Cappell, K. M. & Kochenderfer, J. N. Long-term outcomes following CAR T cell therapy: what we know so far. Nat. Rev. Clin. Oncol. 20, 359–371 (2023).
Ali, A. & DiPersio, J. F. ReCARving the future: bridging CAR T-cell therapy gaps with synthetic biology, engineering, and economic insights. Front. Immunol. 15, 1432799 (2024).
Brudno, J. N. & Kochenderfer, J. N. Current understanding and management of CAR T cell-associated toxicities. Nat. Rev. Clin. Oncol. 21, 501–521 (2024).
Rejeski, K., Hill, J. A., Dahiya, S. & Jain, M. D. Noncanonical and mortality-defining toxicities of CAR T cell therapy. Nat. Med. 31, 2132–2146 (2025).
Fraietta, J. A. et al. Disruption of TET2 promotes the therapeutic efficacy of CD19-targeted T cells. Nature 558, 307–312 (2018).
Shah, N. N. et al. Clonal expansion of CAR T cells harboring lentivector integration in the CBL gene following anti-CD22 CAR T-cell therapy. Blood Adv. 3, 2317–2322 (2019).
Sasu, B. J. et al. Detection of chromosomal alteration after infusion of gene-edited allogeneic CAR T cells. Mol. Ther. 31, 676–685 (2023).
Nobles, C. L. et al. CD19-targeting CAR T cell immunotherapy outcomes correlate with genomic modification by vector integration. J. Clin. Invest. 130, 673–685 (2020).
Ozdemirli, M. et al. Indolent CD4+ CAR T-cell lymphoma after cilta-cel CAR T-cell therapy. N. Engl. J. Med. 390, 2074–2082 (2024).
Perica, K. et al. CD4+ T-cell lymphoma harboring a chimeric antigen receptor integration in TP53. N. Engl. J. Med. 392, 577–583 (2025).
Hosoya, H. et al. Long-term follow-up of gastrointestinal CAR T-cell lymphoma: homing, clonal expansion, and response to cyclosporine. Blood 147, 1191–1198 (2025).
Bezerra, E. et al. Isolated oral CD30+/CD4+ CAR+ T cell lymphoma in long-term remission after radiotherapy. Mol. Ther. 33, 6033–6040 (2025).
Zuniga, L. M. et al. Diarrheal dilemma: a case of post-CAR-T T-cell lymphoma presenting as refractory diarrhea. Am. J. Gastroenterol. 120, S1081–S1081 (2025).
Rolak, S., Patel, C., Cheng, J., Eller, V. & Umar, S. Persistent diarrhea following CAR-T therapy for multiple myeloma: a case of immune effector cell-associated enterocolitis.Am. J. Gastroenterol. 120, S1305–S1305 (2025).
Aleman, A. et al. Targeted therapy of CAR+ T-cell lymphoma after anti-BCMA CAR T-cell therapy. N. Engl. J. Med. 393, 823–825 (2025).
Braun, T. et al. Multiomic profiling of T cell lymphoma after therapy with anti-BCMA CAR T cells and GPRC5D-directed bispecific antibody. Nat. Med. 31, 1145–1153 (2025).
Harrison, S. J. et al. CAR+ T-cell lymphoma after cilta-cel therapy for relapsed or refractory myeloma. N. Engl. J. Med. 392, 677–685 (2025).
Micklethwaite, K. P. et al. Investigation of product-derived lymphoma following infusion of piggyBac-modified CD19 chimeric antigen receptor T cells. Blood 138, 1391–1405 (2021).
Kobbe, G. et al. Aggressive lymphoma after CD19 CAR T-cell therapy. N. Engl. J. Med. 391, 1217–1226 (2024).
Hamilton, M. P. et al. Risk of second tumors and T-cell lymphoma after CAR T-cell therapy. N. Engl. J. Med. 390, 2047–2060 (2024).
Ghilardi, G. et al. T cell lymphoma and secondary primary malignancy risk after commercial CAR T cell therapy. Nat. Med. 30, 984–989 (2024).
Godfrey, J., Querfeld, C. & Song, J. Risk of second tumors and T-cell lymphoma after CAR T-cell therapy. N. Engl. J. Med. 391, 869–870 (2024).
Li, P. et al. C-CAR039, a novel anti-CD20/CD19 bi-specific CAR T-cell therapy shows deep and durable clinical benefits in patients with relapsed or refractory (r/r) B-cell non-Hodgkin lymphoma (B-NHL) in long term follow up. Blood 142, 1025–1025 (2023).
Palomba, M. L. et al. Lisocabtagene maraleucel in patients with relapsed or refractory marginal zone lymphoma (TRANSCEND FL): primary analysis results from the global, multicohort, single-arm, phase 2 study. Lancet 407, 963–975 (2026).
Alaggio, R. et al. The 5th edition of the world health organization classification of haematolymphoid tumours: lymphoid neoplasms. Leukemia 36, 1720–1748 (2022).
Campo, E. et al. The international consensus classification of mature lymphoid neoplasms: a report from the clinical advisory committee. Blood 140, 1229–1253 (2022).
De Leval, L., Gaulard, P. & Dogan, A. A practical approach to the modern diagnosis and classification of T- and NK-cell lymphomas. Blood 144, 1855–1872 (2024).
Savage, K. J. & De Leval, L. Introduction to the peripheral T-cell lymphoma review series: advances in molecular characterization, classification refinement and treatment optimization. Haematologica 108, 3204–3210 (2023).
Dulery, R. et al. T cell malignancies after CAR T cell therapy in the DESCAR-T registry. Nat. Med. 31, 1130–1133 (2025).
Elsallab, M. et al. Second primary malignancies after commercial CAR T-cell therapy: analysis of the FDA Adverse Events Reporting System. Blood 143, 2099–2105 (2024).
Jadlowsky, J. K. et al. Long-term safety of lentiviral or gammaretroviral gene-modified T cell therapies. Nat. Med. 31, 1134–1144 (2025).
Steffin, D. H. M. et al. Long-term follow-up for the development of subsequent malignancies in patients treated with genetically modified IECs. Blood 140, 16–24 (2022).
Cappell, K. M. et al. Long-term follow-up of anti-CD19 chimeric antigen receptor T-cell therapy. J. Clin. Oncol. 38, 3805–3815 (2020).
Umyarova, E. et al. Second primary malignancies following CAR T-cell therapy in patients with hematologic malignancies. J. Hematol. Oncol. 18, 30 (2025).
Camacho-Arteaga, L. et al. Late adverse events after chimeric antigen receptor T-cell therapy for patients with aggressive B-cell non-Hodgkin lymphoma. JAMA Netw. Open 8, e2461683 (2025).
Hsieh, E. M. et al. Low rate of subsequent malignant neoplasms after CD19 CAR T-cell therapy. Blood Adv. 6, 5222–5226 (2022).
Levine, B. L. et al. Unanswered questions following reports of secondary malignancies after CAR-T cell therapy. Nat. Med. 30, 338–341 (2024).
Pagliuca, S. et al. The landscape of immune monitoring in CAR-T cell therapy: a comprehensive review and survey study by the cellular therapy and immunobiology working party of the EBMT. Blood Rev. 71, 101272 (2025).
Andersen, M. Jr. et al. Clonal myeloid disorders following CAR T-cell therapy. Ann. Hematol. 102, 3643–3645 (2023).
Zhao, A. et al. Secondary myeloid neoplasms after CD19 CAR T therapy in patients with refractory/relapsed B-cell lymphoma: case series and review of literature. Front. Immunol. 13, 1063986 (2022).
Shen, J. et al. Characterization of second primary malignancies post CAR T-cell therapy: real-world insights from the two global pharmacovigilance databases of FAERS and VigiBase. EClinicalMedicine 73, 102684 (2024).
Gazeau, N. et al. Myeloid neoplasms after CD19-directed CAR T cells therapy in long-term B-cell lymphoma responders, a rising risk over time? Leukemia 39, 1714–1722 (2025).
Melody, M. et al. Subsequent malignant neoplasms in patients previously treated with anti-CD19 CAR T-cell therapy. Blood Adv. 8, 2327–2331 (2024).
Tix, T. et al. Second primary malignancies after CAR T-cell therapy: a systematic review and meta-analysis of 5,517 lymphoma and myeloma patients. Clin. Cancer Res. 30, 4690–4700 (2024).
Alkhateeb, H. B. et al. Therapy-related myeloid neoplasms following chimeric antigen receptor T-cell therapy for non-Hodgkin lymphoma. Blood Cancer J. 12, 113 (2022).
Gurney, M. et al. Features and factors associated with myeloid neoplasms after chimeric antigen receptor T-cell therapy. JAMA Oncol. 10, 532–535 (2024).
Farina, M. et al. Real-world collection of secondary myeloid neoplasms after CD19 CAR-T cell therapy: first report of the ClonHema study. Bone Marrow Transplant. 60, 702–704 (2025).
Hamilton, M. P. et al. CAR19 therapy drives expansion of clonal hematopoiesis and associated cytopenias. Preprint at Res. Sq. https://doi.org/10.21203/rs.3.rs-7746241/v1 (2025).
Hacein-Bey-Abina, S. et al. A serious adverse event after successful gene therapy for X-linked severe combined immunodeficiency. N. Engl. J. Med. 348, 255–256 (2003).
Hacein-Bey-Abina, S. et al. LMO2-associated clonal T cell proliferation in two patients after gene therapy for SCID-X1. Science 302, 415–419 (2003).
Braun, C. J. et al. Gene therapy for Wiskott–Aldrich syndrome-long-term efficacy and genotoxicity. Sci. Transl. Med. 6, 227ra233 (2014).
Tucci, F., Galimberti, S., Naldini, L., Valsecchi, M. G. & Aiuti, A. A systematic review and meta-analysis of gene therapy with hematopoietic stem and progenitor cells for monogenic disorders. Nat. Commun. 13, 1315 (2022).
Cesana, D. et al. A case of T-cell acute lymphoblastic leukemia in retroviral gene therapy for ADA-SCID. Nat. Commun. 15, 3662 (2024).
Goyal, S. et al. Acute myeloid leukemia case after gene therapy for sickle cell disease. N. Engl. J. Med. 386, 138–147 (2022).
Assessing the risks of engineered T cells. Nat. Med. 31, 1079–1080 (2025).
Sheih, A. et al. Clonal kinetics and single-cell transcriptional profiling of CAR-T cells in patients undergoing CD19 CAR-T immunotherapy. Nat. Commun. 11, 219 (2020).
McLellan, A. D. & Ali Hosseini Rad, S. M. Chimeric antigen receptor T cell persistence and memory cell formation. Immunol. Cell Biol. 97, 664–674 (2019).
Neelapu, S. S. et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N. Engl. J. Med. 377, 2531–2544 (2017).
Locke, F. L. et al. Phase 1 results of ZUMA-1: a multicenter study of KTE-C19 anti-CD19 CAR T cell therapy in refractory aggressive lymphoma. Mol. Ther. 25, 285–295 (2017).
Brentjens, R. J. et al. CD19-targeted T cells rapidly induce molecular remissions in adults with chemotherapy-refractory acute lymphoblastic leukemia. Sci. Transl. Med. 5, 177ra138 (2013).
Turtle, C. J. et al. CD19 CAR-T cells of defined CD4+:CD8+ composition in adult B cell ALL patients. J. Clin. Invest. 126, 2123–2138 (2016).
Kaczanowska, S. et al. Immune determinants of CAR-T cell expansion in solid tumor patients receiving GD2 CAR-T cell therapy. Cancer Cell 42, 35–51.e38 (2024).
Biasco, L. et al. Clonal expansion of T memory stem cells determines early anti-leukemic responses and long-term CAR T cell persistence in patients. Nat. Cancer 2, 629–642 (2021).
Melenhorst, J. J. et al. Decade-long leukaemia remissions with persistence of CD4+ CAR T cells. Nature 602, 503–509 (2022).
Ledergor, G. et al. CD4+ CAR T-cell exhaustion associated with early relapse of multiple myeloma after BCMA CAR T-cell therapy. Blood Adv. 8, 3562–3575 (2024).
Cao, G. et al. Two-stage CD8+ CAR T-cell differentiation in patients with large B-cell lymphoma. Nat. Commun. 16, 4205 (2025).
Ho, M. et al. CD4+ T cells mediate CAR-T cell-associated immune-related adverse events after BCMA CAR-T cell therapy. Nat. Med. 32, 702–716 (2026).
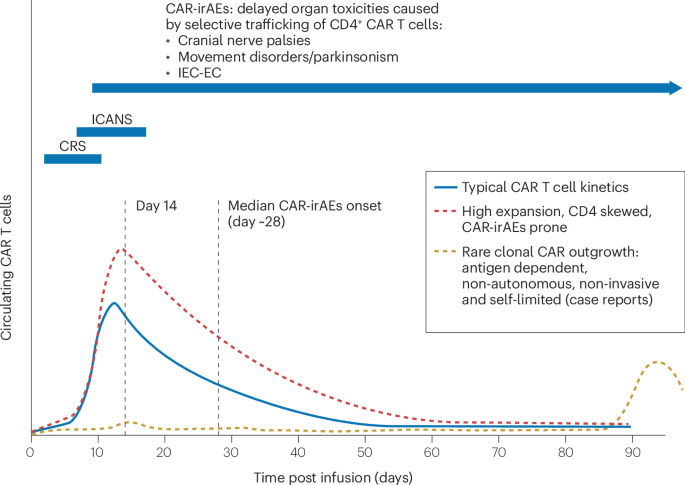
Van De Donk, N. W. C. J. et al. Clinical experience with cranial nerve impairment in the CARTITUDE-1, CARTITUDE-2 cohorts A, B, and C, and cartitude-4 studies of ciltacabtagene autoleucel (cilta-cel). Blood 142, 3501–3501 (2023).
Kumar, A. D. et al. Delayed neurotoxicity after CAR-T in multiple myeloma: results from a global IMWG registry. Blood 144, 4758–4758 (2024).
Iqbal, J., Inghirami, G. & Chan, W. C. New insights into the biology of T-cell lymphomas. Blood 144, 1873–1886 (2024).
Bisig, B., Savage, K. J. & De Leval, L. Pathobiology of nodal peripheral T-cell lymphomas: current understanding and future directions. Haematologica 108, 3227–3243 (2023).
Laurent, C. et al. Impact of expert pathologic review of lymphoma diagnosis: study of patients from the french lymphopath network. J. Clin. Oncol. 35, 2008–2017 (2017).
Chan, J. K. & Kwong, Y. L. Common misdiagnoses in lymphomas and avoidance strategies. Lancet Oncol. 11, 579–588 (2010).
Gratzinger, D. et al. T- and NK-cell lymphomas and systemic lymphoproliferative disorders and the immunodeficiency setting: 2015 SH/EAHP workshop report — part 4. Am. J. Clin. Pathol. 147, 188–203 (2017).
Brown, J. S. et al. Updating the definition of cancer. Mol. Cancer Res. 21, 1142–1147 (2023).
Perry, A. M. et al. Indolent T-cell lymphoproliferative disease of the gastrointestinal tract. Blood 122, 3599–3606 (2013).
US Food & Drug Administration. FDA approves labeling changes that include a boxed warning for immune effector cell-associated enterocolitis following treatment with ciltacabtagene autoleucel (Carvykti, Janssen Biotech, Inc.). Medical product safety information. FDA https://www.fda.gov/safety/medical-product-safety-information/fda-approves-labeling-changes-include-boxed-warning-immune-effector-cell-associated-enterocolitis (2025).
Chihara, D., Dores, G. M., Flowers, C. R. & Morton, L. M. The bidirectional increased risk of B-cell lymphoma and T-cell lymphoma. Blood 138, 785–789 (2021).
Maurer, K. et al. A clonally expanded nodal T-cell population diagnosed as T-cell lymphoma after CAR-T therapy. Nat. Commun. 16, 7462 (2025).
Kumar, B. V., Connors, T. J. & Farber, D. L. Human T cell development, localization, and function throughout life. Immunity 48, 202–213 (2018).
Larson, R. C. & Maus, M. V. Recent advances and discoveries in the mechanisms and functions of CAR T cells. Nat. Rev. Cancer 21, 145–161 (2021).
Niroula, A. et al. Distinction of lymphoid and myeloid clonal hematopoiesis. Nat. Med. 27, 1921–1927 (2021).
Kang, T. G. et al. Epigenetic regulators of clonal hematopoiesis control CD8 T cell stemness during immunotherapy. Science 386, eadl4492 (2024).
Ma, H., Gao, W., Sun, X. & Wang, W. STAT5 and TET2 cooperate to regulate FOXP3–TSDR demethylation in CD4+ T cells of patients with colorectal cancer. J. Immunol. Res. 2018, 6985031 (2018).
Carty, S. A. et al. The loss of TET2 promotes CD8+ T cell memory differentiation. J. Immunol. 200, 82–91 (2018).
Lee, M. et al. Tet2 inactivation enhances the antitumor activity of tumor-infiltrating lymphocytes. Cancer Res. 81, 1965–1976 (2021).
Dimitri, A. J. et al. TET2 regulates early and late transitions in exhausted CD8+ T cell differentiation and limits CAR T cell function. Sci. Adv. 10, eadp9371 (2024).
Prinzing, B. et al. Deleting DNMT3A in CAR T cells prevents exhaustion and enhances antitumor activity. Sci. Transl. Med. 13, eabh0272 (2021).
Zebley, C. C. et al. Proinflammatory cytokines promote TET2-mediated DNA demethylation during CD8 T cell effector differentiation. Cell Rep. 37, 109796 (2021).
Boettcher, S. et al. Clonal hematopoiesis in donors and long-term survivors of related allogeneic hematopoietic stem cell transplantation. Blood 135, 1548–1559 (2020).
Gibson, C. J. et al. Donor clonal hematopoiesis and recipient outcomes after transplantation. J. Clin. Oncol. 40, 189–201 (2022).
Herold, S. et al. Donor cell leukemia: evidence for multiple preleukemic clones and parallel long term clonal evolution in donor and recipient. Leukemia 31, 1637–1640 (2017).
Gondek, L. P. et al. Donor cell leukemia arising from clonal hematopoiesis after bone marrow transplantation. Leukemia 30, 1916–1920 (2016).
Wong, W. H. et al. Engraftment of rare, pathogenic donor hematopoietic mutations in unrelated hematopoietic stem cell transplantation. Sci. Transl. Med. 12, eaax6249 (2020).
Miller, P. G. et al. Clonal hematopoiesis in patients receiving chimeric antigen receptor T-cell therapy. Blood Adv. 5, 2982–2986 (2021).
Panagiota, V. et al. Clinical implications and dynamics of clonal hematopoiesis in anti-CD19 CAR T-cell treated patients. Hemasphere 7, e957 (2023).
Teipel, R. et al. Prevalence and variation of CHIP in patients with aggressive lymphomas undergoing CD19-directed CAR T-cell treatment. Blood Adv. 6, 1941–1946 (2022).
Yi, F. et al. CAR-engineered lymphocyte persistence is governed by a FAS ligand–FAS autoregulatory circuit. Nat. Cancer 6, 1638–1655 (2025).
Long, A. H. et al. 4-1BB costimulation ameliorates T cell exhaustion induced by tonic signaling of chimeric antigen receptors. Nat. Med. 21, 581–590 (2015).
Calderon, H., Mamonkin, M. & Guedan, S. Analysis of CAR-mediated tonic signaling. Methods Mol. Biol. 2086, 223–236 (2020).
Landoni, E. et al. Modifications to the framework regions eliminate chimeric antigen receptor tonic signaling. Cancer Immunol. Res. 9, 441–453 (2021).
Gomes-Silva, D. et al. Tonic 4-1BB costimulation in chimeric antigen receptors impedes T cell survival and is vector-dependent. Cell Rep. 21, 17–26 (2017).
Blumenberg, V. et al. Ruxolitinib for ciltacabtagene autoleucel-associated refractory diarrhea. Blood https://doi.org/10.1182/blood.2025032347 (2026).
Yang, S. et al. Neutrophil activation and clonal CAR-T re-expansion underpinning cytokine release syndrome during ciltacabtagene autoleucel therapy in multiple myeloma. Nat. Commun. 15, 360 (2024).
Luan, D. et al. Dynamics of immune reconstitution and impact on outcomes across CAR-T cell products in large B-cell lymphoma. Blood Cancer Discov. 6, 119–130 (2025).
Riedel, A. et al. Immunological consequences of CAR T-cell therapy: an analysis of infectious complications and immune reconstitution. Blood Adv. 9, 3149–3158 (2025).
Nann-Rütti, S. et al. Large granular lymphocyte expansion after allogeneic hematopoietic stem cell transplant is associated with a cytomegalovirus reactivation and shows an indolent outcome. Biol. Blood Marrow Transplant. 18, 1765–1770 (2012).
Ergas, D., Resnitzky, P. & Berrebi, A. Pure red blood cell aplasia associated with parvovirus B19 infection in large granular lymphocyte leukemia. Blood 87, 3523–3524 (1996).
Silva-Santos, B., Serre, K. & Norell, H. γδ T cells in cancer. Nat. Rev. Immunol. 15, 683–691 (2015).
Yoshie, O. et al. Frequent expression of CCR4 in adult T-cell leukemia and human T-cell leukemia virus type 1-transformed T cells. Blood 99, 1505–1511 (2002).
Campbell, J. J., Clark, R. A., Watanabe, R. & Kupper, T. S. Sezary syndrome and mycosis fungoides arise from distinct T-cell subsets: a biologic rationale for their distinct clinical behaviors. Blood 116, 767–771 (2010).
Jones, D. et al. Expression pattern of T-cell-associated chemokine receptors and their chemokines correlates with specific subtypes of T-cell non-Hodgkin lymphoma. Blood 96, 685–690 (2000).
Fraietta, J. A. et al. Determinants of response and resistance to CD19 chimeric antigen receptor (CAR) T cell therapy of chronic lymphocytic leukemia. Nat. Med. 24, 563–571 (2018).
Bai, Z. et al. Single-cell CAR T atlas reveals type 2 function in 8-year leukaemia remission. Nature 634, 702–711 (2024).
Boztug, K. et al. Stem-cell gene therapy for the Wiskott–Aldrich syndrome. N. Engl. J. Med. 363, 1918–1927 (2010).
Modlich, U. et al. Cell-culture assays reveal the importance of retroviral vector design for insertional genotoxicity. Blood 108, 2545–2553 (2006).
Greig, J. A. et al. Integrated vector genomes may contribute to long-term expression in primate liver after AAV administration. Nat. Biotechnol. 42, 1232–1242 (2024).
Newrzela, S. et al. Resistance of mature T cells to oncogene transformation. Blood 112, 2278–2286 (2008).
Hu, J. & Dunbar, C. E. T-cell lymphomas in recipients of CAR-T cells: assessing risks and causalities. Blood 144, 2473–2481 (2024).
Schmidt, M. et al. High-resolution insertion-site analysis by linear amplification-mediated PCR (LAM-PCR). Nat. Methods 4, 1051–1057 (2007).
Cattoglio, C. et al. High-definition mapping of retroviral integration sites identifies active regulatory elements in human multipotent hematopoietic progenitors. Blood 116, 5507–5517 (2010).
Shao, L. et al. Genome-wide profiling of retroviral DNA integration and its effect on clinical pre-infusion CAR T-cell products. J. Transl. Med. 20, 514 (2022).
Głowacki, P., Tręda, C. & Rieske, P. Regulation of CAR transgene expression to design semiautonomous CAR-T. Mol. Ther. Oncol. 32, 200833 (2024).
Sheehan, M. et al. Comparison and cross-validation of long-read and short-read target-enrichment sequencing methods to assess AAV vector integration into host genome. Mol. Ther. Methods Clin. Dev. 32, 101352 (2024).
Moskowitz, A. J., Stuver, R. N. & Horwitz, S. M. Current and upcoming treatment approaches to common subtypes of PTCL (PTCL, NOS; ALCL; and TFHs). Blood 144, 1887–1897 (2024).
Marchi, E., Craig, J. W. & Kalac, M. Current and upcoming treatment approaches to uncommon subtypes of PTCL (EATL, MEITL, SPTCL, and HSTCL). Blood 144, 1898–1909 (2024).
Ngu, H. S. & Savage, K. J. Past, present and future therapeutic approaches in nodal peripheral T-cell lymphomas. Haematologica 108, 3211–3226 (2023).
US Food & Drug Administration. Workshop on integration site analysis during long-term follow-up for gene therapies with integrating viral vectors. FDA https://www.fda.gov/news-events/otp-events-meetings-and-workshops/workshop-integration-site-analysis-during-long-term-follow-gene-therapies-integrating-viral-vectors (2024).
US Food & Drug Administration. Long term follow-up after administration of human gene therapy products. Guidance for industry. FDA https://www.fda.gov/media/113768/download (2026).
Guerrettaz, R. D., Spellman, S. R. & Page, K. M. Role of the CIBMTR biorepository and registry in precision transplantation research. Bone Marrow Transplant. 60, 161–164 (2025).
Fortuna, G. G. et al. Immune effector cell-associated enterocolitis following chimeric antigen receptor T-cell therapy in multiple myeloma. Blood Cancer J. 14, 180 (2024).
Zundler, S. et al. Case report: IBD-like colitis following CAR T cell therapy for diffuse large B cell lymphoma. Front. Oncol. 13, 1149450 (2023).
Banerjee, R. et al. Managing IEC-associated enterocolitis following CAR-T therapy in multiple myeloma. Blood Cancer J. 15, 112 (2025).
Bar, N. et al. P-008 a rare and unexplored entity of colitis post-BCMA directed CAR T-cell therapy; insights from a multicenter case series. Clin. Lymphoma Myeloma Leuk. 24, S44–S45 (2024).
Lim, K., Lin, Y. & Chowla, N. Diagnosis and management of immune effector cell (CAR-T)-associated enterocolitis in multiple myeloma patients. Am. J. Gastroenterol. 120, S315–S315 (2025).
Belwadi, M. et al. Immune effector cell-associated enterocolitis (IEC-EC) across CAR-T products in myeloma and lymphoma: a real world pharmacovigilance analysis. Blood 146 (Suppl. 1), 6366 (2025).
Li, C. L., Xiong, D., Stamatoyannopoulos, G. & Emery, D. W. Genomic and functional assays demonstrate reduced gammaretroviral vector genotoxicity associated with use of the cHS4 chromatin insulator. Mol. Ther. 17, 716–724 (2009).
Ramezani, A., Hawley, T. S. & Hawley, R. G. Combinatorial incorporation of enhancer-blocking components of the chicken β-globin 5′HS4 and human T-cell receptor α/δBEAD-1 insulators in self-inactivating retroviral vectors reduces their genotoxic potential. Stem Cells 26, 3257–3266 (2008).
Higashimoto, T. et al. The woodchuck hepatitis virus post-transcriptional regulatory element reduces readthrough transcription from retroviral vectors. Gene Ther. 14, 1298–1304 (2007).
Wang, D., Tai, P. W. L. & Gao, G. Adeno-associated virus vector as a platform for gene therapy delivery. Nat. Rev. Drug Discov. 18, 358–378 (2019).
Verghese, S. C., Goloviznina, N. A., Skinner, A. M., Lipps, H. J. & Kurre, P. S/MAR sequence confers long-term mitotic stability on non-integrating lentiviral vector episomes without selection. Nucleic Acids Res. 42, e53 (2014).
Querques, I. et al. A highly soluble sleeping beauty transposase improves control of gene insertion. Nat. Biotechnol. 37, 1502–1512 (2019).
Eyquem, J. et al. Targeting a CAR to the TRAC locus with CRISPR–Cas9 enhances tumour rejection. Nature 543, 113–117 (2017).
Anzalone, A. V. et al. Search-and-replace genome editing without double-strand breaks or donor DNA. Nature 576, 149–157 (2019).
Chiesa, R. et al. Base-edited CAR7 T cells for relapsed T-cell acute lymphoblastic leukemia. N. Engl. J. Med. 389, 899–910 (2023).
Parayath, N. N., Stephan, S. B., Koehne, A. L., Nelson, P. S. & Stephan, M. T. In vitro-transcribed antigen receptor mRNA nanocarriers for transient expression in circulating T cells in vivo. Nat. Commun. 11, 6080 (2020).
Vakulskas, C. A. et al. A high-fidelity Cas9 mutant delivered as a ribonucleoprotein complex enables efficient gene editing in human hematopoietic stem and progenitor cells. Nat. Med. 24, 1216–1224 (2018).
Amengual, J. E. & Pro, B. How I treat posttransplant lymphoproliferative disorder. Blood 142, 1426–1437 (2023).
El-Mallawany, N. K. & Rouce, R. H. EBV and post-transplant lymphoproliferative disorder: a complex relationship. Hematol. Am. Soc. Hematol. Educ. Program 2024, 728–735 (2024).
Ferreiro, J. F. et al. EBV-positive and EBV-negative posttransplant diffuse large B cell lymphomas have distinct genomic and transcriptomic features. Am. J. Transplant. 16, 414–425 (2016).
Margolskee, E. et al. Indolent small intestinal CD4+ T-cell lymphoma is a distinct entity with unique biologic and clinical features. PLoS ONE 8, e68343 (2013).
Perry, A. M., Bailey, N. G., Bonnett, M., Jaffe, E. S. & Chan, W. C. Disease progression in a patient with indolent T-cell lymphoproliferative disease of the gastrointestinal tract. Int. J. Surg. Pathol. 27, 102–107 (2019).
Yuan, D., Liang, N., Wang, D. Y., Wang, J. J. & Jia, C. W. Clinical and pathological analysis of indolent T-cell lymphoproliferative disease of the gastrointestinal tract. Front. Immunol. 16, 1530149 (2025).
Fan, W. et al. Indolent T-cell lymphoproliferative disorder of gastrointestinal tract with unusual clinical courses: report of 6 cases and literature review. Virchows Arch. 482, 729–743 (2023).
Sharma, A. et al. Recurrent STAT3–JAK2 fusions in indolent T-cell lymphoproliferative disorder of the gastrointestinal tract. Blood 131, 2262–2266 (2018).
Soderquist, C. R. et al. Genetic and phenotypic characterization of indolent T-cell lymphoproliferative disorders of the gastrointestinal tract. Haematologica 105, 1895–1906 (2020).
Leberfinger, A. N. et al. Breast implant-associated anaplastic large cell lymphoma: a systematic review. JAMA Surg. 152, 1161–1168 (2017).
Lechner, M. G. et al. Survival signals and targets for therapy in breast implant-associated ALK-anaplastic large cell lymphoma. Clin. Cancer Res. 18, 4549–4559 (2012).
Oishi, N. et al. Genetic subtyping of breast implant-associated anaplastic large cell lymphoma. Blood 132, 544–547 (2018).
Blombery, P. et al. Frequent activating STAT3 mutations and novel recurrent genomic abnormalities detected in breast implant-associated anaplastic large cell lymphoma. Oncotarget 9, 36126–36136 (2018).












Leave a Reply