The majority of this study’s cohort were middle-aged (40–60 years old) with hypertension as the predominant cause of renal failure. This matches epidemiological trends, in which hypertension is the leading contributor to CKD9, and represents the most common phenotype of HD patients in Saudi Arabia2. Chronic hypertension causes long-term damage to renal arterioles and glomeruli. Aging, together with metabolic disorders, increases the impact of hypertension in CKD10. Meanwhile, diabetes frequently coexists with hypertension and accelerates nephron loss, representing a common issue in HD patients11.
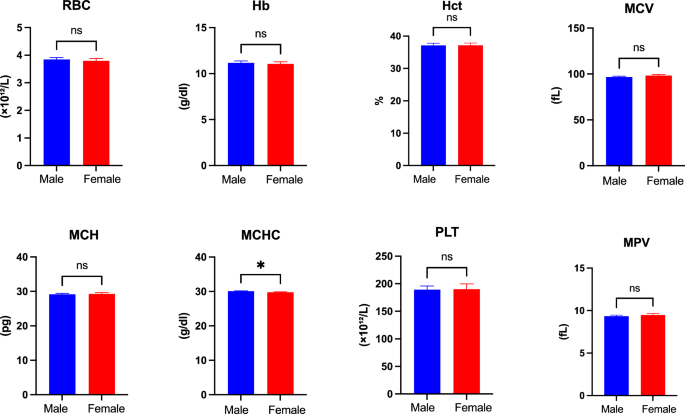
Notably, there is an apparent discrepancy between relative percentages and absolute counts of monocytes and lymphocytes observed in our study. This variation in the percentage from the absolute counts is based on the relative distribution of other leukocyte subtypes. The increase in monocyte and lymphocyte counts in female patients compared to males pre-HD suggests a sex difference in the composition of peripheral leukocytes caused by biological sex rather than HD. Certain lymphocyte subsets and monocytes are upregulated and influenced by X-linked immune genes and sex hormones (estrogens); for instance, females tend to have stronger adaptive immune responses, seen by higher CD4+ T cell counts and distinct B cell responses, which can be reflected in higher absolute lymphocyte and monocyte counts12,13,14,15. Other CBC studies have shown sex-based differences in leukocyte subsets (such as higher CD4+ and some B-cell populations in females), though absolute CBC differences vary by age and comorbidity16. Elevated lymphocyte and monocyte counts may influence inflammatory responses and infection risk in these patients, thus affecting the interpretation of immune-based prognostic markers. Some HD cohorts have reported sex differences in hospitalizations, with increased incidence in females compared to men17, while another study indicated a lower risk for mortality in females18. These contradictions should be further examined while taking into consideration differences in age and comorbidities, diet, infections, autoimmune disorders, and other confounding factors.
The post-HD reduction of WBCs observed in this cohort likely reflects the mechanical stress induced by HD. The lymphocyte reduction observed post-HD is consistent with findings from another study. It was associated with a reduction in the total CD3+ T cell and B lymphocytes, while the CD4+/CD8+ ratio increased. Additionally, the NK cells were decreased in both males and females post-HD. Thus, HD acutely affects both adaptive and innate immunity19. Immune suppression is a common consequence observed in HD patients that increases infection susceptibility and impairs vaccine responses in CKD. This immune dysregulation implicates both the uremic condition and the HD process. Uremia is characterized by the accumulation of toxins in the blood due to renal function decline. Uremia can cause systemic immune dysfunction, in turn increasing susceptibility to infections and cardiovascular disease. Immune dysregulation includes decreased T-cell proliferation, altered cytokine production, and impaired monocyte function. The accumulation of toxins and the inflammatory milieu linked to CKD are linked to these immune changes20,21. The HD process can further exacerbate this immune dysfunction. The blood-dialysis membrane interaction promotes the release of pro-inflammatory cytokines, such as IL-1, IL-6, and TNF-α, which activate mononuclear cells and the complement system. Furthermore, despite this initial inflammatory response, it has been shown that HD can cause an immune suppression, characterized by low T-cell responses and HLA-DR expression on monocytes22. Moreover, synthetic dialysis membranes, while biocompatible, can still trigger an immune response, including monocyte activation and cytokine release; this is considered an important factor in the chronic inflammatory condition of HD patients23. Enhanced membrane materials (such as vitamin E coatings, polymethyl methacrylate, and AN69) and improved HD techniques (such as cool dialysate, high-volume online hemodiafiltration, and hydrogen-enriched dialysate) have been proposed to mitigate these effects24. Therefore, the significant reduction in WBC count observed post-HD in our cohort is possibly multifactorial. One possible factor is cell sequestration and margination, which could result from complement activation triggered by the interaction between blood and the dialyzer membrane, promoting neutrophil adherence to the endothelium and sequestering in the pulmonary vasculature. As a result, temporary decrease in circulating leukocyte counts. Another related mechanism is the mechanical stress that can contribute to leukocyte activation and damage, stimulating premature apoptosis. Therefore, this acute reduction in the long term can result in chronic impairments in lymphocytes and antigen presentation that characterize immunosuppression. This immune dysregulation highlights the importance of continuous monitoring of immune function and necessitates the development of strategies to attenuate immune suppression in this susceptible population. Thus, strategies to mitigate these issues could include the use of more biocompatible dialysis membranes, optimized dialysate composition, and rigorous management of anemia with erythropoiesis-stimulating agents and iron supplementation, tailored to the patient’s sex and comorbidity profile.
Regardless of other comorbidities, older patients with lower WBC counts are more likely to see a rapid decline in kidney function25. This implies that low WBC counts could be an indicator of a malnutrition-inflammation complex, compromised immune surveillance, bone marrow dysfunction, or other pathological conditions that increase susceptibility to CKD progression25. A previous report has indicated that a reduction in WBC count post-HD—if maintained or moving toward the lower normal range—might imply a more favorable prognostic profile26. Together, these findings indicate the importance of careful monitoring and interpretation of WBC indices in the clinical setting. The dynamics behind WBC counts may be useful as potential prognostic markers and risk indicators for the progression of CKD, particularly in older HD patients.
Anemia is the most common comorbidity associated with chronic HD patients27. It typically results from chronic inflammation and decreased erythropoietin production by the kidneys28. In this study, the increase in anemia prevalence observed among male HD patients is exceptional. Especially with the slight increase in MCHC level in males compared to females, indicating an increase in Hb concentration in RBC. This difference between males and females may suggest several factors, such as higher pre-dialysis baseline hemoglobin levels in males, along with increased mechanical hemolysis due to higher blood flow rates, and variation in nutritional status or inflammation. Furthermore, anemia is significantly associated with the severity and duration of CKD29. Thus, further studies should confirm this result and investigate the underlying mechanism that influences gender-specific anemia development in HD patients.
The significant reduction in RBC, Hb, and other blood cell parameters observed post-HD in our study could result from two different causes; volume related effect or true hematological changes. The volume related effect can contribute to increased hemolysis, possibly through mechanical stress. Key dialysis parameters, including ultrafiltration volume, blood and dialysate flow rates, and dialyzer surface area, can impact hematological parameters and could influence the hemoglobin and hematocrit concentration post-HD. In this study, the use of a standardized protocol for Qb, Qd, anticoagulation, and the consistent use of Fresenius polysulfone dialyzers is expected to minimize the variability in these dialysis parameters. Therefore, the constant decrease in hemoglobin and hematocrit across all patients possibly reflects individual differences in ultrafiltration volume, which was not uniformly documented. Ultrafiltration-induced hemoconcentration is predicted to elevate cell counts; however, this effect may be mitigated by hemodilution resulting from saline infusions during intradialytic interventions. Therefore, post-HD reduction in Hb and Hct cannot be justified by ultrafiltration or hemodilution alone. The reduction in RBC parameters could reflect the outcome of true hematological changes, such as the mechanical hemolysis from the blood pump within the dialysis circuit, which can also damage RBC. As a result, the effects of these processes seem to outweigh the anticipated hemoconcentration, which explains the reduction in hemoglobin and hematocrit levels post-HD. This decrease in RBCs contributes to chronic anemia in HD patients30. Therefore, anemia management and transfusion risk should be carefully examined in clinical practice. Personalized approaches are required that ensure adequate iron availability and maintain Hb levels to reduce the need for transfusions and improve patient outcomes31.
The study also assessed the correlations between RBC parameters and the effects upon these relationships of a single HD session. Important indicators of red cell mass, including Hct, Hb, and RBC count, were found to positively correlate with one another. The consistent correlation between these measures before and after HD suggests that the HD session did not significantly alter the relationship between these parameters, confirming their validity and stability in determining red cell mass32. Furthermore, a positive correlation was observed between RDW measurements, RDW-CV and RDW-SD, pre-HD and post-HD. While RDW-SD gives a direct measurement of RBC volume, RDW-CV is affected by MCV32. The consistent positive correlation indicates that they both accurately reflect red cell size variability (anisocytosis), regardless of HD status. This confirms that RBC indices are reliable in determining red cell mass and anisocytosis in HD patients. The stability of these correlations both before and after HD increases the confidence in using these hematological indices for clinical assessment and monitoring in this patient population. Pre-HD analysis also revealed a strong positive correlation between counts of WBCs and their subtypes, including neutrophils, monocytes, and lymphocytes. This shows that these distinct immune cell populations consistently reflect changes in the total WBC count under baseline conditions33. However, with the exception of lymphocytes and monocytes, this correlation essentially vanished after a single HD session, indicating that neutrophil dynamics may be significantly affected by the mechanical stress brought on by HD or changes in plasma volume. Interestingly, the only WBC subtypes that showed a significant sex difference were monocytes and lymphocytes, which were found to be more prevalent in female patients pre-HD. Together, these findings demonstrate that HD can alter the relationship between total WBC and subtype counts; lymphocytes and monocytes are relatively more stable but influenced by sex, while neutrophil counts are more susceptible to changes brought on by HD.
The finding that the percentage change in WBC count was higher in patients with hypertension alone than in those with concurrent diabetes suggests that the latter condition may attenuate the hematological response to HD. Patients with hypertension alone may have a more reactive immune system, which could cause a distinct leukocyte shift during HD. In patients with coexisting diabetes, chronic hyperglycemia and metabolic dysfunction are known to impair leukocyte mobilization, proliferation, and function, impacting the observed WBC change. Leukocyte phenotypes (exhaustion/dysfunction) may be altered by chronic inflammation in diabetic patients, leading the inflammatory state to be underestimated by traditional CBC-based markers (total WBC, neutrophil rise)34. These findings suggest that hypertension alone is associated with a higher WBC response to HD, whereas diabetes reduces this effect. The possibility that this reflects immune dysfunction associated with diabetes underscores the need for more sensitive biomarkers than WBC count to measure inflammation in patients with multiple comorbidities.
Therefore, the results suggest that daily monitoring of hemodialysis patients should evolve from a standardized CBC approach to a stratified methodology that takes into account sex, age, and comorbidities. Early signs of anemia deteriorating or immune suppression in females or patients with diabetes, can be detected by monitoring WBC subtypes and RBC indices before and after dialysis. Thus, early intervention in the management of anemia, dialysis settings, and infection control can lower the risk of side effects during standard HD follow-up. Accordingly, these additional measures can enhance the translational significance of this study by linking the detected hematological changes to individualized clinical follow-up strategies in hemodialysis management.
This study has several limitations. First, the sample size, while sufficient for initial observations, is relatively modest for extensive subgroup analyses. Future multicenter studies with larger cohorts are warranted to confirm and generalize these findings. Secondly, our cohort consisted of prevalent, stable HD patients (on dialysis for > 3 months). Their hematological and immune parameters may have adapted over time, and the acute changes observed in a single session might differ from those in incident patients starting dialysis. Third, potential confounders such as variability in dialysis protocols, medications, and patient heterogeneity (e.g., nutritional status) were not fully adjusted for. Additionally, a standardized dialysis protocol was used for all patients involved in this study; however, the ultrafiltration volume was not systematically recorded, therefore, the study of its potential effect on hematological parameters post-HD was not fully assessed. Finally, the anticoagulant effect may persist due to some residual, which could have influenced the immune parameters measurements, and future studies should aim to minimize these effects.












Leave a Reply