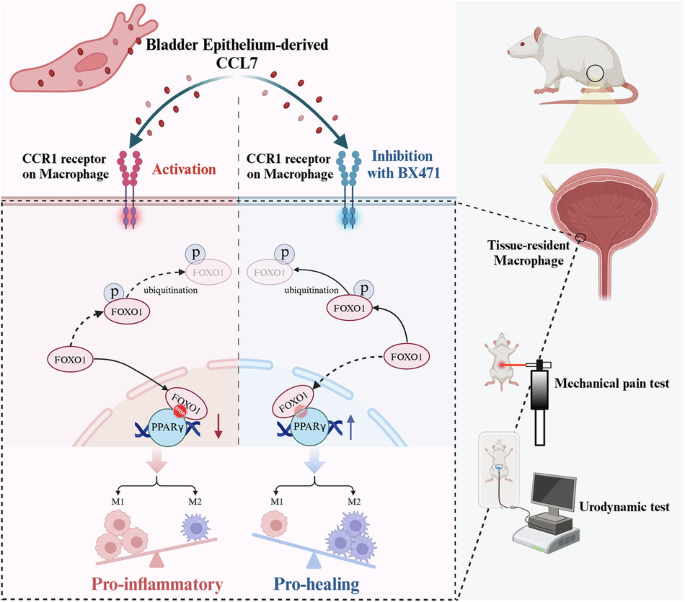
Interstitial cystitis/bladder pain syndrome (IC/BPS) is a chronic inflammatory condition with limited treatments. Although macrophages are implicated in its pathogenesis, the mechanisms driving their phenotypic switching remain unclear. This study identifies CCR1’s role in IC/BPS and evaluates CCR1 inhibition as a therapeutic strategy. Integrated bulk and single-cell RNA sequencing reveal enrichment of pro-inflammatory CCR1⁺ macrophages in bladder tissue from patients with IC/BPS. In a lipopolysaccharide-induced rat model, pharmacological inhibition of CCR1 suppresses M1 polarization, promotes M2 polarization, and improves pain thresholds, urinary symptoms as well as bladder inflammation. Mechanistically, CCR1 knockdown enhances FOXO1 phosphorylation and degradation, reduces its nuclear translocation, and activates PPARγ signaling to promote M2 polarization. Analysis of clinical samples shows increased CCL7 levels in bladder tissue and urine, with urinary levels correlating with symptom severity. These findings identify CCR1 as a candidate target for further therapeutic evaluation in IC/BPS.














Leave a Reply