Sharma, P. et al. Immune checkpoint therapy-current perspectives and future directions. Cell 186, 1652–1669 (2023).
Morad, G., Helmink, B. A., Sharma, P. & Wargo, J. A. Hallmarks of response, resistance, and toxicity to immune checkpoint blockade. Cell 184, 5309–5337 (2021).
Wolchok, J. D. et al. Final, 10-year outcomes with nivolumab plus ipilimumab in advanced melanoma. N. Engl. J. Med. 392, 11–22 (2025).
Postow, M. A., Sidlow, R. & Hellmann, M. D. Immune-related adverse events associated with immune checkpoint blockade. N. Engl. J. Med. 378, 158–168 (2018).
Michot, J. M. et al. Immune-related adverse events with immune checkpoint blockade: a comprehensive review. Eur. J. Cancer 54, 139–148 (2016).
Martins, F. et al. Adverse effects of immune-checkpoint inhibitors: epidemiology, management and surveillance. Nat. Rev. Clin. Oncol. 16, 563–580 (2019).
Brahmer, J. R. et al. Society for Immunotherapy of Cancer (SITC) clinical practice guideline on immune checkpoint inhibitor-related adverse events. J. Immunother. Cancer 9, e002435 (2021).
Haanen, J. et al. Management of toxicities from immunotherapy: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol. 33, 1217–1238 (2022).
Schneider, B. J. et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: ASCO guideline update. J. Clin. Oncol. 39, 4073–4126 (2021).
Thompson, J. A. et al. NCCN guidelines® insights: management of immunotherapy-related toxicities, version 2.2024. J. Natl Compr. Canc. Netw. 22, 582–592 (2024).
Pires da Silva, I. et al. Ipilimumab alone or ipilimumab plus anti-PD-1 therapy in patients with metastatic melanoma resistant to anti-PD-(L)1 monotherapy: a multicentre, retrospective, cohort study. Lancet Oncol. 22, 836–847 (2021).
Xing, P. et al. Incidence rates of immune-related adverse events and their correlation with response in advanced solid tumours treated with NIVO or NIVO+IPI: a systematic review and meta-analysis. J. Immunother. Cancer 7, 341 (2019).
Som, A. et al. Immune checkpoint inhibitor-induced colitis: a comprehensive review. World J. Clin. Cases 7, 405–418 (2019).
Andrews, M. C. et al. Gut microbiota signatures are associated with toxicity to combined CTLA-4 and PD-1 blockade. Nat. Med. 27, 1432–1441 (2021). A clinical study of patients with advanced melanoma and high-grade irAEs resulting from combined treatment of anti-CTLA-4 and anti-PD1, which shows increased B. intestinalis and serum IL-1β, findings that were recapitulated in mice colonized with B. intestinalis.
Chaput, N. et al. Baseline gut microbiota predicts clinical response and colitis in metastatic melanoma patients treated with ipilimumab. Ann. Oncol. 28, 1368–1379 (2017). A prospective clinical study in patients with metastatic melanoma treated with anti-CTLA-4 that correlates the composition of the gut microbiota and circulating Treg cells with incidence of ICI colitis.
Dubin, K. et al. Intestinal microbiome analyses identify melanoma patients at risk for checkpoint-blockade-induced colitis. Nat. Commun. 7, 10391 (2016). A prospective clinical study correlating the composition of the gut microbiome with incidence of ICI colitis in patients with metastatic melanoma treated with anti-CTLA-4.
de Moel, E. C. et al. Autoantibody development under treatment with immune-checkpoint inhibitors. Cancer Immunol. Res. 7, 6–11 (2019).
Mathias, K. et al. Association between rheumatic autoantibodies and immune-related adverse events. Oncologist 28, 440–448 (2023).
Tahir, S. A. et al. Autoimmune antibodies correlate with immune checkpoint therapy-induced toxicities. Proc. Natl Acad. Sci. USA 116, 22246–22251 (2019).
Abu-Sbeih, H. et al. Immune checkpoint inhibitor therapy in patients with preexisting inflammatory bowel disease. J. Clin. Oncol. 38, 576–583 (2020).
Johnson, D. B. et al. Ipilimumab therapy in patients with advanced melanoma and preexisting autoimmune disorders. JAMA Oncol. 2, 234–240 (2016).
Menzies, A. M. et al. Anti-PD-1 therapy in patients with advanced melanoma and preexisting autoimmune disorders or major toxicity with ipilimumab. Ann. Oncol. 28, 368–376 (2017).
Berner, F. et al. Association of checkpoint inhibitor-induced toxic effects with shared cancer and tissue antigens in non-small cell lung cancer. JAMA Oncol. 5, 1043–1047 (2019).
Berner, F. et al. Autoreactive napsin A-specific T cells are enriched in lung tumors and inflammatory lung lesions during immune checkpoint blockade. Sci. Immunol. 7, eabn9644 (2022).
Johnson, D. B. et al. Fulminant myocarditis with combination immune checkpoint blockade. N. Engl. J. Med. 375, 1749–1755 (2016).
Iwama, S. et al. Pituitary expression of CTLA-4 mediates hypophysitis secondary to administration of CTLA-4 blocking antibody. Sci. Transl. Med. 6, 230ra245 (2014).
Kurimoto, C. et al. Predictive and sensitive biomarkers for thyroid dysfunctions during treatment with immune-checkpoint inhibitors. Cancer Sci. 111, 1468–1477 (2020).
Lauwyck, J. et al. C-reactive protein as a biomarker for immune-related adverse events in melanoma patients treated with immune checkpoint inhibitors in the adjuvant setting. Melanoma Res. 31, 371–377 (2021).
Lim, S. Y. et al. Circulating cytokines predict immune-related toxicity in melanoma patients receiving anti-PD-1-based immunotherapy. Clin. Cancer Res. 25, 1557–1563 (2019).
Onodera, R. et al. High level of C-reactive protein as a predictive factor for immune-related adverse events of immune checkpoint inhibitors in non-small cell lung cancer: a retrospective study. J. Thorac. Dis. 15, 4237–4247 (2023).
Tarhini, A. A. et al. Baseline circulating IL-17 predicts toxicity while TGF-β1 and IL-10 are prognostic of relapse in ipilimumab neoadjuvant therapy of melanoma. J. Immunother. Cancer 3, 39 (2015).
Valpione, S. et al. Sex and interleukin-6 are prognostic factors for autoimmune toxicity following treatment with anti-CTLA4 blockade. J. Transl. Med. 16, 94 (2018).
Wang, H. W. et al. Interleukin-10 is a promising marker for immune-related adverse events in patients with non-small cell lung cancer receiving immunotherapy. Front. Immunol. 13, 840313 (2022).
Fukushima, T., Kobayashi, S. & Ueno, M. The correlation between immune-related adverse events and efficacy of immune checkpoint inhibitors. Jpn. J. Clin. Oncol. 54, 949–958 (2024).
Wang, Y. et al. Immune-checkpoint inhibitor-induced diarrhea and colitis in patients with advanced malignancies: retrospective review at MD Anderson. J. Immunother. Cancer 6, 37 (2018).
He, J. et al. Patterns of immune-related adverse events and treatment responses in adolescents and young adults with melanoma during initial and rechallenge immune checkpoint inhibitor therapy. J. Immunother. Cancer 13, e012337 (2025).
Hua, C. et al. Association of vitiligo with tumor response in patients with metastatic melanoma treated with pembrolizumab. JAMA Dermatol. 152, 45–51 (2016).
Teulings, H. E. et al. Vitiligo-like depigmentation in patients with stage III-IV melanoma receiving immunotherapy and its association with survival: a systematic review and meta-analysis. J. Clin. Oncol. 33, 773–781 (2015).
Sivan, A. et al. Commensal Bifidobacterium promotes antitumor immunity and facilitates anti-PD-L1 efficacy. Science 350, 1084–1089 (2015).
Vetizou, M. et al. Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science 350, 1079–1084 (2015).
Gopalakrishnan, V. et al. Gut microbiome modulates response to anti-PD-1 immunotherapy in melanoma patients. Science 359, 97–103 (2018). This study establishes associations between the composition of the gut microbiome and ICI response.
Routy, B. et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science 359, 91–97 (2018). This study establishes associations between the composition of the gut microbiome and ICI response.
Hayase, E. & Jenq, R. R. Role of the intestinal microbiome and microbial-derived metabolites in immune checkpoint blockade immunotherapy of cancer. Genome Med. 13, 107 (2021).
Matson, V., Chervin, C. S. & Gajewski, T. F. Cancer and the microbiome-influence of the commensal microbiota on cancer, immune responses, and immunotherapy. Gastroenterology 160, 600–613 (2021).
Baruch, E. N. et al. Fecal microbiota transplant promotes response in immunotherapy-refractory melanoma patients. Science 371, 602–609 (2021). This study reports results from small-scale clinical trials using FMT to improve response to ICI.
Davar, D. et al. Fecal microbiota transplant overcomes resistance to anti-PD-1 therapy in melanoma patients. Science 371, 595–602 (2021). This study reports results from small-scale clinical trials using FMT to improve response to ICI.
Spencer, C. N. et al. Dietary fiber and probiotics influence the gut microbiome and melanoma immunotherapy response. Science 374, 1632–1640 (2021).
Lee, K. A. et al. Cross-cohort gut microbiome associations with immune checkpoint inhibitor response in advanced melanoma. Nat. Med. 28, 535–544 (2022).
Park, E. M. et al. Targeting the gut and tumor microbiota in cancer. Nat. Med. 28, 690–703 (2022).
Matson, V. et al. The commensal microbiome is associated with anti-PD-1 efficacy in metastatic melanoma patients. Science 359, 104–108 (2018).
Hu, Z. I. et al. Immune checkpoint inhibitors unleash pathogenic immune responses against the microbiota. Proc. Natl Acad. Sci. USA 119, e2200348119 (2022).
Zhang, D. et al. Lower respiratory tract microbiome is associated with checkpoint inhibitor pneumonitis in lung cancer patients. Transl. Lung Cancer Res. 13, 3189–3201 (2024).
Huang, R. et al. Microbiota-indole-3-propionic acid-heart axis mediates the protection of leflunomide against αPD1-induced cardiotoxicity in mice. Nat. Commun. 16, 2651 (2025).
Wang, Y., Jenq, R. R., Wargo, J. A. & Watowich, S. S. Microbiome influencers of checkpoint blockade-associated toxicity. J. Exp. Med. 220, e20220948 (2023).
McCulloch, J. A. et al. Intestinal microbiota signatures of clinical response and immune-related adverse events in melanoma patients treated with anti-PD-1. Nat. Med. 28, 545–556 (2022).
Hamada, K. et al. Turicibacter and Acidaminococcus predict immune-related adverse events and efficacy of immune checkpoint inhibitor. Front. Immunol. 14, 1164724 (2023).
Bjork, J. R. et al. Longitudinal gut microbiome changes in immune checkpoint blockade-treated advanced melanoma. Nat. Med. 30, 785–796 (2024).
Halsey, T., Ologun, G., Wargo, J. & Jenq, R. R. Uncovering the role of the gut microbiota in immune checkpoint blockade therapy: a mini-review. Semin. Hematol. 57, 13–18 (2020).
Abu-Sbeih, H. et al. Impact of antibiotic therapy on the development and response to treatment of immune checkpoint inhibitor-mediated diarrhea and colitis. J. Immunother. Cancer 7, 242 (2019).
Jing, Y. et al. Association of antibiotic treatment with immune-related adverse events in patients with cancer receiving immunotherapy. J. Immunother. Cancer 10, e003779 (2022).
Hou, K. et al. Microbiota in health and diseases. Signal Transduct. Target. Ther. 7, 135 (2022).
Dougan, M., Wang, Y., Rubio-Tapia, A. & Lim, J. K. AGA clinical practice update on diagnosis and management of immune checkpoint inhibitor colitis and hepatitis: expert review. Gastroenterology 160, 1384–1393 (2021).
Colli Cruz, C. et al. Gastrointestinal toxicities associated with immune checkpoint inhibitors therapy: risks and management. Immunotherapy 17, 293–303 (2025).
Shatila, M. et al. Disease outcomes in immune-mediated colitis: a study describing initial endoscopic and histologic presentations and treatment options. J. Natl Compr. Canc. Netw. 23, e257046 (2025).
Wang, D. Y., Ye, F., Zhao, S. & Johnson, D. B. Incidence of immune checkpoint inhibitor-related colitis in solid tumor patients: a systematic review and meta-analysis. Oncoimmunology 6, e1344805 (2017).
Eun, Y. et al. Risk factors for immune-related adverse events associated with anti-PD-1 pembrolizumab. Sci. Rep. 9, 14039 (2019).
Wei, S. C. et al. Distinct cellular mechanisms underlie anti-CTLA-4 and anti-PD-1 checkpoint blockade. Cell 170, 1120–1133 (2017).
Chen, J. H., Pezhouh, M. K., Lauwers, G. Y. & Masia, R. Histopathologic features of colitis due to immunotherapy with anti-PD-1 antibodies. Am. J. Surg. Pathol. 41, 643–654 (2017).
Abu-Sbeih, H. et al. Importance of endoscopic and histological evaluation in the management of immune checkpoint inhibitor-induced colitis. J. Immunother. Cancer 6, 95 (2018).
Choi, K. et al. Can immune checkpoint inhibitors induce microscopic colitis or a brand new entity? Inflamm. Bowel Dis. 25, 385–393 (2019).
Wang, Y. et al. Endoscopic and histologic features of immune checkpoint inhibitor-related colitis. Inflamm. Bowel Dis. 24, 1695–1705 (2018).
Geukes Foppen, M. H. et al. Immune checkpoint inhibition-related colitis: symptoms, endoscopic features, histology and response to management. ESMO Open. 3, e000278 (2018).
Burke, K. E. et al. Microscopic colitis. Nat. Rev. Dis. Primers 7, 39 (2021).
Tarar, Z. I. et al. Are drugs associated with microscopic colitis? a systematic review and meta-analysis. Diseases 11, 6 (2022).
Shirwaikar Thomas, A., Hanauer, S. & Wang, Y. Immune checkpoint inhibitor enterocolitis vs idiopathic inflammatory bowel disease. Clin. Gastroenterol. Hepatol. 21, 878–890 (2023).
Zaher, A. et al. Management of refractory checkpoint inhibitor-induced colitis. Expert Opin. Drug Saf. 25, 49–58 (2025).
Abu-Sbeih, H. et al. Resumption of immune checkpoint inhibitor therapy after immune-mediated colitis. J. Clin. Oncol. 37, 2738–2745 (2019).
Badran, Y. R. et al. Concurrent immune checkpoint inhibition and selective immunosuppressive therapy in patients with immune-related enterocolitis. J. Immunother. Cancer 11, e007195 (2023).
Chang, J. T. Pathophysiology of inflammatory bowel diseases. N. Engl. J. Med. 383, 2652–2664 (2020).
Guan, Q. A comprehensive review and update on the pathogenesis of inflammatory bowel disease. J. Immunol. Res. 2019, 7247238 (2019).
Zhou, Y. et al. Intestinal toxicity to CTLA-4 blockade driven by IL-6 and myeloid infiltration. J. Exp. Med. 220, e20221333 (2023). This study reports mouse models of ICI colitis that reflect immune changes identified in ICI colitis biopsy specimens and shows a causal role for dysregulated APCs and a dysbiotic microbiome in the disease, along with alleviation of mouse ICI colitis via anti-IL-6 therapy and microbiome intervention.
Luoma, A. M. et al. Molecular pathways of colon inflammation induced by cancer immunotherapy. Cell 182, 655–671 (2020). Single-cell analysis of colon immune populations in patients with melanoma who developed ICI colitis after treatment with anti-CTLA-4 and anti-PD1 revealed expansion of cytotoxic T cells, myeloid cells and Treg cells and a reduction in TRM cells with disease.
Sasson, S. C. et al. Mucosal-associated invariant T (MAIT) cells are activated in the gastrointestinal tissue of patients with combination ipilimumab and nivolumab therapy-related colitis in a pathology distinct from ulcerative colitis. Clin. Exp. Immunol. 202, 335–352 (2020).
Sasson, S. C. et al. Interferon-γ-producing CD8+ tissue resident memory T cells are a targetable hallmark of immune checkpoint inhibitor-colitis. Gastroenterology 161, 1229–1244 (2021).
Hailemichael, Y. et al. Interleukin-6 blockade abrogates immunotherapy toxicity and promotes tumor immunity. Cancer Cell 40, 509–523 (2022).
Thomas, M. F. et al. Single-cell transcriptomic analyses reveal distinct immune cell contributions to epithelial barrier dysfunction in checkpoint inhibitor colitis. Nat. Med. 30, 1349–1362 (2024). Single-cell analysis of immune subsets in the colon and blood of patients who developed ICI colitis after treatment with anti-CTLA-4, anti-PD1 or a combination of both showed expansion of cytotoxic T cells, Treg cells and CD8+ TRM cells, along with apoptosis-response and IFN-response genes in luminal epithelial cells.
Wei, S. C., Duffy, C. R. & Allison, J. P. Fundamental mechanisms of immune checkpoint blockade therapy. Cancer Discov. 8, 1069–1086 (2018).
Sharpe, A. H. & Pauken, K. E. The diverse functions of the PD1 inhibitory pathway. Nat. Rev. Immunol. 18, 153–167 (2018).
Bottois, H. et al. KLRG1 and CD103 expressions define distinct intestinal tissue-resident memory CD8 T cell subsets modulated in Crohn’s disease. Front. Immunol. 11, 896 (2020).
Casey, K. A. et al. Antigen-independent differentiation and maintenance of effector-like resident memory T cells in tissues. J. Immunol. 188, 4866–4875 (2012).
Nava, P. et al. Interferon-gamma regulates intestinal epithelial homeostasis through converging beta-catenin signaling pathways. Immunity 32, 392–402 (2010).
Giesler, S., Riemer, R., Lowinus, T. & Zeiser, R. Immune-mediated colitis after immune checkpoint inhibitor therapy. Trends Mol. Med. 31, 265–280 (2025).
Beura, L. K. et al. Normalizing the environment recapitulates adult human immune traits in laboratory mice. Nature 532, 512–516 (2016).
Liu, J., Blake, S. J., Smyth, M. J. & Teng, M. W. Improved mouse models to assess tumour immunity and irAEs after combination cancer immunotherapies. Clin. Transl. Immunology 3, e22 (2014).
Rosshart, S. P. et al. Wild mouse gut microbiota promotes host fitness and improves disease resistance. Cell 171, 1015–1028 (2017).
Klocke, K., Sakaguchi, S., Holmdahl, R. & Wing, K. Induction of autoimmune disease by deletion of CTLA-4 in mice in adulthood. Proc. Natl Acad. Sci. USA 113, E2383–E2392 (2016).
Waterhouse, P. et al. Lymphoproliferative disorders with early lethality in mice deficient in Ctla-4. Science 270, 985–988 (1995).
Wei, S. C. et al. A genetic mouse model recapitulates immune checkpoint inhibitor-associated myocarditis and supports a mechanism-based therapeutic intervention. Cancer Discov. 11, 614–625 (2021).
Tivol, E. A. et al. Loss of CTLA-4 leads to massive lymphoproliferation and fatal multiorgan tissue destruction, revealing a critical negative regulatory role of CTLA-4. Immunity 3, 541–547 (1995).
Lo, B. C. et al. Microbiota-dependent activation of CD4+ T cells induces CTLA-4 blockade-associated colitis via Fcγ receptors. Science 383, 62–70 (2024). This study shows that mice with WildR microbiota that are treated with anti-CTLA-4 develop ICI colitis, characterized by IFNγ-producing CD4+ T cells and depletion of Treg cells; and demonstrates the potential use of Fc-null ICI to prevent development of ICI colitis.
Lo, J. W. et al. Immune checkpoint inhibitor-induced colitis is mediated by polyfunctional lymphocytes and is dependent on an IL23/IFNγ axis. Nat. Commun. 14, 6719 (2023). Assessment of single-cell transcriptional changes in immune cells infiltrating the colon of TRUC mice treated with anti-CTLA-4 plus anti-PD1 shows expansion of IFNγ-producing CD4+ T cells and the potential use of anti-IL-23 therapies to mitigate ICI colitis.
Wang, F., Yin, Q., Chen, L. & Davis, M. M. Bifidobacterium can mitigate intestinal immunopathology in the context of CTLA-4 blockade. Proc. Natl Acad. Sci. USA 115, 157–161 (2018). This study shows that treatment with Bifidobacterium spp. ameliorates ICI colitis in mice treated with DSS and anti-CTLA-4.
Tang, Z. et al. Single-cell multiomics reveals macrophage-derived IL-23 and CXCL9/10 drive pathogenic IFNG+IL17+ T cells in immunotherapy-related colitis. J. Immunother. Cancer 13, e011959 (2025).
Xiong, L. et al. Targeting MLCK1 uncouples immune checkpoint inhibitor-induced colitis from antitumour immunity. Gut https://doi.org/10.1136/gutjnl-2025-337780 (2026).
Gao, Y. et al. Faecalibacterium prausnitzii abrogates intestinal toxicity and promotes tumor immunity to increase the efficacy of dual CTLA4 and PD-1 checkpoint blockade. Cancer Res. 83, 3710–3725 (2023). This study shows a reduction in the commensal Faecalibacterium prausnitzii in the gut microbiomes of patients with ICI colitis and shows that administration of F. prausnitzii can reduce ICI colitis and improve anti-tumour responses in mice treated with DSS and plus anti-CTLA-4 and anti-PD1.
Sun, S. et al. Bifidobacterium alters the gut microbiota and modulates the functional metabolism of T regulatory cells in the context of immune checkpoint blockade. Proc. Natl Acad. Sci. USA 117, 27509–27515 (2020). This study shows that prevention of ICI colitis in mice treated with DSS and anti-CTLA-4 using Bifidobacterium spp. is partly dependent on Treg cells.
Wang, T. et al. Probiotics Lactobacillus reuteri abrogates immune checkpoint blockade-associated colitis by inhibiting group 3 innate lymphoid cells. Front. Immunol. 10, 1235 (2019). This study shows that treatment with Lactobacillus reuteri ameliorates ICI colitis in mice treated with DSS and combination ICI (anti-CTLA-4 and anti-PD1), correlating with a decline in type 3 innate lymphoid cells.
Yan, S. et al. Ligilactobacillus salivarius CCFM 1266 modulates gut microbiota and GPR109a-mediated immune suppression to attenuate immune checkpoint blockade-induced colitis. Food Funct. 14, 10549–10563 (2023).
Yan, S. et al. 2’-Fucosyllactose alleviate immune checkpoint blockade-associated colitis by reshaping gut microbiota and activating AHR pathway. Food Sci. Hum. Wellness 13, 2543–2561 (2024).
Ghosh, S. et al. Inflammation-targeted delivery of urolithin a mitigates chemical- and immune checkpoint inhibitor-induced colitis. J. Nanobiotechnology 22, 701 (2024).
Robles-Vera, I. et al. Microbiota translocation following intestinal barrier disruption promotes Mincle-mediated training of myeloid progenitors in the bone marrow. Immunity 58, 381–396 (2025).
Chrisikos, T. T. et al. STAT3 inhibits autocrine IFN signaling in type I conventional dendritic cells. J. Immunol. 209, 1286–1299 (2022).
Park, J. S., Gazzaniga, F. S., Kasper, D. L. & Sharpe, A. H. Microbiota-dependent regulation of costimulatory and coinhibitory pathways via innate immune sensors and implications for immunotherapy. Exp. Mol. Med. 55, 1913–1921 (2023).
Zeng, M. Y., Inohara, N. & Nuñez, G. Mechanisms of inflammation-driven bacterial dysbiosis in the gut. Mucosal Immunol. 10, 18–26 (2017).
Garrett, W. S. et al. Communicable ulcerative colitis induced by T-bet deficiency in the innate immune system. Cell 131, 33–45 (2007).
Lo, J. W. et al. CTLA-4 expressing innate lymphoid cells modulate mucosal homeostasis in a microbiota dependent manner. Nat. Commun. 15, 9520 (2024).
Shang, J. et al. Baseline colitogenicity and acute perturbations of gut microbiota in immunotherapy-related colitis. J. Exp. Med. 222, e20232079 (2025).
Hopkins, E. G. D. & Frankel, G. Overview of the effect of Citrobacter rodentium infection on host metabolism and the microbiota. Methods Mol. Biol. 2291, 399–418 (2021).
Honda, K. & Littman, D. R. The microbiome in infectious disease and inflammation. Annu. Rev. Immunol. 30, 759–795 (2012).
Fukuda, S. et al. Bifidobacteria can protect from enteropathogenic infection through production of acetate. Nature 469, 543–547 (2011).
Di Vincenzo, F., Del Gaudio, A., Petito, V., Lopetuso, L. R. & Scaldaferri, F. Gut microbiota, intestinal permeability, and systemic inflammation: a narrative review. Intern. Emerg. Med. 19, 275–293 (2024).
Nirschl, C. J. & Drake, C. G. Molecular pathways: coexpression of immune checkpoint molecules: signaling pathways and implications for cancer immunotherapy. Clin. Cancer Res. 19, 4917–4924 (2013).
Devi, K. S. P. et al. PD-1 is requisite for skin TRM cell formation and specification by TGFβ. Nat. Immunol. 26, 1339–1351 (2025).
Jacquelot, N. et al. PD-1 regulates ILC3-driven intestinal immunity and homeostasis. Mucosal Immunol. 17, 371–386 (2024).
Odorizzi, P. M., Pauken, K. E., Paley, M. A., Sharpe, A. & Wherry, E. J. Genetic absence of PD-1 promotes accumulation of terminally differentiated exhausted CD8+ T cells. J. Exp. Med. 212, 1125–1137 (2015).
Reschke, R. et al. Checkpoint blockade-induced dermatitis and colitis are dominated by tissue-resident memory T cells and Th1/Tc1 cytokines. Cancer Immunol. Res. 10, 1167–1174 (2022).
Bamias, G. et al. Immunological characteristics of colitis associated with anti-CTLA-4 antibody therapy. Cancer Invest. 35, 443–455 (2017).
Mowat, A. M. & Agace, W. W. Regional specialization within the intestinal immune system. Nat. Rev. Immunol. 14, 667–685 (2014).
Cao, H. et al. The pathogenicity and synergistic action of Th1 and Th17 cells in inflammatory bowel diseases. Inflamm. Bowel Dis. 29, 818–829 (2023).
Harbour, S. N., Maynard, C. L., Zindl, C. L., Schoeb, T. R. & Weaver, C. T. Th17 cells give rise to Th1 cells that are required for the pathogenesis of colitis. Proc. Natl Acad. Sci. USA 112, 7061–7066 (2015).
Chawla, A. S. et al. Distinct cell death pathways induced by granzymes collectively protect against intestinal Salmonella infection. Mucosal Immunol. 17, 1242–1255 (2024).
Walker, L. S. Treg and CTLA-4: two intertwining pathways to immune tolerance. J. Autoimmun. 45, 49–57 (2013).
Simpson, T. R. et al. Fc-dependent depletion of tumor-infiltrating regulatory T cells co-defines the efficacy of anti-CTLA-4 therapy against melanoma. J. Exp. Med. 210, 1695–1710 (2013).
Selby, M. J. et al. Anti-CTLA-4 antibodies of IgG2a isotype enhance antitumor activity through reduction of intratumoral regulatory T cells. Cancer Immunol. Res. 1, 32–42 (2013).
Elkrief, A. et al. Immune-related colitis is associated with fecal microbial dysbiosis and can be mitigated by fecal microbiota transplantation. Cancer Immunol. Res. 12, 308–321 (2024).
Halsey, T. M. et al. Microbiome alteration via fecal microbiota transplantation is effective for refractory immune checkpoint inhibitor-induced colitis. Sci. Transl. Med. 15, eabq4006 (2023). This study shows that patients with refractory ICI colitis who responded favourably to FMT showed decreased CD8+ T cells infiltrating the colon, along with increased alpha diversity of the microbiome and enrichment of Bifidobacterium and Collinsella.
Wang, Y. et al. Fecal microbiota transplantation for refractory immune checkpoint inhibitor-associated colitis. Nat. Med. 24, 1804–1808 (2018). A clinical study showed the efficacy of FMT in resolving ICI colitis that is refractory to first-line and second-line treatments.
Eberl, G., Colonna, M., Di Santo, J. P. & McKenzie, A. N. Innate lymphoid cells. Innate lymphoid cells: a new paradigm in immunology. Science 348, aaa6566 (2015).
Meininger, I. et al. Tissue-specific features of innate lymphoid cells. Trends Immunol. 41, 902–917 (2020).
Zou, F. et al. Fecal calprotectin concentration to assess endoscopic and histologic remission in patients with cancer with immune-mediated diarrhea and colitis. J. Immunother. Cancer 9, e002058 (2021).
Lucas, M. W. et al. Immune signature-based uncoupling of checkpoint inhibitor efficacy and toxicity. Immunity 59, 29–33 (2026).
Seo, S. U. et al. Distinct commensals induce interleukin-1β via NLRP3 inflammasome in inflammatory monocytes to promote intestinal inflammation in response to injury. Immunity 42, 744–755 (2015).
Scott, C. L., Aumeunier, A. M. & Mowat, A. M. Intestinal CD103+ dendritic cells: master regulators of tolerance? Trends Immunol. 32, 412–419 (2011).
Rooks, M. G. & Garrett, W. S. Gut microbiota, metabolites and host immunity. Nat. Rev. Immunol. 16, 341–352 (2016).
Koh, A., De Vadder, F., Kovatcheva-Datchary, P. & Backhed, F. From dietary fiber to host physiology: short-chain fatty acids as key bacterial metabolites. Cell 165, 1332–1345 (2016).
Roager, H. M. & Licht, T. R. Microbial tryptophan catabolites in health and disease. Nat. Commun. 9, 3294 (2018).
Su, X., Gao, Y. & Yang, R. Gut microbiota derived bile acid metabolites maintain the homeostasis of gut and systemic immunity. Front. Immunol. 14, 1127743 (2023).
Tran, M., Huh, J. R. & Devlin, A. S. The role of gut microbial metabolites in the T cell lifecycle. Nat. Immunol. 26, 1246–1257 (2025).
Shi, Y. et al. Role of gut microbiota in postoperative complications and prognosis of gastrointestinal surgery: a narrative review. Medicine 101, e29826 (2022).
Ahuja, R. et al. Recent abdominal surgery as a risk factor for unfavorable immune-mediated colitis outcomes. Gastro Hep Adv. 5, 100767 (2026).
Mohiuddin, J. J. et al. Association of antibiotic exposure with survival and toxicity in patients with melanoma receiving immunotherapy. J. Natl Cancer Inst. 113, 162–170 (2021).
Stone, S., McPherson, J. P., Kulkarni, R. P. & Young, A. The impact of concomitant medications on treatment outcomes in patients with cancer receiving immune checkpoint inhibitors. Nat. Rev. Cancer 26, 137–158 (2025).
Kiecka, A. & Szczepanik, M. Proton pump inhibitor-induced gut dysbiosis and immunomodulation: current knowledge and potential restoration by probiotics. Pharmacol. Rep. 75, 791–804 (2023).
Shatila, M. et al. Worse survival and gastrointestinal toxicity outcomes among patients receiving proton pump inhibitors during checkpoint inhibitor therapy. J. Natl Compr. Canc. Netw. 23, e257023 (2025).
Eng, L. et al. Impact of antibiotic exposure before immune checkpoint inhibitor treatment on overall survival in older adults with cancer: a population-based study. J. Clin. Oncol. 41, 3122–3134 (2023).
Majeed, U. et al. P13.01 use of antibiotics is associated with an increase in immunotherapy related adverse effects in patients with non-small cell lung cancer. J. Thorac. Oncol. 16, S1010 (2021).
Dar, S. et al. Impact of proton-pump inhibitors on the efficacy of immune checkpoint inhibitors in non-small cell lung cancer: a systematic review and meta-analysis. Ann. Med. Surg. 78, 103752 (2022).
Lopes, S. et al. Do proton pump inhibitors alter the response to immune checkpoint inhibitors in cancer patients? A meta-analysis. Front. Immunol. 14, 1070076 (2023).
Ma, W. et al. Dietary fiber intake, the gut microbiome, and chronic systemic inflammation in a cohort of adult men. Genome Med. 13, 102 (2021).
Majenka, P. et al. Influence of high-fiber diet on ipilimumab-induced gastrointestinal toxicity in metastatic melanoma. Clin. Nutr. ESPEN 67, 660–664 (2025).
Merra, G. et al. Influence of mediterranean diet on human gut microbiota. Nutrients 13, 7 (2020).
Bolte, L. A. et al. Association of a Mediterranean diet with outcomes for patients treated with immune checkpoint blockade for advanced melanoma. JAMA Oncol. 9, 705–709 (2023).
Singh, P., Rawat, A., Alwakeel, M., Sharif, E. & Al Khodor, S. The potential role of vitamin D supplementation as a gut microbiota modifier in healthy individuals. Sci. Rep. 10, 21641 (2020).
Grover, S. et al. Vitamin D intake is associated with decreased risk of immune checkpoint inhibitor-induced colitis. Cancer 126, 3758–3767 (2020).
Groenewegen, B., Terveer, E. M., Joosse, A., Barnhoorn, M. C. & Zwittink, R. D. Fecal microbiota transplantation for immune checkpoint inhibitor-induced colitis is safe and contributes to recovery: two case reports. J. Immunother. 46, 216–220 (2023).
Golčić, M. et al. Fecal microbiota transplantation in refractory immune-mediated colitis: case series and review of the literature. Pharmaceuticals 18, 1719 (2025).
Hearn, J. et al. Refractory immune checkpoint inhibitor colitis treated with biologics, Janus kinase inhibition, plasma exchange, and fecal microbiota transplantation. ACG Case Rep. J. 12, e01847 (2025).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT04038619 (2019).
Simpson, R. C. et al. Diet-driven microbial ecology underpins associations between cancer immunotherapy outcomes and the gut microbiome. Nat. Med. 28, 2344–2352 (2022).
Chang, A. E. et al. Targeting the gut microbiome to mitigate immunotherapy-induced colitis in cancer. Trends Cancer 7, 583–593 (2021).
Deleu, S., Machiels, K., Raes, J., Verbeke, K. & Vermeire, S. Short chain fatty acids and its producing organisms: an overlooked therapy for IBD? EBioMedicine 66, 103293 (2021).
Usyk, M. et al. Bacteroides vulgatus and Bacteroides dorei predict immune-related adverse events in immune checkpoint blockade treatment of metastatic melanoma. Genome Med. 13, 160 (2021).
Mo, C. et al. The influence of Akkermansia muciniphila on intestinal barrier function. Gut Pathog. 16, 41 (2024).
Pena-Cearra, A. et al. Akkermansia muciniphila-induced trained immune phenotype increases bacterial intracellular survival and attenuates inflammation. Commun. Biol. 7, 192 (2024).
Andreu-Sánchez, S. et al. Global genetic diversity of human gut microbiome species is related to geographic location and host health. Cell 188, 3942–3959 (2025).
Asnicar, F. et al. Microbiome connections with host metabolism and habitual diet from 1,098 deeply phenotyped individuals. Nat. Med. 27, 321–332 (2021).
Ghosh, T. S., Shanahan, F. & O’Toole, P. W. The gut microbiome as a modulator of healthy ageing. Nat. Rev. Gastroenterol. Hepatol. 19, 565–584 (2022).
Natalini, J. G., Singh, S. & Segal, L. N. The dynamic lung microbiome in health and disease. Nat. Rev. Microbiol. 21, 222–235 (2023).
Oh, J. & Voigt, A. Y. The human skin microbiome: from metagenomes to therapeutics. Nat. Rev. Microbiol. 23, 771–787 (2025).
Zhou, Z. et al. Metagenomic next-generation sequencing unraveled the characteristic of lung microbiota in patients with checkpoint inhibitor pneumonitis: results from a prospective cohort study. J. Immunother. Cancer 13, e012444 (2025).
Snelson, M. et al. Gut-heart axis: the role of gut microbiota and metabolites in heart failure. Circ. Res. 136, 1382–1406 (2025).
Cao, H. et al. Abnormal gut microbiota may cause PD-1 inhibitor-related cardiotoxicity via suppressing regulatory T cells. Sci. Rep. 15, 20547 (2025).
Desreumaux, P. & Ghosh, S. Review article: mode of action and delivery of 5-aminosalicylic acid – new evidence. Aliment. Pharmacol. Ther. 24, 2–9 (2006).
Ruf, T. et al. Second-line therapies for steroid-refractory immune-related adverse events in patients treated with immune checkpoint inhibitors. Eur. J. Cancer 203, 114028 (2024).
Abu-Sbeih, H. et al. Outcomes of vedolizumab therapy in patients with immune checkpoint inhibitor-induced colitis: a multi-center study. J. Immunother. Cancer 6, 142 (2018).
Johnson, D. H. et al. Infliximab associated with faster symptom resolution compared with corticosteroids alone for the management of immune-related enterocolitis. J. Immunother. Cancer 6, 103 (2018).
Zou, F. et al. Efficacy and safety of vedolizumab and infliximab treatment for immune-mediated diarrhea and colitis in patients with cancer: a two-center observational study. J. Immunother. Cancer 9, e003277 (2021).
Mei, R. et al. Symptomatic and sonographic improvement of immune checkpoint inhibitor enterocolitis with risankizumab. Inflamm. Bowel Dis. 31, 1182–1183 (2025).
Perez Del Nogal, G. & Patel, N. Refractory checkpoint inhibitor colitis responsive to ustekinumab. ACG Case Rep. J. 9, e00946 (2022).
Shirwaikar Thomas, A. et al. IL12/23 blockade for refractory immune-mediated colitis: 2-center experience. Am. J. Gastroenterol. 118, 1679–1683 (2023).
Thomas, A. S., Ma, W. & Wang, Y. Ustekinumab for refractory colitis associated with immune checkpoint inhibitors. N. Engl. J. Med. 384, 581–583 (2021).
Holmstroem, R. B. et al. COLAR: open-label clinical study of IL-6 blockade with tocilizumab for the treatment of immune checkpoint inhibitor-induced colitis and arthritis. J. Immunother. Cancer 10, e005111 (2022).
Bishu, S. et al. Efficacy and outcome of tofacitinib in immune checkpoint inhibitor colitis. Gastroenterology 160, 932–934 (2021).
Holmstroem, R. B. et al. Tofacitinib and faecal microbiota transplantation in treating checkpoint inhibitor-induced enterocolitis: case report. BMJ Open. Gastroenterol. 9, e000989 (2022).
Kono, M. et al. JAK inhibitor upadacitinib induces remission in refractory immune-related colitis triggered by CTLA-4 and PD-1 inhibitor combination therapy in malignant pleural mesothelioma: a case report. Cancer Rep. 7, e70032 (2024).
Bernardi, F. et al. JAK inhibitors and risk of cancer in IBD patients. Cancers 17, 1795 (2025).
Liang, Y., Shao, Y. & Gu, W. Role of interleukin-6 in resistance to tumor therapy. Discov. Oncol. 16, 1791 (2025).
Kumar, A. et al. Identification of medication-microbiome interactions that affect gut infection. Nature 644, 506–515 (2025).
Prasad, R. et al. Antibiotic-induced loss of gut microbiome metabolic output correlates with clinical responses to CAR T-cell therapy. Blood 145, 823–839 (2025).
Smith, M. et al. Gut microbiome correlates of response and toxicity following anti-CD19 CAR T cell therapy. Nat. Med. 28, 713–723 (2022).
Asokan, S., Cullin, N., Stein-Thoeringer, C. K. & Elinav, E. CAR-T cell therapy and the gut microbiota. Cancers 15, 794 (2023).
Wyant, T., Fedyk, E. & Abhyankar, B. An overview of the mechanism of action of the monoclonal antibody vedolizumab. J. Crohns Colitis 10, 1437–1444 (2016).
Levin, A. D., Wildenberg, M. E. & van den Brink, G. R. Mechanism of action of anti-TNF therapy in inflammatory bowel disease. J. Crohns Colitis 10, 989–997 (2016).
Pugliese, D. et al. Targeting IL12/23 in ulcerative colitis: update on the role of ustekinumab. Ther. Adv. Gastroenterol. 15, 17562848221102283 (2022).
Rubbert-Roth, A., Furst, D. E., Nebesky, J. M., Jin, A. & Berber, E. A review of recent advances using tocilizumab in the treatment of rheumatic diseases. Rheumatol. Ther. 5, 21–42 (2018).
Schwartz, D. M. et al. JAK inhibition as a therapeutic strategy for immune and inflammatory diseases. Nat. Rev. Drug. Discov. 16, 843–862 (2017).
Attia, P. et al. Autoimmunity correlates with tumor regression in patients with metastatic melanoma treated with anti-cytotoxic T-lymphocyte antigen-4. J. Clin. Oncol. 23, 6043–6053 (2005).
Downey, S. G. et al. Prognostic factors related to clinical response in patients with metastatic melanoma treated by CTL-associated antigen-4 blockade. Clin. Cancer Res. 13, 6681–6688 (2007).
Weber, J. S. et al. Safety profile of nivolumab monotherapy: a pooled analysis of patients with advanced melanoma. J. Clin. Oncol. 35, 785–792 (2017).
Horvat, T. Z. et al. Immune-related adverse events, need for systemic immunosuppression, and effects on survival and time to treatment failure in patients with melanoma treated with ipilimumab at Memorial Sloan Kettering Cancer Center. J. Clin. Oncol. 33, 3193–3198 (2015).
Anichini, A. et al. Melanoma cells and normal melanocytes share antigens recognized by HLA-A2-restricted cytotoxic T cell clones from melanoma patients. J. Exp. Med. 177, 989–998 (1993).
Bakker, A. B. et al. Melanocyte lineage-specific antigen gp100 is recognized by melanoma-derived tumor-infiltrating lymphocytes. J. Exp. Med. 179, 1005–1009 (1994).
Wohner, M. & Nimmerjahn, F. Cytotoxic IgG: mechanisms, functions, and applications. Immunity 58, 1378–1395 (2025).
Barth, D. A.-O. et al. Evaluation of autoantibodies as predictors of treatment response and immune-related adverse events during the treatment with immune checkpoint inhibitors: a prospective longitudinal pan-cancer study. Cancer Med. 11, 3074–3083 (2022).
Johncilla, M. et al. Ipilimumab-associated hepatitis: clinicopathologic characterization in a series of 11 cases. Am. J. Surg. Pathol. 39, 1075–1084 (2015).
Bevins, C. L. & Salzman, N. H. Paneth cells, antimicrobial peptides and maintenance of intestinal homeostasis. Nat. Rev. Microbiol. 9, 356–368 (2011).
Pracht, K., Wittner, J., Kagerer, F., Jack, H. M. & Schuh, W. The intestine: a highly dynamic microenvironment for IgA plasma cells. Front. Immunol. 14, 1114348 (2023).
Niess, J. H. et al. CX3CR1-mediated dendritic cell access to the intestinal lumen and bacterial clearance. Science 307, 254–258 (2005).
Hickey, J. W. et al. Organization of the human intestine at single-cell resolution. Nature 619, 572–584 (2023).





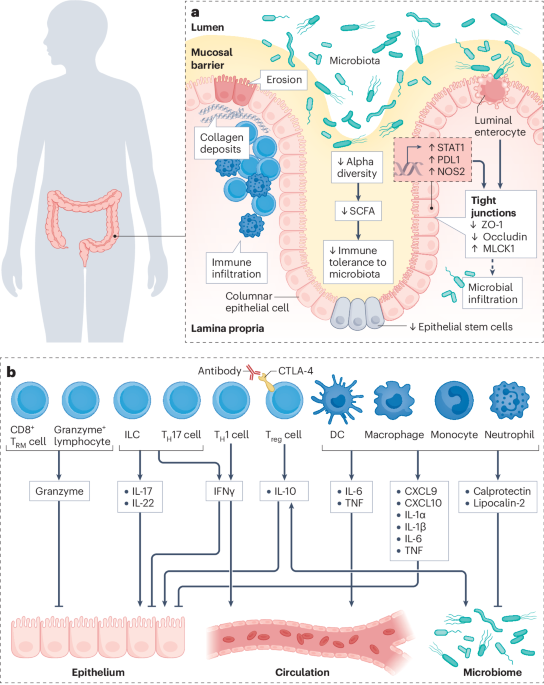







Leave a Reply