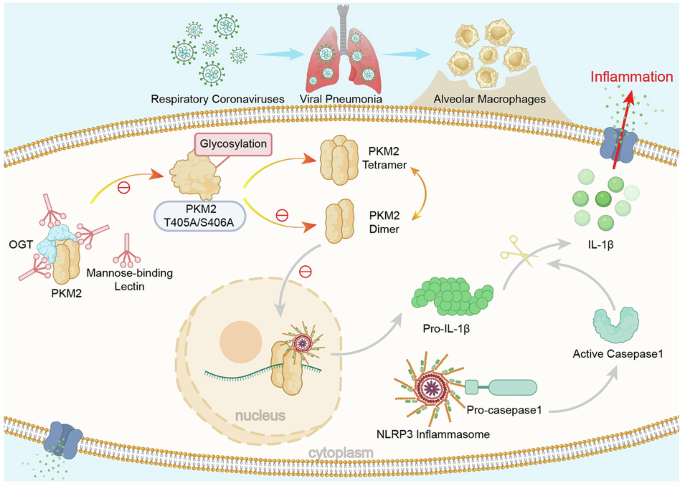
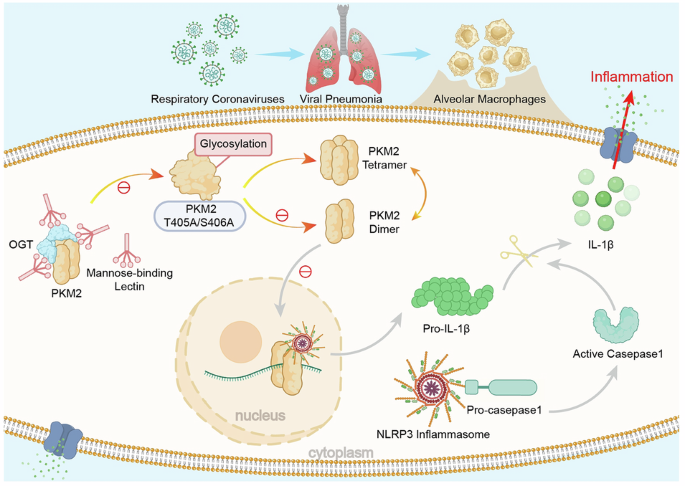
The precise mechanisms governing innate immune response to coronaviruses remain elusive. Our study reveals that mannose-binding lectin (MBL), a key pattern recognition molecule, exhibits significant upregulation in PBMCs from COVID-19 patients, with expression levels positively correlating with clinical severity. Through intranasal coronavirus MHV-A59 challenge in MBL-deficient mice, we demonstrate that macrophage-derived MBL attenuates viral pneumonia by limiting NLRP3 inflammasome hyperactivation. Mechanistically, MBL directly binds pyruvate kinase M2 (PKM2), an interaction significantly enhanced upon viral stimulation. Crucially, MBL competitively inhibits O-GlcNAcylation at PKM2 Thr405/Ser406 residues, thereby disrupting PKM2 tetramer-to-dimer transition and its subsequent nuclear translocation. The MBL-mediated metabolic reprogramming effectively dampens NLRP3 inflammasome activation, with wild-type mice exhibiting markedly reduced caspase-1 cleavage and IL-1β maturation relative to MBL-deficient mice following MHV-A59 infection. Genetic ablation of NLRP3 abolished the differential susceptibility between wild-type and MBL-deficient animals, confirming NLRP3 as the critical downstream effector. Pharmacological inhibition of either PKM2 or NLRP3 restored cytokine homeostasis in virus-infected MBL-deficient mice. These findings establish a previously unrecognized regulatory pathway in which macrophage-specific MBL fine-tunes antiviral immunity through the PKM2-NLRP3 axis. Our work provides crucial insights into the sophisticated interplay between innate immunity and cellular metabolism, laying the groundwork for developing host-directed therapies against coronaviruses.














Leave a Reply