Ethics statement
This study conformed to the Declaration of Helsinki and was approved by the Ethics Committee of the Nanjing First Hospital, Nanjing medical University (Nanjing, China). The experiments involving animals were approved by the Institutional Animal Care and Use Committee of Nanjing Medical University (Nanjing, China). Written informed consent from patients that donated samples for research purposes were obtained. All human sample collections were approved by the Institutional Research Ethics Committee.
All animals were cared for in compliance with institutional guidelines of Nanjing First Hospital, Nanjing Medical University (Nanjing, China). All experimental protocol was approved by the Institutional Animal Care and Use Committee of Nanjing First Hospital, Nanjing Medical University (license number: SYXK(Su) 2021-0007). The NIH guide for the Care and Use of Laboratory Animals (NIH Publications No. 80-23) and ARRIVE 2.0 were also obeyed [51]. Every effort was made to reduce the number of mice and their suffering. Assignments of animals to experimental groups were randomized.
Tumor samples
The fresh primary tumor specimens and corresponding adjacent non-tumor tissues were obtained from colon cancer patients underwent surgery in our center from 2015 to 2021. No patients had received radiotherapy and/or chemotherapy prior to surgery (Detailed information of CRC patients is provided in the supplementary table-2). The specimens were stored in tissue preservation solution (Absin, abs9474) at -80°C immediately after surgery.
Introduction of scRNA-seq
scRNA sequencing was performed by 10X Genomics’ scRNA-seq technology. Eight CAFs samples, CAF1-8, were prepared for scRNA-sequencing assays. CAFs were digested into a single-cell suspension, and the cell density and live cell proportion were detected using a Countess® II Automated Cell Counter. CAF samples with a density of 1×103 cells per microliter and a live cell percentage greater than 80% were used for further scRNA sequencing. Briefly, CAFs samples were loaded on a 10X Genomics Chromium single-cell controller to generate single-cell gel beads-in-emulsion (GEMs). The captured cells were lysed, and the released RNA was barcoded through reverse transcription in individual GEMs. Then, a single-cell cDNA library was prepared according to the manufacturer’s protocols. After quality control via Agilent 4200 Bioanalyzer, the cDNA libraries were sequenced using an Illumina NovaSeq 6000 sequencer with paired-end (PE) reading strategy under the control of data collection software provided by Illumina (Shanghai Biotechnology Corporation, Shanghai, China).
Single-cell data analysis
Sequenced reads were demultiplexed and aligned to the human hg19 reference genome using CellRanger (version 3.0.2) with default parameters. The sequencing depth, unique molecular identifiers (UMI) expression, gene expression, mitochondrial/ribosome ratio associated with the aligned reads were subjected to correction and filtering, and the gene barcode matrix of each CAFs sample was constructed. Further analysis, including data normalization, cell cycle correction, dimensionality reduction, and K-means cell clustering, was performed using R (version 3.6.3) with the Seurat package (version 4.0). Genes were filtered with the following criteria: expressed in more than 10 cells, an average expression value greater than 0.1, and a Qval less than 0.01 in different analyses.
Marker genes for each cluster and subgroup were identified by contrasting gene expression of cells from certain cluster or subgroup to that of others using the Seurat FindMarkers function, and filtered by a detectable expression in more than 20% of all cells from that cluster or subgroup, logFC>0.25 and specifically upregulated expression profile. Additionally, the expression fold change of marker genes in certain cluster or subgroup to others and the difference of detectable expression in that cluster or subgroup with others were both required to be in top 10 of all detected genes.
The total number of UMIs per cell was counted for the number of UMI sequences of high-quality single cells and genes in the sample. Median normalization process was introduced for normalizing the number of UMIs in each cell to the median of the total UMI of cells. The similarity between cells was investigated via Principal component analysis (PCA) reduction dimension. The closer the expression trend of cellular genes, the closer the sample distance was. The protocol of the UMAP presentation method was to recount the sample distance through the conditional probability of random neighbor fitting according to the student’s T distribution in the high dimensional space, so that the top 10 principal components of the PCA resulted in the largest variance explained followed by cell type annotation using Single R was visualized the sample presented in the form of a clearly separated cluster in the low dimensional space.
Gene expression and prognostic performance in colon adenocarcinoma (COAD) samples
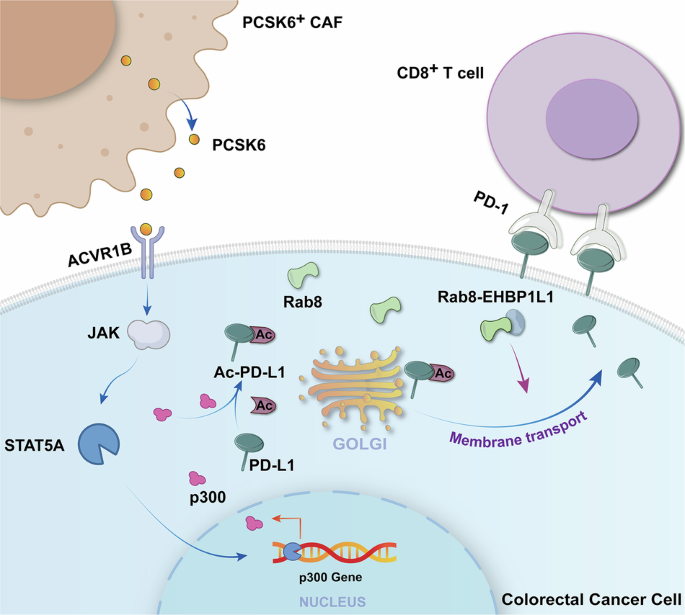
We detected the expression profile of PCSK6 using the GEPIA COAD datasets where 349 healthy people and 275 CRC patients were taken into analysis. Based on gene median expression level, expression of PCSK6 in the CRC samples were significantly higher than that in healthy volunteers.
Based on gene median expression level, the CRC samples were divided into the PCSK6 high- and low-expression groups. To compare the overall survival (OS) among two groups, Kaplan-Meier curve was constructed, and Log-rank P value was calculated.
For verifying that fibroblast is the main source of PCSK6, Tumor Immune Single-cell Hub 2 (TISCH2), a scRNA-seq database focusing on TME, was introduced for providing detailed cell-type annotation of PCSK6 in CRC at the single-cell level.
Isolation of PCSK6+CAFs by fluorescence activated cell sorting
FSP+ cells were then positively selected using a relevant Multisort Microbeads (Miltenyi Biotec). After the removal of the FSP Multisort Microbeads using an enzyme (multisort release reagent), FSP+ cells were further stained with an FITC-conjugated mAb recognizing CXCR4 (Biotechne, USA) and CXCR4+ cells were obtained by positive selection using anti-FITC microbeads through a MACS separation column (Miltenyi Biotec), followed by gradient sorting using anti-ACKR3 microbeads (Invitrogen, USA) and anti-HEG1 microbeads (Sunlong Biotech) step by step based on the results of single-cell data with similar procedure. Aliquots of enriched cells were checked for purity and were consistently more than 96%.
Cre-mediated gene recombination
APCmin/+ spontaneous CRC mice were obtained from GemPharmatech Co., Ltd. COL1A2-CreERT2 mice were kindly provided by Cyagen Biotech Co., Ltd.; PCSK6flox/+(fl/+) mice were purchased from (Cyagen Biosciences Inc, US). Animals were housed in SPF conditions. To target PCSK6 deletion to intestinal fibroblast in APCmin/+ spontaneous CRC mice, APCmin/+ mice were firstly bred to PCSK6fl/+ mice to generate APCmin/+PCSK6fl/+ mice which were then bred to those expressing Cre under the control of the COL1A2 promoter (fibroblast specific promoter), subsequently generating APCmin/+PCSK6fl/flCOL1A2-Cre mice. Offspring were examined by PCR analyses of tail DNA for the presence of Cre and floxed-PCSK6. For Cre activation, 4-week-old mice were gavaged with tamoxifen (120 mg/kg via oral gavage every other day for 10 days). Mice were euthanized using isoflurane, after which the cecum and the inflamed colon tissue were excised, washed with PBS, and frozen in liquid nitrogen until further analysis.
Blood routine index and biochemistry index
Blood biochemistry indexes including red blood cell (RBC), hematocrit (HCT), and hemoglobin (Hb) were examined by the clinical testing platform of the Nanjing first Hospital.
Establishment of orthotopic CRC model
Male C57BL/6 mice and BALB/c Nude mice aged 6–8 weeks were purchased from Charles River (SCXK(Zhe)2019-0001), Zhejiang, China. All mice were allowed to eat and drink freely and kept in a light/dark cycle with humidity-controlled house at 25°C.
For the orthotopic mouse CRC model, mice were placed under anesthesia (2-3% isoflurane) on a heated operating bed, and were unresponsive to touch. Pre-operative buprenorphine (1 mg/kg) was injected subcutaneously in the shank for pain management. 1×105 of cells derived from CAFs or CAFs-PCSK6 knockdown were injected into the cecal wall of the two different mouse strains (C57BL/6 J or BALB/c Nude) together with 1×105 MC38-Luc cells suspended in 100 µL Dulbecco’s modified eagle medium (DMEM, Gibco, USA). Briefly, intercaecal injections were performed using a 31 G needle under binocular guidance, with an approximate 30° angle and its tip 4 mm into the cecal wall (significant bleeding or cell reflux must be avoided). After injection, the gut was returned to the abdominal cavity and closed. Mice were euthanized six weeks after injection and metastasis was scored macroscopically as well as histologically. Post-injection, all mice received analgesia. Orthotopic tumor sizes were measured by caliper after the sacrifice.
1×105 MC38-Luc or MC38-Luc+homogeneous CAFs (5×104 cells transfected with shPCSK9 or shCtrl) were injected into mice spleens for liver metastases monitoring using macroscopical observation and HE staining [52]. Six weeks later, mice were sacrificed and tumor tissue was removed for further analyses. The tumors were stored in liquid nitrogen or embedded in paraffin for pathological examination.
For pharmacodynamic study with same tumor injection operation (such as CRC+CAFs co-injection with or without SB431542 intervention), randomization was conducted by the methods described as follows: mice, named as 1, 2, 3, etc., were listed in the first column of Excel, and randomly numbered corresponding to the animal numbers, a set of random number were generated in the second column using the function of RAND, which were sorted in descending order. The mice were assigned to relevant group based on the sample size in different experiment.
Injection of SB431542 and rPCSK6
rPCSK6 was dissolved in normal saline to prepare a stock solution at a concentration of 500 μg/mL. Mice were injected with rPCSK6 at 320 μg/kg body weight once daily for 4 consecutive weeks.
For pharmacodynamics study, ACVR1B inhibitor SB431542 was purchased from Selleck (Shanghai, China) and dissolved in DMSO. After the establishment of orthotopic CRC in mice. SB431542 (7.5 mg/kg) or DMSO were intraperitoneally injected once daily for four consecutive weeks.
Molecular docking
SB431542 is primarily used as a research-grade inhibitor with limited direct clinical applicability. To address this translational gap, we adopted a strategy involving structure-based virtual screening of FDA-approved small molecules targeting ACVR1B, aiming to identify clinically relevant candidates with high docking scores. Molecular docking was performed using AutoDockTools version 1.5.7. The crystal structure of the candidate target protein ACVR1B was obtained from the UniProt protein database (https://www.uniprot.org/). The protein structure was loaded into AutoDock, followed by standard preprocessing procedures including removal of water molecules, addition of hydrogen atoms, optimization of amino acid side chains, and correction of any missing residues or structural anomalies.
A library of FDA-approved small-molecule compounds was prepared for docking (https://pubchem.ncbi.nlm.nih.gov/). Each compound was structurally optimized and subjected to conformational sampling to ensure chemical diversity and plausible binding poses. The docking simulations were carried out using AutoDock’s molecular docking module, wherein each compound was docked into the binding site of the ACVR1B protein. The binding affinities and interaction patterns were calculated and evaluated.
Docking results were saved in PDB format, and molecular interactions were visualized using PyMOL version 2.5. The top-ranked compounds based on binding energy scores and docking poses were selected for further molecular dynamics (MD) simulations and biological validation experiments.
CETSA
To evaluate the interaction between the candidate small molecule and ACVR1B in a cellular context, a CETSA was performed as previously described with minor modifications. Briefly, cells were seeded in 6-well plates and treated with the indicated concentrations of the small molecule or vehicle control (DMSO) for 1–2 hours at 37 °C. Following treatment, cells were harvested, washed with cold PBS, and resuspended in PBS supplemented with protease inhibitors.
The cell suspension was aliquoted into PCR tubes and subjected to a temperature gradient (38 °C–66 °C, in 4 °C increments) for 3 min using a thermal cycler, followed by cooling at room temperature for 3 min. Cells were then lysed by repeated freeze-thaw cycles (three cycles of liquid nitrogen and 37 °C water bath), followed by centrifugation at 20,000 g for 20 min at 4 °C to separate soluble protein fractions.
The supernatants were collected and analyzed by SDS-PAGE followed by western blot using specific antibodies against the target protein. Band intensities were quantified by densitometry using ImageJ software. Thermal stability of the protein was assessed by comparing the melting curves of the compound-treated and control groups.
Combination therapy with Eletriptan and anti-PD-1
A total of 1×106 MC38 cells and CAFs were mixed (1: 1) and then subcutaneously injected into the axillary region of each C57 mouse. The mice were randomly divided into four groups: vehicle, Eletriptan, anti-PD-1 and anti-PD-1+Eletriptan. Six days after subcutaneous inoculation, mice in Eletriptan and ICB + Eletriptan groups were injected with Eletriptan (10 mg/kg) twice a day for 11 consecutive days (the dosage of Eletriptan was determined via preliminary experiment). Meanwhile, mice in ICB and ICB + Eletriptan groups were injected with anti-PD-1 (10 mg/kg, Bio X Cell) at day 6, day 11 and day 15. The same volume of PBS was used as control for Eletriptan treatment, and isotype control was used as control for ICB treatment. The tumor diameter was calculated during 3 weeks.
Tumor measurements
Tumors were measured weekly using a vernier caliper. The tumor volume was evaluated as [(L×W 2) ×(π/6)], whereas L refers to the length and W to the width. Individual tumor volumes were summed together to calculate total tumor load, and the endpoint was recorded when individual tumor reached 2000 mm 3. Researchers were absolutely blind to the groups of mice during measurements.
Tumor suspension preparation
CRC cell suspensions were prepared from at least 1 mm3 fresh tumor tissue. After the removal of coagulated tissue and blood vessel, tumor tissue was then disrupted using surgical scalpels and digested using 5 U/mL DNase (Invitrogen, USA) and 2.5 mg/mL Collagenase D (Roche, Germany). A single cell suspension was obtained by forcing tumor tissue through a 70 μm Cell Strainer. Tumor-infiltrating immune cells were separated from other cell components by centrifugation in Percoll (Sigma-Aldrich, St-Louis, MO, USA) gradient.
Cell line culture and incubation of reagents
CRC cell lines (LS174T, RKO, HCT116, HT29, DLD1, MC38 and CT26) and HEK293T were cultured in DMEM. All medium contained 10% Fetal Bovine Serum (FBS, Gibco, USA) and a penicillin-streptomycin solution (Gibco, USA). The cells were kept at 37 °C and 5% CO2, and were used during their logarithmic growth phase. All cell lines used in this study were authenticated by short tandem repeat (STR) profiling and were routinely tested to ensure they were free from mycoplasma contamination.
rPCSK6 was dissolved in DMEM containing 10% FBS, and 25 μg/mL (50×) stock solution was obtained, the concentration of which was determined via consulting the serum concentration of PCSK6 in published article[53]. HCT116 were incubated with rPCSK6 (500 ng/mL) for 24 h, followed by the subsequent functional assessment.
Anti-PCSK6 antibody or isotype-matched control antibody (Invitrogen, USA) was added at 5 µg/mL to CRC CM so as to verifying the regulation of PCSK6 in the CRC CM.
For incubation of small-molecule inhibitor, the inhibitors were dissolved in DMSO at a concentration of 50× stock solution, the concentrations of working solution were determined by consulting the published articles and our preliminary experiments. Briefly, cells were incubated with NFκB-IN-1 (50 μM, NFκB inhibitor), LY294002 (20 μM, PI3K/Akt signal inhibitor), IQ3 (1 μM, JNK/JUN inhibitor), XAV939 (10 μM, Wnt/β-catenin inhibitor), TP0427736·HCl (10 μM, Smad2/3 inhibitor), SCH772984 (2 μM, ERK1/2 inhibitor) and Stafia-1 (100 μM, JAK/STAT5a inhibitor) for 24 h, followed by subsequent experiments [54,55,56,57,58,59,60],
Isolation and culture of primary fibroblasts
Isolation and culture of primary CAFs and primary NCFs were performed according to the outgrowth method [61]. Briefly, tumor tissue and normal intestinal tissue were obtained from freshly resected specimens from patients with CRC. Colorectal tumor tissues were first dissected into small fragments (3 mm³) using a razor blade. Tissue fragment was digested using 0.25% trypsin for 10 min, after which the resulting fragments were seeded in 10 cm dishes to allow tight adherence after 48-hour culture. Then, DMEM/F12 medium (Gibco) supplemented with 10% fetal bovine serum (FBS, Gibco), 2 mmol/L L-glutamine (Invitrogen), 0.5% sodium pyruvate (Invitrogen), and 1% antibiotic-antimycotic (Invitrogen) were added in to promote the outgrowth of CAFs. After 7-10 days, primary CAFs were observed outgrowing from the tumor fragments and validated through immunofluorescence, demonstrating positive staining for α-SMA (shown in the left figure of Fig. S29). NCFs were extracted from non-tumor intestinal tissues at least 5 cm away from the tumor margin.
Tumor tissues were harvested from mice subjected to orthotopic MC38 cell inoculation and subsequently dissected into approximately 2 mm³ fragments using a sterile razor blade. These fragments were enzymatically digested with 0.25% trypsin for 10 min to facilitate tissue dissociation. Post-digestion, the tissue pieces were seeded into 10 cm culture dishes and incubated for 48 h to promote firm adherence. Following this initial culture period, Dulbecco’s Modified Eagle Medium/Nutrient Mixture F-12 (DMEM/F12, Gibco), supplemented with 10% fetal bovine serum (FBS, Gibco), 2 mM L-glutamine (Invitrogen), 0.5% sodium pyruvate (Invitrogen), and 1% antibiotic-antimycotic solution (Invitrogen), was added to support the outgrowth of CAFs. After one week, spindle-shaped cells exhibiting fibroblastic morphology were observed migrating from the tumor fragments. These primary CAFs were validated via immunofluorescence staining for α-SMA, confirming their identity (Fig. S29, right).
To monitor cellular senescence throughout passaging, β-galactosidase staining was performed at each passage, ensuring the maintenance of cellular proliferative capacity. For both human and mouse isolations, fibroblast were used at passage 5–10, during which period isolated cells were tested monthly for mycoplasma contamination.
Preparation of CM
CAF CM was prepared by growing cells to 80% confluence in a 37 °C constant temperature incubator with 5% CO2. Subsequently, CAFs were incubated in glutamine/SFM. Cells were allowed to condition the medium for 2 days. The CM was collected and filtered with 0.22 μm membrane sterile filter and used immediately or frozen at −80 °C until use (up to one month).
Establishment of co-culture system (CS)
CS1: CAFs or PCSK6 knockdown CAF cells (1×107 cells/mL) were co-cultured at a ratio of 5:1 with HCT116 cells (2×106 cells/mL) cells in a 24-well plate inserted into a transwell cube for 10 days. HCT116 cells were seeded in the lower chamber and CAFs or PCSK6 knockdown CAF cells were seeded in the upper chamber.
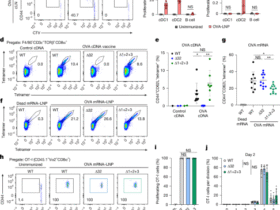
Isolation, activation and expansion of peripheral blood cytotoxic T cells (CD8+T cells) was conducted according to the published article. Briefly, lymphocytes was isolated from total blood from healthy donors by Ficoll gradients, following a negative magnetic selection with a CD8+T cells isolation kit (Miltenyi Biotec). CD8+T cells were activated for 5 days with magnetic Dynabeads (Invitrogen) bound with anti-CD3 (OKT3, BioLegend) and anti-CD28 (CD28.2, BioLegend) antibodies in the presence of IL-2 (100 U/mL). After 48 h, cells were collected, centrifuged and re-suspended at 106 cells/mL in RPMI complete media supplemented with IL-7 (25 ng/mL, InmunoTools). Such cultures were used in experiments between days 10 and 15 of culture [62].
CS2: After the co-culture with CAFs, CRC cells from CS1 were incubated with homogeneous CD8+T cells. Activated CD8+T cells (effector cells, 1×107 cells/mL) were co-cultured with CRC cells (2×105 cells/mL) at a ratio of 50: 1 in 40-well polystyrene plates for 8 h. after which the CD8+T cells were separated from HCT116 via shaking the culture plate at 200 rpm for three times (3 min per time) followed by detection of apoptotic rate with flowcytometry. The remining HCT116 cells were subjected to CCK-8 assay for cell viability determination.
Transient transfection of overexpressing plasmid and short hairpin RNA (shRNA)
ACVR1C-overexpressing plasmids, ACVR1B-overexpressing plasmids, ACVR2B-overexpressing plasmids, ACVR2A-overexpressing plasmids, shRNA against ACVR1B, p300, Rab8, PCSK6, CBP, PCAF, GCN5, Tip60 were designed and constructed by Sangon (Shanghai, China). Required cells were transfected at 60% to 70% confluence by Lipofectamine™ Transfection Reagent (Invitrogen, USA) following the manufacturer’s instructions. The targeting sequences of shRNA were listed as follows (Table 1):
Knockout of p300 or ACVR1B
Single-guide RNA (sgRNA) sequences for CRISPR-Cas9 were designed at CRISPR design website (https://chopchop.cbu.uib.no/). Insert oligonucleotides for human p300 sgRNA was 5’-GTACGACTAGGTACAGGCGAGGG-3’ and 5’-GGTACGACTAGGTACAGGCGAGG-3’. Insert oligonucleotides for human ACVR1B sgRNA was 5’-GAGGGCACTAAGTCGTAATATGG-3’ and 5’-CTTCTCGTGAAGAACGGTCTTGG-3’. The complementary oligonucleotides for sgRNAs were annealed, and cloned into LentiCrispr V2 vector. Green fluorescent protein (GFP) lentivirus vector was used as a control. LentiCrispr V2 vector was transfected to HEK-293T cells using calcium phosphate and media was exchanged after 12 h. The lentivirus was harvested from the media after 72 h, and viral particles were concentrated by centrifugation. HCT116 cells were transfected with CRISPR-Cas9 lentiviruses or the GFP lentivirus control. The GFP-positive cells were counted to ensure the efficiency of transfection. The expression of p300 and ACVR1B was analyzed by western blot. The effect of p300 and ACVR1B knockout on the cell viability, migration, invasion and CD8+T cell apoptosis was further evaluated.
p300 CRISPR/Cas9 KO plasmids (sc-435902) were purchased from Santa Cruz Biotechnology. MC38 cells were co-transfected with p300 CRISPR/Cas9 KO and HDR plasmids using Lipofectamine 3000 (Invitrogen) to generate p300 knockout cells (MC38 p300-/-). Pooled cells were selected using puromycin followed by fluorescence-activated single cell sorting to select cells expressing red fluorescent protein. The successful knockout was validated using western blot (data not shown).
Enzyme-linked immunosorbent assay (ELISA)
The concentration of PCSK6 in the cell culture supernatants were determined using a relevant PCSK6 ELISA kit (Innovative Research, Vancouver, Canada). Briefly, supernatant of CAFs were collected after centrifugation at 3000 g for 10 min and then subjected to ELISA assay.
In vivo imaging of tumor-bearing mice
C57BL/6 mice were anesthetized with isoflurane (3% induction, 1.5-2% maintenance). The cecum was exteriorized via a small midline laparotomy. A suspension of 2 × 106 luciferase-expressing MC38 cells in 50 µL DMEM was slowly injected into the serosal layer of the cecal wall using a 30-gauge needle. The injection site was gently compressed with sterile gauze for 30 s to prevent leakage. The cecum was then returned to the abdominal cavity, and the incision was closed in two layers. Animals were monitored until full recovery.
Tumor growth was monitored using a Tanon ABL X1 Lumina system. For imaging, mice received 150 mg/kg D-luciferin (200 μL, i.p.) in sterile PBS at day 14 post-injection. Ten minutes after luciferin injection, bioluminescence images were acquired with 1-minute exposure.
Cell viability assay
Briefly, CRC cells after corresponding treatment were harvested and seeded in 96-well plates (100 μL, 1×104 cells/well). To test the cell viability, 10 μL of CCK-8 reagent (Solarbio, Beijing, China) was added to each well and incubated for 2 h at 37 °C. Then, the absorption was evaluated by a microplate reader at 450 nm (Tecan, Sweden). Three wells were replicated for each group, and each experiment was repeated at least 3 times.
Then the CRC cells were co-cultured with activated T cells for another 24 h. After the removal of T cells by washing with PBS at 300 rpm for three times, CCK-8 solution (10 μL) was added into each well and incubated for 1 h. Cell viability was analyzed by microplate reader at 450 nm.
Cycloheximide (CHX) degradation assay
To exclude the translational effect, HCT116 were treated with a protein biosynthesis inhibitor CHX (50 μg/mL, Sigma), so that the only changes would be attributed to the already translated proteins. Briefly, for the degradation assays, HCT116 cells were treated with CHX (50 μg/mL, Sigma) to block the protein biosynthesis with or without rPCSK6 for the indicated periods of time (0 h, 1 h, 2 h, 3 h, 4 h, 5 h) and the levels of PD-L1 analyzed by immunoblotting with the indicated antibodies.
Site-directed mutagenesis
Mutants of Myc-tagged PD-L1 were constructed by site-directed mutagenesis using a two-step PCR technique. A deletion mutant of Myc-tagged PD-L1 were constructed from pGEX-4T-PD-L1 plasmid. cDNAs of PD-L1 corresponding to 1-90 amino acid (aa) deletion, 91-180 aa deletion, 181-235 aa deletion and 236-290 aa deletion were amplified by PCR and inserted into pGEX-4T. For construction of stable cell lines, pGEX-4T carrying the cDNAs of mutant Myc-tagged PD-L1 truncations were transfected into HEK293T using relevant kit (Invitrogen™). The potential acetylation site was determined via evaluating the PD-L1 acetylation level.
Quantitative reverse transcription-polymerase chain reaction (qPCR)
Cells were lysed, and then total RNA was extracted, following with the reverse transcribed using TRIzol Reagents (Invitrogen) and the HiScript III RT SuperMix for qPCR (+gDNA wiper) (R323, Vazyme, Nanjing, China) in accordance with the manufacturer’s instruction. The primers for PCSK6 were designed and purchased from Sangon biotech (Shanghai, China): PCSK6 (forward, 5’-ACTCCAGAAGAAGAGGAAGAGTA-3’; reverse, 5’-ACCATCGCAGCCTTTATCA-3’). Real-time qPCR was performed on the ABI 7900 Prism HT (Applied Biosystems, Shanghai, China), followed by melting curve analysis. The fold change in gene expression was normalized to GAPDH mRNA levels (forward, 5’- GAGTCAACGGATTTGGTCGT-3’; reverse, 5’-AATGAAGGGGTCATTGATGG-3’). Each treatment was assayed in triplicate and three independent experiments were performed. The expression levels were calculated using the 2−ΔΔCt method.
Western blot
Cells and tissues were obtained and then lysed on ice for 30 min using radioimmunoprecipitation assay (RIPA) buffer (Solarbio, Beijing, China). Equal amounts of cell lysates (the BCA protein assay kit and uniformized to 2 μg/μL) were subjected to electrophoresis in 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to polyvinylidene fluoride (PVDF) membranes for antibody blotting. The membranes were blocked with 5% BSA in TBST for 1 h at room temperature. the membrane was then incubated with primary antibody overnight at 4 °C, and then incubated with secondary antibody for 1 h. The enhanced chemiluminescence (ECL) reagent was used for detecting the immune-reactive proteins. Details of the antibodies used in the study can be seen in Supplementary Table 1.
Hematoxylin and eosin (HE) staining
The level of hepatic metastases was evaluated via HE staining. Briefly, the livers (harvested from the mice subjected to spleen injection of MC38) were washed, fixed in 4% paraformaldehyde for 24 h, dehydrated and then embedded in paraffin. The liver tissues were cut into sections followed by deparaffinization and rehydration. The sections were stained with hematoxylin and eosin solution (KeyGEN Biotech, Nanjing, China) for 5 min to perform histological analysis. The degree of tumor metastasis was quantified via ImageJ.
Immunohistochemistry (IHC)
Deparaffinized and rehydrated sequential sections were subjected to antigen retrieval by boiling in 10 mM sodium citrate, pH 6.0, for 15 min. Non-specific binding was blocked with 3% normal horse serum. The sections were stained with primary antibody against PCSK6, Ki-67, CD4, CD8, MLH1, MSH2, MSH6 and PMS2 respectively overnight at 4 °C for 48 h and then were washed in 0.1 M PBS for 30 min and incubated with a biotinylated secondary antibody of corresponding species for 2 h at room temperature, rinsed, incubated in 0.1% Extravadin Peroxidase (Sigma, St. Louis, MO) for 1 h, and then rinsed again. Immunolabelling was developed using a nickel-enhanced 3, 3’-diaminobenzidine (DAB) reaction. Tissues from the four experimental groups were performed simultaneously and the DAB reactions were developed for exactly the same length of time following the addition of glucose oxidase (1: 1000). Negative control sections, in which no primary antibodies were added, were developed at the same time to confirm the specificity of labeling. After processing was completed, sections were washed, mounted onto chrome alum-coated slides and cover-slipped.
GST fusion protein pull-down assay and in vitro acetylation assays
Recombinant Human PD-L1 (WT and K263R mutation) protein with a fusion GST was expressed in HEK293 Cells and purified via using glutathione-agarose beads. GST protein retained on the beads (about 0.5 μg protein) and/or 0.5 μg of active p300 recombinant protein (#81158, Active motif) were incubated in acetylation assay buffer (50 mM Tris pH 8.0, 0.1 mM EDTA, 50 ng/μL BSA) with or without 20 μM Acetyl-CoA (#10101893001, Sigma) for 1 h at 30°C. DNase (0.1 U/mL) and ethidium bromide (10 g/mL) were included in the binding reaction to reduce nonspecific DNA binding. Proteins were eluted by boiling in 1×SDS gel loading buffer and analyzed by western blot.
Survival analysis
For Kaplan-Meier survival analysis, percentages of surviving mice in the various groups were recorded daily following MC38 cell implantation (with or without CAFs). All remaining animals were sacrificed 6 weeks after the implantation. Humane end point was defined by significantly reduced physical activity, body weight reduction>15%, loss of righting response, onset of seizures, or pain anxiety symptom.
Flow cytometry staining and analysis
In both human and murine experiments, isolated immune cells were stained for flow cytometry immediately. Cells were stained with antibodies (BioLegend) according to the instruction of manufacturers. Cells were incubated with fluorochrome labeled antibodies to sort out CD45highCD3+T cells with CD4+ and CD8+ subfractions. Next, the ratio of CD8+GranB+T cells (effective cytotoxic T cells) in CD8+T cells and CD4+Foxp3+ T cells (regulatory T cells) in CD4+T cells were further analyzed. Moreover, the apoptosis (GranB+Annexin V+) and PD-1 expression (GranB+PD-1+) of CD8+T cells was analyzed.
The membrane expression of PD-L1 in HCT116 or MC38 cells was also evaluated via flow cytometry after the intervention of PCSK6 with or without shRNA transfection.
Dead cells were excluded with Dead Cell Removal Kit (NovoBiotechnology Co., Ltd., Beijing, China). Labeled cells were analyzed on DxFLEX Clinical Flow Cytometer (Beckman Colter Life Sciences, USA, C47508).
Invasion assay
For the invasion assay, a 24-well Transwell chamber containing a polycarbonate filter with 8 μm pores (Corning Inc) was used. Cancer cells were seeded to inserts with Matrigel coating (BD Biosciences) in 0.5 mL of serum-free DMEM. The lower well was filled with 0.5 mL of CM supplemented with 10% FBS. After 48 h, cells invading to the bottom surface of the membrane were fixed and stained. These cells were counted in five randomly selected areas under a 200× microscope.
Migration assay
The spreading and migration capabilities of CRC were assessed via scratch wound assay which measures the expansion of a cell population on surfaces. The cells were seeded into 24-well tissue culture dishes containing coverslips pre-coated with collagen type I (40 μg/mL) for 2 h at 37 °C, at a density of 3×105 cells/mL and cultured in medium containing 10% FBS to nearly confluent cell monolayers. Then, a linear wound was generated in the monolayer with a sterile 100 μL plastic pipette tip. Any cellular debris was fully removed by washing the coverslips with PBS. Scratched cells were incubated with rPCSK6 or CAFs-CM (with only 1% FBS). Twenty-four hours later, three representative images from each coverslip of the scratched areas under each condition were photographed to estimate the relative migration cells. The data were analyzed using ImageJ software. The experiments were performed at least in duplicate.
Immunofluorescence staining
Briefly, CRC cells were seeded into 24-well plates with glass coverslips. After the cell adhesion, cells were incubated with CAFs-CM, rPCSK6 or transfected with ACVR1B or p300 shRNA. Next, cells were fixed with 4% paraformaldehyde. After permeabilization with 0.1% Triton-X100 at room temperature for 15 min followed by three times washes with PBS, cells were then incubated with anti-PCSK6 (1: 200), anti-PD-L1 (1: 100), anti-ACVR1B (1: 100), anti-GM130 (1: 100) and anti-Rab8 (1: 200) overnight. fluorescein-conjugated goat anti-rabbit IgG (1:100) was used to incubate the cells for 1 h, then Dil (cell membrane orange fluorescent probe) and DAPI was used to stain the cells membrane and nucleus respectively for 5 min. Photographs were taken with a fluorescence microscope after glycerin gelatin was used to seal the coverslips on glass slides.
Immunoprecipitation and co-immunoprecipitation (Co-IP)
Protein was extracted from cells using lysis buffer (HEPES 50 µM, NaCl 150 µM, EDTA 1 mM, 1% Triton, 10% glycerol) supplemented with protease inhibitor cocktail, centrifuged at 4 °C, 10000 g for 30 min. After adding protein A/G agarose and antibody, the mixtures were incubated on rotator at 4 °C overnight. After washing the beads with 500 μL lysis buffer for three times, the beads were added loading buffer and denatured at 99 °C for 10 min. Proteins were isolated by electrophoresis using 10% polyacrylamide gel, then transferred to nitrocellulose membrane. 5% (w/v) milk (non-fat milk powder in TBST) was used to block nonspecific binding sites at room temperature for 2 h. Proteins on PVDF membrane were incubated in primary antibody overnight, antibodies employed in the study were listed in Supplementary Table 1. After washed with TBST for 5 times, membranes were incubated with secondary peroxidase-conjugated antibodies for 1 h, the detection of blots was performed with an enhanced chemiluminescence system (BIO-RAD, USA).
Antibody feeding assay
To investigate PD-L1 endocytosis and recycling in primary human CRC cells under conditions with or without rPCSK6 stimulation and p300 knockdown, an antibody feeding assay was performed with minor modifications. Briefly, primary CRC cells were seeded on glass coverslips and cultured to approximately 60–70% confluence. Live cells were incubated with monoclonal antibody recognizing the extracellular domain of human PD-L1 diluted in pre-warmed SFM at room temperature for 15 min to label surface-expressed PD-L1. After extensive washing to remove unbound antibody, cells were returned to complete medium and incubated at 37 °C for 50 min to allow receptor internalization. To selectively analyze PD-L1 recycling, surface-bound antibodies on non-internalized PD-L1 were blocked by incubation with excess unconjugated Fab fragments against IgG for 30 min at room temperature, followed by thorough washing. Cells were then incubated at 37 °C in the presence of dynasore to inhibit further endocytosis, allowing internalized PD-L1 to recycle back to the plasma membrane. Cells were subsequently fixed with paraformaldehyde without permeabilization and incubated with Alexa Fluor 488 conjugated secondary antibodies to label recycled PD-L1 at the cell surface. After membrane permeabilization, intracellular PD-L1 was visualized using a second Alexa Fluor 594-conjugated secondary antibody. Confocal microscopy was used for image acquisition, and quantitative analysis of PD-L1 internalization and recycling was performed by calculating the relative fluorescence intensity of surface-recycled versus intracellular PD-L1 pools using ImageJ software.
Statistical analysis
The sample size determination was based on experimental designs reported in previously published high-quality studies with similar methodologies [63,64,65,66], Statistical analysis and graph plotting were performed using SPSS 22.0 (SPSS standard version 22.0; SPSS Inc.) and Prism 8.0 software (GraphPad, San Diego, CA). Animals were randomly assigned to experimental groups using a random number table generated in Microsoft Excel. Group allocation, treatment administration, and outcome assessment were all performed under blinded conditions to minimize bias. For survival analysis, Log-rank test was used to determine significance between the experimental groups. The normality of data distribution was assessed using the Shapiro-Wilk test. Differences between two groups were assessed with an F-test and a two-tailed unpaired Student’s t‑test. For comparisons involving three or more groups (≥ 3), Levene’s test was first used to evaluate homogeneity of variances, followed by a one‑way ANOVA with LSD post hoc comparisons. If the assumption of equal variances is violated, Welch’s correction is applied to the ANOVA before conducting pairwise group comparisons. Data are presented as means ± SD (standard deviation) of three independent experiments. For all tests, a value of P < 0.05 was considered statistically significant.













Leave a Reply