Tuesday, May 12, 2026
Comprehensive characterization of islet cells facilitated by national organ donation program.
A National Institutes of Health (NIH)-funded study has identified key differences in human pancreatic islet cells that may help explain why some people are more likely to develop diabetes. Researchers found that the mix of hormone-producing cells in the pancreas varies widely from person to person, and that variation plays a central role in how the body regulates blood sugar.
Researchers analyzed islets from 299 donors collected through the NIH-funded Integrated Islet Distribution Program (IIDP), at City of Hope in Duarte, California, the largest source of human islets for research in the country. Scientists say the dataset will help clarify how genetic and biological factors combine to influence diabetes risk.
“This level of characterization across so many donors has not been done in the past,” said Albert J. Hwa, Ph.D., program director at NIH’s National Institute of Diabetes and Kidney Diseases (NIDDK). “Collection and analysis of this rich trove of data should enable better understanding of how multiple factors come together to contribute to the risk of developing diabetes.”
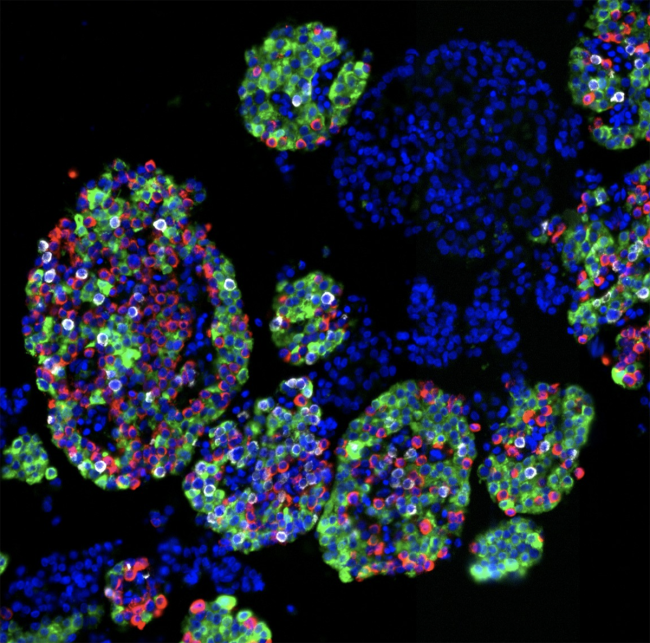
The study analyzed pancreatic islets — clusters of cells that regulate blood glucose by releasing hormones. These include:
- Alpha cells, which produce glucagon to raise blood sugar
- Beta cells, which produce insulin to lower blood sugar
- Delta cells, which produce somatostatin to regulate both
The IIDP team developed the infrastructure for managing and coordinating human pancreatic islet isolation, while maximizing islet yield, viability and purity. The donor group represented in this study reflects a diverse range of race, ethnicity, sex, age and body mass index measurements.
“The composition of the organ donor pool is highly reflective of the population of the United States,” said study co-lead author Carmella Evans-Molina, M.D., Ph.D., director of the Indiana Diabetes Research Center at Indiana University School of Medicine and IIDP co-principal investigator. “This attribute of the study population is important for understanding differences in islet function between people of different backgrounds.”
The study involved a deep dive into islet cell function that is linked to donor traits associated with observable characteristics, or phenotype, such as sex, race and ethnicity, as well as genetic information, or genotype, including predicted ancestry and genetic risk for both type 1 and type 2 diabetes.
On average, islets were comprised of 34% alpha cells, 58% beta cells and 8% delta cells. However, the researchers detected broad variability in the percentage of alpha and beta cells in the study population, and lower but significant variation in the percentage of delta cells. Since each cell has a particular role in sugar metabolism, this variability has complex effects that inform future investigation.
The findings highlight that islet cell composition, rather than the physical size and shape of islets, is a key factor in regulating hormone release. The team found that the makeup of pancreatic islets plays a major role in how effectively they release insulin and glucagon — key hormones that regulate blood glucose. Islets with a higher proportion of insulin-producing beta cells showed stronger insulin secretion in response to various stimuli, while higher levels of alpha and delta cells were generally linked to reduced insulin output.
The researchers observed that cells derived from female donors tended to have a higher percentage of alpha cells and a lower percentage of beta cells. Their findings also suggested differences in beta cell proportions across ancestry groups.
In addition, the researchers found that islet hormone secretion is affected by donor traits, such as sex, race and ethnicity and their genetic makeup, including ancestry predicted from genetic testing and genetic risk for type 2 diabetes. Combined, the findings of the study have significant implications for understanding the factors that may predispose people to diabetes.
“This study is the tip of the iceberg,” said Dr. Evans-Molina. “We hope this dataset becomes useful to the entire diabetes research community and that researchers use it to answer questions about the genotype-phenotype correlation within these data.”
Co-authors of the study include IIDP founder and principal investigator Joyce Niland, Ph.D.; HIPP director Marcela Brissova, Ph.D. and HIPP scientist Yasminye Pettway, Ph.D.; and HIGI director Anna Gloyn, D.Phil.
This research was supported in part by NIDDK and the National Institute of General Medical Sciences, through grants UC4DK098085, U24DK098085, F30DK134041, R01DK129469, T32GM007347 and T32GM152284.
The NIDDK, a component of the NIH, conducts and supports research on diabetes and other endocrine and metabolic diseases; digestive diseases, nutrition, and obesity; and kidney, urologic, and hematologic diseases. Spanning the full spectrum of medicine and afflicting people of all ages and ethnic groups, these diseases encompass some of the most common, severe and disabling conditions affecting Americans. For more information about the NIDDK and its programs, see https://www.niddk.nih.gov.
About the National Institutes of Health (NIH): NIH, the nation’s medical research agency, includes 27 Institutes and Centers and is a component of the U.S. Department of Health and Human Services. NIH is the primary federal agency conducting and supporting basic, clinical, and translational medical research, and is investigating the causes, treatments, and cures for both common and rare diseases. For more information about NIH and its programs, visit www.nih.gov.
NIH…Turning Discovery Into Health®
Reference
C Evans-Molina YD Pettway, DC Saybders, et al. Heterogeneous endocrine cell composition defines human islet functional phenotypes. Nature Communications. DOI: 10.1038/s41467-026-70689-5.














Leave a Reply