Patient samples
The experimental procedure presented here was approved by the Ethics Committee of Aachen University Hospital (Permission No: EK150/09, Oct. 6, 2009). For minors, informed consent was obtained from the parent(s) and/or legal guardian for study participation. All adult participants gave written informed consent prior to having venous blood samples taken. All methods were performed in accordance with the relevant guidelines and regulations.
Only neonates delivered spontaneously and showing no signs of infection, were accepted for this study. Health status was determined by examination of white blood cell count, Interleukin-6 (IL-6), C-reactive protein and clinical status. The results of clinical investigations were discussed with gynecologists and midwives before taking samples. Immediately after cord ligation, umbilical cord blood samples were placed in heparin-coated tubes (10 IU/ml blood). Neither mothers in whom the amnion had become infected or labour had been prolonged (> 12 h) nor SGA neonates (small for gestational age) nor preterm infants before 36 weeks of gestation were accepted for the study.
In this study we collected umbilical cord blood from 25 donors in total. Additionally, we collected sex matched samples from 30 adult donors.
Reagents and antibodies
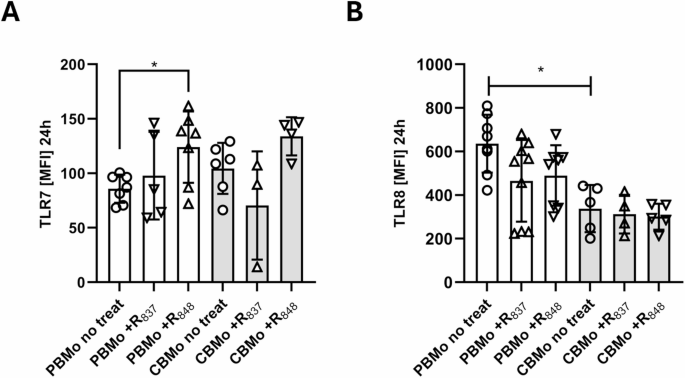
Primary antibodies were purchased by Thermo-Fisher-Scientific (NRW, Germany). The surface antigen antibodies anti-CD14 (clone MEM-18, #21270143, Immunotools, Niedersachsen, Germany), anti-CD32 (clone 3D3, #552884, Becton Dickinson, Switzerland) have been used according to the manufacturers` recommendations. Antibodies specific for intracellular epitopes, namely, anti-IFN-α (clone 7N4-1, 1:100 diluted, #560088, Becton–Dickinson, Switzerland), anti-IFN-ß (clone A1(IFNb), 1:200 diluted, #BMS1044FL), anti-TLR8 (clone 44C143, 1:200 diluted, #MA5-16194), anti-TLR7 (clone 533707, 1:100 diluted,, R&D, Minnesota, USA), anti-IRF7 (clone 23GB2925,1:100 diluted, # MA5-52510), anti-phospho-IRF7 (Ser 470, Ser 471, 1:100 diluted, # BS-3196R), anti-phospho P38 (clone 4NIT4KK, Thr 180, Tyr 182, 1:500 diluted, # 25-9078-42), anti TNF-α (clone Mab11, 1:100 diluted,. # 404-7349-42), anti-NFĸB P65 (clone D14E12, 1:500 diluted, #8242, Cell Signaling Technology, Leiden, the Netherlands) and anti-phospho-NFĸB P65 (clone 93H1, Ser 536, 1:1000 diluted, #3033, Cell Signaling Technology, Leiden, the Netherlands),
The P38/JNK-2 inhibitor Birb 796 (Doramapimod, # S1574, Sellekchem, NRW, Germany) was added at a final concentration of 1 µM40,41. Propidiumiodide (PI, BMS500PI), Kanamycin (J67354.AD, 50 mg/ml) and IPTG (R1171) was purchased fromThermo-Fisher-Scientific (NRW, Germany). LPS was from Thermo Fisher Scientific (00-4976-93, NRW, Germany), Pam3Cys and poly I:C (pIC) from InvivoGen (Toulouse, France, tlrl-pms, vac-pic).
Isolation of mononuclear cells
For this study we collected umbilical blood from 25 donors. Adult blood samples were taken from 30 donors, which were sex-matched and aged 20–50 years. Mononuclear cells were isolated by Ficoll density gradient from whole blood samples and cultivated in RPMI 1640 medium (Gibco, Thermo-Fisher-Scientific, NRW, Germany) supplemented with FCS (Gibco, Thermo-Fisher-Scientific, NRW, Germany, 10% v/v) under standard conditions as already published33,42. In this medium, we seeded 1 × 106 cells/ml prior to stimulation.
Mononuclear cells were designated as PBMc and CBMc, respectively. Both were used in ELISA assays. For FACS based assays Mo were gated from the mononuclear population and designated as PBMo and CBMo respectively (Supplementary Fig. 1). The accuracy of the Mo gate was checked by CD14 staining (Supplementary Fig. 1).
Stimulation of PBMc and CBMc and treatment with inhibitors
Activation of TLR7/8 was initiated for an interval of 4 to 24 h with imiquimod (R837) and resiquimod (R848), respectively. Both base analogues were purchased from Invivogen (Toulouse, France, tlrl-imqs-1, tlrl-r848-1). With exception of the dose kinetic studies (Supplementary Fig. 3), we used 500 µM imiquimod (R837) and 40 µM resiquimod (R848). To compare stimulation efficacy, PBMc and CBMc were also stimulated with LPS (50 ng/ml), Pam3Cys (1 µg/ml) or pIC (100 ng/ml). The inhibitor Birb 796 (Birb) was added 30 min before TLR7/8 stimulation. Samples with Birb 796 were compared to a control using DMSO. For modulating the TLR7/8 stimulation, poly d:T (pdT, #tlrlr-pt17, Invivogen, Toulouse, France) was added together with R837 and R848 in a final concentration of 0.5 µM 13,16. Indicated samples were infected with E. coli with or without combination of imiquimod (R837) and resiquimod (R848) for an additional 24 h. Cells were fixed in final concentration of 1% paraformaldehyde (PFA) in PBS for 1 h and permeabilized with permeabilization buffer (#0083356, Thermo Fisher Scientific NRW, Germany). Afterwards cells were washed with the permeabilization buffer and subsequent primary antibody incubation took place at room temperature (RT). For intracellular staining with non-fluorochrome labelled primary antibodies, cells were permeabilized and stained as described above. The secondary anti-rabbit antibody (Fab fragment #A66788, Thermo Fisher Scientific NRW, Germany) was added in a dilution of 1:500 and incubated for additional 30 min at RT. Prior to FACS analysis all samples were washed once with PBS.
ELISA
The following ELISA kits, namely IL-6 ELISA (#31670069 Immunotools, Niedersachsen, Germany), IL-10 ELISA (#BMS215-2TEN, Thermo-Fisher-Scientific, NRW, Germany), TNF-α ELISA (#BMS223-4, Thermo-Fisher-Scientific, NRW, Germany) and IFN-α ELISA (Invitrogen) were used according to the manufacturers` recommendations.
Phagocytosis assay
The phagocytosis index (CD14+GFP+ Mo %: CD14+ Mo %) and the phagocytic capacity (mean fluorescent intensity (MFI) of CD14+ monocytes) were assessed by flow cytometry after 24 h p.i. (post infection). The cultivation of E. coli and the genetic background is published elsewhere42. The E. coli strain DH5α, an encapsulated K12 laboratory strain, carrying the green fluorescent protein (gfp)-mut2 gene (E. coli-GFP) was a gift of Prof. Dr. C. Gille (University of Heidelberg). In brief, we cultivated GFP expressing E.coli in LB-medium, supplemented with Kanamycine (50 µg/ml) and IPTG (1 mmol/l). Growth was allowed until an OD of 600 was reached. Bacterial suspension was centrifuged at 300xg for 10 min and the pellet resuspended in PBS. For bacterial phagocytosis assays a multiplicity of infection of 25 (MOI 25) was utilized.
Neutralization assay
In brief, E. coli infection took place for 1 h at standard conditions. Afterwards, the medium was replaced by medium containing kanamycine (50 µg/ml) for additional 3 h. PBMc and CBMc were washed in PBS prior to disruption by addition of 0.1%v/v Triton-X 100 and vigorous shaking. After centrifugation, the pellet was resuspended in LB and applied to LB agar plates (PO5309A, Thermo-Fisher-Scientific, NRW, Germany). After 24 h at 37 °C plaques were counted.
Immunoblot analysis
For checking FACS based intracellular staining, we performed immunoblot analysis for NFĸB P65 / phospho-NFĸB P65 expression. To this end, we removed lymphocytes from PBMc and CBMc cultures by using a negative selection kit from Mylteni Biotech (Mylteni Biotech, NRW, Germany). In brief, isolated PBMc and CBMc were incubated with a magnetic-bead coupled mix of antibodies specifically binding to T-cells, B-cells, dendritic cells and NK-cells according to protocols published by the supplier (Mylteni Biotech, NRW, Germany). These cells remained bound to a matrix, whereas the flow through contained Mo with a purity > 90% (See also Supplementary Fig. 1). Afterwards, the purified Mo were washed twice in PBS before being lysed with RIPA buffer (150 mM Sodium chloride, 1% v/v NP-40, 50 mM pH 8 Tris/HCl, 0,5% v/v Sodium deoxycholate, 0,1% v/v SDS). The crude cell lysate was centrifuged two times at 4 °C. The 4-time concentrated Laemmli buffer was added to the supernatants. After boiling for three minutes, the lysates were deployed to SDS-PAGE and transferred to nitrocellulose membranes (according to standard protocols of Lämmli and Khyse Anderson). For imaging and quantification, a LAS 3000 imager (Fujifilm, NRW, Germany) combined with the Multi-Gauge software (Fujifilm, Düsseldorf, Germany) was used.
Detection of apoptosis by hypodiploid nuclei
Hypodiploid nuclei were assessed according to the method described by Nicoletti et al.43. CBMC and PBMc were isolated and preincubated with bacteria for 1 h. After removal of free bacteria, CBMc and PBMc were incubated for 24 h. We then slowly re-suspended the CBMc and PBMc in 2 ml of − 20 °C ethanol 70%, with continuous vortexing, followed by storage for 4 h at − 20 °C. Cells were washed twice in PBS, re-suspended in 50 μl PBS containing 13 kunitz units RNase (EN0531, DNase-free, Thermo-Fisher-Scientific, NRW, Germany), and incubated for 15 min at 37 °C. We then added 180 μl of PI solution (70 μg/ml in PBS). The analysis was done within 20 min of incubation.
Statistical analysis
Results are expressed as mean + /- standard deviation. Error bars represent standard deviations (SD). The pilot experiments in Supplemental Fig. 3 show mean values and standard error of mean (SEM). Values of p < 0.05 were considered significant. Analyses were done with statistical software performing student`s t-test and two-way ANOVA. Experiments where N = 3 were tested according to Mann–Whitney for significant difference. Data which did not pass a test for Gaussian distribution were tested with a Kolmogorov–Smirnov test as provided by Graph Prism Pad Software Statistical Package, La Jolla, CA 92037 USA.












Leave a Reply