Selective scFv screening against mature MSLN (matMSLN) via the yeast display library system
To generate high-affinity antibodies against matMSLN (residues 296–606), 6-week-old BALB/c mice were immunized intraperitoneally with recombinant matMSLN protein (Figs. 1 and 2a, and Supplementary Fig. 1). Upon reaching a serum antibody titer of 1:100,000 (Supplementary Fig. 2), variable heavy (VH) and light (VL) chain genes were amplified from the spleen and bone marrow of sacrificed immunized mice, fused to their respective constant domains, and transformed into Saccharomyces cerevisiae strains JAR200 (MATa) and YVH10 (MATα) to construct heavy-chain (HC) and light-chain (LC) Fab libraries. Mating the HC and LC yeast libraries42,43 yielded a combinatorial Fab repertoire (>4 × 107) (Supplementary Fig. 3a, and Supplementary Table 1). Flow cytometric selection of clones with high antigen-binding activity and subsequent cell-based panning of MSLN+ and MSLN– AsPC-1 cells narrowed the candidates to 96 unique Fab clones (Supplementary Figs. 3 and 4). Most antibody candidates exhibited strong matMSLN binding but negligible affinity for solMSLN (residues 296–592), indicating a favorable selectivity that may reduce interference from shed MSLN (Fig. 2a, b).25,44,45 To identify the most promising clones, we applied sequential selection criteria: retaining the top 90% of expressers to maximize recovery, confirming robust Fab assembly on the basis of diagonal distribution, and isolating the top 5% of matMSLN binders exhibiting preferential specificity for matMSLN over solMSLN. This systematic approach enabled the identification of antibody candidates with optimal expression and selectivity (Fig. 2b, and Supplementary Fig. 4).
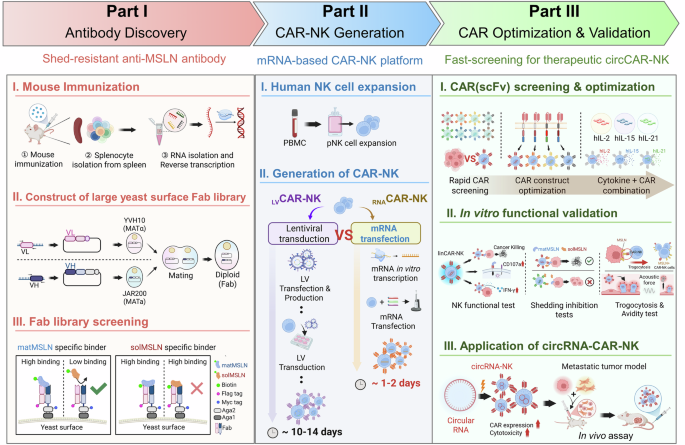
Streamlined chimeric antigen receptor (CAR) screening and circRNA-based CAR (circCAR)-natural killer (NK) cell development to overcome antigen shedding. Schematic workflow illustrating (1) antibody discovery via a yeast surface-display platform to isolate high-affinity clones targeting membrane-bound mesothelin (MSLN); (2) rapid CAR optimization via mRNA-engineering of NK cells for functional screening; and (3) development of persistent shed-resistant CAR-NK cells via circRNAs
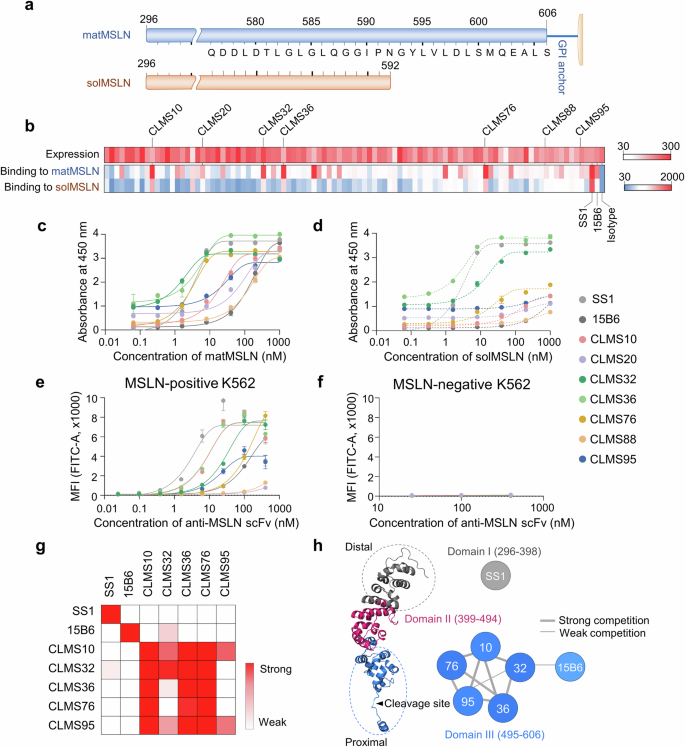
Screening and characterization of mature mesothelin (matMSLN)-specific scFvs. a Schematic representation of matMSLN (residues 296–606) and its soluble counterpart (solMSLN, residues 296–592). The top bar depicts the full-length matMSLN ending in a GPI anchor at residue 606; the bottom bar represents solMSLN, which lacks a C-terminal GPI-anchoring region. The corresponding amino acid sequence is aligned with the annotated residue positions. b Heatmap showing the levels and antigen-binding affinities of individual clones. Yeast-displayed clones were analyzed by flow cytometry using an anti-FLAG antibody (expression) and 10 nM biotinylated matMSLN or solMSLN (binding). SS1 and 15B6 were included as positive controls, and an isotype antibody served as a negative control. Clones are ranked by mean fluorescence intensity (MFI). ELISA-based binding of recombinant anti-MSLN scFvs to matMSLN (c) and solMSLN (d). Binding in immobilized scFvs incubated with serial dilutions of biotinylated antigen and detected via streptavidin-HRP. Note that the yeast-display binding values in (b) represent semiquantitative MFI-based screening at a fixed antigen concentration, whereas the ELISA results in (c, d) reflect the quantitative binding of the purified scFvs. Apparent differences between these assays arise from their distinct experimental formats, with ELISA providing a more accurate measure of solMSLN binding. Flow cytometry of cell surface binding by scFvs to MSLN+ (e) and MSLN– K562 cells (f). Serially diluted antibodies were incubated with the cells, and the bound scFvs were detected via an anti-His-FITC secondary antibody. g Epitope binning of anti-MSLN scFvs (CLMS10, CLMS32, CLMS36, CLMS76, CLMS95, SS1, and 15B6) by competitive ELISA. The immobilized scFvs were challenged with biotinylated matMSLN preincubated with competing antibodies. The heatmap intensity reflects the degree of competitive binding, indicating epitope overlap. h Predicted epitope mapping of anti-MSLN scFvs on the basis of binning data. According to UniProt annotations, the extracellular domain of MSLN is divided into Region I (residues 296–398), Region II (399–494), and Region III (495–606). Three independent experiments were performed; the data are presented as the means ± standard deviations (s.d.)
On the basis of these selection criteria, seven antibody clones—CLMS10, CLMS20, CLMS32, CLMS36, CLMS76, CLMS88, and CLMS95—were selected for further analysis (Fig. 2b). After the production of recombinant scFvs,46,47 their binding activities to both matMSLN and solMSLN were evaluated via ELISA (Fig. 2c, d). Except for CLMS32 and CLMS36, all clones selectively bound to matMSLN (EC50 = 1.7 ± 0.1–151.1 ± 33.3 nM; Supplementary Table 2) over solMSLN. Notably, CLMS10 demonstrated high affinity for matMSLN (EC50 = 18.6 ± 1.1 nM), but minimal binding to solMSLN (EC50 > 1 μM). Surface plasmon resonance (SPR) confirmed that the affinity of CLMS10 for matMSLN was 8.22 ± 0.40 nM (Supplementary Fig. 5). All scFvs, except CLMS20 and CLMS88, specifically bound to MSLN-positive K562 cells, with EC50 values ranging from a few nM to several hundred nM, while negligible binding to MSLN-negative K562 cells was detected (Fig. 2e, f, and Supplementary Table 3). These results indicate that our anti-MSLN antibodies selectively recognize membrane-bound MSLN on the cell surface.
To further define the epitopes of the antibody candidates, we performed competitive ELISA using reference antibodies with well-characterized binding sites: SS1, which targets domain I (residues E296–L398),28 and 15B6, which binds the C-terminal region (residues Y594–L605).25 CLMS10, CLMS32, CLMS36, CLMS76, and CLMS95 competed for antigen binding, suggesting that they either share overlapping epitopes or exhibit steric hindrance that prevents simultaneous binding (Fig. 2g). Notably, CLMS10 and CLMS76 exhibited weak binding to solMSLN, implying that their epitopes are localized near the C-terminus of domain III, which is known to be susceptible to proteolytic shedding (Fig. 2d, h). Collectively, these findings suggest that the epitopes of CLMS10, CLMS32, CLMS36, CLMS76, and CLMS95 are clustered near the C-terminal region of domain III, supporting their therapeutic potential as shed-resistant antibodies (Fig. 2h).
Rapid and efficient live-cell screening of functional scFvs via a linRNA-based CAR-NK (linCAR-NK) cell platform
Standard screening methods often rely on immortalized cell lines (e.g., NK-92), which fail to recapitulate the complex biology of donor-derived cells. To ensure translational relevance, we implemented a “direct-to-primary” screening strategy. This streamlined approach enables rapid live functional screening directly in clinically relevant human primary NK cells, yielding high-fidelity phenotypic data and mitigating the risk of candidate failure during translational scale-up (Figs. 1 and 3a). Each scFv-containing CAR construct was synthesized as a linRNA via in vitro transcription (IVT). The quality of the synthesized linRNAs was validated via agarose gel electrophoresis and purity assessment (Supplementary Fig. 6a). Human primary NK (pNK) cells were electroporated with each linRNA, and CAR expression and cell viability were assessed in a time- and dose-dependent manner. For example, CAR-SS1-NK cells presented over 87% CAR expression at 1 µg linRNA per 106 cells, with detectable expression persisting for up to 5 days, albeit at reduced levels (Supplementary Fig. 6b, c). Following linRNA electroporation, flow cytometry confirmed CAR and GFP expression across all clones, with GFP coexpressed via a P2A linker. CAR expression levels varied among constructs and were quantified by staining with PE- or APC-conjugated streptavidin in combination with biotinylated MSLN extracellular domain proteins (Supplementary Figs. 6d and 7). The cell viability was largely consistent across the clones (Supplementary Fig. 6e).
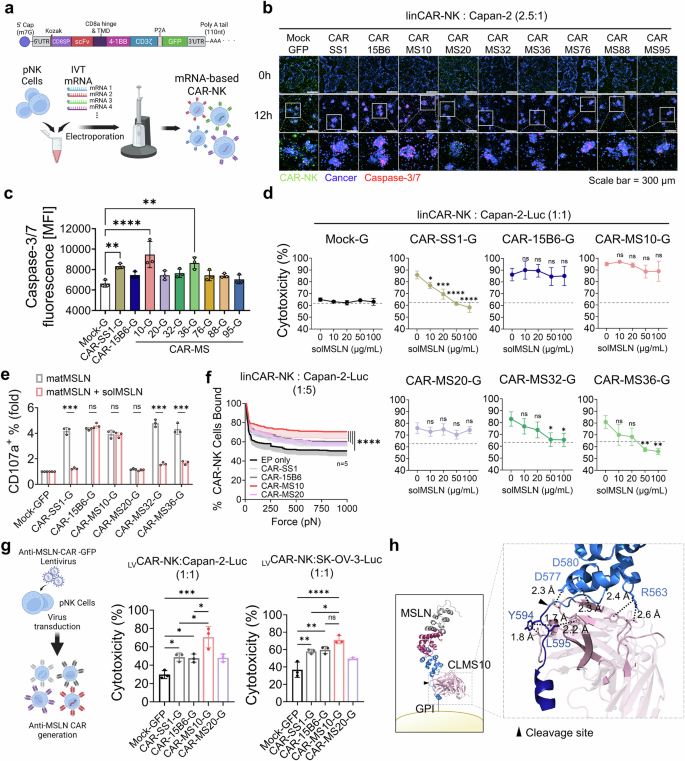
mRNA-based screening identifies CLMS10 as a potent shedding-resistant CAR construct in NK cells. a Generation of linear mRNA-based CAR (linCAR)-NK cells via electroporation (EP) of human primary NK cells. b, c Establishment of a rapid and cost-effective screening platform for evaluating CAR-NK cytotoxicity against Capan-2 cells. b Visualization of CAR-NK-mediated cancer cell killing. Capan-2 cells were stained with CellTrace Violet Cell (blue) and live-cell Caspase-3/7 dye (red), and cocultured with CAR-NK cells (green). c Quantification of apoptotic cancer cells based on Caspase-3/7 activation (n = 3). d Shedding inhibition assay for determining CAR-NK cytotoxicity in the presence of increasing concentrations of solMSLN (residues 296–592); dashed lines indicate the baseline cytotoxicity of allogeneic NK cells. e CD107a activation in CAR-NK cells stimulated with matMSLN-coated plates (20 µg/mL) in the presence or absence of solMSLN (50 µg/mL) was analyzed by flow cytometry (n = 3). f Cell–cell avidity between linCAR-NK and Capan-2 cells was quantified via acoustic force-based microfluidic microscopy (n = 5). g Luciferase-based cytotoxicity of lentivirus (LV)-generated CAR(LVCAR)-NK cells against Capan-2-Luc and SK-OV-3-Luc cells (n = 3). h Predicted docking model of CLMS10 scFv in complex with matMSLN using Schrödinger software. Residues with side chains within 2.5 Å of the binding interface are highlighted, revealing key antigen-antibody interactions. The soluble MSLN regions are indicated in marine blue, and the membrane-bound matMSLN regions are indicated in dark blue. The data are presented as the means ± s.d. Statistical analyses: one-way ANOVA in (c) and (g) and two-way ANOVA in (f); two-tailed Student’s t tests in (d) and (e); *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001
We established a rapid live-cell screening platform to assess the cytotoxic function of linCAR-NK cells against an MSLN-overexpressing pancreatic cancer cell line (Capan-2). Real-time cancer cell death by linCAR-NK cells was monitored by tracking Caspase-3/7 activation during live coculture, with CAR-SS1-NK and CAR-15B6-NK cells used as controls (Fig. 3b, c, and Supplementary Fig. 8a). Notably, CAR-MS10-NK cells caused a significant increase in cancer cell death, as evidenced by a 1.5-fold increase in Caspase-3/7-positive Capan-2 cells compared with mock-NK cells. A luciferase-based cytotoxicity assay revealed that CAR-MS10-NK cells achieved the strongest killing efficacy against both Capan-2-Luc and SK-OV-3-Luc cells, which express different MSLN levels (Supplementary Fig. 8b, c). We assessed the sensitivity of each CAR-NK construct to solMSLN-mediated inhibition. In coculture with solMSLN, CAR-SS1-NK, CAR-MS32-NK, and CAR-MS36-NK cells exhibited a dose-dependent reduction in killing activity. In contrast, CAR-MS10-NK, CAR-15B6-NK, and CAR-MS20-NK cells maintained their cytotoxicity, indicating resistance to solMSLN-mediated inhibition (Fig. 3d). The expression of CD107a, a marker of degranulation, was unaffected by solMSLN treatment in CAR-MS10-NK, CAR-MS20-NK, and CAR-15B6-NK cells activated with matMSLN, further supporting their shedding resistance profiles (Fig. 3e). We examined the extent of trogocytosis associated with impaired CAR function and NK-mediated fratricide in linCAR-NK cells.48,49 Compared with CAR-SS1-NK cells, CAR-MS10-NK cells exhibited significantly reduced trogocytosis (Supplementary Fig. 8d). Furthermore, CAR-MS10-NK cells presented the greatest degree of cell avidity among all the tested CAR constructs, suggesting the formation of robust immunological synapses (Fig. 3f).50 To validate these findings in a more clinically relevant system, we introduced the scFvs into lentivirus-based CAR constructs, which were transduced into pNK cells. The results of functional assays revealed similar cytotoxicity patterns as those observed with linCAR-NK cells, with CAR-MS10-NK cells showing an approximately 22% higher killing rate against Capan-2 cells than CAR-SS1- and CAR-15B6-transduced cells (Fig. 3g and Supplementary Fig. 9a). We evaluated whether this functional trend persisted across immune cell types such as linCAR-T cells. In cytotoxicity assays against Capan-2-Luc cells, linCAR-15B6-T cells showed comparable efficacy to linCAR-MS10-T and linCAR-SS1-T cells (Supplementary Fig. 9b–f). CAR-MS20-T cells showed limited activity, which is consistent with the NK cell results, suggesting that the functional impact of scFvs may vary depending on the immune effector cell type.13,14,32,51
Finally, in silico docking analysis revealed a critical structure–function relationship. Unlike SS1, which binds to distal Region I, CLMS10 interacts with a conformational epitope that overlaps with or lies immediately adjacent to the proteolytic cleavage site (Fig. 3h). This unique binding geometry likely imposes steric hindrance that prevents access by sheddases or stabilizes the membrane-bound conformation of MSLN.
Consequently, CLMS10 selectively engages membrane-anchored MSLN while exhibiting drastically reduced affinity for the shed soluble form, providing a structural basis for its superior performance in shedding inhibition assays. Additionally, our linRNA-based screening platform offers an efficient functional approach for discovering and optimizing clinically relevant MSLN-targeting CARs.
linRNA-driven screening for optimized CAR architecture and cytokine coexpression
We selected CAR-MS10 for further analysis. We employed a linRNA-based system to screen various combinations of transmembrane and costimulatory domains. Specifically, CD8 and NKG2D were chosen as the transmembrane domains (TMDCD8 and TMDNKG2D), and four CAR variants were designed: TMDCD8–4-1BB–CD3ζ (4-1BBζ), TMDCD8–OX40–CD3ζ (OX40ζ), TMDNKG2D–DAP10–DAP12 (DAP10/12), and TMDNKG2D–2B4–CD3ζ (2B4ζ) (Fig. 4a).
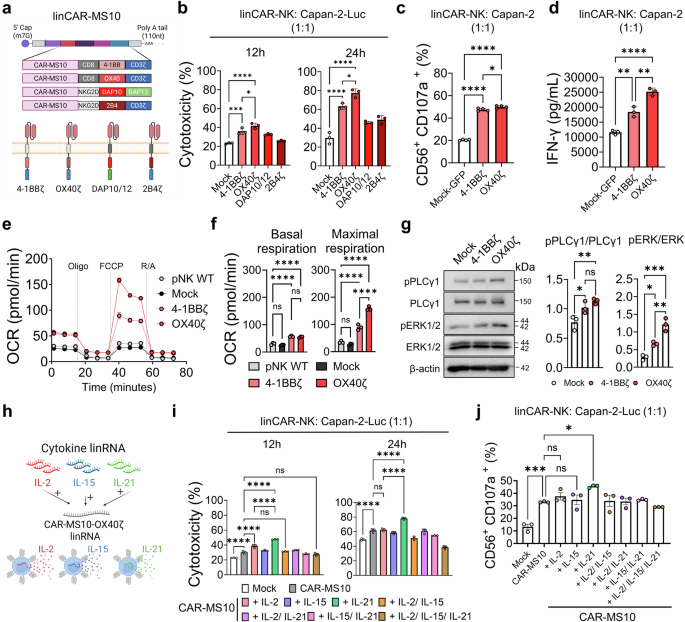
CAR-MS10 structural optimization and cytokine coexpression enhance CAR-NK cytotoxicity and functionality. a Four CAR constructs were designed to evaluate combinations of transmembrane (CD8, NKG2D), costimulatory (4-1BB, OX40, DAP10, and 2B4), and signaling (CD3ζ, DAP12) domains. b Cytotoxicity of linCAR-NK cells at 12 h and 24 h post coculture with Capan-2 cells determined via a luciferase-based assay (n = 3). c CD107a degranulation in CD56+ linCAR-NK cells after coculture with Capan-2 cells was analyzed by flow cytometry (n = 4). d IFN-γ secretion after 24 h of coculture (n = 3). e, f Metabolic activity [oxygen consumption rate (OCR)] after 2 h of stimulation on matMSLN-coated plates (20 µg/mL) (n = 3). g Intracellular signaling pathway activation by the lead construct (linCAR-MS10-OX40ζ), quantified by normalizing the levels of phosphorylated PLCγ and ERK to their respective total forms (n = 3). h–j Functional assessment of primary NK cells cotransfected with linCAR-MS10-OX40ζ and cytokine-encoding linRNAs (IL-2, IL-15, or IL-21). Luciferase-based cytotoxicity (i) and CD107a degranulation (j) were measured in NK cells cotransfected with single or combined cytokines (n = 3). The data are shown as the means ± s.d. One-way ANOVA was used in (b−d, f, g, i, j); *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001
The cytotoxic potential of the CAR variants against Capan-2-Luc cells was evaluated via a luciferase-based killing assay. Among the variants, the CAR-MS10-OX40ζ construct exhibited the strongest cytotoxicity, followed by CAR-MS10-4-1BBζ (Fig. 4b and Supplementary Fig. 10a). A detailed analysis of the functional activities across the 4-1BBζ and OX40ζ constructs revealed that, consistent with the cytotoxicity data, CAR-MS10-OX40ζ resulted in a significant increase in CD107a activation compared with the prototype CAR-MS10-4-1BBζ, accompanied by increased secretion of IFN-γ (Fig. 4c, d). In addition to direct cytotoxicity, metabolic fitness is a key determinant of CAR-NK cell survival in the nutrient-deprived TME.37 The incorporation of the OX40 costimulatory domain significantly reprogrammed NK cell metabolism, enhancing both basal and maximal mitochondrial respiration upon antigen stimulation (Fig. 4e, f). This elevated spare respiratory capacity suggests that CAR-MS10-OX40ζ-NK cells are better equipped to sustain energy production under metabolic stress, contributing to prolonged antitumor activity.
To further investigate the underlying signaling pathways involved in OX40ζ-mediated CAR activation, CAR-MS10-OX40ζ-NK cells were stimulated with an MSLN recombinant protein and analyzed via western blotting (Fig. 4g). Compared with those in the 4-1BBζ group, the phosphorylation of phospholipase C gamma and extracellular signal-regulated kinase, key downstream molecules of CD3ζ signaling, was greater in the OX40ζ group.52,53 Collectively, these results identify OX40ζ as the optimal intracellular domain configuration for CAR-MS10, enhancing both effector function and signaling capacity.
We cotransfected cytokine-encoding linRNAs into CAR-MS10-OX40ζ cells to increase their cytotoxicity and functionality. Cytokines play pivotal roles in modulating NK cell activity, proliferation, and persistence.54,55,56 On the basis of their well-characterized functions, we selected interleukin-2 (IL-2), IL-15, and IL-21 to reinforce NK cell effector function and longevity. The synergistic effects of the selected cytokines were evaluated in CAR-MS10-NK cells, generated by coelectroporating CAR-MS10-OX40ζ linRNAs with linRNAs encoding each cytokine (Fig. 4h and Supplementary Fig. 10b, c). Among the various cytokine combinations, IL-21–CAR-MS10-OX40ζ-coexpressing NK cells presented the strongest antitumor activity against Capan-2-Luc cells, along with significantly increased CD107a expression (Fig. 4i, j). Taken together, the results of the linRNA-based approach demonstrated that the IL-21–CAR-MS10-OX40ζ configuration endows NK cells with superior cytotoxicity and functional activity.
CircRNA-based CAR-NK cells exhibited superior cytotoxicity with prolonged CAR expression
To address the transient nature of linRNA expression in mRNA-based cell therapies, we explored the use of circRNAs for prolonging CAR expression. Compared with linRNAs, circRNAs have previously been used to prolong transgene expression and offer improved structural stability and translational efficiency.57,58,59 We monitored CAR expression and cell viability over time in CAR-NK cells generated from either linRNAs or circRNAs (Fig. 5a). Notably, circCAR-NK cells presented significantly prolonged and sustained CAR expression, as demonstrated by longitudinal flow-cytometry analyses of both the percentage of CAR⁺ cells and the CAR mean fluorescence intensity (MFI) across multiple timepoints (Days 1, 3, and 5) (Fig. 5b, c). In contrast, there was no notable difference in viability between linRNA- and circRNA-transfected cells over the same time period (Fig. 5d). We evaluated the persistence of cytotoxicity using serial killing assays, in which CAR-NK cells were rechallenged with fresh cancer cells for five consecutive rounds (Fig. 5e). Consistent with prolonged CAR expression, circCAR-NK cells maintained superior activity, although a gradual decline was observed over time (Fig. 5f). circCAR-NK cells exhibited robust IFN-γ secretion, supporting a direct association between prolonged CAR expression and sustained effector function (Fig. 5g). circCAR-MS10-NK cells retained strong cytotoxic activity even under solMSLN-shedding conditions, indicating resistance to solMSLN-decoy effects (Fig. 5h). In contrast, circCAR-SS1-NK cells showed limited cytotoxicity and a marked reduction in IFN-γ secretion in the presence of solMSLN, highlighting the importance of target epitope selection and construct design (Fig. 5i). Importantly, circRNA-driven CAR-NK cells consistently demonstrated stronger cytotoxicity and NK-cell activation than linCAR- or LVCAR-NK cells did, further supporting the superior functional durability of the circRNA platform (Supplementary Fig. 11). Together, these findings underscore the superior performance of circCAR-NK cells over their linRNA counterparts, demonstrating enhanced CAR expression, prolonged cytotoxicity, and greater functional resilience under repeated tumor challenge and antigen-shedding stress.
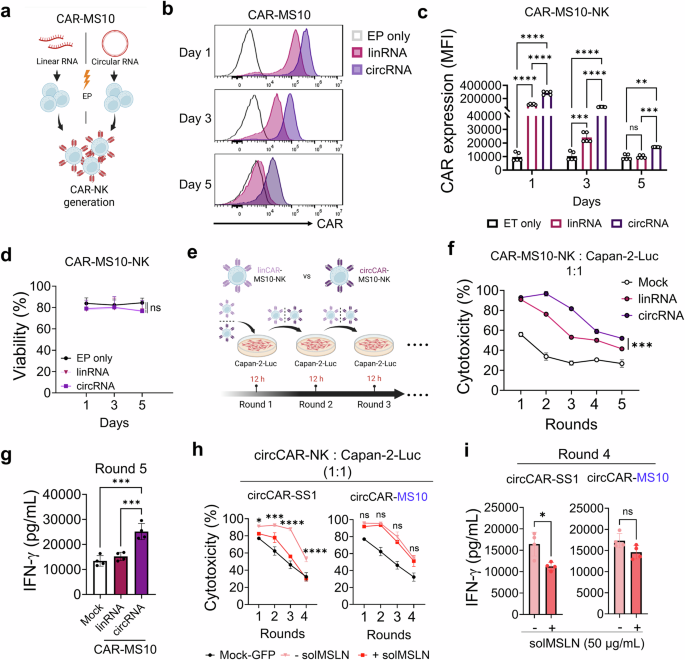
circCAR-MS10-CAR-NK cells exhibit prolonged CAR expression, sustained cytotoxicity, and resistance to antigen shedding. a Generation of circCAR-NK cells via EP. b–d CAR expression and viability of linCAR- and circCAR-NK cells over time. c CAR levels were measured and compared to those of the EP-negative control (n = 5). d NK viability was assessed by live/dead staining following EP (n = 3). e Serial killing assay of CAR-NK cells sequentially exposed to fresh Capan-2-Luc cancer cells at 12 h-intervals. Serial cytotoxicity (f) and IFN-γ secretion (g) were evaluated to determine the sustained antitumor activity of circCAR-NK cells (n = 3). Impact of solMSLN (50 µg/mL) on CAR-NK cell function during repeated tumor challenges. Serial cancer killing (h) and IFN-γ secretion (i) were assessed following solMSLN treatment (50 µg/mL) at each round (n = 3). The data are presented as the means ± s.d. Statistical analyses: one-way ANOVA for (c, d, f, g), and two-tailed Student’s t tests for (h, i); *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001
CircCAR-MS10-NK cells show potent antitumor activity in solMSLN-shedding models in vitro and in vivo
The pancreatic TME is highly immunosuppressive and is characterized by a dense extracellular matrix and abundant TGF-β signaling, which primarily originates from CAFs. This milieu impairs NK cell function and greatly challenges the efficacy of immunotherapies.38,39,40 Recent studies have demonstrated that anti-mesothelin antibodies not only target tumor cells but also disrupt antigen-presenting CAFs, reshaping the TME and enhancing immune responses.60 However, the therapeutic efficacy of MSLN-targeted CAR-T-therapies is undermined by the decoy effect of solMSLN-mediated antigen shedding, impeding CAR-Tcell activity.25 To address the suppression of CAR-Tcell function induced by solMSLN under physiologically relevant conditions, we established an in vitro cancer–CAF coculture model to mimic the TME. Interestingly, we found that the presence of human primary pulmonary fibroblasts or CAFs derived from patients with pancreatic cancer significantly accelerated the secretion of solMSLN compared with that of cancer cells alone (Fig. 6a, b, and Supplementary Fig. 12a, b).
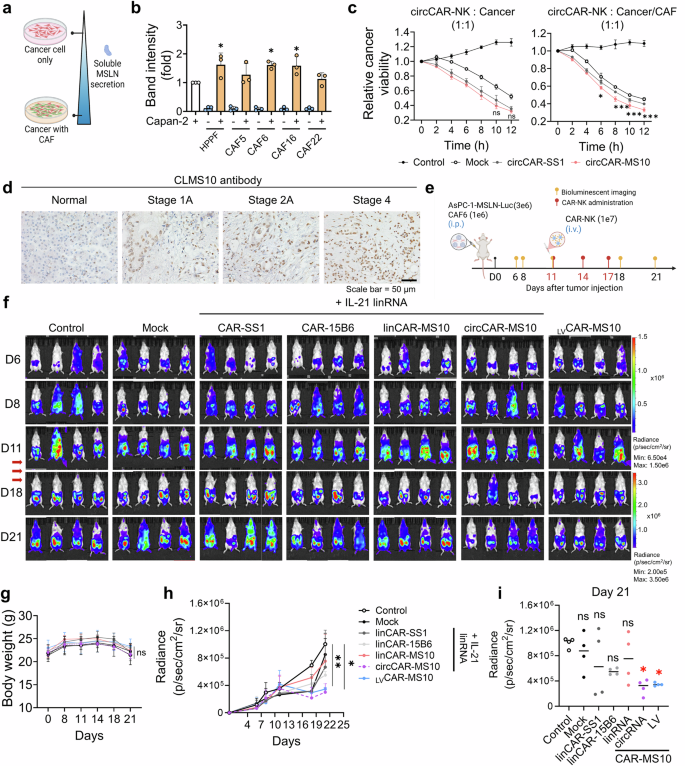
circCAR-MS10-NK cells maintain superior antitumor activity under CAF-induced shedding conditions in vitro and in vivo. a, b Soluble MSLN (solMSLN) levels were elevated in Capan-2 cells cocultured with human pancreatic primary fibroblasts (HPPFs) or CAFs (CAF5, CAF6, CAF16, and CAF22), as shown by western blotting and quantified relative to those in cancer-only controls (n = 3). c In vitro real-time cytotoxicity of circCAR-NK cells cocultured with mCherry-expressing Capan-2 cancer cells, in the absence or presence of CAFs. CAFs were coincubated to mimic the tumor microenvironment characterized by shed MSLN secretion. Cancer cell survival was monitored by quantifying mCherry/RFP fluorescence, presented as relative cancer viability, which was defined as the mCherry signal normalized to the 0 h value (set to 1.0) (n = 3). Statistical comparisons were performed between the circCAR-SS1 and circCAR-MS10 groups at each indicated time point. d IHC of MSLN expression in human pancreatic tumor tissues using a CLMS10-derived antibody across stages 1A, 2A, and 4. e, f In vivo therapeutic effects of CAR-NK cells were evaluated in a metastatic mouse model with intraperitoneal AsPC-1-MSLN-Luc tumors containing CAF6 cells. f Representative IVIS images of mice treated with mock, linCAR-NK (SS1, 15B6, MS10), circCAR-MS10, or LVCAR-MS10 cells (n = 4 per group); all mRNA-CAR groups were coelectroporated with IL-21 mRNA. g Body weight monitoring during treatment. Tumor burden quantified via IVIS imaging over time (h) and on day 21 (i) (n = 4). The data are presented as the means ± s.d. Statistical analyses: one-way ANOVA for (b, i) and two-way ANOVA for (c, g, h); *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001
Based on the observed increase in solMSLN secretion in the presence of CAFs, we further assessed the therapeutic efficacy of CAR-NK cells under CAF-mediated shedding inhibition via real-time live-cell imaging (Fig. 6c, and Supplementary Fig. 12c). The CAF-induced increase in solMSLN levels significantly reduced cancer cell death in the circCAR-SS1-NK cell-treated group. In contrast, the cytotoxic efficacy of circCAR-MS10-NK cells was maintained despite MSLN shedding. Notably, strong MSLN expression was associated with advanced cancer progression, as evidenced by CLMS10-IgG staining in pancreatic tumor tissues, suggesting that MSLN is a therapeutically actionable target (Fig. 6d). We established an in vivo intraperitoneal metastatic tumor model that mimics the TME,5,25 including CAF-mediated MSLN shedding, by coinjecting AsPC-1-MSLN-Luc tumor cells and CAFs into NOD-Prkdcem1BaekIl2rgem1Baek (NSG) mice and treating them with various CAR-NK platforms: linCAR-NK cells expressing CAR-SS1, CAR-15B6, or CAR-MS10; circCAR-MS10-NK cells; and LVCAR-MS10-NK cells (Fig. 6e). To enhance NK cell function and persistence, IL-21 linRNA was coelectroporated into all the mRNA-based CAR-NK groups, but not into the lentivirus-based groups. Compared with the mock group, both the circRNA-based and lentivirus-based CAR-MS10-NK groups demonstrated significant tumor regression (Fig. 6f–i). circCAR-MS10-NK cells showed anticancer activity comparable to that of LVCAR-MS10-NK cells. In contrast, compared with mock cells, linCAR-NK cells induced a modest reduction in tumor size, although the difference was not significant. These findings demonstrate that optimized circCAR-MS10-NK cells retain antitumor efficacy under TME-mimicking conditions and elevated solMSLN levels, supporting the potential applicability of the nonviral CAR-NK platform for treating solid tumors.
Given that IL-21 was co-delivered to increase NK activity, we also assessed potential systemic IL-21-associated toxicity. Following three repeated infusions of CAR-NK cells, we observed no significant changes in body weight, gross liver and kidney morphology, or hepatic and renal function markers, indicating the absence of acute organ toxicity (Supplementary Fig. 13). These findings suggest that cytokine-augmented circCAR-NK cells do not elicit measurable systemic toxicity, further supporting their translational potential as safe and effective nonviral CAR-NK therapies for solid tumors. To further characterize the behavior of the different CAR-NK modalities in vivo, we next examined the distribution and persistence of the infused NK cells (Supplementary Fig. 14). Quantification of CAR⁺ NK cells in the peripheral blood at 72 h confirmed successful engraftment in all the CAR-bearing groups, whereas the mock-NK group presented only background-level events. By day 7, distinct persistence patterns had emerged among the platforms. As expected from the transient nature of linear mRNAs, linCAR-NK cells were largely undetectable in the circulation and secondary lymphoid tissues, highlighting the short-lived in vivo expression driven by linRNAs. In contrast, IL-21–armored circCAR-NK cells demonstrated the most robust persistence among all groups, with clear detection in the spleen 7 days after intravenous injection, which aligns with the known survival- and proliferation-supporting functions of IL-21. Conversely, even LVCAR-NK cells showed limited persistence on day 7. To complement these in vivo persistence data and to directly assess the stability of CAR surface expression under repeated antigen exposure, we next evaluated CAR stability in vitro by repeatedly challenging CAR-NK cells with Capan-2-Luc tumor cells for up to four consecutive rounds (Supplementary Fig. 15). Across all platforms, serial stimulation ultimately resulted in progressive CAR downregulation. Notably, the surface CAR MFI of linCAR-, LVCAR-, and nonarmored circCAR-NK cells rapidly decreased. However, IL-21–incorporated circCAR-NK cells presented a modest transient increase in CAR expression during the first stimulation round, followed by a gradual reduction over subsequent rounds. Overall, compared with nonarmored circCAR controls, IL-21 armoring substantially improved the maintenance of CAR surface expression across stimulation rounds within the CAR⁺ NK-cell population (Supplementary Fig. 15). These findings indicate that transient IL-21 delivery helps to preserve CAR surface expression during repeated antigen encounters—an essential feature for sustaining NK-cell activity in solid-tumor contexts.
















Leave a Reply