Anderson G, Jenkinson EJ. Lymphostromal interactions in thymic development and function. Nat Rev Immunol. 2001;1:31–40.
Takahama Y. Journey through the thymus: stromal guides for T-cell development and selection. Nat Rev Immunol. 2006;6:127–35.
Petrie HT, Zúñiga-Pflücker JC. Zoned out: functional mapping of stromal signaling microenvironments in the thymus. Annu Rev Immunol. 2007;25:649–79.
Rosichini M, Bordoni V, Silvestris DA, Mariotti D, Matusali G, Cardinale A, et al. SARS-CoV-2 infection of thymus induces loss of function that correlates with disease severity. J Allergy Clin Immunol. 2023;151:911–21.
Velardi E, Clave E, Arruda LCM, Benini F, Locatelli F, Toubert A. The role of the thymus in allogeneic bone marrow transplantation and the recovery of the peripheral T-cell compartment. Semin Immunopathol. 2021;43:101–17.
Velardi E, Tsai JJ, van den Brink MRM. T cell regeneration after immunological injury. Nat Rev Immunol. 2021;21:277–91.
Komanduri KV, St John LS, De Lima M, McMannis J, Rosinski S, McNiece I, et al. Delayed immune reconstitution after cord blood transplantation is characterized by impaired thymopoiesis and late memory T-cell skewing. Blood. 2007;110:4543–51.
Clave E, Lisini D, Douay C, Giorgiani G, Busson M, Zecca M, et al. A low thymic function is associated with leukemia relapse in children given T-cell-depleted HLA-haploidentical stem cell transplantation. Leukemia. 2012;26:1886–8.
Söderström A, Vonlanthen S, Jönsson-Videsäter K, Mielke S, Lindahl H, Törlén J, et al. T cell receptor excision circles are potential predictors of survival in adult allogeneic hematopoietic stem cell transplantation recipients with acute myeloid leukemia. Front Immunol. 2022;13:954716.
Troullioud Lucas AG, Lindemans CA, Bhoopalan SV, Dandis R, Prockop SE, Naik S, et al. Early immune reconstitution as predictor for outcomes after allogeneic hematopoietic cell transplant; a tri-institutional analysis. Cytotherapy. 2023;25:977–85.
Goldberg JD, Zheng J, Ratan R, Small TN, Lai K-C, Boulad F, et al. Early recovery of T-cell function predicts improved survival after T-cell depleted allogeneic transplant. Leuk Lymphoma. 2017;58:1859–71.
Bartelink IH, Belitser SV, Knibbe CAJ, Danhof M, De Pagter AJ, Egberts TCG, et al. Immune reconstitution kinetics as an early predictor for mortality using various hematopoietic stem cell sources in children. Biol Blood Marrow Transpl. 2013;19:305–13.
Admiraal R, Chiesa R, Lindemans CA, Nierkens S, Bierings MB, Versluijs AB, et al. Leukemia-free survival in myeloid leukemia, but not in lymphoid leukemia, is predicted by early CD4+ reconstitution following unrelated cord blood transplantation in children: a multicenter retrospective cohort analysis. Bone Marrow Transpl. 2016;51:1376–8.
Clave E, Lisini D, Douay C, Giorgiani G, Busson M, Zecca M et al. Thymic function recovery after unrelated donor cord blood or T-cell depleted HLA-haploidentical stem cell transplantation correlates with leukemia relapse. Front Immunol. 2013;4. https://doi.org/10.3389/fimmu.2013.00054.
Maury S, Mary J, Rabian C, Schwarzinger M, Toubert A, Scieux C, et al. Prolonged immune deficiency following allogeneic stem cell transplantation: risk factors and complications in adult patients. Br J Haematol. 2001;115:630–41.
Tsamadou C, Gowdavally S, Platzbecker U, Sala E, Valerius T, Wagner-Drouet E, et al. Donor genetic determinant of thymopoiesis rs2204985 impacts clinical outcome after single HLA mismatched hematopoietic stem cell transplantation. Bone Marrow Transpl. 2022;57:1539–47.
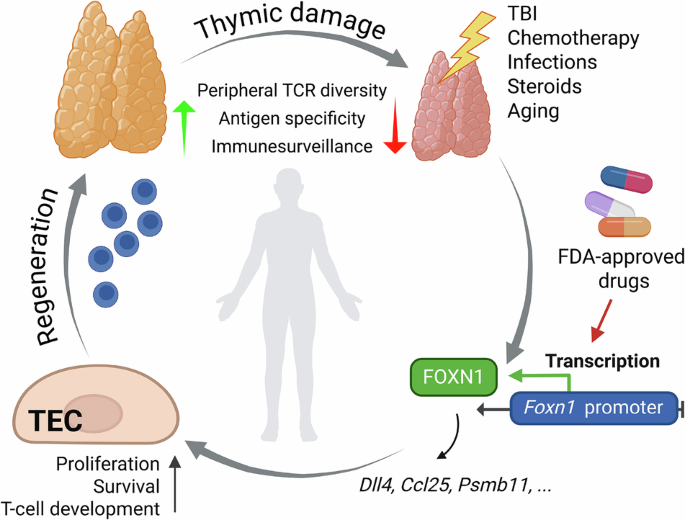
Anderson G, Cosway EJ, James KD, Ohigashi I, Takahama Y. Generation and repair of thymic epithelial cells. J Exp Med. 2024;221:e20230894.
Granadier D, Acenas D, Dudakov JA. Endogenous thymic regeneration: restoring T cell production following injury. Nat Rev Immunol. 2025. https://doi.org/10.1038/s41577-024-01119-0.
Wertheimer T, Velardi E, Tsai J, Cooper K, Xiao S, Kloss CC, et al. Production of BMP4 by endothelial cells is crucial for endogenous thymic regeneration. Sci Immunol. 2018;3:eaal2736.
Vaidya HJ, Briones Leon A, Blackburn CC. FOXN1 in thymus organogenesis and development. Eur J Immunol. 2016;46:1826–37.
Žuklys S, Handel A, Zhanybekova S, Govani F, Keller M, Maio S, et al. Foxn1 regulates key target genes essential for T cell development in postnatal thymic epithelial cells. Nat Immunol. 2016;17:1206–15.
Kinsella S, Evandy CA, Cooper K, Cardinale A, Iovino L, deRoos P et al. Damage-induced pyroptosis drives endogenous thymic regeneration via induction of Foxn1 by purinergic receptor activation. 2023. https://doi.org/10.1101/2023.01.19.524800.
Zhao J, Hu R, Lai KC, Zhang Z, Lai L. Recombinant FOXN1 fusion protein increases T cell generation in old mice. Front Immunol. 2024;15:1423488.
Song Y, Su M, Zhu J, Di W, Liu Y, Hu R, et al. FOXN1 recombinant protein enhances T-cell regeneration after hematopoietic stem cell transplantation in mice. Eur J Immunol. 2016;46:1518–28.
Zook EC, Krishack PA, Zhang S, Zeleznik-Le NJ, Firulli AB, Witte PL, et al. Overexpression of Foxn1 attenuates age-associated thymic involution and prevents the expansion of peripheral CD4 memory T cells. Blood. 2011;118:5723–31.
Bredenkamp N, Nowell CS, Blackburn CC. Regeneration of the aged thymus by a single transcription factor. Development. 2014;141:1627–37.
Chen L, Xiao S, Manley NR. Foxn1 is required to maintain the postnatal thymic microenvironment in a dosage-sensitive manner. Blood. 2009;113:567–74.
Bosticardo M, Yamazaki Y, Cowan J, Giardino G, Corsino C, Scalia G, et al. Heterozygous FOXN1 variants cause low TRECs and severe T cell lymphopenia, revealing a crucial role of FOXN1 in supporting early thymopoiesis. Am J Hum Genet. 2019;105:549–61.
Moses A, Bhalla P, Thompson A, Lai L, Coskun FS, Seroogy CM, et al. Comprehensive phenotypic analysis of diverse FOXN1 variants. J Allergy Clin Immunol. 2023;152:1273–1291.e15.
Larsen BM, Cowan JE, Wang Y, Tanaka Y, Zhao Y, Voisin B, et al. Identification of an intronic regulatory element necessary for tissue-specific expression of Foxn1 in thymic epithelial cells. J Immunol. 2019;203:686–95.
Kadouri N, Givony T, Nevo S, Hey J, Ben Dor S, Damari G, et al. Transcriptional regulation of the thymus master regulator Foxn1. Sci Immunol. 2022;7:eabn8144.
Rossignol J-F. Nitazoxanide: a first-in-class broad-spectrum antiviral agent. Antivir Res. 2014;110:94–103.
Rossignol J-F, Abu-Zekry M, Hussein A, Santoro MG. Effect of nitazoxanide for treatment of severe rotavirus diarrhoea: randomised double-blind placebo-controlled trial. Lancet. 2006;368:124–9.
Rossignol JA, Ayoub A, Ayers MS. Treatment of diarrhea caused by Cryptosporidium parvum: a prospective randomized, double-blind, placebo-controlled study of nitazoxanide. J Infect Dis. 2001;184:103–6.
Rossignol J, Ayoub A, Ayers MS. Treatment of diarrhea caused by Giardia intestinalis and Entamoeba histolytica or E. dispar: a randomized, double-blind, placebo-controlled study of nitazoxanide. J Infect Dis. 2001;184:381–4.
Anderson VR, Curran MP. Nitazoxanide: a review of its use in the treatment of gastrointestinal infections. Drugs. 2007;67:1947–67.
Gilles HM, Hoffman PS. Treatment of intestinal parasitic infections: a review of nitazoxanide. Trends Parasitol. 2002;18:95–97.
Haffizulla J, Hartman A, Hoppers M, Resnick H, Samudrala S, Ginocchio C, et al. Effect of nitazoxanide in adults and adolescents with acute uncomplicated influenza: a double-blind, randomised, placebo-controlled, phase 2b/3 trial. Lancet Infect Dis. 2014;14:609–18.
Lü Z, Li X, Li K, Ripani P, Shi X, Xu F, et al. Nitazoxanide and related thiazolides induce cell death in cancer cells by targeting the 20S proteasome with novel binding modes. Biochem Pharm. 2022;197:114913.
Chen X, Shi C, He M, Xiong S, Xia X. Endoplasmic reticulum stress: molecular mechanism and therapeutic targets. Signal Transduct Target Ther. 2023;8:352.
Hetz C, Zhang K, Kaufman RJ. Mechanisms, regulation and functions of the unfolded protein response. Nat Rev Mol Cell Biol. 2020;21:421–38.
Boyce M, Bryant KF, Jousse C, Long K, Harding HP, Scheuner D, et al. A selective inhibitor of eIF2α dephosphorylation protects cells from ER stress. Science. 2005;307:935–9.
Wortel IMN, Van Der Meer LT, Kilberg MS, Van Leeuwen FN. Surviving stress: modulation of ATF4-mediated stress responses in normal and malignant cells. Trends Endocrinol Metab. 2017;28:794–806.
Neill G, Masson GR. A stay of execution: ATF4 regulation and potential outcomes for the integrated stress response. Front Mol Neurosci. 2023;16:1112253.
Hu Q, Nicol SA, Suen AYW, Baldwin TA. Examination of thymic positive and negative selection by flow cytometry. J Vis Exp. 2012;68:4269.
Li J, Wachsmuth LP, Xiao S, Condie BG, Manley NR. Foxn1 overexpression promotes thymic epithelial progenitor cell proliferation and mTEC maintenance, but does not prevent thymic involution. Dev Camb Engl. 2023;150:dev200995.
Du Q, Huynh LK, Coskun F, Molina E, King MA, Raj P, et al. FOXN1 compound heterozygous mutations cause selective thymic hypoplasia in humans. J Clin Invest. 2019;129:4724–38.
Kousa AI, Jahn L, Zhao K, Flores AE, Acenas D, Lederer E, et al. Age-related epithelial defects limit thymic function and regeneration. Nat Immunol. 2024;25:1593–606.
Horie K, Namiki K, Kinoshita K, Miyauchi M, Ishikawa T, Hayama M, et al. Acute irradiation causes a long-term disturbance in the heterogeneity and gene expression profile of medullary thymic epithelial cells. Front Immunol. 2023;14:1186154.
Bredenkamp N, Nowell CS, Blackburn CC. Regeneration of the aged thymus by a single transcription factor. Dev Camb Engl. 2014;141:1627–37.
Srinivasan J, Lancaster JN, Singarapu N, Hale LP, Ehrlich LIR, Richie ER. Age-related changes in thymic central tolerance. Front Immunol. 2021;12:676236.
Takaba H, Takayanagi H. The mechanisms of T cell selection in the thymus. Trends Immunol. 2017;38:805–16.
Elyahu Y, Monsonego A. Thymus involution sets the clock of the aging T-cell landscape: Implications for declined immunity and tissue repair. Ageing Res Rev. 2021;65:101231.
Liang Z, Dong X, Zhang Z, Zhang Q, Zhao Y. Age-related thymic involution: mechanisms and functional impact. Aging Cell. 2022;21:e13671.
Manasanch EE, Orlowski RZ. Proteasome inhibitors in cancer therapy. Nat Rev Clin Oncol. 2017;14:417–33.
Zhang H, Li X, Liu J, Lin X, Pei L, Boyce BF, et al. Proteasome inhibition-enhanced fracture repair is associated with increased mesenchymal progenitor cells in mice. PloS One. 2022;17:e0263839.
Zhang F, Attarilar S, Xie K, Han C, Qingyang Liang, Huang K, et al. Carfilzomib alleviated osteoporosis by targeting PSME1/2 to activate Wnt/β-catenin signaling. Mol Cell Endocrinol. 2022;540:111520.
Caron AZ, Haroun S, Leblanc É, Trensz F, Guindi C, Amrani A, et al. The proteasome inhibitor MG132 reduces immobilization-induced skeletal muscle atrophy in mice. BMC Musculoskelet Disord. 2011;12:185.
Padrissa-Altés S, Zaouali MA, Boncompagni E, Bonaccorsi-Riani E, Carbonell T, Bardag-Gorce F, et al. The use of a reversible proteasome inhibitor in a model of Reduced-Size Orthotopic Liver transplantation in rats. Exp Mol Pathol. 2012;93:99–110.
Al-Homsi AS, Feng Y, Duffner U, Al Malki MM, Goodyke A, Cole K, et al. Bortezomib for the prevention and treatment of graft-versus-host disease after allogeneic hematopoietic stem cell transplantation. Exp Hematol. 2016;44:771–7.
Magenau JM, Reddy P. Proteasome: target for acute and chronic GVHD?. Blood. 2014;124:1551–2.
Sun K, Welniak LA, Panoskaltsis-Mortari A, O’Shaughnessy MJ, Liu H, Barao I, et al. Inhibition of acute graft-versus-host disease with retention of graft-versus-tumor effects by the proteasome inhibitor bortezomib. Proc Natl Acad Sci USA. 2004;101:8120–5.
Nelson AJ, Clegg CH, Farr AG. In vitro positive selection and anergy induction of class II-restricted TCR transgenic thymocytes by a cortical thymic epithelial cell line. Int Immunol. 1998;10:1335–46.
O’Neil R, Wei Q, Condie B. High efficiency transfection of thymic epithelial cell lines and primary thymic epithelial cells by Nucleofection. Nat Preced. 2011. https://doi.org/10.1038/npre.2011.6283.1.













Leave a Reply