Parisi R, Symmons DPM, Griffiths CEM, Ashcroft DM. Global epidemiology of psoriasis: a systematic review of incidence and prevalence. J Investig Dermatol. 2013;133:377–85. https://doi.org/10.1038/jid.2012.339.
Parisi R, Iskandar IYK, Kontopantelis E, Augustin M, Griffiths CEM, Ashcroft DM. National, regional, and worldwide epidemiology of psoriasis: systematic analysis and modelling study. BMJ. 2020 28;m1590. https://doi.org/10.1136/bmj.m1590
Kelly-Sell M, Gudjonsson JE. Overview of psoriasis. In: Therapy for severe psoriasis. Elsevier; 2016. p. 1–15. https://doi.org/10.1016/B978-0-323-44797-3.00001-3
Gerdes S, Mrowietz U, Boehncke WH. Comorbidity in psoriasis. Hautarzt. 2016;67:438–44. https://doi.org/10.1007/s00105-016-3805-3.
Sommer DM, Jenisch S, Suchan M, Christophers E, Weichenthal M. Increased prevalence of the metabolic syndrome in patients with moderate to severe psoriasis. Arch Dermatol Res. 2006;298:321–8. https://doi.org/10.1007/s00403-006-0703-z.
Ramessur R, Saklatvala J, Budu-Aggrey A, Ostaszewski M, Möbus L, Greco D, et al. Exploring the link between genetic predictors of cardiovascular disease and psoriasis. JAMA Cardiol. 2024;9:1009 https://doi.org/10.1001/jamacardio.2024.2859.
Lynch M, Ahern T, Sweeney CM, Malara A, Tobin AM, O’Shea D, et al. Adipokines, psoriasis, systemic inflammation, and endothelial dysfunction. Int J Dermatol. 2017;56:1103–18. https://doi.org/10.1111/ijd.13699.
Samotij D, Nedoszytko B, Bartosińska J, Batycka-Baran A, Czajkowski R, Dobrucki I, et al. Pathogenesis of psoriasis in the “omic” era. Part I. Epidemiology, clinical manifestation, immunological and neuroendocrine disturbances. Adv Dermatol Allergol. 2020;37:135–53. https://doi.org/10.5114/ada.2020.94832.
Kim YC, Baek YS. Psoriasis and genetics. J Korean Soc Psoriasis. 2022;19:35–9.
Grän F, Kerstan A, Serfling E, Goebeler M, Muhammad K. Current developments in the immunology of psoriasis. Yale J Biol Med. 2020;93:97–110.
Zhu B, Jing M, Yu Q, Ge X, Yuan F, Shi L. Treatments in psoriasis: from standard pharmacotherapy to nanotechnology therapy. Adv Dermatol Allergol. 2022;39:460–71. https://doi.org/10.5114/ada.2021.108445.
Reid C, Griffiths C. Psoriasis and treatment: past, present and future aspects. Acta Derm Venereol. 2020;100:70–80. https://doi.org/10.2340/00015555-3386.
Singh A, Easwari TS. Recent advances in psoriasis therapy: trends and future prospects. Curr Drug Targets. 2021;22:1760–71. https://doi.org/10.2174/1389450122666210118103455.
Armstrong AW, Read C. Pathophysiology, clinical presentation, and treatment of psoriasis: a review. JAMA. 2020;323:1945 https://doi.org/10.1001/jama.2020.4006.
Kaushik SB, Lebwohl MG. Psoriasis: Which therapy for which patient. J Am Acad Dermatol. 2019;80:27–40. https://doi.org/10.1016/j.jaad.2018.06.057.
Gyldenløve M, Alinaghi F, Zachariae C, Skov L, Egeberg A. Combination therapy with apremilast and biologics for psoriasis: a systematic review. Am J Clin Dermatol. 2022;23:605–13. https://doi.org/10.1007/s40257-022-00703-1.
Hsieh TS, Tsai TF. Combination therapy for psoriasis with methotrexate and other oral disease-modifying antirheumatic drugs: a systematic review. Dermatol Ther. 2023;13:891–909. https://doi.org/10.1007/s13555-023-00903-5.
Pourani MR, Abdollahimajd F, Zargari O, Shahidi Dadras M. Soluble biomarkers for diagnosis, monitoring, and therapeutic response assessment in psoriasis. J Dermatol Treat. 2022;33:1967–74. https://doi.org/10.1080/09546634.2021.1966357.
Lwin SM, Snowden JA, Griffiths CEM. The promise and challenges of cell therapy for psoriasis. Br J Dermatol. 2021;185:887–98. https://doi.org/10.1111/bjd.20517.
Lønnberg AS, Skov L, Skytthe A, Kyvik KO, Pedersen OB, Thomsen SF. Heritability of psoriasis in a large twin sample. Br J Dermatol. 2013;169:412–6. https://doi.org/10.1111/bjd.12375.
Griffiths CE, Barker JN. Pathogenesis and clinical features of psoriasis. Lancet. 2007;370:263–71. https://doi.org/10.1016/S0140-6736(07)61128-3.
Enlund F, Samuelsson L, Enerbäck C, Inerot A, Wahlström J, Yhr M, et al. Analysis of three suggested psoriasis susceptibility loci in a large Swedish set of families: confirmation of linkage to chromosome 6p (HLA region), and to 17q, but not to 4q. Hum Hered. 1999;49:2–8. https://doi.org/10.1159/000022832.
Allen MH, Ameen H, Veal C, Evans J, Ramrakha-Jones VS, Marsland AM, et al. The major psoriasis susceptibility locus PSORS1 is not a risk factor for late-onset psoriasis. J Investig Dermatol. 2005;124:103–6. https://doi.org/10.1111/j.0022-202X.2004.23511.x.
Nair RP, Ding J, Duffin KC, Helms C, Voorhees JJ, Krueger GG, et al. Psoriasis bench to bedside: genetics meets immunology. Arch Dermatol. 2009;145:462–4. https://doi.org/10.1001/archdermatol.2009.73.
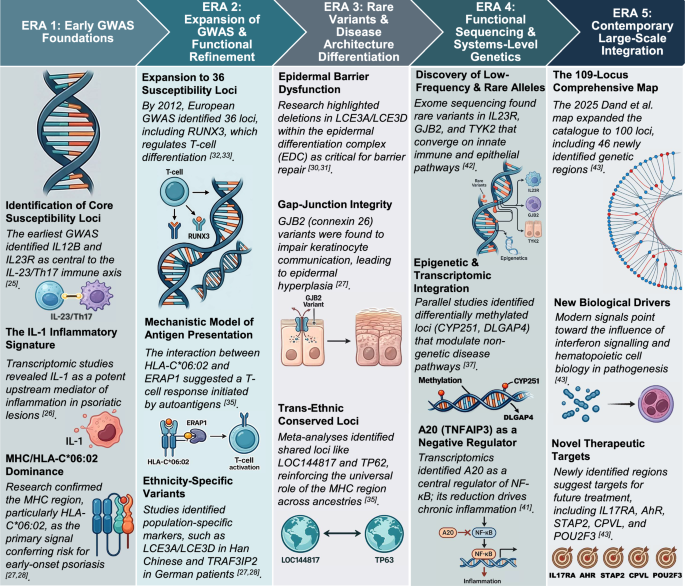
Cargill M, Schrodi SJ, Chang M, Garcia VE, Brandon R, Callis KP, et al. A large-scale genetic association study confirms IL12B and leads to the identification of IL23R as psoriasis-risk genes. Am J Hum Genet. 2007;80:273–90. https://doi.org/10.1086/511051.
Mee JB, Johnson CM, Morar N, Burslem F, Groves RW. The psoriatic transcriptome closely resembles that induced by interleukin-1 in cultured keratinocytes: dominance of innate immune responses in psoriasis. Am J Pathol. 2007;171:32–42. https://doi.org/10.2353/ajpath.2007.061067.
Sun LD, Cheng H, Wang ZX, Zhang AP, Wang PG, Xu JH, et al. Association analyses identify six new psoriasis susceptibility loci in the Chinese population. Nat Genet. 2010;42:1005–9. https://doi.org/10.1038/ng.690.
Genetic Analysis of Psoriasis Consortium & the Wellcome Trust Case Control Consortium 2, Strange A, Capon F, Spencer CCA, Knight J, Weale ME, et al. A genome-wide association study identifies new psoriasis susceptibility loci and an interaction between HLA-C and ERAP1. Nat Genet. 2010;42:985–90. https://doi.org/10.1038/ng.694.
Ran D, Cai M, Zhang X. Genetics of psoriasis: a basis for precision medicine. Precis Clin Med. 2019;2:120–30. https://doi.org/10.1093/pcmedi/pbz011.
Mischke D, Korge BP, Marenholz I, Volz A, Ziegler A. Genes encoding structural proteins of epidermal cornification and S100 calcium-binding proteins form a gene complex (‘epidermal differentiation complex’) on human chromosome 1q21. J Investig Dermatol. 1996;106:989–92. https://doi.org/10.1111/1523-1747.ep12338501.
de Cid R, Riveira-Munoz E, Zeeuwen PLJM, Robarge J, Liao W, Dannhauser EN, et al. Deletion of the late cornified envelope LCE3B and LCE3C genes as a susceptibility factor for psoriasis. Nat Genet. 2009;41:211–5. https://doi.org/10.1038/ng.313.
Zamisch M, Tian L, Grenningloh R, Xiong Y, Wildt KF, Ehlers M, et al. The transcription factor Ets1 is important for CD4 repression and Runx3 up-regulation during CD8 T cell differentiation in the thymus. J Exp Med. 2009;206:2685–99. https://doi.org/10.1084/jem.20092024.
Tsoi LC, Spain SL, Knight J, Ellinghaus E, Stuart PE, Capon F, et al. Identification of 15 new psoriasis susceptibility loci highlights the role of innate immunity. Nat Genet. 2012;44:1341–8. https://doi.org/10.1038/ng.2467.
Tang H, Jin X, Li Y, Jiang H, Tang X, Yang X, et al. A large-scale screen for coding variants predisposing to psoriasis. Nat Genet. 2014;46:45–50. https://doi.org/10.1038/ng.2827.
Yin X, Low HQ, Wang L, Li Y, Ellinghaus E, Han J, et al. Genome-wide meta-analysis identifies multiple novel associations and ethnic heterogeneity of psoriasis susceptibility. Nat Commun. 2015;6:6916. https://doi.org/10.1038/ncomms7916.
Nishikawa R, Nagai H, Bito T, Ikeda T, Horikawa T, Adachi A, et al. Genetic prediction of the effectiveness of biologics for psoriasis treatment. J Dermatol. 2016;43:1273–7. https://doi.org/10.1111/1346-8138.13412.
Zhou F, Wang W, Shen C, Li H, Zuo X, Zheng X, et al. Epigenome-wide association analysis identified nine skin DNA methylation loci for psoriasis. J Investig Dermatol. 2016;136:779–87. https://doi.org/10.1016/j.jid.2015.12.029.
Swindell WR, Sarkar MK, Liang Y, Xing X, Gudjonsson JE. Cross-disease transcriptomics: unique IL-17A signaling in psoriasis lesions and an autoimmune PBMC signature. J Investig Dermatol. 2016;136:1820–30. https://doi.org/10.1016/j.jid.2016.04.035.
Ahn R, Yan D, Chang HW, Lee K, Bhattarai S, Huang ZM, et al. RNA-seq and flow-cytometry of conventional, scalp, and palmoplantar psoriasis reveal shared and distinct molecular pathways. Sci Rep. 2018;8:11368. https://doi.org/10.1038/s41598-018-29472-w.
Nomura I, Gao B, Boguniewicz M, Darst MA, Travers JB, Leung DYM. Distinct patterns of gene expression in the skin lesions of atopic dermatitis and psoriasis: a gene microarray analysis. J Allergy Clin Immunol. 2003;112:1195–202. https://doi.org/10.1016/j.jaci.2003.08.049.
Devos M, Mogilenko DA, Fleury S, Gilbert B, Becquart C, Quemener S, et al. Keratinocyte expression of A20/TNFAIP3 controls skin inflammation associated with atopic dermatitis and psoriasis. J Investig Dermatol. 2019;139:135–45. https://doi.org/10.1016/j.jid.2018.06.191.
Dand N, Mucha S, Tsoi LC, Mahil SK, Stuart PE, Arnold A, et al. Exome-wide association study reveals novel psoriasis susceptibility locus at TNFSF15 and rare protective alleles in genes contributing to type I IFN signalling. Hum Mol Genet. 2017;26:4301–13. https://doi.org/10.1093/hmg/ddx328.
Dand N, Stuart PE, Bowes J, Ellinghaus D, Nititham J, Saklatvala JR, et al. GWAS meta-analysis of psoriasis identifies new susceptibility alleles impacting disease mechanisms and therapeutic targets. Nat Commun. 2025;16:2051. https://doi.org/10.1038/s41467-025-56719-8.
Zheng XF, Sun YD, Liu XY. Correlation of expression of STAT3, VEGF and differentiation of Th17 cells in psoriasis vulgaris of guinea pig. Asian Pac J Trop Med. 2014;7:412–20. https://doi.org/10.1016/S1995-7645(14)60046-8.
Elder JT, Bruce AT, Gudjonsson JE, Johnston A, Stuart PE, Tejasvi T, et al. Molecular dissection of psoriasis: integrating genetics and biology. J Investig Dermatol. 2010;130:1213–26. https://doi.org/10.1038/jid.2009.319.
Ellinghaus E, Ellinghaus D, Stuart PE, Nair RP, Debrus S, Raelson JV, et al. Genome-wide association study identifies a psoriasis susceptibility locus at TRAF3IP2. Nat Genet. 2010;42:991–5. https://doi.org/10.1038/ng.689.
Gong H, Gao S, Pu X, Kang X, Wu X. Association of rs610604 in TNFAIP3 and rs17728338 in TNIP1 gene polymorphisms with psoriasis susceptibility: a meta-analysis of case-control studies. BMC Med Genet. 2020;21:103. https://doi.org/10.1186/s12881-020-01041-x.
Jordan CT, Cao L, Roberson EDO, Pierson KC, Yang CF, Joyce CE, et al. PSORS2 is due to mutations in CARD14. Am J Hum Genet. 2012;90:784–95. https://doi.org/10.1016/j.ajhg.2012.03.012.
Zuo X, Sun L, Yin X, Gao J, Sheng Y, Xu J, et al. Whole-exome SNP array identifies 15 new susceptibility loci for psoriasis. Nat Commun. 2015;6:6793. https://doi.org/10.1038/ncomms7793.
Fu B, Li S, Wang L, Berman MA, Dorf ME. The ubiquitin conjugating enzyme UBE2L3 regulates TNFα-induced linear ubiquitination. Cell Res. 2014;24:376–9. https://doi.org/10.1038/cr.2013.133.
De Molfetta GA, Lucíola Zanette D, Alexandre Panepucci R, dos Santos ARD, da Silva WA, Antonio Zago M. Role of NFKB2 on the early myeloid differentiation of CD34+ hematopoietic stem/progenitor cells. Differentiation. 2010;80:195–203. https://doi.org/10.1016/j.diff.2010.07.004.
Lu T, Jackson MW, Wang B, Yang M, Chance MR, Miyagi M, et al. Regulation of NF-κB by NSD1/FBXL11-dependent reversible lysine methylation of p65. Proc Natl Acad Sci USA. 2010;107:46–51. https://doi.org/10.1073/pnas.0912493107.
Capon F. The genetic basis of psoriasis. Int J Mol Sci. 2017;25:18. https://doi.org/10.3390/ijms18122526.
Imada K, Leonard WJ. The Jak-STAT pathway. Mol Immunol. 2000;37:1–11. https://doi.org/10.1016/s0161-5890(00)00018-3.
Morelli M, Madonna S, Albanesi C. SOCS1 and SOCS3 as key checkpoint molecules in the immune responses associated to skin inflammation and malignant transformation. Front Immunol. 2024;15:1393799. https://doi.org/10.3389/fimmu.2024.1393799.
Madonna S, Scarponi C, Doti N, Carbone T, Cavani A, Scognamiglio PL, et al. Therapeutical potential of a peptide mimicking the SOCS1 kinase inhibitory region in skin immune responses. Eur J Immunol. 2013;43:1883–95. https://doi.org/10.1002/eji.201343370.
Sheng Y, Jin X, Xu J, Gao J, Du X, Duan D, et al. Sequencing-based approach identified three new susceptibility loci for psoriasis. Nat Commun. 2014;5:4331. https://doi.org/10.1038/ncomms5331.
Gotoh K, Tanaka Y, Nishikimi A, Nakamura R, Yamada H, Maeda N, et al. Selective control of type I IFN induction by the Rac activator DOCK2 during TLR-mediated plasmacytoid dendritic cell activation. J Exp Med. 2010;207:721–30. https://doi.org/10.1084/jem.20091776.
Piganis RAR, De Weerd NA, Gould JA, Schindler CW, Mansell A, Nicholson SE, et al. Suppressor of Cytokine Signaling (SOCS) 1 Inhibits Type I Interferon (IFN) Signaling via the Interferon α Receptor (IFNAR1)-associated Tyrosine Kinase Tyk2. J Biol Chem. 2011;286:33811–8. https://doi.org/10.1074/jbc.M111.270207.
Uzé G, Monneron D. IL-28 and IL-29: newcomers to the interferon family. Biochimie. 2007;89:729–34. https://doi.org/10.1016/j.biochi.2007.01.008.
Onomoto K, Jogi M, Yoo JS, Narita R, Morimoto S, Takemura A, et al. Critical role of an antiviral stress granule containing RIG-I and PKR in viral detection and innate immunity. PLoS ONE. 2012;7:e43031. https://doi.org/10.1371/journal.pone.0043031.
Guo H, Gao J, Gong L, Wang Y. Multi-omics analysis reveals novel causal pathways in psoriasis pathogenesis. J Transl Med. 2025;23:100. https://doi.org/10.1186/s12967-025-06099-w.
Leung A, Marquez-Grap G, Kranyak A, Liao W. A review of spatial transcriptomics in psoriasis: new insights into cellular contributions. Curr Opin Immunol. 2025;95:102585 https://doi.org/10.1016/j.coi.2025.102585.
Cui N, Xu X, Zhou F. Single-cell technologies in psoriasis. Clin Immunol. 2024;264:110242. https://doi.org/10.1016/j.clim.2024.110242.
Dong Y, Ge HS, Chang RX, Chu J. Potential marker genes for psoriasis revealed based on single-cell sequencing and Mendelian randomization analysis. Front Genet. 2025;17:16. https://doi.org/10.3389/fgene.2025.1634874
Sieminska I, Pieniawska M, Grzywa TM. The immunology of psoriasis—current concepts in pathogenesis. Clin Rev Allergy Immunol. 2024;66:164–91. https://doi.org/10.1007/s12016-024-08991-7.
Clark RA. Skin-resident T cells: the ups and downs of on site immunity. J Investig Dermatol. 2010;130:362–70. https://doi.org/10.1038/jid.2009.247.
Arican O, Aral M, Sasmaz S, Ciragil P. Serum levels of TNF-alpha, IFN-gamma, IL-6, IL-8, IL-12, IL-17, and IL-18 in patients with active psoriasis and correlation with disease severity. Mediat Inflamm. 2005;2005:273–9. https://doi.org/10.1155/MI.2005.273.
Eyerich S, Eyerich K, Pennino D, Carbone T, Nasorri F, Pallotta S, et al. Th22 cells represent a distinct human T cell subset involved in epidermal immunity and remodeling. J Clin Investig. 2009;119:3573–85. https://doi.org/10.1172/JCI40202.
Zheng Y, Danilenko DM, Valdez P, Kasman I, Eastham-Anderson J, Wu J, et al. Interleukin-22, a T(H)17 cytokine, mediates IL-23-induced dermal inflammation and acanthosis. Nature. 2007;445:648–51. https://doi.org/10.1038/nature05505.
Hawkes JE, Chan TC, Krueger JG. Psoriasis pathogenesis and the development of novel targeted immune therapies. J Allergy Clin Immunol. 2017;140:645–53. https://doi.org/10.1016/j.jaci.2017.07.004.
Girolomoni G, Strohal R, Puig L, Bachelez H, Barker J, Boehncke WH, et al. The role of IL-23 and the IL-23/TH17 immune axis in the pathogenesis and treatment of psoriasis. J Eur Acad Dermatol Venereol. 2017;31:1616–26. https://doi.org/10.1111/jdv.14433.
Liu T, Li S, Ying S, Tang S, Ding Y, Li Y, et al. The IL-23/IL-17 pathway in inflammatory skin diseases: from bench to bedside. Front Immunol. 2020:17;11. https://doi.org/10.3389/fimmu.2020.594735.
Ha HL, Wang H, Pisitkun P, Kim JC, Tassi I, Tang W, et al. IL-17 drives psoriatic inflammation via distinct, target cell-specific mechanisms. Proc Natl Acad Sci. 2014;19:111. https://doi.org/10.1073/pnas.1400513111.
Lowes MA, Kikuchi T, Fuentes-Duculan J, Cardinale I, Zaba LC, Haider AS, et al. Psoriasis vulgaris lesions contain discrete populations of Th1 and Th17 T cells. J Investig Dermatol. 2008;128:1207–11. https://doi.org/10.1038/sj.jid.5701213.
Wang Q, Yan D, Zheng S, Li M, Li J, Fu X, et al. Cytokine profiles and the relationship of disease severity in patients with psoriasis. Indian J Dermatol. 2022;67:204. https://doi.org/10.4103/ijd.ijd_79_22.
Schwandner R, Yamaguchi K, Cao Z. Requirement of tumor necrosis factor receptor-associated factor (TRAF)6 in interleukin 17 signal transduction. J Exp Med. 2000;191:1233–40. https://doi.org/10.1084/jem.191.7.1233.
Bugaut H, Aractingi S. Major role of the IL17/23 axis in psoriasis supports the development of new targeted therapies. Front Immunol. 2021;25:12. https://doi.org/10.3389/fimmu.2021.621956
Ruscitti P, Esposito M, Di Cola I, Pellegrini C, De Berardinis A, Mastrangelo M, et al. Cytokine profile characterization of naïve patients with psoriasis and psoriatic arthritis: implications for a pathogenic disease continuum. Front Immunol. 2023;13:14. https://doi.org/10.3389/fimmu.2023.1229516
Fujita H. The role of IL-22 and Th22 cells in human skin diseases. J Dermatol Sci. 2013;72:3–8. https://doi.org/10.1016/j.jdermsci.2013.04.028.
Yadav D, Sarvetnick N. Cytokines and autoimmunity: redundancy defines their complex nature. Curr Opin Immunol. 2003;15:697–703. https://doi.org/10.1016/j.coi.2003.09.006.
Prignano F, Donetti E. Looking at interleukin-22 from a new dermatological perspective: from epidermal homeostasis to its role in chronic skin diseases. Dermatology. 2022;238:829–36. https://doi.org/10.1159/000524119.
Nikoopour E, Bellemore SM, Singh B. IL-22, cell regeneration and autoimmunity. Cytokine. 2015;74:35–42. https://doi.org/10.1016/j.cyto.2014.09.007.
Wolk K, Kunz S, Witte E, Friedrich M, Asadullah K, Sabat R. IL-22 increases the innate immunity of tissues. Immunity. 2004;21:241–54. https://doi.org/10.1016/j.immuni.2004.07.007.
Wolk K, Haugen HS, Xu W, Witte E, Waggie K, Anderson M, et al. IL-22 and IL-20 are key mediators of the epidermal alterations in psoriasis while IL-17 and IFN-gamma are not. J Mol Med. 2009;87:523–36. https://doi.org/10.1007/s00109-009-0457-0.
Reich K, Ortonne JP, Gottlieb AB, Terpstra IJ, Coteur G, Tasset C, et al. Successful treatment of moderate to severe plaque psoriasis with the PEGylated Fab’ certolizumab pegol: results of a phase II randomized, placebo-controlled trial with a re-treatment extension. Br J Dermatol. 2012;167:180–90. https://doi.org/10.1111/j.1365-2133.2012.10941.x.
McGeough MD, Wree A, Inzaugarat ME, Haimovich A, Johnson CD, Peña CA, et al. TNF regulates transcription of NLRP3 inflammasome components and inflammatory molecules in cryopyrinopathies. J Clin Investig. 2017;127:4488–97. https://doi.org/10.1172/JCI90699.
Grine L, Dejager L, Libert C, Vandenbroucke RE. An inflammatory triangle in psoriasis: TNF, type I IFNs and IL-17. Cytokine Growth Factor Rev. 2015;26:25–33. https://doi.org/10.1016/j.cytogfr.2014.10.009.
Hu P, Wang M, Gao H, Zheng A, Li J, Mu D, et al. The role of helper T cells in psoriasis. Front Immunol. 2021;12:788940. https://doi.org/10.3389/fimmu.2021.788940.
Arakawa A, Siewert K, Stöhr J, Besgen P, Kim SM, Rühl G, et al. Melanocyte antigen triggers autoimmunity in human psoriasis. J Exp Med. 2015;212:2203–12. https://doi.org/10.1084/jem.20151093.
Ganguly D, Chamilos G, Lande R, Gregorio J, Meller S, Facchinetti V, et al. Self-RNA–antimicrobial peptide complexes activate human dendritic cells through TLR7 and TLR8. J Exp Med. 2009;206:1983–94. https://doi.org/10.1084/jem.20090480.
Shimoura N, Nagai H, Fujiwara S, Jimbo H, Yoshimoto T, Nishigori C. Interleukin (IL)-18, cooperatively with IL-23, induces prominent inflammation and enhances psoriasis-like epidermal hyperplasia. Arch Dermatol Res. 2017;309:315–21. https://doi.org/10.1007/s00403-017-1735-2.
Chiricozzi A, Guttman-Yassky E, Suárez-Fariñas M, Nograles KE, Tian S, Cardinale I, et al. Integrative responses to IL-17 and TNF-α in human keratinocytes account for key inflammatory pathogenic circuits in psoriasis. J Investig Dermatol. 2011;131:677–87. https://doi.org/10.1038/jid.2010.340.
Bielecki P, Riesenfeld SJ, Hütter JC, Torlai Triglia E, Kowalczyk MS, Ricardo-Gonzalez RR, et al. Skin-resident innate lymphoid cells converge on a pathogenic effector state. Nature. 2021;592:128–32. https://doi.org/10.1038/s41586-021-03188-w.
Teunissen MBM, Munneke JM, Bernink JH, Spuls PI, Res PCM, te Velde A, et al. Composition of innate lymphoid cell subsets in the human skin: enrichment of NCR + ILC3 in lesional skin and blood of psoriasis patients. J Investig Dermatol. 2014;134:2351–60. https://doi.org/10.1038/jid.2014.146.
Villanova F, Flutter B, Tosi I, Grys K, Sreeneebus H, Perera GK, et al. Characterization of innate lymphoid cells in human skin and blood demonstrates increase of NKp44 + ILC3 in psoriasis. J Investig Dermatol. 2014;134:984–91. https://doi.org/10.1038/jid.2013.477.
Barker JN, Mitra RS, Griffiths CE, Dixit VM, Nickoloff BJ. Keratinocytes as initiators of inflammation. Lancet. 1991;337:211–4. https://doi.org/10.1016/0140-6736(91)92168-2.
Meister M, Tounsi A, Gaffal E, Bald T, Papatriantafyllou M, Ludwig J, et al. Self-antigen presentation by keratinocytes in the inflamed adult skin modulates T-cell auto-reactivity. J Investig Dermatol. 2015;135:1996–2004. https://doi.org/10.1038/jid.2015.130.
Nestle FO, Di Meglio P, Qin JZ, Nickoloff BJ. Skin immune sentinels in health and disease. Nat Rev Immunol. 2009;9:679–91. https://doi.org/10.1038/nri2622.
Wu M, Dai C, Zeng F. Cellular mechanisms of psoriasis pathogenesis: a systemic review. Clin Cosmet Investig Dermatol. 2023;16:2503–15. https://doi.org/10.2147/CCID.S420850.
Lande R, Gregorio J, Facchinetti V, Chatterjee B, Wang YH, Homey B, et al. Plasmacytoid dendritic cells sense self-DNA coupled with antimicrobial peptide. Nature. 2007;449:564–9. https://doi.org/10.1038/nature06116.
Herster F, Bittner Z, Archer NK, Dickhöfer S, Eisel D, Eigenbrod T, et al. Neutrophil extracellular trap-associated RNA and LL37 enable self-amplifying inflammation in psoriasis. Nat Commun. 2020;11:105. https://doi.org/10.1038/s41467-019-13756-4.
Lande R, Botti E, Jandus C, Dojcinovic D, Fanelli G, Conrad C, et al. Corrigendum: the antimicrobial peptide LL37 is a T-cell autoantigen in psoriasis. Nat Commun. 2015;6:6595. https://doi.org/10.1038/ncomms7595.
Fuentes-Duculan J, Bonifacio KM, Hawkes JE, Kunjravia N, Cueto I, Li X, et al. Autoantigens ADAMTSL5 and LL37 are significantly upregulated in active Psoriasis and localized with keratinocytes, dendritic cells and other leukocytes. Exp Dermatol. 2017;26:1075–82. https://doi.org/10.1111/exd.13378.
Cai Y, Fleming C, Yan J. New insights of T cells in the pathogenesis of psoriasis. Cell Mol Immunol. 2012;9:302–9. https://doi.org/10.1038/cmi.2012.15.
Sasson SC, Gordon CL, Christo SN, Klenerman P, Mackay LK. Local heroes or villains: tissue-resident memory T cells in human health and disease. Cell Mol Immunol. 2020;17:113–22. https://doi.org/10.1038/s41423-019-0359-1.
Kurihara K, Fujiyama T, Phadungsaksawasdi P, Ito T, Tokura Y. Significance of IL-17A-producing CD8 + CD103+ skin resident memory T cells in psoriasis lesion and their possible relationship to clinical course. J Dermatol Sci. 2019;95:21–7. https://doi.org/10.1016/j.jdermsci.2019.06.002.
Vo S, Watanabe R, Koguchi-Yoshioka H, Matsumura Y, Ishitsuka Y, Nakamura Y, et al. CD8 resident memory T cells with interleukin 17A-producing potential are accumulated in disease-naïve nonlesional sites of psoriasis possibly in correlation with disease duration. Br J Dermatol. 2019;181:410–2. https://doi.org/10.1111/bjd.17748.
Liu Y, Wang H, Cook C, Taylor MA, North JP, Hailer A, et al. Defining patient-level molecular heterogeneity in psoriasis vulgaris based on single-cell transcriptomics. Front Immunol. 2022;13:842651. https://doi.org/10.3389/fimmu.2022.842651.
Cheuk S, Wikén M, Blomqvist L, Nylén S, Talme T, Ståhle M, et al. Epidermal Th22 and Tc17 cells form a localized disease memory in clinically healed psoriasis. J Immunol. 2014;192:3111–20. https://doi.org/10.4049/jimmunol.1302313.
Fenix K, Wijesundara DK, Cowin AJ, Grubor-Bauk B, Kopecki Z. Immunological memory in imiquimod-induced murine model of psoriasiform dermatitis. Int J Mol Sci. 2020;21. https://doi.org/10.3390/ijms21197228.
Pan Y, Tian T, Park CO, Lofftus SY, Mei S, Liu X, et al. Survival of tissue-resident memory T cells requires exogenous lipid uptake and metabolism. Nature. 2017;543:252–6. https://doi.org/10.1038/nature21379.
Pan Y, Kupper TS. Metabolic reprogramming and longevity of tissue-resident memory T cells. Front Immunol. 2018;9. https://doi.org/10.3389/fimmu.2018.01347.
Diani M, Galasso M, Cozzi C, Sgambelluri F, Altomare A, Cigni C, et al. Blood to skin recirculation of CD4+ memory T cells associates with cutaneous and systemic manifestations of psoriatic disease. Clin Immunol. 2017;180:84–94. https://doi.org/10.1016/j.clim.2017.04.001.
Peng L, Chen L, Wan J, Liu W, Lou S, Shen Z. Single-cell transcriptomic landscape of immunometabolism reveals intervention candidates of ascorbate and aldarate metabolism, fatty-acid degradation and PUFA metabolism of T-cell subsets in healthy controls, psoriasis and psoriatic arthritis. Front Immunol. 2023;14:1179877 https://doi.org/10.3389/fimmu.2023.1179877.
Casciano F, Diani M, Altomare A, Granucci F, Secchiero P, Banfi G, et al. CCR4+ skin-tropic phenotype as a feature of central memory CD8 + T cells in healthy subjects and psoriasis patients. Front Immunol. 2020;11:529. https://doi.org/10.3389/fimmu.2020.00529.
Watanabe R, Gehad A, Yang C, Scott LL, Teague JE, Schlapbach C, et al. Human skin is protected by four functionally and phenotypically discrete populations of resident and recirculating memory T cells. Sci Transl Med. 2015;7:279ra39. https://doi.org/10.1126/scitranslmed.3010302.
Martins C, Darrigade AS, Jacquemin C, Barnetche T, Taieb A, Ezzedine K, et al. Phenotype and function of circulating memory T cells in human vitiligo. Br J Dermatol. 2020;183:899–908. https://doi.org/10.1111/bjd.18902.
Kamiyama T, Watanabe H, Iijima M, Miyazaki A, Iwamoto S. Coexpression of CCR6 and CD146 (MCAM) is a marker of effector memory T-helper 17 cells. J Dermatol. 2012;39:838–42. https://doi.org/10.1111/j.1346-8138.2012.01544.x.
Hughes TK, Wadsworth MH, Gierahn TM, Do T, Weiss D, Andrade PR, et al. Second-strand synthesis-based massively parallel scRNA-Seq reveals cellular states and molecular features of human inflammatory skin pathologies. Immunity. 2020;53:878–894.e7. https://doi.org/10.1016/j.immuni.2020.09.015.
Reynolds G, Vegh P, Fletcher J, Poyner EFM, Stephenson E, Goh I, et al. Developmental cell programs are co-opted in inflammatory skin disease. Science. 2021;371. https://doi.org/10.1126/science.aba6500.
Di Meglio P, Duarte JH, Ahlfors H, Owens NDL, Li Y, Villanova F, et al. Activation of the aryl hydrocarbon receptor dampens the severity of inflammatory skin conditions. Immunity. 2014;40:989–1001. https://doi.org/10.1016/j.immuni.2014.04.019.
Mahil SK, Capon F, Barker JN. Update on psoriasis immunopathogenesis and targeted immunotherapy. Semin Immunopathol. 2016;38:11–27. https://doi.org/10.1007/s00281-015-0539-8.
Gupta RK, Gracias DT, Figueroa DS, Miki H, Miller J, Fung K, et al. TWEAK functions with TNF and IL-17 on keratinocytes and is a potential target for psoriasis therapy. Sci Immunol. 2021;6:eabi8823. https://doi.org/10.1126/sciimmunol.abi8823.
Osmola-Mańkowska A, Teresiak-Mikołajczak E, Skrzypczak-Zielińska M, Adamski Z. Genetic polymorphism in psoriasis and its meaning for the treatment efficacy in the future. Adv Dermatol Allergol. 2018;35:331–7. https://doi.org/10.5114/ada.2018.77661.
van de Kerkhof PC. Psoriasis in the perspective of predictive, preventive participatory and personalized medicine. J Dermatol Treat. 2018;29:107–8. https://doi.org/10.1080/09546634.2018.1437739.
Kar BR, Sathishkumar D, Tahiliani S, Parthasarathi A, Neema S, Ganguly S, et al. Biomarkers in psoriasis: the future of personalised treatment. Indian J Dermatol. 2024;69:256–63. https://doi.org/10.4103/ijd.ijd_167_24.
Camela E, Potestio L, Ruggiero A, Ocampo-Garza SS, Fabbrocini G, Megna M. Towards personalized medicine in psoriasis: current progress. Psoriasis Targets Ther. 2022;ume 12:231–50. https://doi.org/10.2147/PTT.S328460.
Yin X, Cheng H, Wang W, Wang W, Fu H, Liu L, et al. TNIP1/ANXA6 and CSMD1 variants interacting with cigarette smoking, alcohol intake affect risk of psoriasis. J Dermatol Sci. 2013;70:94–8. https://doi.org/10.1016/j.jdermsci.2013.02.006.
Prieto-Pérez R, Solano-López G, Cabaleiro T, Román M, Ochoa D, Talegón M, et al. Polymorphisms associated with age at onset in patients with moderate-to-severe plaque psoriasis. J Immunol Res. 2015;2015:1–8. https://doi.org/10.1155/2015/101879.
Atasoy M, Pirim I, Bayrak OF, Ozdemir S, Ikbal M, Erdem T, et al. Association of HLA class I and class II alleles with psoriasis vulgaris in Turkish population. Influence of type I and II psoriasis. Saudi Med J. 2006;27:373–6.
Weng HJ, Tsai TF. ABCB1 in dermatology: roles in skin diseases and their treatment. J Mol Med. 2021;99:1527–38. https://doi.org/10.1007/s00109-021-02105-y.
Morelli M, Galluzzo M, Scarponi C, Madonna S, Scaglione GL, Girolomoni G, et al. Allelic variants of HLA-C upstream region, PSORS1C3, MICA, TNFA and genes involved in epidermal homeostasis and barrier function influence the clinical response to anti-IL-12/IL-23 treatment of patients with psoriasis. Vaccines. 2022;10. https://doi.org/10.3390/vaccines10111977.
Morelli M, Galluzzo M, Madonna S, Scarponi C, Scaglione GL, Galluccio T, et al. HLA-Cw6 and other HLA-C alleles, as well as MICB-DT, DDX58, and TYK2 genetic variants associate with optimal response to anti-IL-17A treatment in patients with psoriasis. Expert Opin Biol Ther. 2021;21:259–70. https://doi.org/10.1080/14712598.2021.1862082.
Lee CC, Huang YH, Chi CC, Chung WH, Chen CB. Generalized pustular psoriasis: immunological mechanisms, genetics, and emerging therapeutics. Trends Immunol. 2025;46:74–89. https://doi.org/10.1016/j.it.2024.12.001.
Wu X, Zhao Z. Associations between ERAP1 gene polymorphisms and psoriasis susceptibility: a meta-analysis of case-control studies. Biomed Res Int. 2021;2021. https://doi.org/10.1155/2021/5515868.
Zaba LC, Fuentes-Duculan J, Eungdamrong NJ, Abello MV, Novitskaya I, Pierson KC, et al. Psoriasis is characterized by accumulation of immunostimulatory and Th1/Th17 cell-polarizing myeloid dendritic cells. J Investig. Dermatol. 2009;129:79–88. https://doi.org/10.1038/jid.2008.194.
Yang L, Li B, Dang E, Jin L, Fan X, Wang G. Impaired function of regulatory T cells in patients with psoriasis is mediated by phosphorylation of STAT3. J Dermatol Sci. 2016;81:85–92. https://doi.org/10.1016/j.jdermsci.2015.11.007.
Hayashida S, Uchi H, Moroi Y, Furue M. Decrease in circulating Th17 cells correlates with increased levels of CCL17, IgE and eosinophils in atopic dermatitis. J Dermatol Sci. 2011;61:180–6. https://doi.org/10.1016/j.jdermsci.2010.10.013.
Furue K, Ito T, Tsuji G, Nakahara T, Furue M. The CCL20 and CCR6 axis in psoriasis. Scand J Immunol. 2020;91. https://doi.org/10.1111/sji.12846.
Morizane S, Yamasaki K, Mühleisen B, Kotol PF, Murakami M, Aoyama Y, et al. Cathelicidin antimicrobial peptide LL-37 in psoriasis enables keratinocyte reactivity against TLR9 ligands. J Investig Dermatol. 2012;132:135–43. https://doi.org/10.1038/jid.2011.259.
Mabuchi T, Hirayama N. Binding Affinity and Interaction of LL-37 with HLA-C*06:02 in Psoriasis. J Investig Dermatol. 2016;136:1901–3. https://doi.org/10.1016/j.jid.2016.04.033.
Seyhan AA, Carini C. Are innovation and new technologies in precision medicine paving a new era in patients centric care. J Transl Med. 2019;17:114. https://doi.org/10.1186/s12967-019-1864-9.
Hasin Y, Seldin M, Lusis A. Multi-omics approaches to disease. Genome Biol. 2017;18:83. https://doi.org/10.1186/s13059-017-1215-1.
Dand N, Mahil S, Capon F, Smith C, Simpson M, Barker J. Psoriasis and Genetics. Acta Derm Venereol. 2020;100:55–65. https://doi.org/10.2340/00015555-3384.
Dopytalska K, Ciechanowicz P, Wiszniewski K, Szymańska E, Walecka I. The role of epigenetic factors in psoriasis. Int J Mol Sci. 2021;22:9294. https://doi.org/10.3390/ijms22179294.
Cui J, Zhang X, Deng J, Yan Y, Yao D, Deng H, et al. Potential biomarkers for psoriasis topical treatment by in-depth serum proteomics. J Dermatol Treat. 2023;34. https://doi.org/10.1080/09546634.2023.2248318.
Łuczaj W, Gęgotek A, Skrzydlewska E. Analytical approaches to assess metabolic changes in psoriasis. J Pharm Biomed Anal. 2021;205:114359. https://doi.org/10.1016/j.jpba.2021.114359.
Daugaard C, Iversen L, Hjuler KF. Comorbidity in adult psoriasis: considerations for the clinician. Psoriasis Targets Ther. 2022;ume 12:139–50. https://doi.org/10.2147/PTT.S328572.
Yu K, Syed MN, Bernardis E, Gelfand JM. Machine learning applications in the evaluation and management of psoriasis: a systematic review. J Psoriasis Psoriatic Arthritis. 2020;5:147–59. https://doi.org/10.1177/2475530320950267.
Smith P, Johnson CE, Haran K, Orcales F, Kranyak A, Bhutani T, et al. Advancing psoriasis care through artificial intelligence: a comprehensive review. Curr Dermatol Rep. 2024;13:141–7. https://doi.org/10.1007/s13671-024-00434-y.
den Braanker H, Razawy W, Wervers K, Mus AMC, Davelaar N, Kok MR, et al. Characterizing memory T helper cells in patients with psoriasis, subclinical, or early psoriatic arthritis using a machine learning algorithm. Arthritis Res Ther. 2022;24:28. https://doi.org/10.1186/s13075-021-02714-5.
ten Bergen LL, Petrovic A, Krogh Aarebrot A, Appel S. The TNF/IL-23/IL-17 axis—Head-to-head trials comparing different biologics in psoriasis treatment. Scand J Immunol. 2020;92. https://doi.org/10.1111/sji.12946.
Leonardi CL, Kimball AB, Papp KA, Yeilding N, Guzzo C, Wang Y, et al. Efficacy and safety of ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with psoriasis: 76-week results from a randomised, double-blind, placebo-controlled trial (PHOENIX 1. Lancet. 2008;371:1665–74. https://doi.org/10.1016/S0140-6736(08)60725-4.
Li K, Huang CC, Randazzo B, Li S, Szapary P, Curran M, et al. HLA-C*06:02 allele and response to IL-12/23 inhibition: results from the ustekinumab phase 3 psoriasis program. J Investig Dermatol. 2016;136:2364–71. https://doi.org/10.1016/j.jid.2016.06.631.
Dand N, Duckworth M, Baudry D, Russell A, Curtis CJ, Lee SH, et al. HLA-C*06:02 genotype is a predictive biomarker of biologic treatment response in psoriasis. J Allergy Clin Immunol. 2019;143:2120–30. https://doi.org/10.1016/j.jaci.2018.11.038.
Gönülal M, Balcı DD, Öztürk A, Doğan S. Effectiveness and safety of ustekinumab for the treatment of psoriasis; six years of clinical experience. J Dermatol Treat. 2023;34:2241941. https://doi.org/10.1080/09546634.2023.2241941.
Schwarz CW, Loft N, Bryld LE, Nissen CV, Ajgeiy KK, Bertelsen T, et al. Drug survival of biologics in bionaive and bioexperienced patients with psoriasis. JAMA Dermatol. 2026;162:166. https://doi.org/10.1001/jamadermatol.2025.5205.
Kamata M, Tada Y. Efficacy and safety of biologics for psoriasis and psoriatic arthritis and their impact on comorbidities: a literature review. Int J Mol Sci. 2020;21:1690. https://doi.org/10.3390/ijms21051690.
Schäkel K, Reich K, Asadullah K, Pinter A, Jullien D, Weisenseel P, et al. Early disease intervention with guselkumab in psoriasis leads to a higher rate of stable complete skin clearance (‘clinical super response’): Week 28 results from the ongoing phase IIIb randomized, double-blind, parallel-group, GUIDE study. J Eur. Acad Dermatol Venereol. 2023;37:2016–27. https://doi.org/10.1111/jdv.19236.
Prajapati VH, Seyger MMB, Wilsmann-Theis D, Szakos E, Kaszuba A, van Hartingsveldt B, et al. Guselkumab for the treatment of moderate-to-severe plaque psoriasis in paediatric patients: results of the phase III randomized placebo-controlled PROTOSTAR study. Br J Dermatol. 2025;192:618–28. https://doi.org/10.1093/bjd/ljae502.
Papp KA, Blauvelt A, Puig L, Ohtsuki M, Beissert S, Gooderham M, et al. Long-term safety and efficacy of risankizumab for the treatment of moderate-to-severe plaque psoriasis: interim analysis of the LIMMitless open-label extension trial up to 5 years of follow-up. J Am Acad Dermatol. 2023;89:1149–58. https://doi.org/10.1016/j.jaad.2023.07.1024.
Haugh I, Preston A, Kivelevitch D, Menter A. Risankizumab: an anti-IL-23 antibody for the treatment of psoriasis. Drug Des Devel Ther. 2018;ume 12:3879–83. https://doi.org/10.2147/DDDT.S167149.
Gordon KB, Strober B, Lebwohl M, Augustin M, Blauvelt A, Poulin Y, et al. Efficacy and safety of risankizumab in moderate-to-severe plaque psoriasis (UltIMMa-1 and UltIMMa-2): results from two double-blind, randomised, placebo-controlled and ustekinumab-controlled phase 3 trials. Lancet. 2018;392:650–61. https://doi.org/10.1016/S0140-6736(18)31713-6.
Blauvelt A, Gooderham M, Griffiths CEM, Armstrong AW, Zhu B, Burge R, et al. Cumulative clinical benefits of biologics in the treatment of patients with moderate-to-severe psoriasis over 1 year: a network meta-analysis. Dermatol Ther. 2022;12:727–40. https://doi.org/10.1007/s13555-022-00690-5.
Camiña-Conforto G, Mateu-Arrom L, López-Ferrer A, Puig L. Bimekizumab in the treatment of plaque psoriasis: focus on patient selection and perspectives. Patient Prefer Adherence. 2023;17:1541–9. https://doi.org/10.2147/PPA.S350760.
Langley RG, Sofen H, Dei-Cas I, Reich K, Sigurgeirsson B, Warren RB, et al. Secukinumab long-term efficacy and safety in psoriasis through to year 5 of treatment: results of a randomized extension of the phase III ERASURE and FIXTURE trials. Br J Dermatol. 2023;188:198–207. https://doi.org/10.1093/bjd/ljac040.
Craig S, Warren RB. Ixekizumab for the treatment of psoriasis: up-to-date. Expert Opin Biol Ther. 2020;20:549–57. https://doi.org/10.1080/14712598.2020.1729736.
Deodhar A, Blauvelt A, Lebwohl M, Feely M, Kronbergs A, Eberhart N, et al. Long-term safety of Ixekizumab in adults with psoriasis, psoriatic arthritis, or axial spondyloarthritis: a post-hoc analysis of final safety data from 25 randomized clinical trials. Arthritis Res Ther. 2024;26:49. https://doi.org/10.1186/s13075-023-03257-7.
Papp KA, Reid C, Foley P, Sinclair R, Salinger DH, Williams G, et al. Anti-IL-17 receptor antibody AMG 827 leads to rapid clinical response in subjects with moderate to severe psoriasis: results from a phase I, randomized, placebo-controlled trial. J Investig Dermatol. 2012;132:2466–9. https://doi.org/10.1038/jid.2012.163.
Papp KA, Reich K, Paul C, Blauvelt A, Baran W, Bolduc C, et al. A prospective phase III, randomized, double-blind, placebo-controlled study of brodalumab in patients with moderate-to-severe plaque psoriasis. Br J Dermatol. 2016;175:273–86. https://doi.org/10.1111/bjd.14493.
Lebwohl M, Strober B, Menter A, Gordon K, Weglowska J, Puig L, et al. Phase 3 studies comparing brodalumab with ustekinumab in psoriasis. N Engl J Med. 2015;373:1318–28. https://doi.org/10.1056/NEJMoa1503824.
Babaei N, Gill M, Cervantes M, Kim D, Yang S, Wu JJ. Brodalumab for moderate-to-severe psoriasis: a comprehensive review of efficacy, safety, and clinical positioning. Biologics. 2025;19:415–22. https://doi.org/10.2147/BTT.S532526.
Warren RB, Donnelly K, Kiri S, Taieb V, Slim M, Fahrbach K, et al. Long-term efficacy and safety of bimekizumab and other biologics in moderate to severe plaque psoriasis: updated systematic literature review and network meta-analysis. Dermatol Ther. 2024;14:3133–47. https://doi.org/10.1007/s13555-024-01302-0.
Azimi SI, Jafarzadeh A, Goodarzi A. Systematic review of comparative studies on emerging psoriasis treatments: comparing biologics with biologics, small molecule inhibitors with small molecule inhibitors, and biologics with small molecule inhibitors. Inflammopharmacology. 2025;33:2377–407. https://doi.org/10.1007/s10787-025-01758-2.
Gottlieb AB, Matheson RT, Lowe N, Krueger GG, Kang S, Goffe BS, et al. A randomized trial of etanercept as monotherapy for psoriasis. Arch Dermatol. 2003;139:1627–32. https://doi.org/10.1001/archderm.139.12.1627.
Leonardi C, Langley RG, Papp K, Tyring SK, Wasel N, Vender R, et al. Adalimumab for treatment of moderate to severe chronic plaque psoriasis of the hands and feet: efficacy and safety results from REACH, a randomized, placebo-controlled, double-blind trial. Arch Dermatol. 2011;147:429–36. https://doi.org/10.1001/archdermatol.2010.384.
Masouri S, Stefanaki I, Ntritsos G, Kypreou KP, Drakaki E, Evangelou E, et al. A pharmacogenetic study of psoriasis risk variants in a Greek population and prediction of responses to anti-TNF-α and anti-IL-12/23 agents. Mol Diagn Ther. 2016;20:221–5. https://doi.org/10.1007/s40291-016-0198-z.
Papp K, Gordon K, Thaçi D, Morita A, Gooderham M, Foley P, et al. Phase 2 trial of selective tyrosine kinase 2 inhibition in psoriasis. N Engl J Med. 2018;379:1313–21. https://doi.org/10.1056/NEJMoa1806382.
Mease P, Deodhar A, van der Heijde D, Behrens F, Kivitz A, Kim J, et al. Efficacy and safety of deucravacitinib (BMS-986165), an oral, selective tyrosine kinase 2 inhibitor, in patients with active psoriatic arthritis: results from a Phase 2, randomized, double-blind, placebo-controlled trial. Arth Rheumatol. 2020 [cited 2023 Feb 24];72. Available from: https://acrabstracts.org/abstract/efficacy-and-safety-of-deucravacitinib-bms-986165-an-oral-selective-tyrosine-kinase-2-inhibitor-in-patients-with-active-psoriatic-arthritis-results-from-a-phase-2-randomized-double-blind-plac/.
Bristol-Myers Squibb. Bristol Myers Squibb Announces Deucravacitinib (BMS-986165) Demonstrated Superiority to Placebo and Otezla® (apremilast) in Pivotal Phase 3 Psoriasis Study [press release]. 2020 [cited 2023 Feb 24]. Available from: https://news.bms.com/news/details/2020/Bristol-Myers-Squibb-Announces-Deucravacitinib-BMS-986165-Demonstrated-Superiority-to-Placebo-and-Otezla-apremilast-in-Pivotal-Phase-3-Psoriasis-Study/default.aspx.
Truong TM, Pathak GN, Singal A, Taranto V, Rao BK. Deucravacitinib: the first FDA-approved oral TYK2 inhibitor for moderate to severe plaque psoriasis. Ann Pharmacother. 2024;58:416–27. https://doi.org/10.1177/10600280231153863.
Zheng Y, Han Y, Chen J, Huang J, Zhu C, Lin L, et al. Comparative efficacy and safety of JAK/TYK2 inhibitors and other oral drugs for moderate-to-severe plaque psoriasis: systematic review and network meta-analysis. Indian J Dermatol Venereol Leprol. 2024;90:590–8. https://doi.org/10.25259/IJDVL_775_2023.
Hagino T, Saeki H, Fujimoto E, Kanda N. Effectiveness and safety of deucravacitinib treatment for moderate-to-severe psoriasis in real-world clinical practice in Japan. J Dermatol Treat. 2024;35:2307489 https://doi.org/10.1080/09546634.2024.2307489.
Rahman MDT, Afrin S, Antora FZ, Akter J, Nila SR, Wahida F. Efficacy and safety of deucravacitinib in the treatment of psoriasis: a systematic review. Saudi J Med. 2024;9:455–9. https://doi.org/10.36348/sjm.2024.v09i10.007.
Markham A. Baricitinib: first global approval. Drugs. 2017;77:697–704. https://doi.org/10.1007/s40265-017-0723-3.
Choy EH. Clinical significance of Janus Kinase inhibitor selectivity. Rheumatology. 2019;58:1122. https://doi.org/10.1093/rheumatology/kez002.
Papp KA, Menter MA, Raman M, Disch D, Schlichting DE, Gaich C, et al. A randomized phase 2b trial of baricitinib, an oral Janus kinase (JAK) 1/JAK2 inhibitor, in patients with moderate-to-severe psoriasis. Br J Dermatol. 2016;174:1266–76. https://doi.org/10.1111/bjd.14403.
AbbVie. A phase 3, randomized, double-blind, study comparing upadacitinib (ABT-494) to Placebo in subjects with active psoriatic arthritis who have a history of inadequate response to at least one biologic disease modifying anti-rheumatic drug (bDMARD). 2017 [cited 2023 Feb 17]. Available from: https://clinicaltrials.gov/ct2/show/study/NCT03104374.
RINVOQ® (upadacitinib) Receives U.S. FDA Approval for Active Psoriatic Arthritis.
Tokareva K, Reid P, Yang V, Liew D, Peterson AC, Baraff A, et al. JAK inhibitors and black box warnings: what is the future for JAK inhibitors. Expert Rev Clin Immunol. 2023;19:1385–97. https://doi.org/10.1080/1744666X.2023.2249237.
Smith SH, Jayawickreme C, Rickard DJ, Nicodeme E, Bui T, Simmons C, et al. Tapinarof is a natural AhR agonist that resolves skin inflammation in mice and humans. J Investig Dermatol. 2017;137:2110–9. https://doi.org/10.1016/j.jid.2017.05.004.
Furue M, Hashimoto-Hachiya A, Tsuji G. Aryl hydrocarbon receptor in atopic dermatitis and psoriasis. Int J Mol Sci. 2019;20. https://doi.org/10.3390/ijms20215424.
Bissonnette R, Stein Gold L, Rubenstein DS, Tallman AM, Armstrong A. Tapinarof in the treatment of psoriasis: a review of the unique mechanism of action of a novel therapeutic aryl hydrocarbon receptor–modulating agent. J Am Acad Dermatol. 2021;84:1059–67. https://doi.org/10.1016/j.jaad.2020.10.085.
Stein Gold L, Bhatia N, Tallman AM, Rubenstein DS. A phase 2b, randomized clinical trial of tapinarof cream for the treatment of plaque psoriasis: secondary efficacy and patient-reported outcomes. J Am Acad Dermatol. 2021;84:624–31. https://doi.org/10.1016/j.jaad.2020.04.181.
Robbins K, Bissonnette R, Maeda-Chubachi T, Ye L, Peppers J, Gallagher K, et al. Phase 2, randomized dose-finding study of tapinarof (GSK2894512 cream) for the treatment of plaque psoriasis. J Am Acad Dermatol. 2019;80:714–21. https://doi.org/10.1016/j.jaad.2018.10.037.
FDA Approves Dermavant’s VTAMA® (tapinarof) cream. 1% for the treatment of plaque psoriasis in adults: first topical novel chemical entity launched for psoriasis in the U.S. in 25 Years.
Balogh B, Pető Á, Fehér P, Ujhelyi Z, Bácskay I. Tapinarof nanogels as a promising therapeutic approach. Pharmaceutics. 2025;17. https://doi.org/10.3390/pharmaceutics17060731.
Jandhyala S. Hit hard and early: Can the march of psoriasis be halted?. Indian Dermatol Online J. 2013;4:177–9. https://doi.org/10.4103/2229-5178.115510.
Lu X, Kuai L, Huang F, Jiang J, Song J, Liu Y, et al. Single-atom catalysts-based catalytic ROS clearance for efficient psoriasis treatment and relapse prevention via restoring ESR1. Nat Commun. 2023;14:6767. https://doi.org/10.1038/s41467-023-42477-y.
Chen Y, Yan Y, Liu H, Qiu F, Liang CL, Zhang Q, et al. Dihydroartemisinin ameliorates psoriatic skin inflammation and its relapse by diminishing CD8 + T-cell memory in wild-type and humanized mice. Theranostics. 2020;10:10466–82. https://doi.org/10.7150/thno.45211.
Kundu-Raychaudhuri S, Chen YJ, Wulff H, Raychaudhuri SP. Kv1.3 in psoriatic disease: PAP-1, a small molecule inhibitor of Kv1.3 is effective in the SCID mouse psoriasis-xenograft model. J Autoimmun. 2014;55:63–72. https://doi.org/10.1016/j.jaut.2014.07.003.
Jones PA, Taylor SM, Wilson VL. Inhibition of DNA methylation by 5-azacytidine. In: Modified nucleosides and cancer. Berlin, Heidelberg: Springer Berlin Heidelberg; 1983. p. 202–11. https://doi.org/10.1007/978-3-642-81947-6_15.
Thatikonda S, Pooladanda V, Sigalapalli DK, Godugu C. Piperlongumine regulates epigenetic modulation and alleviates psoriasis-like skin inflammation via inhibition of hyperproliferation and inflammation. Cell Death Dis. 2020;11:21. https://doi.org/10.1038/s41419-019-2212-y.
Soler DC, McCormick TS. The dark side of regulatory T cells in psoriasis. J Investig Dermatol. 2011;131:1785–6. https://doi.org/10.1038/jid.2011.200.
Krueger JG, Suárez-Fariñas M, Cueto I, Khacherian A, Matheson R, Parish LC, et al. A randomized, placebo-controlled study of SRT2104, a SIRT1 activator, in patients with moderate to severe psoriasis. PLoS ONE. 2015;10:e0142081. https://doi.org/10.1371/journal.pone.0142081.
Feng H, Wu R, Zhang S, Kong Y, Liu Z, Wu H, et al. Topical administration of nanocarrier miRNA-210 antisense ameliorates imiquimod-induced psoriasis-like dermatitis in mice. J Dermatol. 2020;47:147–54. https://doi.org/10.1111/1346-8138.15149.
Lifshiz Zimon R, Lerman G, Elharrar E, Meningher T, Barzilai A, Masalha M, et al. Ultrasound targeting of Q-starch/miR-197 complexes for topical treatment of psoriasis. J Control Release. 2018;284:103–11. https://doi.org/10.1016/j.jconrel.2018.05.040.












Leave a Reply