Lanier, L. L. Five decades of natural killer cell discovery. J. Exp. Med. 221, e20231222 (2024).
Narni-Mancinelli, E. & Vivier, E. Advancing natural killer therapies against cancer. Cell 185, 1451–1454 (2022).
Kyrysyuk, O. & Wucherpfennig, K. W. Designing cancer immunotherapies that engage T cells and NK cells. Annu. Rev. Immunol. 41, 17–38 (2023).
Sun, J. C. & Lanier, L. L. NK cell development, homeostasis and function: parallels with CD8+ T cells. Nat. Rev. Immunol. 11, 645–657 (2011).
Dean, I. et al. Rapid functional impairment of natural killer cells following tumor entry limits anti-tumor immunity. Nat. Commun. 15, 683 (2024).
Zhang, X. et al. IRF4 expression by NK precursors predetermines exhaustion of NK cells during tumor metastasis. Nat. Immunol. 26, 1062–1073 (2025).
Lupo, K. B. & Matosevic, S. CD155 immunoregulation as a target for natural killer cell immunotherapy in glioblastoma. J. Hematol. Oncol. 13, 76 (2020).
Sivori, S. et al. Human NK cells: surface receptors, inhibitory checkpoints, and translational applications. Cell. Mol. Immunol. 16, 430–441 (2019).
Wolf, N. K., Kissiov, D. U. & Raulet, D. H. Roles of natural killer cells in immunity to cancer, and applications to immunotherapy. Nat. Rev. Immunol. 23, 90–105 (2023).
Cao, Y. et al. Immune checkpoint molecules in natural killer cells as potential targets for cancer immunotherapy. Signal Transduct. Target. Ther. 5, 250 (2020).
Reis, E. S. C., Yamasaki, S. & Brown, G. D. Myeloid C-type lectin receptors in innate immune recognition. Immunity 57, 700–717 (2024).
Brown, G. D., Willment, J. A. & Whitehead, L. C-type lectins in immunity and homeostasis. Nat. Rev. Immunol. 18, 374–389 (2018).
Piersma, S. J. et al. Expression of a single inhibitory member of the Ly49 receptor family is sufficient to license NK cells for effector functions. Elife https://doi.org/10.7554/eLife.100218.3 (2025).
Babic, M. et al. NK cell receptor NKG2D enforces proinflammatory features and pathogenicity of Th1 and Th17 cells. J. Exp. Med. 217, e20190133 (2020).
Li, K. et al. The uric acid crystal receptor Clec12A potentiates type I interferon responses. Proc. Natl Acad. Sci. USA 116, 18544–18549 (2019).
Neumann, K. et al. Clec12a is an inhibitory receptor for uric acid crystals that regulates inflammation in response to cell death. Immunity 40, 389–399 (2014).
Hoffmann, S. C. et al. Identification of CLEC12B, an inhibitory receptor on myeloid cells. J. Biol. Chem. 282, 22370–22375 (2007).
Montaudie, H. et al. CLEC12B decreases melanoma proliferation by repressing signal transducer and activator of transcription 3. J. Invest. Dermatol. 142, 425–434 (2022).
Chi, D. et al. CLEC12B suppresses lung cancer progression by inducing SHP-1 expression and inactivating the PI3K/AKT signaling pathway. Exp. Cell. Res. 409, 112914 (2021).
Chen, C. H. et al. Dendritic-cell-associated C-type lectin 2 (DCAL-2) alters dendritic-cell maturation and cytokine production. Blood 107, 1459–1467 (2006).
Strack, R. Nanobodies made versatile. Nat. Methods 20, 37 (2023).
Ding, Z. et al. Nanobody-based trispecific T cell engager (Nb-TriTE) enhances therapeutic efficacy by overcoming tumor-mediated immunosuppression. J. Hematol. Oncol. 16, 115 (2023).
Zhang, F. et al. Structural basis of a novel PD-L1 nanobody for immune checkpoint blockade. Cell Discov. 3, 17004 (2017).
Bernard, N. J. CTLA-4 nanobody stops colitis. Nat. Immunol. 25, 190 (2024).
Blot, L. et al. CLEC12B regulates melanocyte immunity and homeostasis in the skin through the STAT1/IRF1 axis. J. Invest. Dermatol. 145, 2007–2020 (2025).
Tang, F. et al. A pan-cancer single-cell panorama of human natural killer cells. Cell 186, 4235–4251 (2023).
Ohs, I. et al. Restoration of natural killer cell antimetastatic activity by IL12 and checkpoint blockade. Cancer Res. 77, 7059–7071 (2017).
Liu, W. N. et al. Single-cell RNA sequencing reveals anti-tumor potency of CD56+ NK cells and CD8+ T cells in humanized mice via PD-1 and TIGIT co-targeting. Mol. Ther. 32, 3895–3914 (2024).
Hockemeyer, K. et al. The stress response regulator HSF1 modulates natural killer cell anti-tumour immunity. Nat. Cell Biol. 26, 1734–1744 (2024).
Goh, W. et al. IKAROS and AIOLOS directly regulate AP-1 transcriptional complexes and are essential for NK cell development. Nat. Immunol. 25, 240–255 (2024).
Duan, S. & Paulson, J. C. Siglecs as immune cell checkpoints in disease. Annu. Rev. Immunol. 38, 365–395 (2020).
Awwad, M. H. S. et al. Selective elimination of immunosuppressive T cells in patients with multiple myeloma. Leukemia 35, 2602–2615 (2021).
Zhang, Q. et al. Blockade of the checkpoint receptor TIGIT prevents NK cell exhaustion and elicits potent anti-tumor immunity. Nat. Immunol. 19, 723–732 (2018).
Zhang, H., Yang, L., Wang, T. & Li, Z. NK cell-based tumor immunotherapy. Bioact. Mater. 31, 63–86 (2024).
Chan, W. K. et al. Antibody-dependent cell-mediated cytotoxicity overcomes NK cell resistance in MLL-rearranged leukemia expressing inhibitory KIR ligands but not activating ligands. Clin. Cancer Res. 18, 6296–6305 (2012).
Kim, N., Yi, E., Lee, E., Park, H. J. & Kim, H. S. Interleukin-2 is required for NKp30-dependent NK cell cytotoxicity by preferentially regulating NKp30 expression. Front Immunol. 15, 1388018 (2024).
Deuse, T. et al. The SIRPα–CD47 immune checkpoint in NK cells. J. Exp. Med. 218, e20200839 (2021).
Porgador, A. Unveiling the relevance of immune checkpoints for innate and adaptive response to hepatocellular carcinoma using improved model of humanized mice. Mol. Ther. 32, 3761–3762 (2024).
Zhang, H. et al. Silencing of SIRPα enhances the antitumor efficacy of CAR-M in solid tumors. Cell. Mol. Immunol. 21, 1335–1349 (2024).
Ma, S. et al. Identification of a small-molecule Tim-3 inhibitor to potentiate T cell-mediated antitumor immunotherapy in preclinical mouse models. Sci. Transl. Med. 15, eadg6752 (2023).
Anania, C. Reprogramming-based gene therapy promotes anti-tumor immunity in vivo. Nat. Genet. 56, 1999 (2024).
Verhaar, E. R., Woodham, A. W. & Ploegh, H. L. Nanobodies in cancer. Semin. Immunol. 52, 101425 (2021).
Wu, S. Y., Wu, F. G. & Chen, X. Antibody-incorporated nanomedicines for cancer therapy. Adv. Mater. 34, e2109210 (2022).
Jiang, C. et al. Engineering a smart agent for enhanced immunotherapy effect by simultaneously blocking PD-L1 and CTLA-4. Adv. Sci. 8, e2102500 (2021).
Boukhaled, G. M. et al. Pre-encoded responsiveness to type I interferon in the peripheral immune system defines outcome of PD-1 blockade therapy. Nat. Immunol. 23, 1273–1283 (2022).
Chen, G. et al. Chemotherapy-induced neoantigen nanovaccines enhance checkpoint blockade cancer immunotherapy. ACS Nano 17, 18818–18831 (2023).
Tsao, L. C., Force, J. & Hartman, Z. C. Mechanisms of therapeutic antitumor monoclonal antibodies. Cancer Res. 81, 4641–4651 (2021).
Ma, L. et al. Multiregional single-cell dissection of tumor and immune cells reveals stable lock-and-key features in liver cancer. Nat. Commun. 13, 7533 (2022).
Ma, L. et al. Single-cell atlas of tumor cell evolution in response to therapy in hepatocellular carcinoma and intrahepatic cholangiocarcinoma. J. Hepatol. 75, 1397–1408 (2021).
Ma, L. et al. Tumor cell biodiversity drives microenvironmental reprogramming in liver cancer. Cancer Cell 36, 418–430 (2019).
Sharma, A. et al. Onco-fetal reprogramming of endothelial cells drives immunosuppressive macrophages in hepatocellular carcinoma. Cell 183, 377–394 (2020).
Losic, B. et al. Intratumoral heterogeneity and clonal evolution in liver cancer. Nat. Commun. 11, 291 (2020).
Zhu, G. Q. et al. CD36+ cancer-associated fibroblasts provide immunosuppressive microenvironment for hepatocellular carcinoma via secretion of macrophage migration inhibitory factor. Cell Discov. 9, 25 (2023).
Giraud, J. et al. THBS1(+) myeloid cells expand in SLD hepatocellular carcinoma and contribute to immunosuppression and unfavorable prognosis through TREM1. Cell Rep 43, 113773 (2024).
Liu, Z. et al. Viral-Track integrated single-cell RNA-sequencing reveals HBV lymphotropism and immunosuppressive microenvironment in HBV-associated hepatocellular carcinoma. Commun. Biol. 8, 1030 (2025).
Alvarez, M. et al. Human liver single nucleus and single cell RNA sequencing identify a hepatocellular carcinoma-associated cell-type affecting survival. Genome Med. 14, 50 (2022).
Darci-Maher, N. et al. Cross-tissue omics analysis discovers ten adipose genes encoding secreted proteins in obesity-related non-alcoholic fatty liver disease. EBioMedicine 92, 104620 (2023).
Lee, S. H. T. et al. Single nucleus RNA-sequencing integrated into risk variant colocalization discovers 17 cell-type-specific abdominal obesity genes for metabolic dysfunction-associated steatotic liver disease. EBioMedicine 106, 105232 (2024).
Netskar, H. et al. Pan-cancer profiling of tumor-infiltrating natural killer cells through transcriptional reference mapping. Nat. Immunol. 25, 1445–1459 (2024).
Bi, J. et al. Checkpoint TIPE2 limits the helper functions of NK cells in supporting antitumor CD8+ T cells. Adv. Sci. 10, e2207499 (2023).
Ma, H. et al. Hetero-bivalent nanobodies provide broad-spectrum protection against SARS-CoV-2 variants of concern including Omicron. Cell Res. 32, 831–842 (2022).
Yakkala, C. et al. Cryoablation does not significantly contribute to systemic effector immune responses in a poorly immunogenic B16F10 melanoma model. Clin. Cancer Res. 30, 4190–4200 (2024).
Wu, L. et al. Camouflaging attenuated Salmonella by cryo-shocked macrophages for tumor-targeted therapy. Signal Transduct. Target. Ther. 9, 14 (2024).
Zhao, L. et al. Antitumor efficacy and potential mechanism of FAP-targeted radioligand therapy combined with immune checkpoint blockade. Signal Transduct. Target. Ther. 9, 142 (2024).





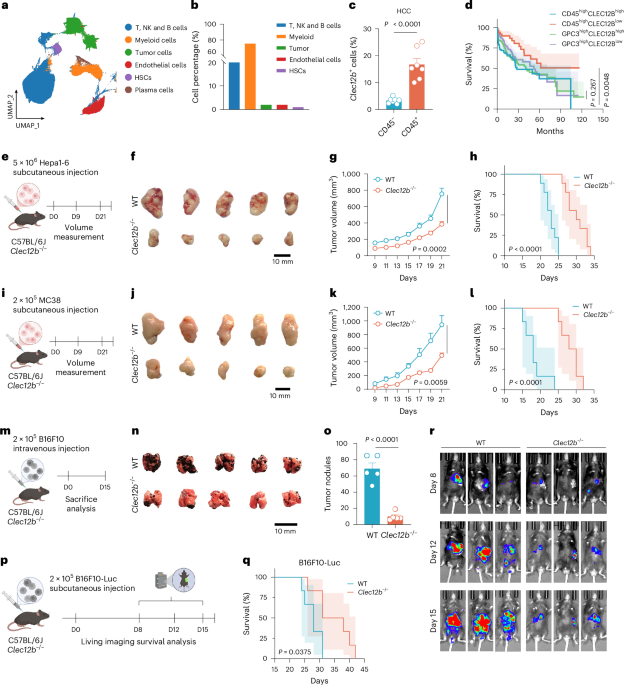









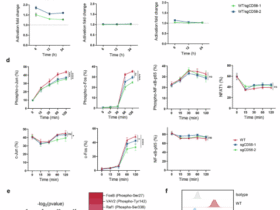
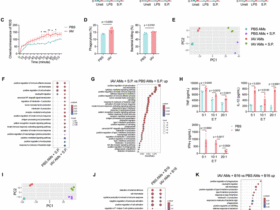
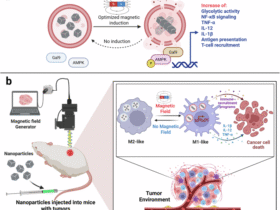
Leave a Reply