Flaxman, S. R. et al. Global causes of blindness and distance vision impairment 1990-2020: a systematic review and meta-analysis. Lancet Glob. Health 5, e1221–e1234 (2017).
Whitcher, J. P. & Srinivasan, M. Corneal ulceration in the developing world—a silent epidemic. Br. J. Ophthalmol. 81, 622–623 (1997).
Bourne, R. R. A. et al. The UK clinical eye research strategy: refreshing research priorities for clinical eye research in the UK. Eye 38, 1947–1957 (2024).
Wilgus, T. A., Roy, S. & McDaniel, J. C. Neutrophils and wound repair: positive actions and negative reactions. Adv. Wound Care 2, 379–388 (2013).
Cheng, K. K. W. et al. In vitro and ex vivo models of microbial keratitis: present and future. Prog. Retin. Eye Res. 102, 101287 (2024).
Howe, K. et al. The zebrafish reference genome sequence and its relationship to the human genome. Nature 496, 498–503 (2013).
Kaveh, A. et al. Selective Cdk9 inhibition resolves neutrophilic inflammation and enhances cardiac regeneration in larval zebrafish. Development 149, https://doi.org/10.1242/dev.199636 (2022).
Hoodless, L. J. et al. Genetic and pharmacological inhibition of CDK9 drives neutrophil apoptosis to resolve inflammation in zebrafish in vivo. Sci. Rep. 5, 36980 (2016).
Isles, H. M. et al. Pioneer neutrophils release chromatin within in vivo swarms. Elife 10, https://doi.org/10.7554/eLife.68755 (2021).
Renshaw, S. A., Loynes, C. A., Elworthy, S., Ingham, P. W. & Whyte, M. K. Modeling inflammation in the zebrafish: how a fish can help us understand lung disease. Exp. Lung Res. 33, 549–554 (2007).
Rebelo de Almeida, C. et al. Zebrafish xenografts as a fast screening platform for bevacizumab cancer therapy. Commun. Biol. 3, 299 (2020).
Crouzier, L. et al. Use of zebrafish models to boost research in rare genetic diseases. Int. J. Mol. Sci. 22, https://doi.org/10.3390/ijms222413356 (2021).
Bournele, D. & Beis, D. Zebrafish models of cardiovascular disease. Heart Fail. Rev. 21, 803–813 (2016).
Hubrecht, R. C. & Carter, E. The 3Rs and humane experimental technique: implementing change. Animals 9, https://doi.org/10.3390/ani9100754 (2019).
Xie, Y., Meijer, A. H. & Schaaf, M. J. M. Modeling Inflammation in zebrafish for the development of anti-inflammatory drugs. Front. Cell Dev. Biol. 8, 620984 (2020).
Kaveh, A. et al. Live imaging of heart injury in larval zebrafish reveals a multi-stage model of neutrophil and macrophage migration. Front. Cell Dev. Biol. 8, 579943 (2020).
Bruton, F. A. et al. Macrophages trigger cardiomyocyte proliferation by increasing epicardial Vegfaa expression during larval zebrafish heart regeneration. Dev. Cell 57, 1512–1528 (2022).
Huang, C. & Niethammer, P. Tissue damage signaling is a prerequisite for protective neutrophil recruitment to microbial infection in zebrafish. Immunity 48, 1006–1013 (2018).
Ma, Y., Hui, K. L., Gelashvili, Z. & Niethammer, P. Oxoeicosanoid signaling mediates early antimicrobial defense in zebrafish. Cell Rep. 42, 111974 (2023).
Meijer, A. H. Protection and pathology in TB: learning from the zebrafish model. Semin. Immunopathol. 38, 261–273 (2016).
Renshaw, S. A. et al. A transgenic zebrafish model of neutrophilic inflammation. Blood 108, 3976–3978 (2006).
Patton, E. E., Zon, L. I. & Langenau, D. M. Zebrafish disease models in drug discovery: from preclinical modelling to clinical trials. Nat. Rev. Drug Discov. 20, 611–628 (2021).
Starnes, T. W. & Huttenlocher, A. Neutrophil reverse migration becomes transparent with zebrafish. Adv. Hematol. 2012, 398640 (2012).
Lam, S. H., Chua, H. L., Gong, Z., Lam, T. J. & Sin, Y. M. Development and maturation of the immune system in zebrafish, Danio rerio: a gene expression profiling, in situ hybridization and immunological study. Dev. Comp. Immunol. 28, 9–28 (2004).
Ikkala, K., Raatikainen, S., Koivula, H. & Michon, F. Zebrafish cornea formation and homeostasis reveal a slow maturation process, similarly to terrestrial vertebrates’ corneas. Front. Physiol. 13, 906155 (2022).
Bill, B. R., Petzold, A. M., Clark, K. J., Schimmenti, L. A. & Ekker, S. C. A primer for morpholino use in zebrafish. Zebrafish 6, 69–77 (2009).
Ting, D. S. J., Ho, C. S., Deshmukh, R., Said, D. G. & Dua, H. S. Infectious keratitis: an update on epidemiology, causative microorganisms, risk factors, and antimicrobial resistance. Eye 35, 1084–1101 (2021).
Rinaldi, G. et al. Fish erythrocyte extracellular traps (FEETs) are an evolutionarily conserved cellular process triggered by different stimuli. Fish Shellfish Immunol. 136, 108638 (2023).
Yousefi, S., Mihalache, C., Kozlowski, E., Schmid, I. & Simon, H. U. Viable neutrophils release mitochondrial DNA to form neutrophil extracellular traps. Cell Death Differ. 16, 1438–1444 (2009).
Yipp, B. G. & Kubes, P. NETosis: how vital is it? Blood 122, 2784–2794 (2013).
Casari, A. et al. A Smad3 transgenic reporter reveals TGF-beta control of zebrafish spinal cord development. Dev. Biol. 396, 81–93 (2014).
Carrington, L. M., Albon, J., Anderson, I., Kamma, C. & Boulton, M. Differential regulation of key stages in early corneal wound healing by TGF-β isoforms and their inhibitors. Investig. Ophthalmol. Vis. Sci. 47, 1886–1894 (2006).
O’Kane, S. & Ferguson, M. W. J. Transforming growth factor βs and wound healing. Int. J. Biochem. Cell Biol. 29, 63–78 (1997).
Jester, J. V., Barry-Lane, P. A., Petroll, W. M., Olsen, D. R. & Cavanagh, H. D. Inhibition of corneal fibrosis by topical application of blocking antibodies to TGF beta in the rabbit. Cornea 16, 177–187 (1997).
Møller-Pedersen, T., Cavanagh, H. D., Petroll, W. M. & Jester, J. V. Neutralizing antibody to TGFbeta modulates stromal fibrosis but not regression of photoablative effect following PRK. Curr. Eye Res. 17, 736–747 (1998).
Leitch, A. E., Lucas, C. D. & Rossi, A. G. Editorial: neutrophil apoptosis: hot on the TRAIL of inflammatory resolution. J. Leukoc. Biol. 90, 841–843 (2011).
Gilroy, D. W., Lawrence, T., Perretti, M. & Rossi, A. G. Inflammatory resolution: new opportunities for drug discovery. Nat. Rev. Drug Discov. 3, 401–416 (2004).
Li, Z., Burns, A. R. & Smith, C. W. Two waves of neutrophil emigration in response to corneal epithelial abrasion: distinct adhesion molecule requirements. Investig. Ophthalmol. Vis. Sci. 47, 1947–1955 (2006).
Chang, J. H., Gabison, E. E., Kato, T. & Azar, D. T. Corneal neovascularization. Curr. Opin. Ophthalmol. 12, 242–249 (2001).
Cursiefen, C. Immune Response and the Eye Vol. 92 (eds Niederkorn, J. Y. & Kaplan, H. J.) 0 (S. Karger AG, 2007).
Hall, C., Flores, M. V., Storm, T., Crosier, K. & Crosier, P. The zebrafish lysozyme C promoter drives myeloid-specific expression in transgenic fish. BMC Dev. Biol. 7, 42 (2007).
Ong, S. L. M., de Vos, I., Meroshini, M., Poobalan, Y. & Dunn, N. R. Microfibril-associated glycoprotein 4 (Mfap4) regulates haematopoiesis in zebrafish. Sci. Rep. 10, 11801 (2020).
Clarke, D. et al. Biologics in peripheral ulcerative keratitis. Semin. Arthritis Rheum. 63, 152269 (2023).
Straub, M., Bron, A. M., Muselier-Mathieu, A. & Creuzot-Garcher, C. Long-term outcome after topical ciclosporin in severe dry eye disease with a 10-year follow-up. Br. J. Ophthalmol. 100, 1547 (2016).
Taube, M. A., del Mar Cendra, M., Elsahn, A., Christodoulides, M. & Hossain, P. Pattern recognition receptors in microbial keratitis. Eye 29, 1399–1415 (2015).
Subramanian, B. C., Majumdar, R. & Parent, C. A. The role of the LTB(4)-BLT1 axis in chemotactic gradient sensing and directed leukocyte migration. Semin. Immunol. 33, 16–29 (2017).
Zhao, X. C. et al. The zebrafish cornea: structure and development. Investig. Ophthalmol. Vis. Sci. 47, 4341–4348 (2006).
Lubin, A. et al. A versatile, automated and high-throughput drug screening platform for zebrafish embryos. Biol Open 10, https://doi.org/10.1242/bio.058513 (2021).
Westhoff, J. H. et al. Development of an automated imaging pipeline for the analysis of the zebrafish larval kidney. PLoS ONE 8, e82137 (2013).
Kleinhans, D. S. & Lecaudey, V. Standardized mounting method of (zebrafish) embryos using a 3D-printed stamp for high-content, semi-automated confocal imaging. BMC Biotechnol. 19, 68 (2019).
Lin, X. et al. Autonomous system for cross-organ investigation of ethanol-induced acute response in behaving larval zebrafish. Biomicrofluidics 10, 024123 (2016).
Carvalho, R. et al. A high-throughput screen for tuberculosis progression. PLoS ONE 6, e16779 (2011).
Huemer, K. et al. zWEDGI: wounding and entrapment device for imaging live zebrafish larvae. Zebrafish 14, 42–50 (2017).
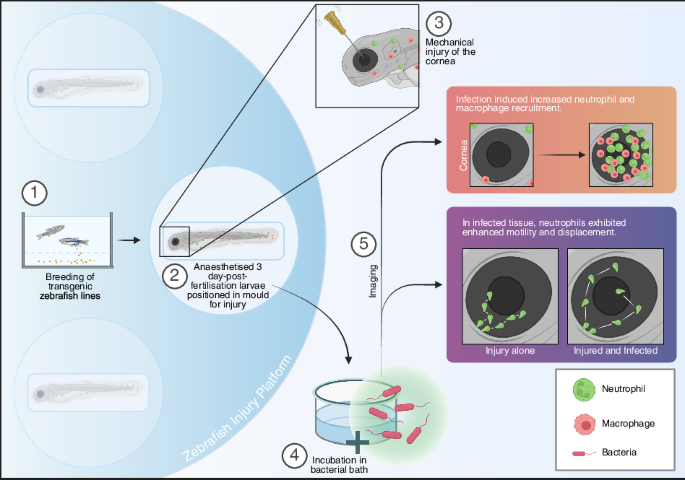
Cheng, K. Zebrafish Injury Platform (ZIP). https://doi.org/10.6084/m9.figshare.31007692.v1 (2026).
Gillies, S. et al. Transgenic zebrafish larvae as a non-rodent alternative model to assess pro-inflammatory (neutrophil) responses to nanomaterials. Nanotoxicology 16, 333–354 (2022).
Lucas, C. D. et al. Flavones induce neutrophil apoptosis by down-regulation of Mcl-1 via a proteasomal-dependent pathway. FASEB J. 27, 1084–1094 (2013).
Mader, D., Rabiet, M.-J., Boulay, F. & Peschel, A. Formyl peptide receptor-mediated proinflammatory consequences of peptide deformylase inhibition in Staphylococcus aureus. Microbes Infect. 12, 415–419 (2010).
Dorward, D. A. et al. The role of formylated peptides and formyl peptide receptor 1 in governing neutrophil function during acute inflammation. Am. J. Pathol. 185, 1172–1184 (2015).
Sahin, A., Kam, W. R., Darabad, R. R., Topilow, K. & Sullivan, D. A. Regulation of leukotriene B4 secretion by human corneal, conjunctival, and meibomian gland epithelial cells. Arch. Ophthalmol. 130, 1013–1018 (2012).
Thakur, A. & Willcox, M. D. Cytokine and lipid inflammatory mediator profile of human tears during contact lens associated inflammatory diseases. Exp. Eye Res. 67, 9–19 (1998).
Kernacki, K. A. & Berk, R. S. Characterization of arachidonic acid metabolism and the polymorphonuclear leukocyte response in mice infected intracorneally with Pseudomonas aeruginosa. Investig. Ophthalmol. Vis. Sci. 36, 16–23 (1995).
Lin, M., Carlson, E., Diaconu, E. & Pearlman, E. CXCL1/KC and CXCL5/LIX are selectively produced by corneal fibroblasts and mediate neutrophil infiltration to the corneal stroma in LPS keratitis. J. Leukoc. Biol. 81, 786–792 (2007).
Carlson, E. C. et al. Regulation of corneal inflammation by neutrophil-dependent cleavage of keratan sulfate proteoglycans as a model for breakdown of the chemokine gradient. J. Leukoc. Biol. 88, 517–522 (2010).
Liu, J. et al. Trade-off between branching and polarity controls decision-making during cell migration. Sci. Adv. 12, eads2734 (2026).
Yoo, S. K. & Huttenlocher, A. Spatiotemporal photolabeling of neutrophil trafficking during inflammation in live zebrafish. J. Leukoc. Biol. 89, 661–667 (2011).
Carlson, E. C., Drazba, J., Yang, X. & Perez, V. L. Visualization and characterization of inflammatory cell recruitment and migration through the corneal stroma in endotoxin-induced keratitis. Investig. Ophthalmol. Vis. Sci. 47, 241–248 (2006).
Chinnery, H. R. et al. TLR9 ligand CpG-ODN applied to the injured mouse cornea elicits retinal inflammation. Am. J. Pathol. 180, 209–220 (2012).
Lundgren, S. M. et al. Signaling dynamics distinguish high- and low-priority neutrophil chemoattractant receptors. Sci. Signal. 16, eadd1845 (2023).
Richardson, I. M. et al. Diverse bacteria elicit distinct neutrophil responses in a physiologically relevant model of infection. iScience 27, 108627 (2024).
Snäll, J. et al. Differential neutrophil responses to bacterial stimuli: streptococcal strains are potent inducers of heparin-binding protein and resistin-release. Sci. Rep. 6, 21288 (2016).
Ikkala, K., Stratoulias, V. & Michon, F. Unilateral zebrafish corneal injury induces bilateral cell plasticity supporting wound closure. Sci. Rep. 12, 161 (2022).
Alper, S. R. & Dorsky, R. I. Unique advantages of zebrafish larvae as a model for spinal cord regeneration. Front. Mol. Neurosci. 15, 983336 (2022).
Shankar, J. et al. Genotypic analysis of UK keratitis-associated Pseudomonas aeruginosa suggests adaptation to environmental water as a key component in the development of eye infections. FEMS Microbiol. Lett. 334, 79–86 (2012).
Sun, Y. et al. A murine model of contact lens-associated Fusarium keratitis. Investig. Ophthalmol. Vis. Sci. 51, 1511–1516 (2010).
Sharma, C., Thakur, A., Bhatia, A., Gupta, A. & Khurana, S. Acanthamoeba keratitis in a mouse model using a novel approach. Indian J. Med. Microbiol. 39, 523–527 (2021).
Nguyen-Chi, M. et al. TNF signaling and macrophages govern fin regeneration in zebrafish larvae. Cell Death Dis. 8, e2979–e2979 (2017).
Tsarouchas, T. M. et al. Dynamic control of proinflammatory cytokines Il-1beta and Tnf-alpha by macrophages in zebrafish spinal cord regeneration. Nat. Commun. 9, 4670 (2018).
Hammond, F. R. et al. An arginase 2 promoter transgenic line illuminates immune cell polarisation in zebrafish. Dis. Model Mech. 16, https://doi.org/10.1242/dmm.049966 (2023).
Konstantopoulos, A. et al. Morphological and cytokine profiles as key parameters to distinguish between Gram-negative and Gram-positive bacterial keratitis. Sci. Rep. 10, 20092 (2020).
Shiau, C. E., Kaufman, Z., Meireles, A. M. & Talbot, W. S. Differential requirement for irf8 in formation of embryonic and adult macrophages in zebrafish. PLoS ONE 10, e0117513 (2015).
Lian, J. et al. Alas1 is essential for neutrophil maturation in zebrafish. Haematologica 103, 1785–1795 (2018).
Hall, C. J., Astin, J. W., Mumm, J. S. & Ackerley, D. F. A new transgenic line for rapid and complete neutrophil ablation. Zebrafish 19, 109–113 (2022).
Hwang, W. Y. et al. Efficient genome editing in zebrafish using a CRISPR-Cas system. Nat. Biotechnol. 31, 227–229 (2013).
Peters, J. M. et al. A comprehensive, CRISPR-based functional analysis of essential genes in bacteria. Cell 165, 1493–1506 (2016).
Yu, M. A. et al. Investigating Pseudomonas aeruginosa gene function during pathogenesis using mobile-CRISPRi. Methods Mol. Biol. 2721, 13–32 (2024).
Qu, J. et al. Modulating pathogenesis with mobile-CRISPRi. J. Bacteriol. 201, https://doi.org/10.1128/jb.00304-19 (2019).
Ellett, F., Pase, L., Hayman, J. W., Andrianopoulos, A. & Lieschke, G. J. mpeg1 promoter transgenes direct macrophage-lineage expression in zebrafish. Blood 117, e49–e56 (2011).
Lee, R. T., Asharani, P. V. & Carney, T. J. Basal keratinocytes contribute to all strata of the adult zebrafish epidermis. PLoS ONE 9, e84858 (2014).
Westerfield, M. The Zebrafish Book: a Guide for the Laboratory Use of Zebrafish (University of Oregon, 1993).
Tinevez, J. Y. et al. TrackMate: an open and extensible platform for single-particle tracking. Methods 115, 80–90 (2017).













Leave a Reply