We start with a case report, because it illustrates well the current challenges and possibilities in m-ccRCC patients with BrM.
Case report: impact of cabozantinib on brain metastases and oedema
A 61-year old male patient (patient 15 in the tables) underwent a nephrectomy for a pT1bN0M0 Fuhrman Grade 1 ccRCC. Twenty months later, brain, liver, lung, adrenal, peritoneal and paravertebral metastases were diagnosed. Cabozantinib (40 mg/day) and nivolumab (240 mg Q2W) were started since cabozantinib is very active intracranially and has antiphlogistic activity. We aimed to avoid the use of corticosteroids for control of the perilesional oedema, because of the specific adverse events of corticosteroids and the potential negative impact on nivolumab efficacy. We also aimed to avoid RT to avoid radionecrosis with oedema necessitating even more and longer exposure to corticosteroids. The BrM were asymptomatic.
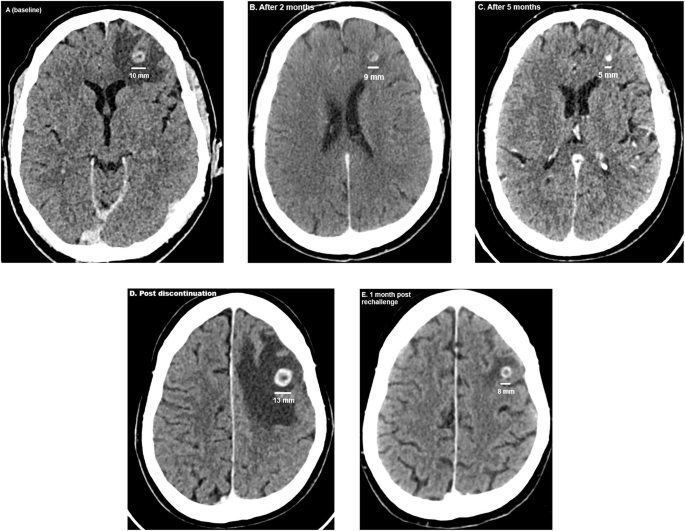
At baseline, the two BrM had a diameter of 10 and 5 millimeters (mm) surrounded by oedema (Fig. 1A). After two months of cabozantinib/nivolumab, on CT-scanner the largest BrM measured 9 mm and the smallest BrM was barely detectable (Fig. 1B). After 5 months, the largest BrM had shrunk to 5 mm and the smaller BrM was no longer visible, with significantly diminished oedema (Fig. 1C). The peritoneal implants showed a decrease in volume, while extracranial disease in other locations remained unchanged. After 8 months, the diameter of the largest BrM was 4 mm. Progression at the level of fifth dorsal vertebra led to embolization and tumor debulking with hemilaminectomy and pediculectomy. Cabozantinib was interrupted peri-operatively. Due to postoperative complications adjuvant RT on the fifth dorsal vertebra was postponed and cabozantinib discontinued longer. Two months later, the patient presented with speech disorders, dysarthria and word-finding problems. On CT scan, the largest BrM had increased to 13 mm and multiple new BrM had emerged (Fig. 1D). Cabozantinib was restarted at 40 mg/day. After 1 month of cabozantinib, the largest lesion had decreased to 8 mm (Fig. 1E). After 4 months of cabozantinib, this BrM measured 5 mm and the other BrMs as well as the oedema had resolved. At the thoracoabdominal level, the disease remained stable.
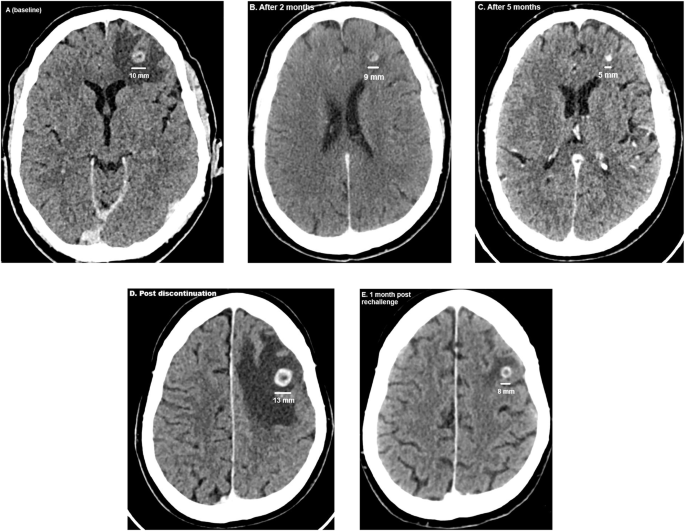
A–E Evolution of the largest brain metastasis. A At start of cabozantinib. B and C Response on cabozantinib. D: Increase of the diameter after two months of cabozantinib discontinuation. E Response after rechallenge of cabozantinib.
Two months later, fulminant thoraco-abdominal disease progression led to a switch to axitinib. Four months later, disease progression was observed with concomitant acute renal insufficiency. Together with the patient, we opted for a best supportive care. Soon after, the patient died.
This case demonstrates a clear, dose-dependent benefit of cabozantinib, both on the size of the BrM as on oedema, without use of corticosteroids or RT. The impact of nivolumab on the BrM seems of less importance, given the fact that the size of the BrM evolved depending on the dose of cabozantinib, while nivolumab administration was continued at the same dose since the start till the final progression on cabozantinib/nivolumab.
Included patients
According to the inclusion criteria, 30 patients were included in this study (patients characteristics in Table 1). In 6 patients, the intracranial efficacy of 2 sequential therapies could be studied and in one patient, the intracranial efficacy of 4 sequential therapies could be studied. Hence, a total of 39 observations were possible: 7 upon nivolumab, 1 upon ipilimumab/nivolumab, 4 upon sunitinib, 2 upon pazopanib, 3 upon axitinib/pembrolizumab, 7 upon axitinib, 4 upon cabozantinib/nivolumab and 11 upon cabozantinib. Systemic therapies were used in the approved setting: ipilimumab/nivolumab, axitinib/pembrolizumab, cabozantinib/nivolumab, sunitinib and pazopanib in first-line and axitinib, nivolumab and cabozantinib in monotherapy in second- or further line. The only exception was patient 2 treated with nivolumab in first-line.
Baseline characteristics at start of the systemic therapy in these 39 cases are reported in Table 2. A single BrM was present in 10 patients, 12 patients had two BrM and 17 three or more BrM.
Starting dose of the immunotherapy was the standard approved dose. Starting VEGFR-TKI dose was the standard approved dose in 23 cases and a lower dose in 8 cases: two patients started with axitinib at 2 * 3 mg/day (together with pembrolizumab), 5 patients with cabozantinib in monotherapy at 40 mg/day and one patient with cabozantinib in monotherapy at 20 mg/day. Hence, in cabozantinib-treated patients, the starting dose was more often inferior to standard of care compared to patients treated with other VEGFR-TKIs.
We excluded 31 cases from the study (Flowchart in Supplemental Figure. 1). Although these patients also had BrM at start of systemic therapy, they were not included for the following reasons. In 26 patients, radiation therapy was performed within one month before start of systemic therapy. In 4 patients, BrM were still decreasing after previous radiation therapy performed more than one month before the start of systemic therapy. Finally, one patient died before the second CT scan of the brain could take place in order to check intracranial response. These data show that there has been an evolution over the last years. As older therapies (sunitinib, axitinib, nivolumab and pazopanib), were considered to be less efficient on BrM, patients underwent more often radiation therapy before the start of systemic therapy. Nowadays, with the higher intracranial efficacy of cabozantinib, patients do not undergo upfront radiation therapy anymore.
Intracranial responses following systemic therapy
Intracranial response rate (RR) is reported for each systemic treatment in Table 3. No partial responses (PR) were seen in patients treated with ipilimumab/nivolumab (0%) and pazopanib (0%). PR were rare with nivolumab (14%), sunitinib (25%), axitinib (28%) or axitinib/pembrolizumab (33%), but higher upon cabozantinib/nivolumab (50%) or cabozantinib as monotherapy (64%). Two patients, treated with cabozantinib or cabozantinib/nivolumab, reached a complete intracranial response. The waterfall plot (Fig. 2A) depicts best response in each case. The swimmers plot in Fig. 2B depicts the duration on systemic therapy in each case.
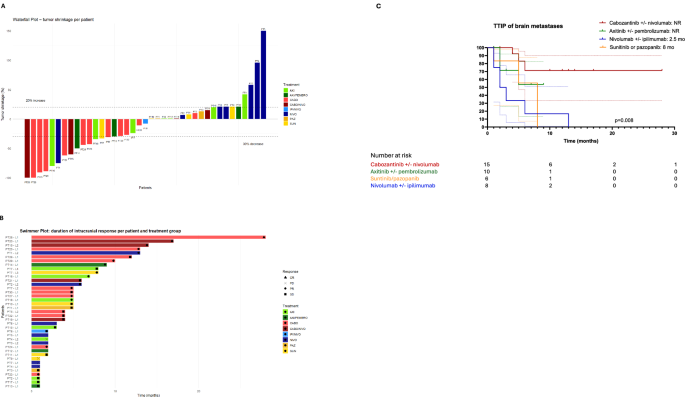
A Waterfall plot with intracranial best response depending on type of systemic therapy. B Swimmers plot of intracranial disease control in each patient. C Kaplan-Meier estimates of time-to-intracranial progression (TTIP) of brain metastases upon different systemic therapies.
We performed a Kaplan-Meier estimate of TTIP of the BrM depending on type of systemic therapy (Fig. 2C). Poorest intracranial disease control duration was observed with ICPs (2.5 months), followed by sunitinib and pazopanib (8 months) and axitinib with or without pembrolizumab (not reached). Median intracranial disease control was the longest (not reached) in patients treated with cabozantinib with or without nivolumab (p = 0.008).
In preparation of a multivariate analysis, we checked the impact of possible confounders on univariate analysis (Supplemental Table 1). Given the reduced number of events and given the reduced number of factors associated with TTIP on univariate analysis, we performed in parallel two bivariate analysis. Patients were divided in two groups: cases treated with a regimen including cabozantinib (n = 15) and cases treated with other therapies (n = 24). Supplemental Figure. 2 shows that in both bivariate analysis (including corticosteroid use or previous radiation therapy), cabozantinib remained as independently associated with TTIP. Note that previous radiation therapy is correlated with poorer TTIP. This is due to the fact that with the availability of cabozantinib, we are less prone to irradiate BrM. BrM were more often irradiated in the era where less efficient systemic therapies were used. Note also that intracranial response on cabozantinib was not driven by more favorable patient characteristics such as reduced number of BrM, lower IMDC risk or neurologic symptoms.
In four patients (patients 15, 20, 21 and 28), we noticed a cabozantinib dose-dependent response: upon cabozantinib interruption or dose reduction, progression was noticed, while upon dose re-escalation or rechallenge, intracranial disease control was again observed. Patient 15 is reported extensively above. Patient 21 was treated with cabozantinib 40 mg/day plus nivolumab. He required a cabozantinib dose reduction to 20 mg/day due to toxic hepatitis. The clinical consequence of this reduction was progressive BrM. Good disease control was regained after the dose was re-escalated to 30 mg/day. Patient 20, treated with cabozantinib 40 mg/day, had a dose reduction to 20 mg/day after two months of therapy due to skin blisters. This led to the emergence of new BrM. Disease regression of the BrM was subsequently achieved after re-escalating cabozantinib to 40 mg/day. Patient 28, treated with cabozantinib in monotherapy, discontinued cabozantinib for two months due to toxic hepatitis, resulting in progressive BrM. While control was initially regained upon restarting at 20 mg/day, progression recurred eight months later. After a dose increase to 30 mg/day, the BrM were again under control.
We checked whether the poor response of BrM on ICPs could be attributed to pseudoprogression. Of the 5 patients whose BrM displayed progressive disease (PD) as best response, four received whole brain radiotherapy after the first CT scan showed PD. Hence, pseudoprogression could not be assessed. The other patient underwent a subsequent CT scan evaluation, that confirmed PD, and hence, excluded pseudoprogression.
Finally, we also studied intracranial response with RANO-criteria, taking in consideration, on top of RECIST, corticosteroid tapering and clinical improvement. Results are shown in Table 3. Briefly, the number of PR were identical between RECIST and RANO, because in all RECIST PR patients, there was no increase of corticosteroid use nor clinical degradation. All patients with RECIST PD as best response had also RANO PD, by definition. Only 4 out of 17 patients with RECIST SD switched to RANO PD because of an increase of corticosteroid use or clinical degradation.
Analyses of the molecular targets of systemic therapies
A total of 716 samples were included: 9 BrM (among them 2 pre-treated, one with ipilimumab/nivolumab and one with pazopanib followed by nivolumab), 345 non-BrM, 12 normal kidney samples and 350 primary kidney tumors. For the paired samples analysis, between BrM and primary kidney tumor, 6 patients were available (all not-pretreated samples).
Median cMET-expression was significantly higher in BrM, compared to non-BrM, normal kidney samples and primary kidney tumors (p = 0.0002). Among all metastatic sites, cMET-expression was the highest in BrM. In a paired approach, cMET-expression was higher in BrM compared to primary tumor. Median AXL-expression was significantly higher in BrM, compared to non-BrM, normal kidney samples and primary kidney tumors (p = 0.003). Among all metastatic sites, AXL-expression was the highest in BrM. In a paired approach, AXL-expression was higher in BrM compared to primary tumor. The high cMET– and AXL-expression in BrM may explain high intracranial cabozantinib efficacy (Table 4, Fig. 3, Supplemental Figure. 3 and Supplemental Table 2).
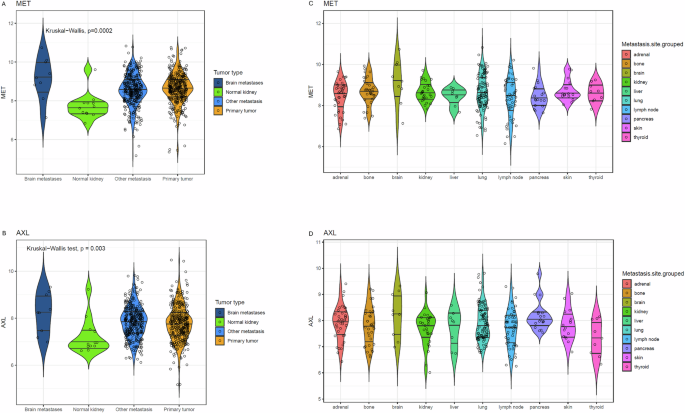
A, B cMET- and AXL-expression in brain metastases compared to non-brain metastases, primary kidney tumor and normal kidney tissue. C, D cMET- and AXL- expression in distinct metastatic sites.
Median VEGFR2-expression (p = 0.012), the IMmotion-angio score (p < 0.001) and the JAVELIN101-angio score (p = 0.052) were lower in BrM compared to metastases at other sites, primary tumor and normal kidney tumor. In a paired analysis, VEGFR1-, VEGFR2– and VEGFR3-expression as well as the IMmotion-angio and JAVELIN101-angio score were lower in BrM compared to primary tumor. The observed lower angiogenesis expression in BrM could explain the reduced intracranial activity of VEGFR-TKIs, cabozantinib being an exception, because it also targets cMET and AXL (Table 4, Fig. 4 and Supplemental Figure. 3).
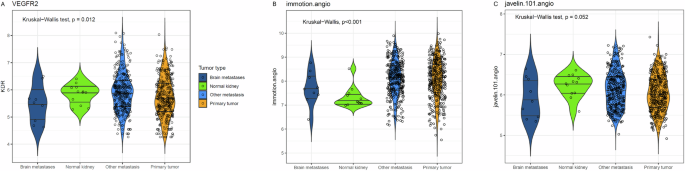
A VEGFR2-expression in brain metastases, normal kidney tissue, non-brain metastases and primary kidney tumor. B Score of the IMmotion-angio score in brain metastases, normal kidney tissue, non-brain metastases and primary kidney tumor. C Score of the JAVELIN101-angio score in brain metastases, normal kidney tissue, non-brain metastases and primary kidney tumor.
Median PDL1-, PD1– and CTLA4-expression were not significantly different in BrM compared to other metastatic sites, primary tumor and normal kidney samples. In a paired analysis, PDL1-expression was lower in BrM compared to primary tumor. The JAVELIN101-immune (p = 0.004) and IMmotion-T-effector score (p = 0.002) were lower in BrM compared to primary tumor and other metastatic sites, while the IMmotion-myeloid score was not different. In a paired analysis, the IMmotion-T-effector, JAVELIN101-immune and IMmotion-myeloid score were lower in BrM compared to primary tumor. On immune cell type deconvolution by CIBERSORTX, in BrM compared to primary tumor, other metastatic sites and normal kidney tissue, we observed less CD8 + T-cells (p = 0.02) and less activated Natural Killer (NK)-cells (p = 0.02), but more monocytes (p = 0.03) and M2-like (anti-inflammatory) macrophages (<0.001). The increased presence of M2-like macrophages was confirmed in a paired analysis between BrM and primary kidney tumor (p = 0.03). In summary, lower JAVELIN101-immune and IMmotion-T-effector scores and less CD8 + T-cells paralleled by more M2-like macrophages could explain possible resistance to ICPs (Table 4, Fig. 5, Supplemental Figure. 3 and Supplemental Table 3) [8,9,10].
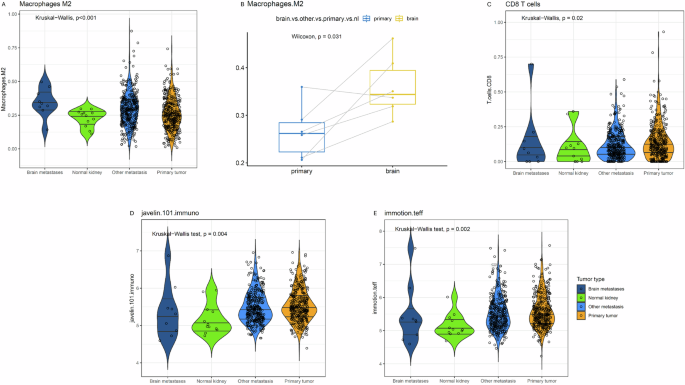
A Presence of M2-like (anti-inflammatory) macrophages in brain metastases, normal kidney tissue, non-brain metastases and primary kidney tumor. B Presence of M2-like macrophages in primary kidney tumor and brain metastases in a paired approach. C Presence of CD8 + T-cells in brain metastases, normal kidney tissue, non-brain metastases and primary kidney tumor. D, E JAVELIN101-immune score and IMmotion T-effector score in brain metastases, normal kidney tissue, non-brain metastases and primary kidney tumor.
Finally, we checked also FGFR1-, FGFR2-, and FGFR3-expression in BrM, other metastatic sites, primary tumor and normal kidney samples (Supplemental Figure. 3 and Table 4). This could be useful when treating patients with VEGFR-TKIs than also target FGFR1-3 such as lenvatinib. FGFR1-expression was similar at all sites. FGFR2-expression was higher at BrM compared to other metastatic sites, while FGFR3-expression was lower compared to other metastatic sites. In our clinical database, we do not have patients with BrM treated with lenvatinib.
GSEA did not show significantly enriched pathways between BrM and metastases at other sites (Supplemental Figure. 4).
















Leave a Reply