Andersen, B. M. et al. Barcoded viral tracing identifies immunosuppressive astrocyte–glioma interactions. Nature 644, 1097–1106 (2025).
Faust Akl, C. et al. Glioblastoma-instructed astrocytes suppress tumour-specific T cell immunity. Nature 643, 219–229 (2025).
Perelroizen, R. et al. Astrocyte immunometabolic regulation of the tumour microenvironment drives glioblastoma pathogenicity. Brain 145, 3288–3307 (2022).
Priego, N. et al. TIMP1 mediates astrocyte-dependent local immunosuppression in brain metastasis acting on infiltrating CD8+ T cells. Cancer Discov. 15, 179–201 (2025).
Priego, N. et al. STAT3 labels a subpopulation of reactive astrocytes required for brain metastasis. Nat. Med. 24, 1024–1035 (2018).
Heiland, D. H. et al. Tumor-associated reactive astrocytes aid the evolution of immunosuppressive environment in glioblastoma. Nat. Commun. 10, 2541 (2019).
Cui, S., Guan, F., Li, X., Long, X. & Wu, M. Astrocytes in glioblastoma tumor microenvironment. Biochim. Biophys. Acta Rev. Cancer 1881, 189518 (2026).
Mellinghoff, I. K. et al. Vorasidenib in IDH1- or IDH2-mutant low-grade glioma. N. Engl. J. Med. 389, 589–601 (2023).
Rajaratnam, V. et al. Glioblastoma: pathogenesis and current status of chemotherapy and other novel treatments. Cancers 12, 937 (2020).
Aizer, A. A. et al. Brain metastases: a Society for Neuro-Oncology (SNO) consensus review on current management and future directions. Neuro Oncol. 24, 1613–1646 (2022).
Bunse, L., Bunse, T., Kilian, M., Quintana, F. J. & Platten, M. The immunology of brain tumors. Sci. Immunol. 10, eads0449 (2025).
Schonfeld, E., Choi, J., Tran, A., Kim, L. H. & Lim, M. The landscape of immune checkpoint inhibitor clinical trials in glioblastoma: a systematic review. Neurooncol. Adv. 6, vdae174 (2024).
Fares, J., Ulasov, I., Timashev, P. & Lesniak, M. S. Emerging principles of brain immunology and immune checkpoint blockade in brain metastases. Brain 144, 1046–1066 (2021).
Escartin, C. et al. Reactive astrocyte nomenclature, definitions, and future directions. Nat. Neurosci. 24, 312–325 (2021).
Lee, H.-G., Wheeler, M. A. & Quintana, F. J. Function and therapeutic value of astrocytes in neurological diseases. Nat. Rev. Drug Discov. 21, 339–358 (2022).
Lee, H.-G. et al. Disease-associated astrocyte epigenetic memory promotes CNS pathology. Nature 627, 865–872 (2024).
Burda, J. E. et al. Divergent transcriptional regulation of astrocyte reactivity across disorders. Nature 606, 557–564 (2022).
Rothhammer, V. et al. Microglial control of astrocytes in response to microbial metabolites. Nature 557, 724–728 (2018).
Linnerbauer, M. et al. PD-L1 positive astrocytes attenuate inflammatory functions of PD-1 positive microglia in models of autoimmune neuroinflammation. Nat. Commun. 14, 5555 (2023).
Karimi, E. et al. Single-cell spatial immune landscapes of primary and metastatic brain tumours. Nature 614, 555–563 (2023).
Greenwald, A. C. et al. Integrative spatial analysis reveals a multi-layered organization of glioblastoma. Cell 187, 2485–2501 (2024).
Campbell, S. C. et al. Potassium and glutamate transport is impaired in scar-forming tumor-associated astrocytes. Neurochem. Int. 133, 104628 (2020).
Ghosh, M. et al. Identification and characterization of tumor-associated astrocyte subpopulations and their interactions with the tumor microenvironment in experimental glioblastomas. PLoS Biol. 23, e3002893 (2025).
Yang, G. et al. Glioblastoma exploits ATP from leading-edge astrocytes to fuel its infiltrative growth revealed by spatially resolved chimeric analysis. Sci. Adv. 11, eadv5673 (2025).
Watkins, S. et al. Disruption of astrocyte–vascular coupling and the blood–brain barrier by invading glioma cells. Nat. Commun. 5, 4196 (2014).
Grimaldi, A. et al. Kv1.3 activity perturbs the homeostatic properties of astrocytes in glioma. Sci. Rep. 8, 7654 (2018).
Ballestín, A., Armocida, D., Ribecco, V. & Seano, G. Peritumoral brain zone in glioblastoma: biological, clinical and mechanical features. Front. Immunol. 15, 1347877 (2024).
Berghoff, A. S. et al. Invasion patterns in brain metastases of solid cancers. Neuro Oncol. 15, 1664–1672 (2013).
Valiente, M. et al. Serpins promote cancer cell survival and vascular co-option in brain metastasis. Cell 156, 1002–1016 (2014).
Fitzgerald, D. P. et al. Reactive glia are recruited by highly proliferative brain metastases of breast cancer and promote tumor cell colonization. Clin. Exp. Metastasis 25, 799–810 (2008).
Lorger, M. & Felding-Habermann, B. Capturing changes in the brain microenvironment during initial steps of breast cancer brain metastasis. Am. J. Pathol. 176, 2958–2971 (2010).
Sierra, A. et al. Astrocyte-derived cytokines contribute to the metastatic brain specificity of breast cancer cells. Lab. Invest. 77, 357–368 (1997).
Marchetti, D., Li, J. & Shen, R. Astrocytes contribute to the brain-metastatic specificity of melanoma cells by producing heparanase. Cancer Res. 60, 4767–4770 (2000).
Andersen, B. M. et al. Glial and myeloid heterogeneity in the brain tumour microenvironment. Nat. Rev. Cancer 21, 786–802 (2021).
Mega, A. et al. Astrocytes enhance glioblastoma growth. Glia 68, 316–327 (2020).
Kim, S.-J. et al. Astrocytes upregulate survival genes in tumor cells and induce protection from chemotherapy. Neoplasia 13, 286–298 (2011).
Genc, A. et al. Connexin 43 inhibitors reduce cell viability in glioblastoma and astrocyte co-culture systems. Mol. Biol. Rep. 52, 803 (2025).
Sin, W. C. et al. Astrocytes promote glioma invasion via the gap junction protein connexin43. Oncogene 35, 1504–1516 (2016).
Chen, Q. et al. Carcinoma–astrocyte gap junctions promote brain metastasis by cGAMP transfer. Nature 533, 493–498 (2016).
Hong, X., Sin, W. C., Harris, A. L. & Naus, C. C. Gap junctions modulate glioma invasion by direct transfer of microRNA. Oncotarget 6, 15566–15577 (2015).
Lin, Q. et al. Reactive astrocytes protect melanoma cells from chemotherapy by sequestering intracellular calcium through gap junction communication channels. Neoplasia 12, 748–754 (2010).
Zhang, W. et al. Direct gap junction communication between malignant glioma cells and astrocytes. Cancer Res. 59, 1994–2003 (1999).
Watson, D. C. et al. GAP43-dependent mitochondria transfer from astrocytes enhances glioblastoma tumorigenicity. Nat. Cancer 4, 648–664 (2023).
Valdebenito, S. et al. Tunneling nanotubes, TNT, communicate glioblastoma with surrounding non-tumor astrocytes to adapt them to hypoxic and metabolic tumor conditions. Sci. Rep. 11, 14556 (2021).
Zhang, L. et al. Microenvironment-induced PTEN loss by exosomal microRNA primes brain metastasis outgrowth. Nature 527, 100–104 (2015).
Villa, G. R. et al. An LXR–cholesterol axis creates a metabolic co-dependency for brain cancers. Cancer Cell 30, 683–693 (2016).
Zou, Y. et al. Polyunsaturated fatty acids from astrocytes activate PPARγ signaling in cancer cells to promote brain metastasis. Cancer Discov. 9, 1720–1735 (2019).
Wurm, J. et al. Astrogliosis releases pro-oncogenic chitinase 3-like 1 causing MAPK signaling in glioblastoma. Cancers 11, 1437 (2019).
Okolie, O. et al. Reactive astrocytes potentiate tumor aggressiveness in a murine glioma resection and recurrence model. Neuro Oncol. 18, 1622–1633 (2016).
Malone, K. et al. Astrocytes and the tumor microenvironment inflammatory state dictate the killing of glioblastoma cells by Smac mimetic compounds. Cell Death Dis. 15, 592 (2024).
Qu, F. et al. Crosstalk between small-cell lung cancer cells and astrocytes mimics brain development to promote brain metastasis. Nat. Cell Biol. 25, 1506–1519 (2023).
Oushy, S. et al. Glioblastoma multiforme-derived extracellular vesicles drive normal astrocytes towards a tumour-enhancing phenotype. Philos. Trans. R. Soc. Lond. B Biol. Sci. 373, 20160477 (2018).
Taheri, B., Soleimani, M., Aval, S. F., Memari, F. & Zarghami, N. C6 glioma-derived microvesicles stimulate the proliferative and metastatic gene expression of normal astrocytes. Neurosci. Lett. 685, 173–178 (2018).
Colangelo, N. W. & Azzam, E. I. Extracellular vesicles originating from glioblastoma cells increase metalloproteinase release by astrocytes: the role of CD147 (EMMPRIN) and ionizing radiation. Cell Commun. Signal. 18, 21 (2020).
Tang, Z. et al. Inhibition of hypoxic exosomal miR-423-3p decreases glioma progression by restricting autophagy in astrocytes. Cell Death Dis. 16, 265 (2025).
Thompson, C. E. et al. MiR-146b-5p decreases cytokine release from astrocytes and preserves oligodendrocyte progenitor cell complexity during inflammation. Eur. J. Neurosci. 62, e70248 (2025).
Loeffler, S., Fayard, B., Weis, J. & Weissenberger, J. Interleukin-6 induces transcriptional activation of vascular endothelial growth factor (VEGF) in astrocytes in vivo and regulates VEGF promoter activity in glioblastoma cells via direct interaction between STAT3 and Sp1. Int. J. Cancer 115, 202–213 (2005).
Seike, T. et al. Interaction between lung cancer cells and astrocytes via specific inflammatory cytokines in the microenvironment of brain metastasis. Clin. Exp. Metastasis 28, 13–25 (2011).
Gui, Y. et al. Glioma–astrocyte connexin43 confers temozolomide resistance through activation of the E2F1/ERCC1 axis. Neuro Oncol. 27, 711–726 (2025).
Yang, N. et al. A co-culture model with brain tumor-specific bioluminescence demonstrates astrocyte-induced drug resistance in glioblastoma. J. Transl. Med. 12, 278 (2014).
Pustchi, S. E., Avci, N. G., Akay, Y. M. & Akay, M. Astrocytes decreased the sensitivity of glioblastoma cells to temozolomide and Bay 11-7082. Int. J. Mol. Sci. 21, 7154 (2020).
Kim, S. W. et al. Role of the endothelin axis in astrocyte- and endothelial cell-mediated chemoprotection of cancer cells. Neuro Oncol. 16, 1585–1598 (2014).
Munoz, J. L. et al. Temozolomide resistance in glioblastoma cells occurs partly through epidermal growth factor receptor-mediated induction of connexin 43. Cell Death Dis. 5, e1145 (2014).
Shabtay-Orbach, A., Amit, M., Binenbaum, Y., Na’ara, S. & Gil, Z. Paracrine regulation of glioma cells invasion by astrocytes is mediated by glial-derived neurotrophic factor. Int. J. Cancer 137, 1012–1020 (2015).
Rath, B. H., Fair, J. M., Jamal, M., Camphausen, K. & Tofilon, P. J. Astrocytes enhance the invasion potential of glioblastoma stem-like cells. PLoS ONE 8, e54752 (2013).
Herrera-Perez, R. M. et al. Presence of stromal cells in a bioengineered tumor microenvironment alters glioblastoma migration and response to STAT3 inhibition. PLoS ONE 13, e0194183 (2018).
Chen, W. et al. Human astrocytes secrete IL-6 to promote glioma migration and invasion through upregulation of cytomembrane MMP14. Oncotarget 7, 62425–62438 (2016).
Gagliano, N. et al. Glioma–astrocyte interaction modifies the astrocyte phenotype in a co-culture experimental model. Oncol. Rep. 22, 1349–1356 (2009).
Le, D. M. et al. Exploitation of astrocytes by glioma cells to facilitate invasiveness: a mechanism involving matrix metalloproteinase-2 and the urokinase-type plasminogen activator–plasmin cascade. J. Neurosci. 23, 4034–4043 (2003).
Kim, J.-K. et al. Tumoral RANKL activates astrocytes that promote glioma cell invasion through cytokine signaling. Cancer Lett. 353, 194–200 (2014).
Lu, Y. et al. TGF-β1 promotes motility and invasiveness of glioma cells through activation of ADAM17. Oncol. Rep. 25, 1329–1335 (2011).
Dankner, M. et al. Invasive growth of brain metastases is linked to CHI3L1 release from pSTAT3-positive astrocytes. Neuro Oncol. 26, 1052–1066 (2024).
Huang, G. et al. Role of GPX3+ astrocytes in breast cancer brain metastasis activated by circulating tumor cell exosomes. npj Precis. Oncol. 9, 64 (2025).
Doron, H. et al. Inflammatory activation of astrocytes facilitates melanoma brain tropism via the CXCL10–CXCR3 signaling axis. Cell Rep. 28, 1785–1798 (2019).
Gril, B. et al. Reactive astrocytic S1P3 signaling modulates the blood–tumor barrier in brain metastases. Nat. Commun. 9, 2705 (2018).
Ishibashi, K. et al. Astrocyte-induced mGluR1 activates human lung cancer brain metastasis via glutamate-dependent stabilization of EGFR. Dev. Cell 59, 579–594 (2024).
Xing, F. et al. Reactive astrocytes promote the metastatic growth of breast cancer stem-like cells by activating Notch signalling in brain. EMBO Mol. Med. 5, 384–396 (2013).
Jandial, R., Choy, C., Levy, D. M., Chen, M. Y. & Ansari, K. I. Astrocyte-induced Reelin expression drives proliferation of Her2+ breast cancer metastases. Clin. Exp. Metastasis 34, 185–196 (2017).
Monteiro, C. et al. Stratification of radiosensitive brain metastases based on an actionable S100A9/RAGE resistance mechanism. Nat. Med. 28, 752–765 (2022).
Mészáros, A. et al. Inflammasome activation in peritumoral astrocytes is a key player in breast cancer brain metastasis development. Acta Neuropathol. Commun. 11, 155 (2023).
Serpe, C. et al. Astrocytes-derived small extracellular vesicles hinder glioma growth. Biomedicines 10, 2952 (2022).
Diep, Y. N. et al. A NEuroimmune-Oncology Microphysiological Analysis Platform (NEO-MAP) for evaluating astrocytic scar formation and microgliosis in glioblastoma microenvironment. Adv. Healthc. Mater. 14, e2404821 (2025).
Silver, D. J. et al. Chondroitin sulfate proteoglycans potently inhibit invasion and serve as a central organizer of the brain tumor microenvironment. J. Neurosci. 33, 15603–15617 (2013).
Yao, P.-S. et al. Glutamate/glutamine metabolism coupling between astrocytes and glioma cells: neuroprotection and inhibition of glioma growth. Biochem. Biophys. Res. Commun. 450, 295–299 (2014).
Rosito, M. et al. Ketogenic diet induces an inflammatory reactive astrocytes phenotype reducing glioma growth. Cell. Mol. Life Sci. 82, 73 (2025).
Zhang, C.-B., Wang, Z.-L., Liu, H.-J., Wang, Z. & Jia, W. Characterization of tumor-associated reactive astrocytes in gliomas by single-cell and bulk tumor sequencing. Front. Neurol. 14, 1193844 (2023).
Katz, A. M. et al. Astrocyte-specific expression patterns associated with the PDGF-induced glioma microenvironment. PLoS ONE 7, e32453 (2012).
Rigg, E. et al. Inhibition of extracellular vesicle-derived miR-146a-5p decreases progression of melanoma brain metastasis via Notch pathway dysregulation in astrocytes. J. Extracell. Vesicles 12, e12363 (2023).
Klein, A. et al. Astrocytes facilitate melanoma brain metastasis via secretion of IL-23. J. Pathol. 236, 116–127 (2015).
Ji, J. et al. Radiotherapy-induced astrocyte senescence promotes an immunosuppressive microenvironment in glioblastoma to facilitate tumor regrowth. Adv. Sci. 11, e2304609 (2024).
Yang, F. et al. Synergistic immunotherapy of glioblastoma by dual targeting of IL-6 and CD40. Nat. Commun. 12, 3424 (2021).
Gong, X. et al. Interaction of tumor cells and astrocytes promotes breast cancer brain metastases through TGF-β2/ANGPTL4 axes. npj Precis. Oncol. 3, 24 (2019).
Tang, M. et al. Brain metastasis from EGFR-mutated non-small cell lung cancer: secretion of IL11 from astrocytes up-regulates PDL1 and promotes immune escape. Adv. Sci. 11, e2306348 (2024).
Chen, Z. et al. A paracrine circuit of IL-1β/IL-1R1 between myeloid and tumor cells drives genotype-dependent glioblastoma progression. J. Clin. Invest. 133, e163802 (2023).
Ji, H. et al. IL-18, a therapeutic target for immunotherapy boosting, promotes temozolomide chemoresistance via the PI3K/AKT pathway in glioma. J. Transl. Med. 22, 951 (2024).
Sanmarco, L. M. et al. Gut-licensed IFNγ+ NK cells drive LAMP1+TRAIL+ anti-inflammatory astrocytes. Nature 590, 473–479 (2021).
Steelman, A. J., Smith, R. 3rd, Welsh, C. J. & Li, J. Galectin-9 protein is up-regulated in astrocytes by tumor necrosis factor and promotes encephalitogenic T-cell apoptosis. J. Biol. Chem. 288, 23776–23787 (2013).
Castello-Pons, M. et al. VP3.15, a dual GSK-3β/PDE7 inhibitor, reduces glioblastoma tumor growth though changes in the tumor microenvironment in a PTEN wild-type context. Neurotherapeutics 22, e00576 (2025).
Chahlavi, A. et al. Glioblastomas induce T-lymphocyte death by two distinct pathways involving gangliosides and CD70. Cancer Res. 65, 5428–5438 (2005).
Schachtele, S. J., Hu, S., Sheng, W. S., Mutnal, M. B. & Lokensgard, J. R. Glial cells suppress postencephalitic CD8+ T lymphocytes through PD-L1. Glia 62, 1582–1594 (2014).
Vardjan, N. et al. IFN-γ-induced increase in the mobility of MHC class II compartments in astrocytes depends on intermediate filaments. J. Neuroinflammation 9, 144 (2012).
Abdel-Haq, N., Hao, H. N. & Lyman, W. D. Cytokine regulation of CD40 expression in fetal human astrocyte cultures. J. Neuroimmunol. 101, 7–14 (1999).
Smith, B. C. et al. Spatial protein profiling reveals active roles for astrocytes in the chronic active lesion core during multiple sclerosis. Acta Neuropathol. 150, 47 (2025).
Li, S. et al. Microglia mediated by LAG3 regulate astrocyte morphogenesis in the brain white matter. Cell Rep. 44, 116112 (2025).
Miska, J. et al. Anti-GITR therapy promotes immunity against malignant glioma in a murine model. Cancer Immunol. Immunother. 65, 1555–1567 (2016).
Yuzhalin, A. E. et al. Astrocyte-induced Cdk5 expedites breast cancer brain metastasis by suppressing MHC-I expression to evade immune recognition. Nat. Cell Biol. 26, 1773–1789 (2024).
Wheeler, M. A. et al. Droplet-based forward genetic screening of astrocyte–microglia cross-talk. Science 379, 1023–1030 (2023).
Ma, W. et al. Type I interferon response in astrocytes promotes brain metastasis by enhancing monocytic myeloid cell recruitment. Nat. Commun. 14, 2632 (2023).
Rothhammer, V. et al. Type I interferons and microbial metabolites of tryptophan modulate astrocyte activity and central nervous system inflammation via the aryl hydrocarbon receptor. Nat. Med. 22, 586–597 (2016).
Liu, L. et al. Blocking the MIF–CD74 axis augments radiotherapy efficacy for brain metastasis in NSCLC via synergistically promoting microglia M1 polarization. J. Exp. Clin. Cancer Res. 43, 128 (2024).
Steelman, A. J. & Li, J. Astrocyte galectin-9 potentiates microglial TNF secretion. J. Neuroinflammation 11, 144 (2014).
Kang, S. et al. Glioblastoma shift from bulk to infiltrative growth is guided by plexin-B2-mediated microglia alignment in invasive niches. Nat. Cancer 6, 1505–1523 (2025).
Clark, I. C. et al. Barcoded viral tracing of single-cell interactions in central nervous system inflammation. Science 372, eabf1230 (2021).
Kim, S.-J. et al. Macitentan, a dual endothelin receptor antagonist, in combination with temozolomide leads to glioblastoma regression and long-term survival in mice. Clin. Cancer Res. 21, 4630–4641 (2015).
Demers, M. et al. Solid peripheral tumor leads to systemic inflammation, astrocyte activation and signs of behavioral despair in mice. PLoS ONE 13, e0207241 (2018).
Gril, B. et al. Pazopanib inhibits the activation of PDGFRβ-expressing astrocytes in the brain metastatic microenvironment of breast cancer cells. Am. J. Pathol. 182, 2368–2379 (2013).
Gril, B. et al. Pazopanib reveals a role for tumor cell B-Raf in the prevention of HER2+ breast cancer brain metastasis. Clin. Cancer Res. 17, 142–153 (2011).
Chen, Z. et al. Doxorubicin–polyglycerol–nanodiamond conjugates disrupt STAT3/IL-6-mediated reciprocal activation loop between glioblastoma cells and astrocytes. J. Control. Release 320, 469–483 (2020).
Tinkey, R. A., Smith, B. C., Habean, M. L. & Williams, J. L. BATF2 is a regulator of interferon-γ signaling in astrocytes during neuroinflammation. Cell Rep. 44, 115393 (2025).
Wheeler, M. A. et al. MAFG-driven astrocytes promote CNS inflammation. Nature 578, 593–599 (2020).
Ren, Y. et al. Spatial transcriptomics reveals niche-specific enrichment and vulnerabilities of radial glial stem-like cells in malignant gliomas. Nat. Commun. 14, 1028 (2023).
Soni, N. et al. Single-cell dissection of the genotype–immunophenotype relationship in glioblastoma. Brain 148, 3153–3169 (2025).
Tagore, S. et al. Single-cell and spatial genomic landscape of non-small cell lung cancer brain metastases. Nat. Med. 31, 1351–1363 (2025).
Ha, W. et al. Ibudilast sensitizes glioblastoma to temozolomide by targeting macrophage migration inhibitory factor (MIF). Sci. Rep. 9, 2905 (2019).
Contreras-Zárate, M. J. et al. Estradiol induces BDNF/TrkB signaling in triple-negative breast cancer to promote brain metastases. Oncogene 38, 4685–4699 (2019).
Kanwore, K. et al. Testosterone upregulates glial cell line-derived neurotrophic factor (GDNF) and promotes neuroinflammation to enhance glioma cell survival and proliferation. Inflamm. Regen. 43, 49 (2023).
Arvanitis, C. D., Ferraro, G. B. & Jain, R. K. The blood–brain barrier and blood–tumour barrier in brain tumours and metastases. Nat. Rev. Cancer 20, 26–41 (2020).
van Bussel, M. T. J., Beijnen, J. H. & Brandsma, D. Intracranial antitumor responses of nivolumab and ipilimumab: a pharmacodynamic and pharmacokinetic perspective, a scoping systematic review. BMC Cancer 19, 519 (2019).
Edavettal, S. et al. Enhanced delivery of antibodies across the blood–brain barrier via TEMs with inherent receptor-mediated phagocytosis. Med 3, 860–882 (2022).
Huang, Q. et al. An AAV capsid reprogrammed to bind human transferrin receptor mediates brain-wide gene delivery. Science 384, 1220–1227 (2024).
von Roemeling, C. A. et al. Adeno-associated virus delivered CXCL9 sensitizes glioblastoma to anti-PD-1 immune checkpoint blockade. Nat. Commun. 15, 5871 (2024).
Ling, A. L. et al. Clinical trial links oncolytic immunoactivation to survival in glioblastoma. Nature 623, 157–166 (2023).
Giovannoni, F. et al. Retargeted oncolytic viruses engineered to remodel the tumor microenvironment for glioblastoma immunotherapy. Nat. Cancer 6, 1994–2010 (2025).
Fletcher-Sananikone, E. et al. Elimination of radiation-induced senescence in the brain tumor microenvironment attenuates glioblastoma recurrence. Cancer Res. 81, 5935–5947 (2021).
Berg, T. J. et al. The irradiated brain microenvironment supports glioma stemness and survival via astrocyte-derived transglutaminase 2. Cancer Res. 81, 2101–2115 (2021).
Garbow, J. R. et al. Irradiation-modulated murine brain microenvironment enhances GL261-tumor growth and inhibits anti-PD-L1 immunotherapy. Front. Oncol. 11, 693146 (2021).
Weathers, S.-P. et al. Results of a phase I trial to assess the safety of macitentan in combination with temozolomide for the treatment of recurrent glioblastoma. Neurooncol. Adv. 3, vdab141 (2021).
Hingorani, M., Dixit, S. & Maraveyas, A. Pazopanib-induced regression of brain metastasis after whole brain palliative radiotherapy in metastatic renal cell cancer progressing on first-line sunitinib: a case report. World J. Oncol. 5, 223–227 (2014).





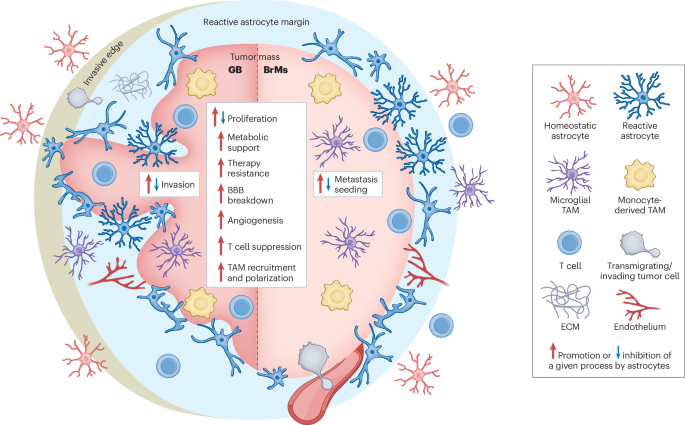

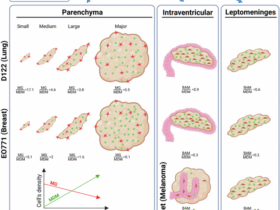
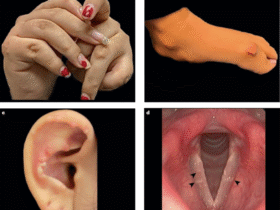





Leave a Reply