419–1427 (2018).
Malvehy, J. et al. Talimogene laherparepvec upregulates immune-cell populations in non-injected lesions: findings from a phase II, multicenter, open-label study in patients with stage IIIB–IVM1c melanoma. J. Immunother. Cancer https://doi.org/10.1136/jitc-2020-001621 (2021).
Lin, D., Shen, Y. & Liang, T. Oncolytic virotherapy: basic principles, recent advances and future directions. Signal Transduct. Target. Ther. 8, 156 (2023).
Zamarin, D. et al. Localized oncolytic virotherapy overcomes systemic tumor resistance to immune checkpoint blockade immunotherapy. Sci. Transl. Med. 6, 226ra32 (2014).
Kaufman, H. L., Kohlhapp, F. J. & Zloza, A. Oncolytic viruses: a new class of immunotherapy drugs. Nat. Rev. Drug Discov. 14, 642–662 (2015).
Ling, A. L. et al. Clinical trial links oncolytic immunoactivation to survival in glioblastoma. Nature 623, 157–166 (2023).
Nadafi, R., Dong, W. & van Beusechem, V. W. Immunological impact of oncolytic adenoviruses on cancer therapy: clinical insights. Eur. J. Immunol. 55, e70024 (2025).
Vanderlugt, C. L. & Miller, S. D. Epitope spreading in immune-mediated diseases: implications for immunotherapy. Nat. Rev. Immunol. 2, 85–95 (2002).
Chen, X. et al. An oncolytic virus delivering tumor-irrelevant bystander T cell epitopes induces anti-tumor immunity and potentiates cancer immunotherapy. Nat. Cancer 5, 1063–1081 (2024).
Delaunay, T. et al. Oncolytic viruses sensitize human tumor cells for NY-ESO-1 tumor antigen recognition by CD4+ effector T cells. Oncoimmunology 7, e1407897 (2017).
Brossart, P. The role of antigen spreading in the efficacy of immunotherapies. Clin. Cancer Res. 26, 4442–4447 (2020).
Keskin, D. B. et al. Neoantigen vaccine generates intratumoral T cell responses in phase Ib glioblastoma trial. Nature 565, 234–239 (2019).
Lu, Y. & Zhao, F. Strategies to overcome tumour relapse caused by antigen escape after CAR T therapy. Mol. Cancer 24, 126 (2025).
Markert, J. M. et al. Phase Ib trial of mutant herpes simplex virus G207 inoculated pre-and post-tumor resection for recurrent GBM. Mol. Ther. 17, 199–207 (2009).
Todo, T. et al. Intratumoral oncolytic herpes virus G47∆ for residual or recurrent glioblastoma: a phase 2 trial. Nat. Med. 28, 1630–1639 (2022).
Chiocca, E. A. et al. A phase I open-label, dose-escalation, multi-institutional trial of injection with an E1B-attenuated adenovirus, ONYX-015, into the peritumoral region of recurrent malignant gliomas, in the adjuvant setting. Mol. Ther. 10, 958–966 (2004).
Andtbacka, R. H. et al. Talimogene laherparepvec improves durable response rate in patients with advanced melanoma. J. Clin. Oncol. 33, 2780–2788 (2015).
Kaufman, H. L. et al. Local and distant immunity induced by intralesional vaccination with an oncolytic herpes virus encoding GM-CSF in patients with stage IIIc and IV melanoma. Ann. Surg. Oncol. 17, 718–730 (2010).
Hofbauer, G. F. L. et al. Clinical phase I intratumoral administration of two recombinant ALVAC canarypox viruses expressing human granulocyte–macrophage colony-stimulating factor or interleukin-2: the transgene determines the composition of the inflammatory infiltrate. Melanoma Res. 18, 104–111 (2008).
Chiocca, E. A. et al. A phase I trial of Ad.hIFN-β gene therapy for glioma. Mol. Ther. 16, 618–626 (2008).
Algazi, A. et al. Intratumoral delivery of tavokinogene telseplasmid yields systemic immune responses in metastatic melanoma patients. Ann. Oncol. 31, 532–540 (2020).
Colombo, F. et al. Combined HSV-TK/IL-2 gene therapy in patients with recurrent glioblastoma multiforme: biological and clinical results. Cancer Gene Ther. 12, 835–848 (2005).
Umemura, Y. et al. Combined cytotoxic and immune-stimulatory gene therapy for primary adult high-grade glioma: a phase 1, first-in-human trial. Lancet Oncol. 24, 1042–1052 (2023).
Park, S. H. et al. Phase 1b trial of biweekly intravenous pexa-vec (JX-594), an oncolytic and immunotherapeutic vaccinia virus in colorectal cancer. Mol. Ther. 23, 1532–1540 (2015).
Monberg, T. J. et al. Safety and efficacy of combined treatment with tumor-infiltrating lymphocytes and oncolytic adenovirus TILT-123 in metastatic melanoma. Cell Rep. Med. https://doi.org/10.1016/j.xcrm.2025.102016 (2025).
Ramesh, N. et al. CG0070, a conditionally replicating granulocyte macrophage colony-stimulating factor-armed oncolytic adenovirus for the treatment of bladder cancer. Clin. Cancer Res. 12, 305–313 (2006).
Patel, D. M. et al. Design of a phase I clinical trial to evaluate M032, a genetically engineered HSV-1 expressing IL-12, in patients with recurrent/progressive glioblastoma multiforme, anaplastic astrocytoma, or gliosarcoma. Hum. Gene Ther. Clin. Dev. 27, 69–78 (2016).
Garber, K. China approves world’s first oncolytic virus therapy for cancer treatment. J. Natl Cancer Inst. 98, 298–300 (2006).
Rigvir marketing authorisation suspended; information for current patients. State Agency of Medicines of Latvia https://www.zva.gov.lv/en/news-and-publications/news/rigvir-marketing-authorisation-suspended-information-current-patients (2019).
Alberts, P., Tilgase, A., Rasa, A., Bandere, K. & Venskus, D. The advent of oncolytic virotherapy in oncology: the Rigvir® story. Eur. J. Pharmacol. 837, 117–126 (2018).
Andtbacka, R. H. I. et al. Final analyses of OPTiM: a randomized phase III trial of talimogene laherparepvec versus granulocyte–macrophage colony-stimulating factor in unresectable stage III–IV melanoma. J. Immunother. Cancer 7, 145 (2019).
Dummer, R. et al. Neoadjuvant talimogene laherparepvec plus surgery versus surgery alone for resectable stage IIIB–IVM1a melanoma: a randomized, open-label, phase 2 trial. Nat. Med. 27, 1789–1796 (2021).
Amgen seeks a second chance for Imlygic in the competitive melanoma market. Pharmaceutical Technology https://www.pharmaceutical-technology.com/comment/amgen-seeks-second-chance-imlygic-competitive-melanoma-market/ (2018).
Chesney, J. A. et al. Randomized, double-blind, placebo-controlled, global phase III trial of talimogene laherparepvec combined with pembrolizumab for advanced melanoma. J. Clin. Oncol. 41, 528–540 (2023).
Colbert, L. et al. FDA approval summary: nadofaragene firadenovec-vncg for Bacillus Calmette–Guérin-unresponsive non-muscle-invasive bladder cancer. Clin. Cancer Res. 31, 1182–1185 (2025).
Ferring bolsters global gene therapy supply chain with European manufacturing facility. Ferring Pharmaceuticals https://www.ferring.com/ferring-bolsters-global-gene-therapy-supply-chain-with-european-manufacturing-facility/ (2024).
Ning, W. et al. Non-secreting IL12 expressing oncolytic adenovirus Ad-TD-nsIL12 in recurrent high-grade glioma: a phase I trial. Nat. Commun. 15, 9299 (2024).
Noonan, A. M. et al. Randomized phase 2 trial of the oncolytic virus pelareorep (Reolysin) in upfront treatment of metastatic pancreatic adenocarcinoma. Mol. Ther. 24, 1150–1158 (2016).
Noh, M. H. et al. Targeting IGF2 to reprogram the tumor microenvironment for enhanced viro-immunotherapy. Neuro Oncol. 26, 1602–1616 (2024).
Toker, J. et al. Clinical importance of the lncRNA NEAT1 in cancer patients treated with immune checkpoint inhibitors. Clin. Cancer Res. 29, 2226–2238 (2023).
Mineo, M. et al. Tumor interferon signaling is regulated by a lncRNA INCR1 transcribed from the PD-L1 locus. Mol. Cell 78, 1207–1223 (2020).
Meylan, M. et al. Persistent T cell activation and cytotoxicity against glioblastoma following single oncolytic virus treatment in a clinical trial. Cell https://doi.org/10.1016/j.cell.2025.12.055 (2026).
Gulley, J. L. et al. Role of antigen spread and distinctive characteristics of immunotherapy in cancer treatment. J. Natl Cancer Inst. https://doi.org/10.1093/jnci/djw261 (2017).
Katayama, Y. et al. Oncolytic reovirus inhibits immunosuppressive activity of myeloid-derived suppressor cells in a TLR3-dependent manner. J. Immunol. 200, 2987–2999 (2018).
Breitbach, C. J. et al. Targeting tumor vasculature with an oncolytic virus. Mol. Ther. 19, 886–894 (2011).
Bazan-Peregrino, M. et al. VCN-01 disrupts pancreatic cancer stroma and exerts antitumor effects. J. Immunother. Cancer https://doi.org/10.1136/jitc-2021-003254 (2021).
Cervera, S. T., Martínez, S., Iranzo-Martínez, M., de Mera, R. M. M.-F. & Alonso, J. Suicide gene therapy targeting Ewing sarcoma via an Ewing-specific GGAA promoter. Sci. Rep. 15, 29020 (2025).
Haines, B. B. et al. ONCR-177, an oncolytic HSV-1 designed to potently activate systemic antitumor immunity. Cancer Immunol. Res. 9, 291–308 (2021).
Gujar, S., Pol, J. G., Kim, Y., Lee, P. W. & Kroemer, G. Antitumor benefits of antiviral immunity: an underappreciated aspect of oncolytic virotherapies. Trends Immunol. 39, 209–221 (2018).
Chaurasiya, S., Chen, N. G. & Fong, Y. Oncolytic viruses and immunity. Curr. Opin. Immunol. 51, 83–90 (2018).
Taipale, K. et al. Predictive and prognostic clinical variables in cancer patients treated with adenoviral oncolytic immunotherapy. Mol. Ther. 24, 1323–1332 (2016).
Ikeda, K. et al. Oncolytic virus therapy of multiple tumors in the brain requires suppression of innate and elicited antiviral responses. Nat. Med. 5, 881–887 (1999).
Wakimoto, H., Fulci, G., Tyminski, E. & Chiocca, E. A. Altered expression of antiviral cytokine mRNAs associated with cyclophosphamide’s enhancement of viral oncolysis. Gene Ther. 11, 214–223 (2004).
Johnson, D. C. & Huber, M. T. Directed egress of animal viruses promotes cell-to-cell spread. J. Virol. 76, 1–8 (2002).
Ding, J. et al. Pre-existing HSV-1 immunity enhances anticancer efficacy of a novel immune-stimulating oncolytic virus. Viruses https://doi.org/10.3390/v14112327 (2022).
Ricca, J. M. et al. Pre-existing immunity to oncolytic virus potentiates its immunotherapeutic efficacy. Mol. Ther. 26, 1008–1019 (2018).
Gujar, S. A., Pan, D. A., Marcato, P., Garant, K. A. & Lee, P. W. K. Oncolytic virus-initiated protective immunity against prostate cancer. Mol. Ther. 19, 797–804 (2011).
Li, X. et al. The efficacy of oncolytic adenovirus is mediated by T-cell responses against virus and tumor in Syrian hamster model. Clin. Cancer Res. 23, 239–249 (2017).
Cao, D. et al. Redirecting anti-Vaccinia virus T cell immunity for cancer treatment by AAV-mediated delivery of the VV B8R gene. Mol. Ther. Oncolytics 25, 264–275 (2022).
Pourchet, A. et al. CD8+ T-cell immune evasion enables oncolytic virus immunotherapy. EBioMedicine 5, 59–67 (2016).
Todo, T., Martuza, R. L., Rabkin, S. D. & Johnson, P. A. Oncolytic herpes simplex virus vector with enhanced MHC class I presentation and tumor cell killing. Proc. Natl Acad. Sci. USA 98, 6396–6401 (2001).
Tähtinen, S. et al. Exploiting preexisting immunity to enhance oncolytic cancer immunotherapy. Cancer Res. 80, 2575–2585 (2020).
Chuang, C.-M., Monie, A., Wu, A., Pai, S. I. & Hung, C.-F. Combination of viral oncolysis and tumor-specific immunity to control established tumors. Clin. Cancer Res. 15, 4581–4588 (2009).
Hecht, J. R. et al. A phase I/II trial of intratumoral endoscopic ultrasound injection of ONYX-015 with intravenous gemcitabine in unresectable pancreatic carcinoma. Clin. Cancer Res. 9, 555–561 (2003).
Li, R. et al. Oncolytic adenoviral therapy plus pembrolizumab in BCG-unresponsive non-muscle-invasive bladder cancer: the phase 2 CORE-001 trial. Nat. Med. 30, 2216–2223 (2024).
Li, R. et al. Oncolytic immunotherapy with nivolumab in muscle-invasive bladder cancer: a phase 1b trial. Nat. Med. 31, 176–188 (2025).
Boorjian, S. A. et al. Intravesical nadofaragene firadenovec gene therapy for BCG-unresponsive non-muscle-invasive bladder cancer: a single-arm, open-label, repeat-dose clinical trial. Lancet Oncol. 22, 107–117 (2021).
Kim, K. H. et al. A phase I clinical trial of Ad5/3-Δ24, a novel serotype-chimeric, infectivity-enhanced, conditionally-replicative adenovirus (CRAd), in patients with recurrent ovarian cancer. Gynecol. Oncol. 130, 518–524 (2013).
Kimball, K. J. et al. A phase I study of a tropism-modified conditionally replicative adenovirus for recurrent malignant gynecologic diseases. Clin. Cancer Res. 16, 5277–5287 (2010).
Galanis, E. et al. Phase I trial of intraperitoneal administration of an oncolytic measles virus strain engineered to express carcinoembryonic antigen for recurrent ovarian cancer. Cancer Res. 70, 875–882 (2010).
Geevarghese, S. K. et al. Phase I/II study of oncolytic herpes simplex virus NV1020 in patients with extensively pretreated refractory colorectal cancer metastatic to the liver. Hum. Gene Ther. 21, 1119–1128 (2010).
Reid, T. R., Freeman, S., Post, L., McCormick, F. & Sze, D. Y. Effects of Onyx-015 among metastatic colorectal cancer patients that have failed prior treatment with 5-FU/leucovorin. Cancer Gene Ther. 12, 673–681 (2005).
Kang, K.-D. et al. Safety and efficacy of intraventricular immunovirotherapy with oncolytic HSV-1 for CNS cancers. Clin. Cancer Res. 28, 5419–5430 (2022).
Wagner, S. et al. Combined treatment of pediatric high-grade glioma with the oncolytic viral strain MTH-68/H and oral valproic acid. APMIS 114, 731–743 (2006).
Ruano, D. et al. First-in-human, first-in-child trial of autologous MSCs carrying the oncolytic virus Icovir-5 in patients with advanced tumors. Mol. Ther. 28, 1033–1042 (2020).
Fares, J. et al. Neural stem cell delivery of an oncolytic adenovirus in newly diagnosed malignant glioma: a first-in-human, phase 1, dose-escalation trial. Lancet Oncol. 22, 1103–1114 (2021).
Du, Y.-N. et al. Hydrogel-based co-delivery of CIK cells and oncolytic adenovirus armed with IL12 and IL15 for cancer immunotherapy. Biomed. Pharmacother. https://doi.org/10.1016/j.biopha.2022.113110 (2022).
Garcia-Carbonero, R. et al. Phase I, multicenter, open-label study of intravenous VCN-01 oncolytic adenovirus with or without nab-paclitaxel plus gemcitabine in patients with advanced solid tumors. J. Immunother. Cancer https://doi.org/10.1136/jitc-2021-003255 (2022).
Hajda, J. et al. Phase 2 trial of oncolytic H-1 parvovirus therapy shows safety and signs of immune system activation in patients with metastatic pancreatic ductal adenocarcinoma. Clin. Cancer Res. 27, 5546–5556 (2021).
Mahalingam, D. et al. Pembrolizumab in combination with the oncolytic virus pelareorep and chemotherapy in patients with advanced pancreatic adenocarcinoma: a phase Ib study. Clin. Cancer Res. 26, 71–81 (2020).
Fakih, M. et al. Safety and efficacy of the tumor-selective adenovirus enadenotucirev, in combination with nivolumab, in patients with advanced/metastatic epithelial cancer: a phase I clinical trial (SPICE). J. Immunother. Cancer https://doi.org/10.1136/jitc-2022-006561 (2023).
García, M. et al. A phase 1 trial of oncolytic adenovirus ICOVIR-5 administered intravenously to cutaneous and uveal melanoma patients. Hum. Gene Ther. 30, 352–364 (2019).
Msaouel, P. et al. Clinical trials with oncolytic measles virus: current status and future prospects. Curr. Cancer Drug Targets 18, 177–187 (2018).
Wakimoto, H. et al. The complement response against an oncolytic virus is species-specific in its activation pathways. Mol. Ther. 5, 275–282 (2002).
Jirovec, E. et al. Single intravenous administration of oncolytic adenovirus TILT-123 results in systemic tumor transduction and immune response in patients with advanced solid tumors. J. Exp. Clin. Cancer Res. 43, 297 (2024).
Wakimoto, H., Johnson, P. R., Knipe, D. M. & Chiocca, E. A. Effects of innate immunity on herpes simplex virus and its ability to kill tumor cells. Gene Ther. 10, 983–990 (2003).
Ikeda, K. et al. Complement depletion facilitates the infection of multiple brain tumors by an intravascular, replication-conditional herpes simplex virus mutant. J. Virol. 74, 4765–4775 (2000).
Geletneky, K. et al. Oncolytic H-1 parvovirus shows safety and signs of immunogenic activity in a first phase I/IIa glioblastoma trial. Mol. Ther. 25, 2620–2634 (2017).
Lawler, S. E., Speranza, M.-C., Cho, C.-F. & Chiocca, E. A. Oncolytic viruses in cancer treatment: a review. JAMA Oncol. 3, 841–849 (2017).
Perez, M. C. et al. Observational study of talimogene laherparepvec use for melanoma in clinical practice in the United States (COSMUS-1). Melanoma Manag. https://doi.org/10.2217/mmt-2019-0012 (2019).
Smith, K. E. R. et al. A phase I oncolytic virus trial with vesicular stomatitis virus expressing human interferon beta and tyrosinase related protein 1 administered intratumorally and intravenously in uveal melanoma: safety, efficacy, and T cell responses. Front. Immunol. 14, 1279387 (2023).
Shoaf, M. L. & Desjardins, A. Oncolytic viral therapy for malignant glioma and their application in clinical practice. Neurotherapeutics 19, 1818–1831 (2022).
Oncolytic virus therapy market size, share, growth, and industry analysis, by type (HSV-based oncolytic viruses, adenoviruses-based oncolytic viruses, vaccinia virus-based oncolytic viruses, vesicular stomatitis virus-based oncolytic viruses, Newcastle disease virus-based oncolytic viruses), by application (melanoma, prostate cancer, breast cancer, ovarian cancer, others), regional insights and forecast to 2034. 360 Research Reports. https://www.360researchreports.com/market-reports/oncolytic-virus-therapy-market-200773 (2025).
Ungerechts, G. et al. Moving oncolytic viruses into the clinic: clinical-grade production, purification, and characterization of diverse oncolytic viruses. Mol. Ther. Methods Clin. Dev. 3, 16018 (2016).
Liu, J. S. et al. Spatial and temporal heterogeneity of tumor immune microenvironment between primary tumor and brain metastases in NSCLC. BMC Cancer 24, 123 (2024).
Gerlinger, M. et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N. Engl. J. Med. 366, 883–892 (2012).
Kurokawa, C. et al. Constitutive interferon pathway activation in tumors as an efficacy determinant following oncolytic virotherapy. J. Natl Cancer Inst. 110, 1123–1132 (2018).
Galanis, E. et al. Carcinoembryonic antigen-expressing oncolytic measles virus derivative in recurrent glioblastoma: a phase 1 trial. Nat. Commun. 15, 493 (2024).
Nassiri, F. et al. Oncolytic DNX-2401 virotherapy plus pembrolizumab in recurrent glioblastoma: a phase 1/2 trial. Nat. Med. 29, 1370–1378 (2023).
Wen, P. Y. et al. A multi-institutional phase 1 clinical trial exploring upfront multimodal standard of care and combined immunotherapies for newly diagnosed glioblastoma. Neuro Oncol. https://doi.org/10.1093/neuonc/noaf079 (2025).
Mahalingam, D. et al. A phase II study of pelareorep (REOLYSIN®) in combination with gemcitabine for patients with advanced pancreatic adenocarcinoma. Cancers (Basel) https://doi.org/10.3390/cancers10060160 (2018).
Karapanagiotou, E. M. et al. Phase I/II trial of carboplatin and paclitaxel chemotherapy in combination with intravenous oncolytic reovirus in patients with advanced malignancies. Clin. Cancer Res. 18, 2080–2089 (2012).
Jonker, D. J. et al. A randomized phase II study of FOLFOX6/bevacizumab with or without pelareorep in patients with metastatic colorectal cancer: IND.210, a Canadian Cancer Trials Group trial. Clin. Colorectal Cancer 17, 231–239 (2018).
Cohn, D. E. et al. Randomized phase IIB evaluation of weekly paclitaxel versus weekly paclitaxel with oncolytic reovirus (Reolysin®) in recurrent ovarian, tubal, or peritoneal cancer: an NRG Oncology/Gynecologic Oncology Group study. Gynecol. Oncol. 146, 477–483 (2017).
Eigl, B. J. et al. A randomized phase II study of pelareorep and docetaxel or docetaxel alone in men with metastatic castration resistant prostate cancer: CCTG study IND 209. Oncotarget 9, 8155–8164 (2018).
Xie, R., Huang, H., Chen, T., Huang, X. & Chen, C. Effectiveness and safety of pelareorep plus chemotherapy versus chemotherapy alone for advanced solid tumors: a meta-analysis. Front. Pharmacol. 14, 1228225 (2023).
Li, Y. et al. Oncolytic virus combined with traditional treatment versus traditional treatment alone in patients with cancer: a meta-analysis. Int. J. Clin. Oncol. 25, 1901–1913 (2020).
Shirakawa, Y. et al. Phase I dose-escalation study of endoscopic intratumoral injection of OBP-301 (telomelysin) with radiotherapy in oesophageal cancer patients unfit for standard treatments. Eur. J. Cancer 153, 98–108 (2021).
Meng, X., Feng, R., Yang, L., Xing, L. & Yu, J. The role of radiation oncology in immuno-oncology. Oncologist 24, S42–S52 (2019).
Markert, J. M. et al. A phase 1 trial of oncolytic HSV-1, G207, given in combination with radiation for recurrent GBM demonstrates safety and radiographic responses. Mol. Ther. 22, 1048–1055 (2014).
O’Cathail, S. M. et al. A phase 1 trial of the safety, tolerability, and biological effects of intravenous enadenotucirev (EnAd), a novel oncolytic virus, in combination with chemoradiotherapy in locally advanced rectal cancer (CEDAR). Int. J. Radiat. Oncol. Biol. Phys. 117, e329–e330 (2023).
Gállego Pérez-Larraya, J. et al. Oncolytic DNX-2401 virus for pediatric diffuse intrinsic pontine glioma. N. Engl. J. Med. 386, 2471–2481 (2022).
Bernstock, J. D. et al. A novel in situ multiplex immunofluorescence panel for the assessment of tumor immunopathology and response to virotherapy in pediatric glioblastoma reveals a role for checkpoint protein inhibition. Oncoimmunology 8, e1678921 (2019).
Puzanov, I. et al. Talimogene laherparepvec in combination with ipilimumab in previously untreated, unresectable stage IIIB–IV melanoma. J. Clin. Oncol. 34, 2619–2626 (2016).
Dummer, R. et al. Neoadjuvant anti-PD-1 alone or in combination with anti-TIGIT or an oncolytic virus in resectable stage IIIB–D melanoma: a phase 1/2 trial. Nat. Med. 31, 144–151 (2025).
Rudin, C. M. et al. Phase 1, open-label, dose-escalation study on the safety, pharmacokinetics, and preliminary efficacy of intravenous Coxsackievirus A21 (V937), with or without pembrolizumab, in patients with advanced solid tumors. J. Immunother. Cancer https://doi.org/10.1136/jitc-2022-005007 (2023).
Schuelke, M. R. et al. Phase I trial of sargramostim/pelareorep therapy in pediatric patients with recurrent or refractory high-grade brain tumors. Neurooncol. Adv. https://doi.org/10.1093/noajnl/vdac085 (2022).
Cyrelle Ornella, M. S., Kim, J.-J., Cho, E., Cho, M. & Hwang, T.-H. Dose considerations for vaccinia oncolytic virus based on retrospective reanalysis of early and late clinical trials. Vaccines (Basel) https://doi.org/10.3390/vaccines12091010 (2024).
Chahlavi, A., Rabkin, S., Todo, T., Sundaresan, P. & Martuza, R. Effect of prior exposure to herpes simplex virus 1 on viral vector-mediated tumor therapy in immunocompetent mice. Gene Ther. 6, 1751–1758 (1999).
Heo, J. et al. Randomized dose-finding clinical trial of oncolytic immunotherapeutic vaccinia JX-594 in liver cancer. Nat. Med. 19, 329–336 (2013).
Bhatnagar, A. R. et al. Long-term follow-up of phase I trial of oncolytic adenovirus-mediated cytotoxic and interleukin-12 gene therapy for treatment of metastatic pancreatic cancer. Biomedicines https://doi.org/10.3390/biomedicines12051065 (2024).
Ling, A. L. et al. Serial multiomics uncovers anti-glioblastoma responses not evident by routine clinical analyses. Sci. Transl. Med. 17, eadv2881 (2025).
Brenner, A. J. et al. Safety and efficacy of VB-111, an anticancer gene therapy, in patients with recurrent glioblastoma: results of a phase I/II study. Neuro Oncol. 22, 694–704 (2020).
Kelly, E. & Russell, S. J. History of oncolytic viruses: genesis to genetic engineering. Mol. Ther. 15, 651–659 (2007).
Stepanenko, A. A. et al. Superior infectivity of the fiber chimeric oncolytic adenoviruses Ad5/35 and Ad5/3 over Ad5-delta-24-RGD in primary glioma cultures. Mol. Ther. Oncolytics 24, 230–248 (2021).
Ludwig, B. S., Kessler, H., Kossatz, S. & Reuning, U. RGD-binding integrins revisited: how recently discovered functions and novel synthetic ligands (re-)shape an ever-evolving field. Cancers (Basel) https://doi.org/10.3390/cancers13071711 (2021).
Liszewski, M. K. & Atkinson, J. P. The multiverse of CD46 and oncologic interactions. J. Clin. Invest. https://doi.org/10.1172/jci188355 (2025).
Mineta, T., Rabkin, S. D., Yazaki, T., Hunter, W. D. & Martuza, R. L. Attenuated multi-mutated herpes simplex virus-1 for the treatment of malignant gliomas. Nat. Med. 1, 938–943 (1995).
Markert, J. M. et al. Conditionally replicating herpes simplex virus mutant, G207 for the treatment of malignant glioma: results of a phase I trial. Gene Ther. 7, 867–874 (2000).
Chiocca, E. A., Nakashima, H., Kasai, K., Fernandez, S. A. & Oglesbee, M. Preclinical toxicology of rQNestin34.5v.2: an oncolytic herpes virus with transcriptional regulation of the ICP34.5 neurovirulence gene. Mol. Ther. Methods Clin. Dev. 17, 871–893 (2020).
Kambara, H., Okano, H., Chiocca, E. A. & Saeki, Y. An oncolytic HSV-1 mutant expressing ICP34.5 under control of a nestin promoter increases survival of animals even when symptomatic from a brain tumor. Cancer Res. 65, 2832–2839 (2005).
Chiocca, E. A. et al. Combined immunotherapy with controlled interleukin-12 gene therapy and immune checkpoint blockade in recurrent glioblastoma: an open-label, multi-institutional phase I trial. Neuro Oncol. 24, 951–963 (2022).
Qian, X. et al. The oncolytic adenovirus Ad-TD-nsIL12 in primary or progressive pediatric IDH wild-type diffuse intrinsic pontine glioma results of two phase I clinical trials. Nat. Commun. 16, 6934 (2025).
Shen, Y. et al. Oncolytic virus VG161 in refractory hepatocellular carcinoma. Nature 641, 503–511 (2025).
Chu, Y. et al. Combinatorial immunotherapy with anti-ROR1 CAR NK cells and an IL-21 secreting oncolytic virus against neuroblastoma. Mol. Ther. Oncol. 33, 200927 (2025).
Guo, Y., Luan, L., Patil, N. K. & Sherwood, E. R. Immunobiology of the IL-15/IL-15Rα complex as an antitumor and antiviral agent. Cytokine Growth Factor Rev. 38, 10–21 (2017).
Kowalsky, S. J. et al. Superagonist IL-15-armed oncolytic virus elicits potent antitumor immunity and therapy that are enhanced with PD-1 blockade. Mol. Ther. 26, 2476–2486 (2018).
Zhu, W., Wei, L., Zhang, H., Chen, J. & Qin, X. Oncolytic adenovirus armed with IL-24 inhibits the growth of breast cancer in vitro and in vivo. J. Exp. Clin. Cancer Res. 31, 51 (2012).
West, E. J. et al. A phase I clinical trial of intrahepatic artery delivery of TG6002 in combination with oral 5-fluorocytosine in patients with liver-dominant metastatic colorectal cancer. Clin. Cancer Res. 31, 1243–1256 (2025).
Bernstock, J. D. et al. Recent oncolytic virotherapy clinical trials outline a roadmap for the treatment of high-grade glioma. Neurooncol. Adv. 5, vdad081 (2023).
Park, H. J. et al. Incidence of pseudoprogression during immune checkpoint inhibitor therapy for solid tumors: a systematic review and meta-analysis. Radiology 297, 87–96 (2020).
Chen, E., Ling, A. L., Reardon, D. A. & Chiocca, E. A. Lessons learned from phase 3 trials of immunotherapy for glioblastoma: time for longitudinal sampling? Neuro Oncol. 26, 211–225 (2024).
Yu, K. K. H. et al. Investigative needle core biopsies support multimodal deep-data generation in glioblastoma. Nat. Commun. 16, 3957 (2025).
Lowenstein, P. R. Now you see me; now you don’t. Sci. Transl. Med. 17, eadz1286 (2025).
Geoerger, B. et al. Oncolytic activity of the E1B-55 kDa-deleted adenovirus ONYX-015 is independent of cellular p53 status in human malignant glioma xenografts. Cancer Res. 62, 764–772 (2002).
Wong, M. K. et al. RP1 combined with nivolumab in advanced anti-PD-1-failed melanoma (IGNYTE). J. Clin. Oncol. https://doi.org/10.1200/jco-25-01346 (2025).
Cui, C. et al. OrienX010, an oncolytic virus, in patients with unresectable stage IIIC–IV melanoma: a phase Ib study. J. Immunother. Cancer https://doi.org/10.1136/jitc-2021-004307 (2022).
Liu, J. et al. Neoadjuvant oncolytic virus orienx010 and toripalimab in resectable acral melanoma: a phase Ib trial. Signal Transduct. Target. Ther. 9, 318 (2024).
Andtbacka, R. H. I. et al. Clinical responses of oncolytic coxsackievirus A21 (V937) in patients with unresectable melanoma. J. Clin. Oncol. 39, 3829–3838 (2021).
Curti, B. D. et al. Intratumoral oncolytic virus V937 plus ipilimumab in patients with advanced melanoma: the phase 1b MITCI study. J. Immunother. Cancer https://doi.org/10.1136/jitc-2022-005224 (2022).
Silk, A. W. et al. A phase 1b single-arm trial of intratumoral oncolytic virus V937 in combination with pembrolizumab in patients with advanced melanoma: results from the CAPRA study. Cancer Immunol. Immunother. 72, 1405–1415 (2023).
Schiza, A. et al. Adenovirus-mediated CD40L gene transfer increases Teffector/Tregulatory cell ratio and upregulates death receptors in metastatic melanoma patients. J. Transl. Med. 15, 79 (2017).
Desjardins, A. et al. Recurrent glioblastoma treated with recombinant poliovirus. N. Engl. J. Med. 379, 150–161 (2018).
Thompson, E. M. et al. Recombinant polio–rhinovirus immunotherapy for recurrent paediatric high-grade glioma: a phase 1b trial. Lancet Child. Adolesc. Health 7, 471–478 (2023).
Kurokawa, C. & Galanis, E. Interferon signaling predicts response to oncolytic virotherapy. Oncotarget 10, 1544–1545 (2019).
Wheeler, L. A. et al. Phase II multicenter study of gene-mediated cytotoxic immunotherapy as adjuvant to surgical resection for newly diagnosed malignant glioma. Neuro Oncol. 18, 1137–1145 (2016).
Burke, J. M. et al. A first in human phase 1 study of CG0070, a GM-CSF expressing oncolytic adenovirus, for the treatment of nonmuscle invasive bladder cancer. J. Urol. 188, 2391–2397 (2012).
Packiam, V. T. et al. An open label, single-arm, phase II multicenter study of the safety and efficacy of CG0070 oncolytic vector regimen in patients with BCG-unresponsive non-muscle-invasive bladder cancer: interim results. Urol. Oncol. 36, 440–447 (2018).
Abou-Alfa, G. K. et al. PHOCUS: a phase 3, randomized, open-label study of sequential treatment with pexa-vec (JX-594) and sorafenib in patients with advanced hepatocellular carcinoma. Liver Cancer 13, 248–264 (2023).





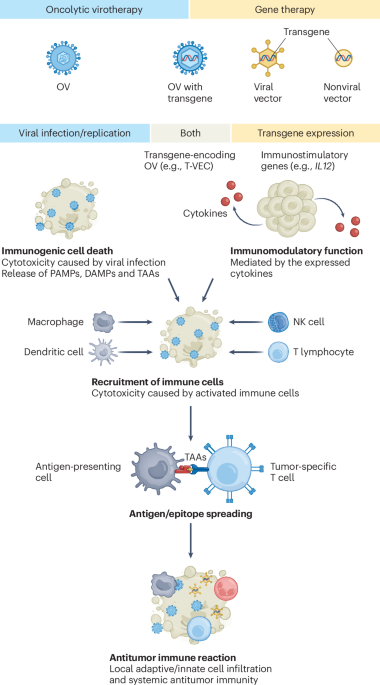









Leave a Reply